Abstract
Oestrogens and their receptors contribute broadly to physiology and diseases. In premenopausal women, endogenous oestrogens protect against cardiovascular, metabolic and neurological diseases and are involved in hormone-sensitive cancers such as breast cancer. Oestrogens and oestrogen mimetics mediate their effects via the cytosolic and nuclear receptors oestrogen receptor-α (ERα) and oestrogen receptor-β (ERβ) and membrane subpopulations as well as the 7-transmembrane G protein-coupled oestrogen receptor (GPER). GPER, which dates back more than 450 million years in evolution, mediates both rapid signalling and transcriptional regulation. Oestrogen mimetics (such as phytooestrogens and xenooestrogens including endocrine disruptors) and licensed drugs such as selective oestrogen receptor modulators (SERMs) and downregulators (SERDs) also modulate oestrogen receptor activity in both health and disease. Following up on our previous Review of 2011, we herein summarize the progress made in the field of GPER research over the past decade. We will review molecular, cellular and pharmacological aspects of GPER signalling and function, its contribution to physiology, health and disease, and the potential of GPER to serve as a therapeutic target and prognostic indicator of numerous diseases. We also discuss the first clinical trial evaluating a GPER-selective drug and the opportunity of repurposing licensed drugs for the targeting of GPER in clinical medicine.
Key points
-
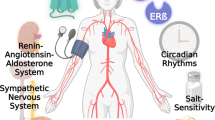
Oestrogens exert multiple activities in physiology, including reproduction, immunity, cardiovascular and endocrine functions, and ageing, as well as in diseases such as hormone-sensitive cancers, arterial hypertension, atherosclerosis and osteoporosis.
-
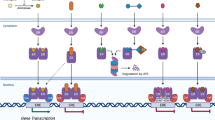
Oestrogen signalling mediates both acute (non-genomic) and chronic (transcriptional) effects through cytosolic or nuclear oestrogen receptors ERα and ERβ and membrane subpopulations and the G protein‐coupled oestrogen receptor (GPER), which is a 7-transmembrane protein.
-
Molecules that activate oestrogen receptors include natural oestrogens, phytooestrogens, mycooestrogens and synthetic compounds, such as selective oestrogen receptor modulators and downregulators and xenooestrogens (also known as endocrine disruptors), activate oestrogen receptors and/or GPER.
-
Research using Gper-deficient animals, GPER‐selective agonists and antagonists, and non-selective compounds has revealed multiple roles of GPER in physiology and disease, including as a constitutive activator of the reactive oxygen species-producing enzyme NOX1.
-
GPER holds potential to become a diagnostic, prognostic and therapeutic target in clinical medicine, including the repurposing of licensed drugs targeting GPER and the ongoing first-in-human clinical trial of the GPER-selective agonist G-1.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$32.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$189.00 per year
only $15.75 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Aristotélēs. Historia Animalium Books 1–10 (transl. and ed. Peck, A. L., Balme, D. M. & Gotthelf, A.) 1–624 (Harvard Univ. Press, 1965).
Starling, E. H. Croonian lecture: on the chemical correlation of the functions of the body. Lancet 2, 579–583 (1905).
Allen, E. & Doisy, E. A. An ovarian hormone: preliminary report on its localization, extraction and partial purification, and action in test animals. JAMA 81, 819–821 (1923).
Butenandt, A. Über “Progynon”, ein krystallisiertes weibliches Sexualhormon. Die Naturwissenschaften 17, 879 (1929).
Jensen, E. A conversation with Elwood Jensen. Interview by David D. Moore. Annu. Rev. Physiol. 74, 1–11 (2012).
Soloff, M. S. & Szego, C. M. Purification of estradiol receptor from rat uterus and blockade of its estrogen-binding function by specific antibody. Biochem. Biophys. Res. Commun. 34, 141–147 (1969).
Barton, M. et al. Twenty years of the G protein-coupled estrogen receptor GPER: historical and personal perspectives. J. Steroid Biochem. Mol. Biol. 176, 4–15 (2018).
Walter, P. et al. Cloning of the human estrogen receptor cDNA. Proc. Natl Acad. Sci. USA 82, 7889–7893 (1985). The cloning of ERα was a landmark accomplishment in the oestrogen and nuclear hormone receptor fields.
Kuiper, G. G., Enmark, E., Pelto-Huikko, M., Nilsson, S. & Gustafsson, J. A. Cloning of a novel receptor expressed in rat prostate and ovary. Proc. Natl Acad. Sci. USA 93, 5925–5930 (1996). One of two papers reporting the cloning of ERβ, revealing the existence of a second oestrogen receptor.
Mosselman, S., Polman, J. & Dijkema, R. ERβ: identification and characterization of a novel human estrogen receptor. FEBS Lett. 392, 49–53 (1996). One of two papers reporting the cloning of ERβ, revealing the existence of a second oestrogen receptor.
Prossnitz, E. R. & Barton, M. The G-protein-coupled estrogen receptor GPER in health and disease. Nat. Rev. Endocrinol. 7, 715–726 (2011). A comprehensive review summarizing knowledge in the GPER field in the first 10 years after GPER was identified as an oestrogen receptor.
Cabas, I., Chaves-Pozo, E., Mulero, V. & Garcia-Ayala, A. Role of estrogens in fish immunity with special emphasis on GPER1. Dev. Comp. Immunol. 89, 102–110 (2018).
Filardo, E. J., Quinn, J. A., Bland, K. I. & Frackelton, A. R. Jr. Estrogen-induced activation of Erk-1 and Erk-2 requires the G protein-coupled receptor homolog, GPR30, and occurs via trans-activation of the epidermal growth factor receptor through release of HB-EGF. Mol. Endocrinol. 14, 1649–1660 (2000). A landmark paper reporting the discovery of GPR30/GPER activation by oestrogen.
Revankar, C. M., Cimino, D. F., Sklar, L. A., Arterburn, J. B. & Prossnitz, E. R. A transmembrane intracellular estrogen receptor mediates rapid cell signaling. Science 307, 1625–1630 (2005). This is one of two key studies that first reported oestrogen binding to and signalling via GPER.
Thomas, P., Pang, Y., Filardo, E. J. & Dong, J. Identity of an oestrogen membrane receptor coupled to a G protein in human breast cancer cells. Endocrinology 146, 624–632 (2005). This is one of two key studies that first reported oestrogen binding to and signalling via GPER.
Alexander, S. P. H., Mathie, A. & Peters, J. A. Guide to Receptors and Channels (GRAC), 3rd edn. Br. J. Pharmacol. 153 (Suppl. 2), 1–209 (2008).
Gaudet, H. M., Cheng, S. B., Christensen, E. M. & Filardo, E. J. The G-protein coupled estrogen receptor, GPER: the inside and inside-out story. Mol. Cell Endocrinol. 418, 207–219 (2015).
Madeo, A. & Maggiolini, M. Nuclear alternate estrogen receptor GPR30 mediates 17β-estradiol-induced gene expression and migration in breast cancer-associated fibroblasts. Cancer Res. 70, 6036–6046 (2010).
Filardo, E. J., Quinn, J. A., Frackelton, A. R. Jr. & Bland, K. I. Estrogen action via the G protein-coupled receptor, GPR30: stimulation of adenylyl cyclase and cAMP-mediated attenuation of the epidermal growth factor receptor-to-MAPK signaling axis. Mol. Endocrinol. 16, 70–84 (2002).
Lucas, T. F., Royer, C., Siu, E. R., Lazari, M. F. & Porto, C. S. Expression and signaling of G protein-coupled estrogen receptor 1 (GPER) in rat Sertoli cells. Biol. Reprod. 83, 307–317 (2010).
Deng, Q. et al. GPER/Hippo-YAP signal is involved in bisphenol S induced migration of triple negative breast cancer (TNBC) cells. J. Hazard. Mater. 355, 1–9 (2018).
Daub, H., Weiss, F. U., Wallasch, C. & Ullrich, A. Role of transactivation of the EGF receptor in signalling by G-protein-coupled receptors. Nature 379, 557–560 (1996).
Zekas, E. & Prossnitz, E. R. Estrogen-mediated inactivation of FOXO3a by the G protein-coupled estrogen receptor GPER. BMC Cancer 15, 702 (2015).
Fredette, N. C., Meyer, M. R. & Prossnitz, E. R. Role of GPER in estrogen-dependent nitric oxide formation and vasodilation. J. Steroid Biochem. Mol. Biol. 176, 65–72 (2018).
Peixoto, P., Aires, R. D., Lemos, V. S., Bissoli, N. S. & Santos, R. L. D. GPER agonist dilates mesenteric arteries via PI3K-Akt-eNOS and potassium channels in both sexes. Life Sci. 183, 21–27 (2017).
Sharma, G. & Prossnitz, E. R. Mechanisms of estradiol-induced insulin secretion by the G protein-coupled estrogen receptor GPR30/GPER in pancreatic β-cells. Endocrinology 152, 3030–3039 (2011).
Greenlee, M. M. et al. Estradiol activates epithelial sodium channels in rat alveolar cells through the G protein-coupled estrogen receptor. Am. J. Physiol. Lung Cell Mol. Physiol. 305, L878–889 (2013).
Evanson, K. W., Goldsmith, J. A., Ghosh, P. & Delp, M. D. The G protein-coupled estrogen receptor agonist, G-1, attenuates BK channel activation in cerebral arterial smooth muscle cells. Pharmacol. Res. Perspect. 6, e00409 (2018).
Yue, J. et al. Activation of G-protein-coupled receptor 30 protects neurons against excitotoxicity through inhibiting excessive autophagy induced by glutamate. ACS Chem. Neurosci. 10, 4227–4236 (2019).
Zhang, H. et al. Mechanisms of estradiol-induced EGF-like factor expression and oocyte maturation via G protein-coupled estrogen receptor. Endocrinology 161, bqaa190 (2020).
Sun, M. et al. G protein-coupled estrogen receptor enhances melanogenesis via cAMP-protein kinase (PKA) by upregulating microphthalmia-related transcription factor-tyrosinase in melanoma. J. Steroid Biochem. Mol. Biol. 165, 236–246 (2017).
Pandey, D. P. et al. Estrogenic GPR30 signalling induces proliferation and migration of breast cancer cells through CTGF. EMBO J. 28, 523–532 (2009).
Cirillo, F. et al. GPER is involved in the regulation of the estrogen-metabolizing cyp1b1 enzyme in breast cancer. Oncotarget 8, 106608–106624 (2017).
Zhu, P. et al. GPER/ERK&AKT/NF-κB pathway is involved in cadmium-induced proliferation, invasion and migration of GPER-positive thyroid cancer cells. Mol. Cell Endocrinol. 442, 68–80 (2017).
Chen, Z. J. et al. Activation of GPER suppresses epithelial mesenchymal transition of triple negative breast cancer cells via NF-κB signals. Mol. Oncol. 10, 775–788 (2016).
De Francesco, E. M., Maggiolini, M. & Musti, A. M. Crosstalk between Notch, HIF-1α and GPER in breast cancer EMT. Int. J. Mol. Sci. 19, 2011 (2018).
Zhou, X. et al. Estrogen regulates Hippo signaling via GPER in breast cancer. J. Clin. Invest. 125, 2123–2135 (2015).
Zhu, L. et al. MicroRNA-2861 and microRNA-5115 regulates myocardial ischemia-reperfusion injury through the GPR30/mTOR signaling pathway by binding to GPR30. J. Cell Physiol. 235, 7791–7802 (2020).
Meyer, M. R. et al. Obligatory role for GPER in cardiovascular aging and disease. Sci. Signal. 9, ra105 (2016). This study reports regulation of the NADPH oxidase NOX1 by GPER expression and the discovery of GPER antagonists as NOX1 downregulators.
Barton, M., Meyer, M. R. & Prossnitz, E. R. Nox1 downregulators: a new class of therapeutics. Steroids 152, 108494 (2019).
Jensen, E. V. & Jordan, V. C. The estrogen receptor: a model for molecular medicine. Clin. Cancer Res. 9, 1980–1989 (2003).
Cheng, S. B. et al. Anatomical location and redistribution of G protein-coupled estrogen receptor-1 during the estrus cycle in mouse kidney and specific binding to estrogens but not aldosterone. Mol. Cell Endocrinol. 382, 950–959 (2014).
Rigiracciolo, D. C. et al. GPER is involved in the stimulatory effects of aldosterone in breast cancer cells and breast tumor-derived endothelial cells. Oncotarget 7, 94–111 (2016).
Lu, A. S., Rouhimoghadam, M., Arnatt, C. K., Filardo, E. J. & Salem, A. K. Proteolytic targeting chimeras with specificity for plasma membrane and intracellular estrogen receptors. Mol. Pharm. 18, 1455–1469 (2021).
Koganti, S., Snyder, R., Gumaste, U., Karamyan, V. T. & Thekkumkara, T. 2-Methoxyestradiol binding of GPR30 down-regulates angiotensin AT(1) receptor. Eur. J. Pharmacol. 723, 131–140 (2014).
Zucchetti, A. E. et al. G-protein-coupled receptor 30/adenylyl cyclase/protein kinase A pathway is involved in estradiol 17ss-D-glucuronide-induced cholestasis. Hepatology 59, 1016–1029 (2014).
Chourasia, T. K., Pang, Y. & Thomas, P. The catecholestrogen, 2-hydroxyestradiol-17beta, acts as a G protein-coupled estrogen receptor 1 (GPER/GPR30) antagonist to promote the resumption of meiosis in zebrafish oocytes. Biol. Reprod. 92(69), 1–13 (2015).
Teng, Y. et al. Dehydroepiandrosterone activation of G-protein-coupled estrogen receptor rapidly stimulates MicroRNA-21 transcription in human hepatocellular carcinoma cells. J. Biol. Chem. 290, 15799–15811 (2015).
Cao, J., Lu, M., Yan, W., Li, L. & Ma, H. Dehydroepiandrosterone alleviates intestinal inflammatory damage via GPR30-mediated nrf2 activation and NLRP3 inflammasome inhibition in colitis mice. Free Radic. Biol. Med. 172, 386–402 (2021).
Sandra, N., Ester, P., Marie-Agnes, P., Robert, M. & Olivier, H. The DHEA metabolite 7β-hydroxy-epiandrosterone exerts anti-estrogenic effects on breast cancer cell lines. Steroids 77, 542–551 (2012).
Avena, P. et al. 27-Hydroxycholesterol binds GPER and induces progression of estrogen receptor-negative breast cancer. Cancers 14, 1521 (2022).
Diamanti-Kandarakis, E. et al. Endocrine-disrupting chemicals: an Endocrine Society scientific statement. Endocr. Rev. 30, 293–342 (2009).
Lorand, T., Vigh, E. & Garai, J. Hormonal action of plant derived and anthropogenic non-steroidal estrogenic compounds: phytoestrogens and xenoestrogens. Curr. Med. Chem. 17, 3542–3574 (2010).
Thomas, P. & Dong, J. Binding and activation of the seven-transmembrane estrogen receptor GPR30 by environmental estrogens: a potential novel mechanism of endocrine disruption. J. Steroid Biochem. Mol. Biol. 102, 175–179 (2006).
Zhou, C., Li, P., Han, M. & Gao, X. Daidzein stimulates fatty acid-induced fat deposition in C2C12 myoblast cells via the G protein-coupled receptor 30 pathway. Anim. Biotechnol. 33, 851–863 (2020).
Moriyama, M. et al. S-equol, a major isoflavone from soybean, inhibits nitric oxide production in lipopolysaccharide-stimulated rat astrocytes partially via the GPR30-mediated pathway. Int. J. Inflam. 2018, 8496973 (2018).
Maggiolini, M. et al. The G protein-coupled receptor GPR30 mediates c-fos up-regulation by 17β-estradiol and phytoestrogens in breast cancer cells. J. Biol. Chem. 279, 27008–27016 (2004).
Dong, W. H., Chen, J. C., He, Y. L., Xu, J. J. & Mei, Y. A. Resveratrol inhibits K(v)2.2 currents through the estrogen receptor GPR30-mediated PKC pathway. Am. J. Physiol. Cell Physiol. 305, C547–557 (2013).
Chimento, A. et al. Oleuropein and hydroxytyrosol activate GPER/ GPR30-dependent pathways leading to apoptosis of ER-negative SKBR3 breast cancer cells. Mol. Nutr. Food Res. 58, 478–489 (2013).
Li, Y. C., Ding, X. S., Li, H. M. & Zhang, C. Icariin attenuates high glucose-induced type IV collagen and fibronectin accumulation in glomerular mesangial cells by inhibiting transforming growth factor-β production and signalling through G protein-coupled oestrogen receptor 1. Clin. Exp. Pharmacol. Physiol. 40, 635–643 (2013).
Moreno-Ulloa, A. et al. (-)-Epicatechin stimulates mitochondrial biogenesis and cell growth in C2C12 myotubes via the G-protein coupled estrogen receptor. Eur. J. Pharmacol. 822, 95–107 (2018).
Lo, E. K., Lee, J. C., Turner, P. C. & El-Nezami, H. Low dose of zearalenone elevated colon cancer cell growth through G protein-coupled estrogenic receptor. Sci. Rep. 11, 7403 (2021).
Paterni, I., Granchi, C., Katzenellenbogen, J. A. & Minutolo, F. Estrogen receptors alpha (ERα) and beta (ERβ): subtype-selective ligands and clinical potential. Steroids 90, 13–29 (2014).
Stauffer, S. R. et al. Pyrazole ligands: structure-affinity/activity relationships and estrogen receptor-α-selective agonists. J. Med. Chem. 43, 4934–4947 (2000).
Meyers, M. J. et al. Estrogen receptor-β potency-selective ligands: structure-activity relationship studies of diarylpropionitriles and their acetylene and polar analogues. J. Med. Chem. 44, 4230–4251 (2001).
Petrie, W. K. et al. G protein-coupled estrogen receptor-selective ligands modulate endometrial tumor growth. Obstet. Gynecol. Int. 2013, 472720 (2013).
Bologa, C. G. et al. Virtual and biomolecular screening converge on a selective agonist for GPR30. Nat. Chem. Biol. 2, 207–212 (2006). This study reports the first GPER-selective ligand G-1.
Dennis, M. K. et al. Identification of a GPER/GPR30 antagonist with improved estrogen receptor counterselectivity. J. Steroid Biochem. Mol. Biol. 127, 358–366 (2011).
Dennis, M. K. et al. In vivo effects of a GPR30 antagonist. Nat. Chem. Biol. 5, 421–427 (2009). This study reports the discovery of the first GPER-selective antagonist.
Urban, N., Leonhardt, M. & Schaefer, M. Multiplex GPCR screen reveals reliably acting agonists and a Gq-phospholipase C coupling mode of GPR30/GPER1. Mol. Pharmacol. 103, 48–62 (2023).
Tutzauer, J. et al. Ligand-independent G protein-coupled estrogen receptor/G protein-coupled receptor 30 activity: lack of receptor-dependent effects of G-1 and 17β-estradiol. Mol. Pharmacol. 100, 271–282 (2021).
Lappano, R. et al. Two novel GPER agonists induce gene expression changes and growth effects in cancer cells. Curr. Cancer Drug Targets 12, 531–542 (2012).
O’Dea, A., Sondergard, C., Sweeney, P. & Arnatt, C. K. A series of indole-thiazole derivatives act as GPER agonists and inhibit breast cancer cell growth. ACS Med. Chem. Lett. 9, 901–906 (2018).
DeLeon, C. et al. A novel GPER antagonist protects against the formation of estrogen-induced cholesterol gallstones in female mice. J. Lipid Res. 61, 767–777 (2020).
Lappano, R. et al. MIBE acts as antagonist ligand of both estrogen receptor α and GPER in breast cancer cells. Breast Cancer Res. 14, R12 (2012).
Bargagna-Mohan, P., Baek, S. H., Lee, H., Kim, K. & Mohan, R. Use of PROTACS as molecular probes of angiogenesis. Bioorg. Med. Chem. Lett. 15, 2724–2727 (2005).
Revankar, C. M. et al. A selective ligand for estrogen receptor proteins discriminates rapid and genomic signaling. Cell. Chem. Biol. 26, 1692–1702 (2019). This study reports the first oestrogen receptor-selective ligand (agonist) that does not bind GPER.
US National Library of Medicine. ClinicalTrials.gov https://clinicaltrials.gov/ct2/show/NCT04130516 (2019).
Muller, C. et al. Phase 1 trial of a novel, first-in-class G protein-coupled estrogen receptor (GPER) agonist, LNS8801 in patients with advanced or recurrent treatment-refractory solid malignancies. J. Clin. Oncol. 39, 3084 (2021).
Muller, C. et al. Phase 1b study of the novel first-in-class G protein-coupled estrogen receptor (GPER) agonist, LNS8801 in combination with pembrolizumab in patients with immune checkpoint inhibitor (ICI)-relapsed and refractory solid malignancies and dose escalation update. J. Clin. Oncol. 40, 2574 (2022).
Prossnitz, E. R. & Hathaway, H. J. What have we learned about GPER function in physiology and disease from knockout mice? J. Steroid Biochem. Mol. Biol. 153, 114–126 (2015). A review of GPER physiology based on studies with different Gper-knockout mouse strains.
Mielke, M. M. & Miller, V. M. Improving clinical outcomes through attention to sex and hormones in research. Nat. Rev. Endocrinol. 17, 625–635 (2021).
Mauvais-Jarvis, F. et al. Sex and gender: modifiers of health, disease, and medicine. Lancet 396, 565–582 (2020). An excellent overview of the roles of sex and sex steroids and their receptors in human physiology and disease.
Lafferty, A. R. et al. A novel genetic locus for low renin hypertension: familial hyperaldosteronism type II maps to chromosome 7 (7p22). J. Med. Genet. 37, 831–835 (2000).
Feldman, R. D. et al. A common hypofunctional genetic variant of GPER is associated with increased blood pressure in women. Br. J. Clin. Pharmacol. 78, 1441–1452 (2014). The first clinical genetics study to suggest a role of GPER in the regulation of arterial blood pressure and possibly in the pathogenesis of arterial hypertension in humans.
Fredette, N. C., Malik, E., Mukhtar, M. L., Prossnitz, E. R. & Terada, N. A hypertension patient derived induced pluripotent stem cell model demonstrates a role for GPER in hypertension risk and development. Am. J. Physiol. Cell. Physiol. 319, C825–C838 (2020).
Hussain, Y. et al. G-protein estrogen receptor as a regulator of low-density lipoprotein cholesterol metabolism: cellular and population genetic studies. Arterioscler. Thromb. Vasc. Biol. 35, 213–221 (2015).
Pupo, M. et al. A genetic polymorphism repurposes the G-protein coupled and membrane-associated estrogen receptor GPER to a transcription factor-like molecule promoting paracrine signaling between stroma and breast carcinoma cells. Oncotarget 8, 46728–46744 (2017).
Meyer, M. R. & Barton, M. Estrogens and coronary artery disease: new clinical perspectives. Adv. Pharmacol. 77, 307–360 (2016).
Barton, M. & Meyer, M. R. Heart failure with preserved ejection fraction in women: new clues to causes and treatment. JACC Basic Transl. Sci. 5, 296–299 (2020).
Haas, E. et al. Regulatory role of G protein-coupled estrogen receptor for vascular function and obesity. Circ. Res. 104, 288–291 (2009). The first study reporting acute changes in vascular tone and arterial blood pressure following activation of GPER and a regulatory role for endogenous GPER in obesity.
Meyer, M. R., Baretella, O., Prossnitz, E. R. & Barton, M. Dilation of epicardial coronary arteries by the G protein-coupled estrogen receptor agonists G-1 and ICI 182,780. Pharmacology 86, 58–64 (2010).
Arefin, S. et al. Vasodilatory effects of the selective GPER agonist G-1 is maximal in arteries of postmenopausal women. Maturitas 78, 123–130 (2014).
Yu, X. et al. Activation of G protein-coupled estrogen receptor induces endothelium-independent relaxation of coronary artery smooth muscle. Am. J. Physiol. Endocrinol. Metab. 301, E882–888 (2011).
Lindsey, S. H., Liu, L. & Chappell, M. C. Vasodilation by GPER in mesenteric arteries involves both endothelial nitric oxide and smooth muscle cAMP signaling. Steroids 81, 99–102 (2014).
Pang, Y. & Thomas, P. Additive effects of low concentrations of estradiol-17β and progesterone on nitric oxide production by human vascular endothelial cells through shared signaling pathways. J. Steroid Biochem. Mol. Biol. 165, 258–267 (2017).
Yu, X., Stallone, J. N., Heaps, C. L. & Han, G. The activation of G protein-coupled estrogen receptor induces relaxation via cAMP as well as potentiates contraction via EGFR transactivation in porcine coronary arteries. PLoS ONE 13, e0191418 (2018).
Yu, X. et al. Activation of G protein-coupled estrogen receptor 1 induces coronary artery relaxation via Epac/Rap1-mediated inhibition of RhoA/Rho kinase pathway in parallel with PKA. PLoS ONE 12, e0173085 (2017).
Meyer, M. R., Fredette, N. C., Barton, M. & Prossnitz, E. R. G protein-coupled estrogen receptor inhibits vascular prostanoid production and activity. J. Endocrinol. 227, 61–69 (2015).
Meyer, M. R., Field, A. S., Kanagy, N. L., Barton, M. & Prossnitz, E. R. GPER regulates endothelin-dependent vascular tone and intracellular calcium. Life Sci. 91, 623–627 (2012).
Tropea, T. et al. Pregnancy augments G protein estrogen receptor (GPER) induced vasodilation in rat uterine arteries via the nitric oxide–cGMP signaling pathway. PLoS ONE 10, e0141997 (2015).
Gurrala, R. et al. Alterations in the estrogen receptor profile of cardiovascular tissues during aging. Geroscience 43, 433–442 (2021).
Meyer, M. R., Rosemann, T., Barton, M. & Prossnitz, E. R. GPER mediates functional endothelial aging in renal arteries. Pharmacology 100, 188–193 (2017).
Davis, G. K., Newsome, A. D., Cole, A. B., Ojeda, N. B. & Alexander, B. T. Chronic estrogen supplementation prevents the increase in blood pressure in female intrauterine growth-restricted offspring at 12 months of age. Hypertension 73, 1128–1136 (2019).
Luo, P. et al. Stress-related arterial hypertension in Gper-deficient rats. Acta Physiol. Sin. 69, 532–540 (2017).
Waghulde, H. et al. Attenuation of microbiotal dysbiosis and hypertension in a CRISPR/Cas9 gene ablation rat model of GPER1. Hypertension 72, 1125–1132 (2018).
Gohar, E. Y. et al. Evidence for G-protein-coupled estrogen receptor as a pronatriuretic factor. J. Am. Heart Assoc. 9, e015110 (2020). This study reports the identification of GPER as a modulator of natriuresis.
Barton, M. & Meyer, M. R. Nicolaus Copernicus and the rapid vascular responses to aldosterone. Trends Endocrinol. Metab. 26, 396–398 (2015).
Wehling, M. Rapid actions of aldosterone revisited: receptors in the limelight. J. Steroid Biochem. Mol. Biol. 176, 94–98 (2018).
Dinh, Q. N. et al. Aldosterone-induced hypertension is sex-dependent, mediated by T cells and sensitive to GPER activation. Cardiovasc. Res. 117, 960–970 (2021). An important study identifying T cell-mediated mechanisms involving GPER as sex-dependent modifiers of aldosterone-dependent arterial hypertension.
Caroccia, B. et al. Aldosterone stimulates its biosynthesis via a novel GPER-mediated mechanism. J. Clin. Endocrinol. Metab. 104, 6316–6324 (2019). This study reports the regulation of adrenal aldosterone biosynthesis involving GPER-dependent mechanisms.
Wang, D., Wang, M., Sun, P. & Gao, Q. Eplerenone inhibits oxidized low-density lipoprotein-induced proliferation and migration of vascular smooth muscle cells by downregulating GPER expression. Adv. Clin. Exp. Med. 30, 405–412 (2021).
Cheng, L. et al. Rapid aldosterone-mediated signaling in the DCT increases activity of the thiazide-sensitive NaCl cotransporter. J. Am. Soc. Nephrol. 30, 1454–1470 (2019).
Sharma, G. et al. GPER deficiency in male mice results in insulin resistance, dyslipidemia, and a proinflammatory state. Endocrinology 154, 4136–4145 (2013).
Meoli, L. et al. Sex- and age-dependent effects of Gpr30 genetic deletion on the metabolic and cardiovascular profiles of diet-induced obese mice. Gene 540, 210–216 (2014).
Ghaffari, S., Naderi Nabi, F., Sugiyama, M. G. & Lee, W. L. Estrogen inhibits LDL (low-density lipoprotein) transcytosis by human coronary artery endothelial cells via GPER (G-protein-coupled estrogen receptor) and SR-BI (scavenger receptor class B type 1). Arterioscler. Thromb. Vasc. Biol. 38, 2283–2294 (2018). This study demonstrated that oestrogen-dependent inhibition of LDL cholesterol transcytosis, which is involved in early atherogenesis, is mediated by GPER and scavenger receptor B1.
Jafarynezhad, F. et al. The G-protein-coupled estrogen receptor agonist prevents cardiac lipid accumulation by stimulating cardiac peroxisome proliferator-activated receptor alpha: a preclinical study in ovariectomized-diabetic rat model. Int. J. Endocrinol. Metab. 20, e123560 (2022).
Locher, R., Emmanuele, L., Suter, P. M., Vetter, W. & Barton, M. Green tea polyphenols inhibit human vascular smooth muscle cell proliferation stimulated by native low-density lipoprotein. Eur. J. Pharmacol. 434, 1–7 (2002).
Blasko, E. et al. Beneficial role of the GPR30 agonist G-1 in an animal model of multiple sclerosis. J. Neuroimmunol. 214, 67–77 (2009). One of two studies to first suggest a role for GPER in experimental multiple sclerosis.
Pelekanou, V. et al. Estrogen anti-inflammatory activity on human monocytes is mediated through cross-talk between estrogen receptor ERα36 and GPR30/GPER1. J. Leukoc. Biol. 99, 333–347 (2016).
Haas, E. et al. Differential effects of 17β-estradiol on function and expression of estrogen receptor alpha, estrogen receptor beta, and GPR30 in arteries and veins of patients with atherosclerosis. Hypertension 49, 1358–1363 (2007).
Li, F. et al. Activation of GPER induces differentiation and inhibition of coronary artery smooth muscle cell proliferation. PLoS ONE 8, e64771 (2013).
Jehle, J. et al. G protein-coupled estrogen receptor GPR30 exerts vasoprotective effects in apolipoprotein E-deficient mice. Arch. Med. Sci. https://doi.org/10.5114/aoms/127200 (2021).
Moreno-Ulloa, A. et al. The effects of (-)-epicatechin on endothelial cells involve the G protein-coupled estrogen receptor (GPER). Pharmacol. Res. 100, 309–320 (2015).
Meyer, M. R., Fredette, N. C., Barton, M. & Prossnitz, E. R. Regulation of vascular smooth muscle tone by adipose-derived contracting factor. PLoS ONE 8, e79245 (2013).
Meyer, M. R. et al. G protein-coupled estrogen receptor protects from atherosclerosis. Sci. Rep. 4, 7564 (2014).
Bopassa, J. C., Eghbali, M., Toro, L. & Stefani, E. A novel estrogen receptor GPER inhibits mitochondria permeability transition pore opening and protects the heart against ischemia-reperfusion injury. Am. J. Physiol. Heart Circ. Physiol. 298, H16–23 (2010). First study reporting a protective role of GPER in myocardial reperfusion injury from myocardial ischaemia following coronary occlusion.
Kabir, M. E. et al. G protein-coupled estrogen receptor 1 mediates acute estrogen-induced cardioprotection via MEK/ERK/GSK-3β pathway after ischemia/reperfusion. PLoS ONE 10, e0135988 (2015).
Feng, Y., Madungwe, N. B., da Cruz Junho, C. V. & Bopassa, J. C. Activation of G protein-coupled oestrogen receptor 1 at the onset of reperfusion protects the myocardium against ischemia/reperfusion injury by reducing mitochondrial dysfunction and mitophagy. Br. J. Pharmacol. 174, 4329–4344 (2017).
Iorga, A. et al. Rescue of pressure overload-induced heart failure by estrogen therapy. J. Am. Heart Assoc. 5, e002482 (2016).
Goncalves, G. K. et al. Neonatal cardiomyocyte hypertrophy induced by endothelin-1 is blocked by estradiol acting on GPER. Am. J. Physiol. Cell Physiol. 314, C310–C322 (2018).
Watanabe, T. et al. 17β-Estradiol inhibits cardiac fibroblast growth through both subtypes of estrogen receptor. Biochem. Biophys. Res. Commun. 311, 454–459 (2003).
Mercier, I., Mader, S. & Calderone, A. Tamoxifen and ICI 182,780 negatively influenced cardiac cell growth via an estrogen receptor-independent mechanism. Cardiovasc. Res. 59, 883–892 (2003).
Recchia, A. G. et al. The G protein-coupled receptor 30 is up-regulated by hypoxia-inducible factor-1α (HIF-1α) in breast cancer cells and cardiomyocytes. J. Biol. Chem. 286, 10773–10782 (2011).
Whitcomb, V. et al. Regulation of beta adrenoceptor-mediated myocardial contraction and calcium dynamics by the G protein-coupled estrogen receptor 1. Biochem. Pharmacol. 171, 113727 (2020).
Wang, H., Sun, X., Hodge, H. S., Ferrario, C. M. & Groban, L. NLRP3 inhibition improves heart function in GPER knockout mice. Biochem. Biophys. Res. Commun. 514, 998–1003 (2019). This study reports that inhibition of the NLRP3 inflammasome ameliorates impaired systolic and diastolic myocardial function in mice lacking GPER.
Wang, H. et al. Activation of GPR30 attenuates diastolic dysfunction and left ventricle remodelling in oophorectomized mRen2.Lewis rats. Cardiovasc. Res. 94, 96–104 (2012).
Jessup, J. A., Lindsey, S. H., Wang, H., Chappell, M. C. & Groban, L. Attenuation of salt-induced cardiac remodeling and diastolic dysfunction by the GPER agonist G-1 in female mRen2.Lewis rats. PLoS ONE 5, e15433 (2010). The first report to suggest that pharmacological activation of GPER reduces functional and structural cardiac injury in salt-sensitive hypertension.
Alencar, A. K. et al. Effect of age, estrogen status, and late-life GPER activation on cardiac structure and function in the Fischer344xBrown Norway female rat. J. Gerontol. A Biol. Sci. Med. Sci. 72, 152–162 (2017).
Bairey Merz, C. N. et al. Sex and the kidneys: current understanding and research opportunities. Nat. Rev. Nephrol. 15, 776–783 (2019).
Chang, Y. et al. G protein-coupled estrogen receptor activation improves contractile and diastolic functions in rat renal interlobular artery to protect against renal ischemia reperfusion injury. Biomed. Pharmacother. 112, 108666 (2019).
Hofmeister, M. V. et al. 17β-Estradiol induces nongenomic effects in renal intercalated cells through G protein-coupled estrogen receptor 1. Am. J. Physiol. Ren. Physiol. 302, F358–368 (2012).
Gohar, E. Y. & Pollock, D. M. Functional interaction of endothelin receptors in mediating natriuresis evoked by G protein-coupled estrogen receptor 1. J. Pharmacol. Exp. Ther. 376, 98–105 (2021).
Meyer, M. R. et al. GPER is required for age-dependent albuminuria and glomerulosclerosis: evidence for its role in podocyte injury and mesangial Nox1 regulation. Hypertension 72, AP261 (2018).
Meyer, M. R. et al. Deletion of GPER protects from age-related renovascular dysfunction and tubulo-interstitial injury. Hypertension 66, AP607 (2015).
Qiao, C., Ye, W., Li, S., Wang, H. & Ding, X. Icariin modulates mitochondrial function and apoptosis in high glucose-induced glomerular podocytes through G protein-coupled estrogen receptors. Mol. Cell. Endocrinol. 473, 146–155 (2018).
Lindsey, S. H., Yamaleyeva, L. M., Brosnihan, K. B., Gallagher, P. E. & Chappell, M. C. Estrogen receptor GPR30 reduces oxidative stress and proteinuria in the salt-sensitive female mRen2.Lewis rat. Hypertension 58, 665–671 (2011). The first study to report the antiproteinuric effects of pharmacological activation of GPER.
Gohar, E. Y. et al. Activation of G protein-coupled estrogen receptor 1 ameliorates proximal tubular injury and proteinuria in Dahl salt-sensitive female rats. Am. J. Physiol. Regul. Integr. Comp. Physiol. 320, R297–R306 (2021).
Sanchez, D. S. et al. Estradiol stimulates cell proliferation via classic estrogen receptor-alpha and G protein-coupled estrogen receptor-1 in human renal tubular epithelial cell primary cultures. Biochem. Biophys. Res. Commun. 512, 170–175 (2019).
Kurt, A. H., Bozkus, F., Uremis, N. & Uremis, M. M. The protective role of G protein-coupled estrogen receptor 1 (GPER-1) on methotrexate-induced nephrotoxicity in human renal epithelium cells. Ren. Fail. 38, 686–692 (2016).
Hutchens, M. P., Fujiyoshi, T., Komers, R., Herson, P. S. & Anderson, S. Estrogen protects renal endothelial barrier function from ischemia-reperfusion in vitro and in vivo. Am. J. Physiol. Ren. Physiol. 303, F377–385 (2012).
Tofovic, S. P., Zhang, X., Jackson, E. K., Zhu, H. & Petrusevska, G. 2-Methoxyestradiol attenuates bleomycin-induced pulmonary hypertension and fibrosis in estrogen-deficient rats. Vasc. Pharmacol. 51, 190–197 (2009).
Umar, S. et al. Estrogen rescues preexisting severe pulmonary hypertension in rats. Am. J. Respir. Crit. Care Med. 184, 715–723 (2011).
Alencar, A. K. N. et al. Cardioprotection induced by activation of GPER in ovariectomized rats with pulmonary hypertension. J. Gerontol. A Biol. Sci. Med. Sci. 73, 1158–1166 (2018).
Alencar, A. K. et al. Activation of GPER ameliorates experimental pulmonary hypertension in male rats. Eur. J. Pharm. Sci. 97, 208–217 (2017). This is the first study to report therapeutic efficacy and protection of the right ventricle of the heart by pharmacological activation of GPER in experimental pulmonary arterial hypertension.
Ahmadian, R. et al. GPER contributes to the development of pulmonary hypertension in female rats. FASEB J. 34, 1 (2020).
Meyer, M. R. & Barton, M. GPER blockers as Nox downregulators: a new drug class to target chronic non-communicable diseases. J. Steroid Biochem. Mol. Biol. 176, 82–87 (2018).
Mauvais-Jarvis, F., Clegg, D. J. & Hevener, A. L. The role of estrogens in control of energy balance and glucose homeostasis. Endocr. Rev. 34, 309–338 (2013). A comprehensive review of oestrogen receptor-dependent and GPER-dependent regulation glucose homeostasis and energy balance.
Meyer, M. R., Clegg, D. J., Prossnitz, E. R. & Barton, M. Obesity, insulin resistance and diabetes: sex differences and role of oestrogen receptors. Acta Physiol. 203, 259–269 (2011).
Madak-Erdogan, Z. et al. Design of pathway preferential estrogens that provide beneficial metabolic and vascular effects without stimulating reproductive tissues. Sci. Signal. 9, ra53 (2016). This study reports the identification of ‘pathway-preferential’ oestrogens with beneficial vascular and metabolic effects without stimulating reproductive tissues.
Gurney, E. P., Nachtigall, M. J., Nachtigall, L. E. & Naftolin, F. The Women’s Health Initiative Trial and related studies: 10 years later: a clinician’s view. J. Steroid Biochem. Mol. Biol. 142, 4–11 (2014).
Stubbins, R. E., Holcomb, V. B., Hong, J. & Nunez, N. P. Estrogen modulates abdominal adiposity and protects female mice from obesity and impaired glucose tolerance. Eur. J. Nutr. 51, 861–870 (2012).
Bonds, D. E. et al. The effect of conjugated equine oestrogen on diabetes incidence: the women’s health initiative randomised trial. Diabetologia 49, 459–468 (2006).
Mårtensson, U. E. et al. Deletion of the G protein-coupled receptor 30 impairs glucose tolerance, reduces bone growth, increases blood pressure, and eliminates estradiol-stimulated insulin release in female mice. Endocrinology 150, 687–698 (2009).
Davis, K. E. et al. Sexually dimorphic role of G protein-coupled estrogen receptor (GPER) in modulating energy homeostasis. Horm. Behav. 66, 196–207 (2014).
Le May, C. et al. Estrogens protect pancreatic β-cells from apoptosis and prevent insulin-deficient diabetes mellitus in mice. Proc. Natl Acad. Sci. USA 103, 9232–9237 (2006).
Liu, S. et al. Importance of extranuclear estrogen receptor-α and membrane G protein-coupled estrogen receptor in pancreatic islet survival. Diabetes 58, 2292–2302 (2009).This study is the first to report a role of GPER in the regulation of pancreatic islet survival.
Sharma, G. et al. Preclinical efficacy of the GPER-selective agonist G-1 in mouse models of obesity and diabetes. Sci. Transl Med. 12, eaau5956 (2020). The study reports efficacy of the GPER agonist G-1 for the treatment of obesity, and the associated insulin resistance and diabetes mellitus.
Butler, M. J., Hildebrandt, R. P. & Eckel, L. A. Selective activation of estrogen receptors, ERα and GPER-1, rapidly decreases food intake in female rats. Horm. Behav. 103, 54–61 (2018).
Azizian, H., Khaksari, M., Asadi Karam, G., Esmailidehaj, M. & Farhadi, Z. Cardioprotective and anti-inflammatory effects of G-protein coupled receptor 30 (GPR30) on postmenopausal type 2 diabetic rats. Biomed. Pharmacother. 108, 153–164 (2018).
Balhuizen, A., Kumar, R., Amisten, S., Lundquist, I. & Salehi, A. Activation of G protein-coupled receptor 30 modulates hormone secretion and counteracts cytokine-induced apoptosis in pancreatic islets of female mice. Mol. Cell. Endocrinol. 320, 16–24 (2010).
Kumar, R., Balhuizen, A., Amisten, S., Lundquist, I. & Salehi, A. Insulinotropic and antidiabetic effects of 17β-estradiol and the GPR30 agonist G-1 on human pancreatic islets. Endocrinology 152, 2568–2579 (2011).
Jacenik, D., Beswick, E. J., Krajewska, W. M. & Prossnitz, E. R. G protein-coupled estrogen receptor in colon function, immune regulation and carcinogenesis. World J. Gastroenterol. 25, 4092–4104 (2019).
Hata, M. et al. GPR30-expressing gastric chief cells do not dedifferentiate but are eliminated via PDK-dependent cell competition during development of metaplasia. Gastroenterology 158, 1650–1666.e15 (2020).
Tsai, C. C. et al. Estradiol mediates relaxation of porcine lower esophageal sphincter. Steroids 136, 56–62 (2018).
Zielinska, M. et al. G protein-coupled estrogen receptor and estrogen receptor ligands regulate colonic motility and visceral pain. Neurogastroenterol. Motil. 29, 13025 (2017).
Li, Y. et al. G protein-coupled estrogen receptor is involved in modulating colonic motor function via nitric oxide release in C57BL/6 female mice. Neurogastroenterol. Motil. 28, 432–442 (2016).
Jacenik, D. et al. G protein-coupled estrogen receptor mediates anti-inflammatory action in Crohn’s disease. Sci. Rep. 9, 6749 (2019). This study reports that pharmacological activation of GPER reduces inflammatory activation and mortality in experimental Crohn’s disease.
Wlodarczyk, M. et al. G protein-coupled receptor 30 (GPR30) expression pattern in inflammatory bowel disease patients suggests its key role in the inflammatory process. A preliminary study. J. Gastrointestin. Liver. Dis. 26, 29–35 (2017).
Qin, B. et al. Expression of G protein-coupled estrogen receptor in irritable bowel syndrome and its clinical significance. Int. J. Clin. Exp. Pathol. 7, 2238–2246 (2014).
Shao, X., Li, J., Xu, F., Chen, D. & Liu, K. Mir-155-mediated deregulation of GPER1 plays an important role in the gender differences related to inflammatory bowel disease. Can. J. Infect. Dis. Med. Microbiol. 2020, 8811477 (2020).
Jacenik, D. et al. Estrogen signaling deregulation related with local immune response modulation in irritable bowel syndrome. Mol. Cell. Endocrinol. 471, 89–96 (2018).
Chai, S. et al. Activation of G protein-coupled estrogen receptor protects intestine from ischemia/reperfusion injury in mice by protecting the crypt cell proliferation. Clin. Sci. 133, 449–464 (2019).
Wang, Q. et al. Activation of the G protein-coupled estrogen receptor prevented the development of acute colitis by protecting the crypt cell. J. Pharmacol. Exp. Ther. 376, 281–293 (2021).
Chaturantabut, S. et al. Estrogen activation of G-protein-coupled estrogen receptor 1 regulates phosphoinositide 3-kinase and mTOR signaling to promote liver growth in zebrafish and proliferation of human hepatocytes. Gastroenterology 156, 1788–1804.e13 (2019). An important paper reporting that pharmacological activation of GPER stimulates hepatocyte proliferation and thus liver regeneration.
Tian, L. et al. The developmental wnt signaling pathway effector β-catenin/TCF mediates hepatic functions of the sex hormone estradiol in regulating lipid metabolism. PLoS Biol. 17, e3000444 (2019).
Farruggio, S. et al. Genistein improves viability, proliferation and mitochondrial function of cardiomyoblasts cultured in physiologic and peroxidative conditions. Int. J. Mol. Med. 44, 2298–2310 (2019).
Wang, H. H., Liu, M., Clegg, D. J., Portincasa, P. & Wang, D. Q. New insights into the molecular mechanisms underlying effects of estrogen on cholesterol gallstone formation. Biochim. Biophys. Acta 1791, 1037–1047 (2009).
de Bari, O., Wang, T. Y., Liu, M., Portincasa, P. & Wang, D. Q. Estrogen induces two distinct cholesterol crystallization pathways by activating ERα and GPR30 in female mice. J. Lipid Res. 56, 1691–1700 (2015).
Wang, H. H. et al. Activation of a novel estrogen receptor GPR30 enhances cholesterol cholelithogenesis in female mice. Hepatology 72, 2077–2089 (2020). This study causally links GPER to oestrogen-dependent gallstone formation in female mice.
Filardo, E. J. et al. Distribution of GPR30, a seven membrane-spanning estrogen receptor, in primary breast cancer and its association with clinicopathologic determinants of tumor progression. Clin. Cancer Res. 12, 6359–6366 (2006). The first report showing an association between GPER expression and clinical outcome in breast cancer.
Ignatov, T., Treeck, O., Kalinski, T., Ortmann, O. & Ignatov, A. GPER-1 expression is associated with a decreased response rate to primary tamoxifen therapy of breast cancer patients. Arch. Gynecol. Obstet. 301, 565–571 (2020).
Ignatov, T. et al. G-protein-coupled estrogen receptor GPER-1 expression in hormone receptor-positive breast cancer is associated with poor benefit of tamoxifen. Breast Cancer Res. Treat. 174, 121–127 (2019).
Pepermans, R. A., Sharma, G. & Prossnitz, E. R. G protein-coupled estrogen receptor in cancer and stromal cells: functions and novel therapeutic perspectives. Cells 10, 672 (2021).
De Francesco, E. M. et al. GPER mediates activation of HIF1α/VEGF signaling by estrogens. Cancer Res. 74, 4053–4064 (2014).
Smith, H. O. et al. GPR30: a novel indicator of poor survival for endometrial carcinoma. Am. J. Obstet. Gynecol. 196 (386), e381–e389 (2007). This study was the first to suggest an association between GPER expression and clinical outcome and survival in patients with endometrial cancer.
Smith, H. O. et al. GPR30 predicts poor survival for ovarian cancer. Gynecol. Oncol. 114, 465–471 (2009). This study was the first to suggest an association between GPER expression and clinical outcome and survival in patients with ovarian cancer.
Chan, Q. K. et al. Activation of GPR30 inhibits the growth of prostate cancer cells through sustained activation of Erk1/2, c-jun/c-fos-dependent upregulation of p21, and induction of G(2) cell-cycle arrest. Cell. Death Differ. 17, 1511–1523 (2010).
Natale, C. A. et al. Pharmacologic activation of the G protein-coupled estrogen receptor inhibits pancreatic ductal adenocarcinoma. Cell. Mol. Gastroenterol. Hepatol. 10, 868–880 (2020). This study reports that pharmacological activation of GPER enhances the efficacy of immune checkpoint inhibitors (ICIs) and prolongs survival in a model of pancreatic cancer.
Vivacqua, A. et al. 17β-Estradiol, genistein, and 4-hydroxytamoxifen induce the proliferation of thyroid cancer cells through the G protein-coupled receptor GPR30. Mol. Pharmacol. 70, 1414–1423 (2006).
Gilligan, L. C. et al. Estrogen activation by steroid sulfatase increases colorectal cancer proliferation via GPER. J. Clin. Endocrinol. Metab. 102, 4435–4447 (2017).
Liu, C. et al. G-protein-coupled estrogen receptor antagonist G15 decreases estrogen-induced development of non-small cell lung cancer. Oncol. Res. 27, 283–292 (2019).
Scaling, A. L., Prossnitz, E. R. & Hathaway, H. J. GPER mediates estrogen-induced signaling and proliferation in human breast epithelial cells and normal and malignant breast. Horm. Cancer 5, 146–160 (2014).
Vivacqua, A. et al. The G protein-coupled receptor GPR30 mediates the proliferative effects induced by 17β-estradiol and hydroxytamoxifen in endometrial cancer cells. Mol. Endocrinol. 20, 631–646 (2006).
Albanito, L. et al. G protein-coupled receptor 30 (GPR30) mediates gene expression changes and growth response to 17β-estradiol and selective GPR30 ligand G-1 in ovarian cancer cells. Cancer Res. 67, 1859–1866 (2007).
Ariazi, E. A. et al. The G protein-coupled receptor GPR30 inhibits proliferation of estrogen receptor-positive breast cancer cells. Cancer Res. 70, 1184–1194 (2010).
Natale, C. A. et al. Activation of G protein-coupled estrogen receptor signaling inhibits melanoma and improves response to immune checkpoint blockade. eLife 7, e31770 (2018). This study reports that pharmacological activation of GPER enhances the efficacy of ICI and prolongs survival in a mouse model of malignant melanoma.
Wei, T. et al. G protein-coupled estrogen receptor deficiency accelerates liver tumorigenesis by enhancing inflammation and fibrosis. Cancer Lett. 382, 195–202 (2016).
McDonnell, D. P., Wardell, S. E., Chang, C. Y. & Norris, J. D. Next-generation endocrine therapies for breast cancer. J. Clin. Oncol. 39, 1383–1388 (2021).
Mo, Z. et al. GPR30 as an initiator of tamoxifen resistance in hormone-dependent breast cancer. Breast Cancer Res. 15, R114 (2013).
Ignatov, A. et al. G-protein-coupled estrogen receptor GPR30 and tamoxifen resistance in breast cancer. Breast Cancer Res. Treat. 128, 457–466 (2011).
Marjon, N. A., Hu, C., Hathaway, H. J. & Prossnitz, E. R. G protein-coupled estrogen receptor regulates mammary tumorigenesis and metastasis. Mol. Cancer Res. 12, 1644–1654 (2014).
Ignatov, A., Ignatov, T., Roessner, A., Costa, S. D. & Kalinski, T. Role of GPR30 in the mechanisms of tamoxifen resistance in breast cancer MCF-7 cells. Breast Cancer Res. Treat. 123, 87–96 (2010).
Prossnitz, E. R. & Arterburn, J. B. International Union of Basic and Clinical Pharmacology. XCVII. G protein-coupled estrogen receptor and its pharmacologic modulators. Pharmacol. Rev. 67, 505–540 (2015).
Li, Y. et al. 4-Hydroxytamoxifen-stimulated processing of cyclin E is mediated via G protein-coupled receptor 30 (GPR30) and accompanied by enhanced migration in MCF-7 breast cancer cells. Toxicology 309, 61–65 (2013).
Catalano, S. et al. Tamoxifen through GPER upregulates aromatase expression: a novel mechanism sustaining tamoxifen-resistant breast cancer cell growth. Breast Cancer Res. Treat. 146, 273–285 (2014).
Liu, Y. et al. G15 sensitizes epithelial breast cancer cells to doxorubicin by preventing epithelial-mesenchymal transition through inhibition of GPR30. Am. J. Transl Res. 7, 967–975 (2015).
Wolfson, B., Padget, M. R., Schlom, J. & Hodge, J. W. Exploiting off-target effects of estrogen deprivation to sensitize estrogen receptor negative breast cancer to immune killing. J. Immunother. Cancer 9, e002258 (2021).
Luo, H. et al. GPER-mediated proliferation and estradiol production in breast cancer-associated fibroblasts. Endocr. Relat. Cancer 21, 355–369 (2014).
Yang, K. & Yao, Y. Mechanism of GPER promoting proliferation, migration and invasion of triple-negative breast cancer cells through CAF. Am. J. Transl Res. 11, 5858–5868 (2019).
Ren, J. et al. GPER in CAFs regulates hypoxia-driven breast cancer invasion in a CTGF-dependent manner. Oncol. Rep. 33, 1929–1937 (2015).
Santolla, M. F. et al. GPER mediates a feedforward FGF2/FGFR1 paracrine activation coupling CAFs to cancer cells toward breast tumor progression. Cells 8, 223 (2019).
De Marco, P. et al. GPER signalling in both cancer-associated fibroblasts and breast cancer cells mediates a feedforward IL1β/IL1R1 response. Sci. Rep. 6, 24354 (2016). This study provides evidence for a role of GPER in inflammatory signalling in breast cancer in cancer-associated fibroblasts.
Divella, R., De Luca, R., Abbate, I., Naglieri, E. & Daniele, A. Obesity and cancer: the role of adipose tissue and adipo-cytokines-induced chronic inflammation. J. Cancer 7, 2346–2359 (2016).
De Francesco, E. M. et al. Protective role of GPER agonist G-1 on cardiotoxicity induced by doxorubicin. J. Cell Physiol. 232, 1640–1649 (2017).
Smalley, K. S. Why do women with melanoma do better than men? eLife 7, e33511 (2018).
Chakraborty, B. et al. Inhibition of estrogen signaling in myeloid cells increases tumor immunity in melanoma. J. Clin. Invest. 131, e151347 (2021).
Natale, C. A. et al. Sex steroids regulate skin pigmentation through nonclassical membrane-bound receptors. eLife 5, e15104 (2016).
Ribeiro, M. P. C., Santos, A. E. & Custodio, J. B. A. The activation of the G protein-coupled estrogen receptor (GPER) inhibits the proliferation of mouse melanoma K1735-M2 cells. Chem. Biol. Interact. 277, 176–184 (2017).
Shen, Y., Li, C., Zhou, L. & Huang, J. A. G protein-coupled oestrogen receptor promotes cell growth of non-small cell lung cancer cells via YAP1/QKI/circNOTCH1/m6A methylated NOTCH1 signalling. J. Cell. Mol. Med. 25, 284–296 (2021).
Lam, H. M. et al. Targeting GPR30 with G-1: a new therapeutic target for castration-resistant prostate cancer. Endocr. Relat. Cancer 21, 903–914 (2014).
Lee, S. J. et al. G protein-coupled estrogen receptor-1 agonist induces chemotherapeutic effect via ER stress signaling in gastric cancer. BMB Rep. 52, 647–652 (2019).
Zheng, S. et al. Screening and survival analysis of hub genes in gastric cancer based on bioinformatics. J. Comput. Biol. 26, 1316–1325 (2019).
Bustos, V. et al. GPER mediates differential effects of estrogen on colon cancer cell proliferation and migration under normoxic and hypoxic conditions. Oncotarget 8, 84258–84275 (2017).
Cortes, E. et al. GPER is a mechanoregulator of pancreatic stellate cells and the tumor microenvironment. EMBO Rep. 20, e46556 (2019).
Morelli, E. et al. Therapeutic activation of G protein-coupled estrogen receptor 1 in Waldenstrom macroglobulinemia. Exp. Hematol. Oncol. 11, 54 (2022).
Klein, S. L. & Flanagan, K. L. Sex differences in immune responses. Nat. Rev. Immunol. 16, 626–638 (2016).
Chakraborty, B. et al. Estrogen receptor signaling in the immune system. Endocr. Rev. 44, 117–141 (2023).
Tamaki, M. et al. Expression and functional roles of G-protein-coupled estrogen receptor (GPER) in human eosinophils. Immunol. Lett. 160, 72–78 (2014).
Itoga, M. et al. G-protein-coupled estrogen receptor agonist suppresses airway inflammation in a mouse model of asthma through IL-10. PLoS ONE 10, e0123210 (2015).
Brunsing, R. L., Owens, K. S. & Prossnitz, E. R. The G protein-coupled estrogen receptor (GPER) agonist G-1 expands the regulatory T-cell population under TH17-polarizing conditions. J. Immunother. 36, 190–196 (2013).
Brunsing, R. L. & Prossnitz, E. R. Induction of interleukin-10 in the T helper type 17 effector population by the G protein coupled estrogen receptor (GPER) agonist G-1. Immunology 134, 93–106 (2011).
Rettew, J. A., McCall, S. H. & Marriott, I. GPR30/GPER-1 mediates rapid decreases in TLR4 expression on murine macrophages. Mol. Cell. Endocrinol. 328, 87–92 (2010).
Rodenas, M. C. et al. G protein-coupled estrogen receptor 1 regulates human neutrophil functions. Biomed. Hub. 2, 1–13 (2017).
Yasuda, H. et al. 17-β-Estradiol enhances neutrophil extracellular trap formation by interaction with estrogen membrane receptor. Arch. Biochem. Biophys. 663, 64–70 (2019).
Castleman, M. J. et al. Innate sex bias of Staphylococcus aureus skin infection is driven by α-hemolysin. J. Immunol. 200, 657–668 (2018).
Triplett, K. D. et al. GPER activation protects against epithelial barrier disruption by Staphylococcus aureus α-toxin. Sci. Rep. 9, 1343 (2019).
Costa, A. J. et al. Overexpression of estrogen receptor GPER1 and G1 treatment reduces SARS-CoV-2 infection in BEAS-2B bronchial cells. Mol. Cell. Endocrinol. 558, 111775 (2022).
Wang, C. et al. Membrane estrogen receptor regulates experimental autoimmune encephalomyelitis through up-regulation of programmed death 1. J. Immunol. 182, 3294–3303 (2009). One of two studies to first suggest a role for GPER in multiple sclerosis.
Du, Z. R. et al. G protein-coupled estrogen receptor is involved in the anti-inflammatory effects of genistein in microglia. Phytomedicine 43, 11–20 (2018).
Guan, J., Yang, B., Fan, Y. & Zhang, J. GPER agonist G1 attenuates neuroinflammation and dopaminergic neurodegeneration in parkinson disease. Neuroimmunomodulation 24, 60–66 (2017).
Harding, A. T., Goff, M. A., Froggatt, H. M., Lim, J. K. & Heaton, N. S. GPER1 is required to protect fetal health from maternal inflammation. Science 371, 271–276 (2021). This study reveals a crucial immune regulatory role for GPER in protecting the fetus from maternal inflammation and infection.
Meyer, M. R., Fredette, N. C., Sharma, G., Barton, M. & Prossnitz, E. R. GPER is required for the age-dependent upregulation of the myocardial endothelin system. Life Sci. 159, 61–65 (2016).
Robison, L. S., Gannon, O. J., Salinero, A. E. & Zuloaga, K. L. Contributions of sex to cerebrovascular function and pathology. Brain Res. 1710, 43–60 (2019).
Patkar, S., Farr, T. D., Cooper, E., Dowell, F. J. & Carswell, H. V. Differential vasoactive effects of oestrogen, oestrogen receptor agonists and selective oestrogen receptor modulators in rat middle cerebral artery. Neurosci. Res. 71, 78–84 (2011).
Tang, H. et al. GPR30 mediates estrogen rapid signaling and neuroprotection. Mol. Cell. Endocrinol. 387, 52–58 (2014).
Murata, T., Dietrich, H. H., Xiang, C. & Dacey, R. G. Jr. G protein-coupled estrogen receptor agonist improves cerebral microvascular function after hypoxia/reoxygenation injury in male and female rats. Stroke 44, 779–785 (2013).
Zhang, Z. et al. The novel estrogenic receptor GPR30 alleviates ischemic injury by inhibiting TLR4-mediated microglial inflammation. J. Neuroinflammation 15, 206 (2018).
Han, Z. W. et al. GPER agonist G1 suppresses neuronal apoptosis mediated by endoplasmic reticulum stress after cerebral ischemia/reperfusion injury. Neural Regen. Res. 14, 1221–1229 (2019).
Bai, N. et al. G-protein-coupled estrogen receptor activation upregulates interleukin-1 receptor antagonist in the hippocampus after global cerebral ischemia: implications for neuronal self-defense. J. Neuroinflammation 17, 45 (2020).
Wang, X. S. et al. Activation of G protein-coupled receptor 30 protects neurons by regulating autophagy in astrocytes. Glia 68, 27–43 (2020).
Peng, J. et al. Activation of GPR30 with G1 attenuates neuronal apoptosis via src/EGFR/stat3 signaling pathway after subarachnoid hemorrhage in male rats. Exp. Neurol. 320, 113008 (2019).
Lu, D. et al. Activation of G protein-coupled estrogen receptor 1 (GPER-1) ameliorates blood-brain barrier permeability after global cerebral ischemia in ovariectomized rats. Biochem. Biophys. Res. Commun. 477, 209–214 (2016).
Zhang, B. et al. Estradiol and G1 reduce infarct size and improve immunosuppression after experimental stroke. J. Immunol. 184, 4087–4094 (2010). The first study to suggest protective effects of GPER activation in a preclinical model of ischaemic stroke.
Zheng, Y. et al. GPER-deficient rats exhibit lower serum corticosterone level and increased anxiety-like behavior. Neural Plast. 2020, 8866187 (2020).
Wang, J. et al. The antidepressant and anxiolytic effect of GPER on translocator protein (TSPO) via protein kinase a (PKA) signaling in menopausal female rats. J. Steroid Biochem. Mol. Biol. 207, 105807 (2021).
Bourque, M., Morissette, M. & Di Paolo, T. Neuroprotection in Parkinsonian-treated mice via estrogen receptor alpha activation requires G protein-coupled estrogen receptor 1. Neuropharmacology 95, 343–352 (2015).
Jiao, Y. et al. Molecular identification of bulbospinal on neurons by GPER which drives pain and morphine tolerance. J. Clin. Invest. 133, e154588 (2023).
Yuan, L. J. et al. G protein-coupled estrogen receptor is involved in the neuroprotective effect of IGF-1 against MPTP/MPP+-induced dopaminergic neuronal injury. J. Steroid Biochem. Mol. Biol. 192, 105384 (2019).
de Souza, L. O. et al. The G protein-coupled estrogen receptor (GPER) regulates recognition and aversively-motivated memory in male rats. Neurobiol. Learn. Mem. 184, 107499 (2021).
Kubota, T., Matsumoto, H. & Kirino, Y. Ameliorative effect of membrane-associated estrogen receptor G protein coupled receptor 30 activation on object recognition memory in mouse models of Alzheimer’s disease. J. Pharmacol. Sci. 131, 219–222 (2016).
Wang, Z. F., Pan, Z. Y., Xu, C. S. & Li, Z. Q. Activation of G-protein coupled estrogen receptor 1 improves early-onset cognitive impairment via PI3K/Akt pathway in rats with traumatic brain injury. Biochem. Biophys. Res. Commun. 482, 948–953 (2017).
Amirkhosravi, L. et al. E2-BSA and G1 exert neuroprotective effects and improve behavioral abnormalities following traumatic brain injury: the role of classic and non-classic estrogen receptors. Brain Res. 1750, 147168 (2021).
Confavreux, C., Hutchinson, M., Hours, M. M., Cortinovis-Tourniaire, P. & Moreau, T. Rate of pregnancy-related relapse in multiple sclerosis. Pregnancy in multiple sclerosis group. N. Engl. J. Med. 339, 285–291 (1998).
Seifert, H. A. et al. Estrogen protects both sexes against EAE by promoting common regulatory cell subtypes independent of endogenous estrogen. Metab. Brain Dis. 32, 1747–1754 (2017).
Subramanian, S., Miller, L. M., Grafe, M. R., Vandenbark, A. A. & Offner, H. Contribution of GPR30 for 1,25 dihydroxyvitamin D(3) protection in EAE. Metab. Brain Dis. 27, 29–35 (2012).
Markee, J. E. Rhythmic uterine vascular changes. Am. J. Physiol. 100, 32–39 (1932).
Carloni, E. L’azione degli estratti ovarici sull’atteggiamento dei capillari e sulla loro pressione, nelle varie fasi della rivoluzione funziozionale utero-ovarica. Arch. Ostet. Ginecol. 17, 327i (1930).
Collin, J. B., Browne, J. S. & Thomson, D. L. The chemical nature of emmenin. Endocrinology 18, 71–74 (1934).
Dawson, R. F. & Robson, J. M. The pharmacological actions of diethylstilboestrol and other oestrogenic and non-oestrogenic substances. J. Physiol. 95, 420–430 (1939).
Reynolds, S. R. & Foster, F. I. Peripheral vascular action of estrogen in the human male. J. Clin. Invest. 18, 649–655 (1939).
Vance, D. A. Premarin: the intriguing history of a controversial drug. Int. J. Pharm. Compd. 1, 282–286 (2007).
Veurink, M., Koster, M. & Berg, L. T. The history of DES, lessons to be learned. Pharm. World Sci. 27, 139–43 (2005).
Pincus, G., Rock, J., Chang, M. C. & Garcia, C. R. Effects of certain 19-nor steroids on reproductive processes and fertility. Fed. Proc. 18, 1051–1056 (1959).
Lerner, L. J., Holthaus, F. J. & Thompson, C. R. A non-steroidal estrogen antagonist 1-(p-2-diethylaminoethoxyphenyl)-1-phenyl-2-p-methoxyphenyl ethanol. Endocrinology 63, 295–318 (1958).
Jordan, V. C. Tamoxifen: a most unlikely pioneering medicine. Nat. Rev. Drug Discov. 2, 205–13 (2003).
Toft, D. & Gorski, J. A receptor molecule for estrogens: isolation from the rat uterus and preliminary characterization. Proc. Natl Acad. Sci. USA 55, 1574–1581 (1966).
Jensen, E. V. et al. A two-step mechanism for the interaction of estradiol with rat uterus. Proc. Natl Acad. Sci. USA 59, 632–638 (1968).
Szego, C. M. & Davis, J. S. Adenosine 3’,5’-monophosphate in rat uterus: acute elevation by estrogen. Proc. Natl Acad. Sci. USA 58, 1711–1718 (1967).
Pietras, R. J. & Szego, C. M. Endometrial cell calcium and oestrogen action. Nature 253, 357–359 (1975).
Jordan, V. C. & Dowse, L. J. Tamoxifen as an anti-tumour agent: effect on oestrogen binding. J. Endocrinol. 68, 297–303 (1976).
Pietras, R. J. & Szego, C. M. Estrogen receptors in uterine plasma membrane. J. Steroid Biochem. 11, 1471–1483 (1979).
Owman, C., Blay, P., Nilsson, C. & Lolait, S. J. Cloning of human cDNA encoding a novel heptahelix receptor expressed in Burkitt’s lymphoma and widely distributed in brain and peripheral tissues. Biochem. Biophys. Res. Commun. 228, 285–292 (1996).
Carmeci, C., Thompson, D. A., Ring, H. Z., Francke, U. & Weigel, R. J. Identification of a gene (GPR30) with homology to the G-protein-coupled receptor superfamily associated with estrogen receptor expression in breast cancer. Genomics 45, 607–617 (1997).
Feng, Y. & Gregor, P. Cloning of a novel member of the G protein-coupled receptor family related to peptide receptors. Biochem. Biophys. Res. Commun. 231, 651–654 (1997).
Takada, Y., Kato, C., Kondo, S., Korenaga, R. & Ando, J. Cloning of cDNAs encoding G protein-coupled receptor expressed in human endothelial cells exposed to fluid shear stress. Biochem. Biophys. Res. Commun. 240, 737–741 (1997).
Kvingedal, A. M. & Smeland, E. B. A novel putative G-protein-coupled receptor expressed in lung, heart and lymphoid tissue. FEBS Lett. 407, 59–62 (1997).
O’Dowd, B. F. et al. Discovery of three novel G-protein-coupled receptor genes. Genomics 47, 310–313 (1998).
Bonini, J. A., Anderson, S. M. & Steiner, D. F. Molecular cloning and tissue expression of a novel orphan G protein-coupled receptor from rat lung. Biochem. Biophys. Res. Commun. 234, 190–193 (1997).
Acknowledgements
E.R.P. is supported by grants from the US National Institutes of Health (R01 CA163890 and R01 CA194496), from Dialysis Clinic, Inc., and by the UNM Comprehensive Cancer Center (NIH P30 CA118100) and the Autophagy, Inflammation and Metabolism (AIM) Center of Biomedical Research Excellence (CoBRE, NIH P20 GM121176). M.B. is supported by grants 108 258 and 122 504 from the Swiss National Science Foundation (SNSF).
Author information
Authors and Affiliations
Contributions
Both authors contributed equally to all aspects of this manuscript.
Corresponding authors
Ethics declarations
Competing interests
M.B. and E.R.P. are inventors on U.S. patent Nos. 10,251,870, 10,682,341 and 10,980,785, and E.R.P. is an inventor on U.S. Patent Nos. 10,471,047 and 10,561,648, all for the therapeutic use of compounds targeting GPER (“Method for treating obesity, diabetes, cardiovascular and kidney diseases by regulating GPR30/GPER”). E.R.P. is an inventor on U.S. Patent Nos. 7,875,721 and 8,487,100 for GPER-selective ligands and imaging agents (“Compounds for binding to ERα/β and GPR30, methods of treating disease states and conditions mediated through these receptors and identification thereof”). M.B. has served or serves as a consultant to Abbott, Inc., Abbvie, Inc., Travere, Inc. and Pharmazz, Inc.
Peer review
Peer review information
Nature Reviews Endocrinology thanks Guichun Han and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Review criteria
Abstracts of all articles published on GPR30 or GPER, published between January 1996 and February 2023, were retrieved from the U.S. National Library of Medicine (PubMed.gov). Articles were assessed for relevance, importance and scientific rigour, with a focus on publication in the past 10 years. The authors apologize to their colleagues whose work could not be included due to space and reference restrictions.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Prossnitz, E.R., Barton, M. The G protein-coupled oestrogen receptor GPER in health and disease: an update. Nat Rev Endocrinol 19, 407–424 (2023). https://doi.org/10.1038/s41574-023-00822-7
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41574-023-00822-7
This article is cited by
-
Reproductive history and later-life health: examining the link between birth intervals and CKD in postmenopausal women
BMC Nephrology (2026)
-
Health of adipose tissue: oestrogen matters
Nature Reviews Endocrinology (2026)
-
GPR30-driven fatty acid oxidation targeted by ginsenoside Rd maintains mitochondrial redox homeostasis to restore vascular barrier in diabetic retinopathy
Cardiovascular Diabetology (2025)
-
Sex differences in bladder cancer: understanding biological and clinical implications
Biology of Sex Differences (2025)
-
Depletion of mmu-miR-185 enhances osteocyte connectivity and suppresses bone fragility through the interaction between ARL8B and RAB5A
Stem Cell Research & Therapy (2025)