Abstract
Inflammatory bowel disease (IBD) is a growing global health challenge affecting more than 7 million people worldwide. With increasing prevalence across all age groups, including children and adolescents, IBD places substantial strain on health-care systems and society, resulting in high direct medical costs, lost productivity and reduced quality of life. Despite therapeutic advances, suboptimal disease control and delays in timely diagnosis and adequate treatment persist. Regional disparities in health-care access contribute to these challenges, especially in low-income countries. Addressing these inequities is crucial for improving global IBD outcomes. Using a Delphi methodology, experts from clinical care, research, public health and advocacy (including patient representation) identified priorities across six domains (37 statements in total): epidemiology, care models, treatment strategies, education and awareness, patient and community engagement, and leadership to promote health equity. These priorities emphasize quantifying the burden of IBD, addressing health-care disparities, validating care models, exploring novel treatments, advancing education, engaging patients and advocating for health equity policies. The comprehensive approach seeks to optimize care models, promote patient engagement and ensure equitable access to health care. The identified priorities serve as a guide for both clinical and non-clinical researchers, and funders dedicated to IBD-related initiatives, fostering international collaboration to improve IBD management and reduce its impact globally.
Key points
-
This Consensus Statement provides an evidence-based road map to guide global initiatives, ensuring inflammatory bowel disease (IBD) receives the attention and resources needed to mitigate its growing impact on individuals and society.
-
Understanding the global epidemiological burden remains a priority, with the need for high-quality population-based research on health-care system delivery to prepare personnel, resources and medical infrastructure.
-
Health-care systems should implement timely IBD referral pathways, optimize diagnosis and treatment and assess combination treatments; advancing fibrosis evaluation and novel treatments beyond inflammation is key to improving patient outcomes.
-
IBD education should be expanded in medical curricula and continuing education, emphasizing diet, nutrition and patient awareness of early diagnosis, intervention, disease modification and functional remission through multidisciplinary collaboration.
-
Research should develop effective IBD care models for resource-limited settings; inclusive leadership must address health-care disparities while prioritizing psychosocial support and mental health to improve patients’ overall well-being beyond gastrointestinal symptoms.
-
The future of IBD care demands a holistic, multidisciplinary approach; stakeholders must collaborate to advance personalized treatments, equitable access, mental health support and adaptable health-care models, ensuring inclusive, patient-centred and globally accessible care.
Similar content being viewed by others
Introduction
Inflammatory bowel disease (IBD), encompassing Crohn’s disease and ulcerative colitis, is a chronic, progressive condition that affects millions of individuals worldwide. The prevalence of IBD has been steadily rising in both developed and developing nations, with an estimated 7 million individuals globally affected by the disease1,2. IBD affects people of all ages, with a rising number of diagnoses in children and adolescents, especially in areas where the disease was previously uncommon such as Eastern Asia countries3. The condition imposes a substantial burden on global health-care systems, not only through direct medical expenses but also due to indirect costs, such as decreased productivity, disability and a reduced quality of life for affected individuals4,5. Despite the availability of advanced therapeutic options, many individuals with IBD continue to experience suboptimal disease control, and substantial gaps remain in the timely diagnosis and appropriate management of the condition. The unpredictability of the disease and the potential for severe complications underscore the urgent need for improved health-care policies, better access to care and enhanced patient support systems.
As the global burden of IBD continues to rise, many jurisdictions are increasingly ill-prepared to effectively address the growing needs of individuals living with this condition. A survey of 397 participants published in 2021 highlighted that the increasing prevalence of IBD is the most substantial challenge for gastroenterologists globally6. Perceived challenges differ notably between those in high-income and those in low-income and middle-income countries, primarily reflecting disparities in health-care access. For instance, respondents from high-income countries reported greater difficulty accessing IBD specialists and surgeons, whereas those in low-income countries faced substantial barriers to obtaining simple treatments such as 5-aminosalicylic acid, steroids and immunomodulators, as well as anti-TNF therapy and dose escalation. Addressing these issues in developing regions could reduce inequities and improve global patient outcomes.
To tackle this urgent public health concern, a worldwide team of experts collaborated to establish a consensus on actionable strategies to address the challenges posed by IBD. Policy and programmatic initiatives in various medical fields beyond IBD, such as neurology, cardiology, hepatology and nephrology, have highlighted key global research agenda priorities7,8,9,10. These initiatives focus on advancing public health policies, improving early diagnosis and disease management, enhancing access to care and fostering collaboration across critical areas, including epidemiology, care models, treatment strategies, education and awareness, patient and community perspectives, and leadership to promote health equity.
This Consensus Statement aims to provide a comprehensive, evidence-based road map to guide initiatives at a global level, ensuring that IBD receives the attention and resources it requires to reduce its growing impact on individuals and society worldwide. Through a multidisciplinary and collaborative approach, we aim to reshape how IBD is understood, treated and prevented, working towards a future where the burden of this complex disease is markedly reduced.
Methods
This Consensus Statement was conducted under the framework and with the support of the International Organization for the Study of Inflammatory Bowel Disease (IOIBD), which served as the collaborative platform for this project. This study employed a Delphi methodology to develop consensus action. The four co-chairs (V.S., V.J., L.P.-B. and S.D.) identified 16 experts, covering clinical care and research, public health and policy, and advocacy (C.N.B., I.D., A.D., M.C.D., R.B.G., A.H., G.G.K., C.M., F.M., J.W.Y.M., S.C.N., R.P., S.R., D.T.R. and C.A.S. and patient advocate R.D.) who collectively formed the core author group. The core group identified domain-specific experts to provide critical input on the initial draft of statements (R1). Participants had expertise in IBD, immune-mediated inflammatory diseases (IMID), non-communicable diseases, public health, patient advocacy and/or consensus methodologies. To ensure a comprehensive perspective among survey participants, the core group intentionally included both experienced professionals and emerging voices in the field, aiming to capture diverse expertise and enhance the quality of the findings. A sample size of least 300 participants was considered to be appropriate to facilitate consensus rather than statistical representation. A combination of purposive, snowball and targeted sampling methods was employed to assemble a diverse, multidisciplinary panel with broad geographical representation.
Drafting of action priorities
The core author group carried out an extensive analysis of the literature and available evidence, producing a collection of evidence briefs focused on six major topics. These briefs provided an overview of current knowledge, articulated a vision for progress over the next 10 years, highlighted essential questions and recommended priority actions in the following areas: (1) epidemiology; (2) developing and implementing care models; (3) strategies for treatment and care; (4) educational and awareness programmes; (5) patient and community engagement; and (6) leadership and policies to promote health equity.
The statements were discussed and refined during a virtual meeting held on 17 July 2024. Following this meeting, the core group members updated the priorities to incorporate insights from the discussions in preparation for the first round of the Delphi survey, conducted from 24 July to 15 August 2024.
Delphi method data collection and analysis
The study design consisted of two Delphi survey rounds (R1 and R2, both in English), each anticipated by a virtual meeting within the core group. In both rounds, respondents indicated their agreement with each priority using a four-point Likert-type scale (that is, ‘agree’, ‘somewhat agree’, ‘somewhat disagree’ and ‘disagree’). Given the multidisciplinary nature of the panel, the survey included a fifth option: ‘not qualified to respond’. Panellists could provide comments and suggest edits to individual priorities and provide overall comments at the end of R1. Demographic data were collected in R1 and R2. The survey was distributed using the Qualtrics XM platform.
An analytical team of core group members (V.S., S.R., V.J., L.P.-B., S.D.) reviewed the R1 data, including 333 open-ended comments, and initiated revisions; the core group subsequently reviewed the revised priorities and provided additional inputs in a virtual meeting (23 October 2024) ahead of R2. In R2 (1–30 November 2024), panellists voted on the revised priorities and ranked at least half of the priorities within each of the following six domains: epidemiology; models of care; treatment and care; education and awareness; patient and community perspectives; and leadership and public health policy. Each action priority was graded to indicate the level of combined agreement (‘agree’ + ‘somewhat agree’), using a system that has been used in other Delphi studies9 in which ‘U’ denotes unanimous (100%) agreement, ‘A’ denotes 90–99% combined agreement, ‘B’ denotes 78–89% combined agreement, and ‘C’ denotes 67–77% combined agreement. For the ranking, scores were calculated and normalized in Microsoft Excel (v.16.70) to compare rankings within each domain.
Results
A total of 330 individuals were invited to participate in R1, and 307 (93.0%) completed the survey. These respondents were invited to participate in R2, of whom 300 (90.9%) completed the survey. Table 1 details the demographics of all expert panellists involved in the study. Most respondents were aged 30–50 years (64%), male (51.3%), based in high-income countries (64.7%) and worked as health-care providers (54.7%). In relation to sector of employment, the largest group was employed in the public sector (35%). Respondents represented regions across the world, including East Asia and Pacific, Europe and Central Asia, Latin America and Caribbean, Middle East and North Africa, North America, South Asia and sub-Saharan Africa. Europe and Central Asia had the highest representation (46.7%), whereas sub-Saharan Africa had the lowest representation (1.0%). Most respondents reported national professional association or society membership in the field of gastroenterology (75.7%), whereas primary care was the least represented area (1.3%). During the revisions for R2, 16 statements were rephrased to address free-text comments and suggestions from R1, as well as to incorporate insights from the virtual meeting within the core group. The panel reviewed 37 priorities in R2. Across both Delphi rounds, the combined agreement (‘agree’ + ‘somewhat agree’) remained high in all domains. The mean percentage of ‘agree’ responses slightly decreased from 96.6% in R1 to 94.7% in R2, following the consideration of substantive feedback received in R1. However, two of three statements that initially received a grade B achieved a grade A after R2 (6.1 and 6.2). Table 2 presents the final priorities, agreement grades and rankings for each of the six domains. The core author group established a priority ranking (first, second, third), as presented in the last column of Table 2, based on expert judgment. In the final priorities for R2, the panel reached over 90% combined agreement for all priorities, except one which received a grade B (6.5).
Domain 1: understanding the global epidemiological burden
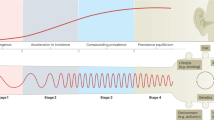
Domain 1 identified several interrelated research priorities essential for advancing our understanding of the epidemiological burden of the disease across diverse populations and settings. A critical area highlighted is the need for geospatial and temporal analyses of the global incidence and prevalence of IBD, which can help to track its evolution through distinct stages. These stages — emergence, acceleration in incidence, compounding prevalence and prevalence equilibrium — are vital for studying how genetic, environmental and microbial factors contribute to disease progression (1.1, ranked second in the domain)11,12. As IBD prevalence rises, particularly in developed nations, there is an increasing need for comprehensive population-based research on health-care systems to ensure that medical infrastructure, personnel and resources are prepared for the growing IBD burden, including the challenges posed by an ageing population and the rise in comorbidities (1.2, ranked first in the domain)13.
Further emphasizing this need for nuanced understanding, standardizing study designs and methodologies across regions is vital. This approach would address the variability in epidemiological data, enabling more accurate comparisons and facilitating the identification of whether observed trends are due to improved detection or true changes in IBD biology (1.3)14,15. Additionally, the rise in paediatric-onset IBD increased by 22.8% from 1990 to 2019, particularly in regions with a high sociodemographic index16, including very early-onset forms of the disease, calls for in-depth studies examining the genetic, environmental, dietary and microbial exposures during early life (1.4)17,18. Understanding these risk factors in children is critical because they are often exposed to fewer environmental factors, which might enable researchers to pinpoint modifiable determinants that could be targeted for preventive interventions.
Another important research priority involves identifying biomarkers from cohorts of individuals at high risk of developing IBD (1.5). These biomarkers, whether genetic, serological, proteomic or microbial, could lead to targeted interventional studies that modify environmental and behavioural factors or support clinical trials aimed at preventing the onset of IBD altogether19,20. Furthermore, the need for epidemiological research on under-represented populations, such as ethnic minorities, low socioeconomic status groups and migrants, is becoming increasingly important (1.6, ranked third in the domain)21,22. These populations are often disproportionately affected by IBD with higher disease burden and limited access to quality care, and conducting studies with diverse groups ensures that we can identify factors unique to different subgroups and improve health-care equity. This aspect is particularly important given the substantial differences in ancestry, genetics, environment and diet, which can also influence variations in IBD epidemiology, phenotype and outcomes across these regions23,24. For instance, the incidence of IBD has markedly increased among Black and Indigenous individuals, people of colour and Hispanic individuals, with a particularly notable rise among Black people, over the past three decades in the USA. However, data show that they experience worse clinical disease, lower health-related quality of life scores, more medication concerns and more limited access to specialists compared with white or non-Hispanic people. Additionally, they report having less social and emotional support and utilize emergency department services more frequently25. Moreover, sex-specific differences in the phenotype and progression of IBD have been observed, with women experiencing longer diagnostic delays than men26, underscoring the need for further epidemiological research to explore the genetic, hormonal, microbiota-related and environmental factors involved27. Lastly, global surveillance of IBD through standardized phenotyping, diagnosis and management definitions is crucial (1.7). Standardization will enable more effective comparisons of IBD across different regions, helping to build cascading guidelines that reflect both global insights and local health-care resource limitations. Overall, these research priorities underscore a comprehensive and inclusive approach to improving IBD understanding, management and prevention across the globe, focusing on both macro trends and micro-level insights.
Domain 2: defining and implementing care models
Domain 2 emphasizes the importance of defining and implementing patient-centred care models for IBD, for which collaboration among all relevant stakeholders is paramount. A key priority (2.1) is ensuring the engagement of patients, their families, caregivers, patient organizations and health-care providers in the development of IBD care models, emphasizing the need for models that reflect patient needs and perspectives28,29. Specialized care models (2.2) are crucial for ensuring timely diagnosis, accurate prognostic evaluation and evidence-based management, including innovative hub-and-spoke frameworks that facilitate access to multidisciplinary care and improve outcomes through technological integration30. Additionally, financial sustainability and efficiency are core concerns, with research suggesting that health-care decision-makers should be well-informed about the direct and indirect costs of IBD care models (2.3, ranked third in the domain). Developing context and resource-specific multidisciplinary care models (2.4) is vital for ensuring that all patients benefit from individualized care, tailored to both the specific needs of the population and available resources31. Furthermore, professional bodies and health authorities should establish clear and actionable care pathways (2.5, ranked second in the domain), ensuring that patients with IBD are swiftly referred to the appropriate specialized care settings. The effectiveness of these pathways depends on an appreciation of local conditions, ensuring that diagnostic and therapeutic measures are adapted to the needs of both patients and health-care providers. Strategies should involve direct input from all stakeholders to create a system that is both practical and sustainable. A successful example comes from Canada, where financial constraints and geographical barriers in the health-care system have prompted the implementation of telemedicine programmes, such as the PACE Telemedicine Program. This initiative has helped to optimize health-care utilization by reducing unnecessary emergency visits and minimizing travel burdens for patients in non-urban areas32. Similar adaptive models should be considered globally, ensuring that health-care delivery remains responsive, accessible and tailored to the realities of each health-care system. Local settings and conditions need to be appreciated, and context-specific adaptations to the local needs and cascading diagnostic and therapeutic measures should be considered based on the input of patients and health-care providers. Finally, multidisciplinary collaboration is critical (2.6, ranked first in the domain); IBD specialists must work closely with primary care providers to identify patients exhibiting alarm symptoms or poor prognostic indicators and direct them to specialized treatment teams to optimize early intervention and care33. Together, these priorities support the goal of creating a health-care environment in which patients with IBD can receive the best care, irrespective of their location, resources or personal circumstances.
Domain 3: defining and implementing treatment strategies
Domain 3 focuses on enhancing treatment and care strategies for IBD, with an emphasis on optimizing clinical pathways, tailored therapeutic approaches (towards precision medicine) and addressing unique patient populations. A major priority (3.1, ranked first in the domain) is the development of clinical pathways that ensure early identification, accurate diagnosis and timely medical or surgical interventions, which are crucial for improving long-term outcomes and quality of life, particularly in individuals with Crohn’s disease due to its well-documented progression when intervention is delayed beyond the window of opportunity34. With scientific advances and improved understanding of immunological changes that occur years before the onset of symptoms and diagnosis20,35, the role of presymptomatic diagnosis and prevention (3.2) is also of emerging interest, with efforts aimed at studying interventions capable of delaying disease onset, mitigating disease progression and/or potentially modifying the natural course of IBD. Whilst the therapeutic armamentarium for IBD has expanded and there has been a paradigm shift towards ‘treating to target’ in IBD, clearly defining treatment thresholds and strategies across the spectrum of disease, from mild-to-moderate to moderate-to-severe disease, is essential for optimizing cost-effective care worldwide (3.3). A key area of exploration will be the use of advanced combination treatment (ACT), which explores pharmacological treatment combinations to identify the most effective and safe regimens for maximizing efficacy whilst minimizing risks for patients (3.4, ranked third in the domain). The goal is not only to address the underlying pathophysiology of IBD but also to improve long-term remission rates and reduce adverse effects36,37.
As clinical evidence evolves including ACT randomized trials and real-world evidence data, more attention will be given to identifying the most effective combination therapies for different patient subgroups, considering factors such as disease severity, previous treatments and genetic or microbiome profiles. Furthermore, incorporating public health and social measures, such as dietary interventions and psychological support, in combination approaches will have a crucial role in providing comprehensive, holistic care for patients with IBD38,39. Along these lines, recognition of the unique needs of specific patient populations (3.5) such as paediatric patients, older patients, those who are pregnant and individuals with comorbid immune-mediated disorders or primary sclerosing cholangitis is essential for providing personalized care that accounts for an individual patient’s distinct profile and risks, and recognition of the effects of different sexual orientation, and racial, ethnic and minority backgrounds40. Currently, many gastroenterologists lack training in assessing sexual dysfunction in patients with IBD, particularly those in sexual and gender minorities. This gap in training leads to frustration for many patients belonging to sexual and gender minority groups, who report feeling ignored and stigmatized41. Additionally, developing new methods to evaluate complications such as fibrosis and penetrating disease (3.6, ranked second in the domain) could lead to more targeted therapies that go beyond controlling inflammation and improving care for patients with Crohn’s disease. Together, these priorities aim to improve treatment outcomes and quality of life for all individuals living with IBD through more comprehensive and innovative care strategies.
Domain 4: promoting education and awareness initiatives
Domain 4 highlights the importance of education and awareness initiatives in improving the understanding and management of IBD among both health-care providers and patients42. One primary focus (4.1) is the development, evaluation and funding of updated curricula for medical education at both undergraduate and postgraduate levels. These updates include training for specialists and the involvement of relevant societies to ensure that future health-care professionals are equipped with current, evidence-based knowledge of IBD. Expanding the availability of educational courses and toolkits (4.2, ranked third in the domain) that emphasize IBD management, particularly the importance of diet and nutrition43, is another priority. Collaboration across disciplines, including medical and surgical specialties, is essential to provide comprehensive guidance on IBD care. In addition, developing best practice educational resources (4.3, ranked second in the domain) for primary-care providers and specialists is critical to emphasize the importance of early diagnosis and proper referral for timely IBD care. Another key initiative is raising awareness among health-care providers and patients about the holistic nature of IBD management (4.4), ensuring that biopsychosocial factors are systematically addressed alongside physical symptoms. Raising awareness is not just about acknowledging these elements but implementing concrete actions to integrate them into routine care44. Patients with IBD frequently experience challenges beyond their physical symptoms, including altered illness perceptions, concerns about body image and self-consciousness, sexual health issues and the psychological burden of chronic disease41. These factors are often overlooked in routine care, yet they markedly affect treatment adherence, disease management and overall well-being. For instance, research has shown that untreated anxiety and depression in patients with IBD can lead to more severe disease symptoms, increased flare-ups, poor medication adherence and higher hospitalization rates45. Importantly, disparities in depression and anxiety diagnoses among socially vulnerable patients with IBD have emerged, with higher levels of social vulnerability associated with an increased likelihood of depression and low overall mental health screening rates, particularly among the most socially vulnerable individuals46.
Furthermore, strategies to improve the transition from paediatric to adult care (4.5) should be prioritized, with an emphasis on supporting both parents and young adults in taking ownership of their health care47. Lastly, education should be targeted at the general population to enable and optimize early diagnosis, and all patients with IBD should be educated on the path to achieving sustained quality of life, disease modification and functional remission (4.6, ranked first in the domain)48. Although early diagnosis is a critical factor in optimizing long-term outcomes, this aspect is particularly relevant for community engagement efforts and for the unaffected relatives of patients with IBD, for whom awareness initiatives might facilitate timely detection and intervention. These initiatives collectively aim to improve the overall knowledge, approach and patient outcomes in IBD care.
Domain 5: integrating patients and community perspectives
Domain 5 emphasizes enhancing patient-centred care by addressing the clinical and psychosocial needs of individuals with IBD. A key aspect (5.1, ranked second in the domain) highlights the importance of understanding patients’ care goals and aligning them with clinical objectives, such as achieving endoscopic healing, characterized by the disappearance of ulcers and normalization of the mucosa. By tailoring treatment plans to balance clinical targets with individual patient priorities, this approach enhances treatment effectiveness, improves patient satisfaction and minimizes the risk of overtreatment49,50. Additionally, the partnership with primary care providers is vital for the ongoing management of IBD (5.2), particularly through models such as nurse-led advice lines and dedicated ‘flare clinics’, which offer continuously available support and appropriate intervention51.
To address access challenges, novel health-care delivery models should be developed (5.3, ranked third in the domain) to ensure equitable access to specialty IBD centres. This step includes the creation of coordinated pathways that allow patients to transition smoothly between primary gastroenterology care and specialized centres, as well as the development of standardized care models for patients moving from paediatric to adult care. Furthermore, community resources (5.4) are essential for better patient empowerment to ensure that patients have the tools and support systems they need for self-management.
Recognizing the broader effect of IBD (5.5, ranked first in the domain) on patients’ lives beyond gastrointestinal symptoms is essential52. Unfortunately, only a small proportion of patients have access to specialist psychiatric consultation and psychological therapy, with only 6% of IBD services meeting resourcing recommendations for mental health support according to the last 2024 IBD UK report53. Barriers include the stigma surrounding mental health services, limited expertise and training in psychological assessment among health-care providers and health-care system structures that separate physical and mental health care. Providing resources for psychosocial support to address mental health needs fosters a holistic approach to care. Additionally, the burden of IBD on families, friends and caregivers (5.6) should not be underestimated. Efforts must focus on understanding and easing the emotional and practical challenges faced by those supporting patients. This comprehensive approach enhances patients’ well-being and quality of life by addressing all aspects of their care.
Domain 6: promoting leadership and policies for health equity
Domain 6 focuses on the leadership and policies necessary to address health equity in IBD care. It emphasizes the need for research that highlights health-care disparities across various socioeconomic, demographic, regional and cultural dimensions (6.1)24,54. Understanding the effects of governance models and policies on health-care access, experience and outcomes in IBD is critical for fostering equitable care. Additionally, the development of core outcome sets or benchmarks related to health equity would provide clear and measurable end points for research, tailored to different strata of health-care inequity (6.2, ranked third in the domain)55.
It is vital to address the broader social determinants of health (SDOH) that influence IBD outcomes (6.3)56. These factors extend beyond traditional health-care systems and include socioeconomic and contextual influences on health. An example of this issue is the 2015 National Health Interview Survey, which found that 12% of adults with IBD experience both food insecurity and a lack of social support. Notably, food insecurity is significantly associated with increased financial hardship, greater distress and higher rates of cost-related medication non-adherence, highlighting the profound effect of these social factors on disease management57. Collaborative national and international research should focus on understanding how these factors affect outcomes in marginalized populations and creating strategies to alleviate their effects.
The rising prevalence of IBD in newly industrialized and developing countries (6.4, ranked first in the domain) underscores the need for developing care models that are adapted to resource-constrained environments, aligning with the anticipated priorities outlined in statements 2.4 and 2.5. One example is the implementation of task-shifting models, in which specialized IBD nurses or trained health-care workers take on roles in patient education, monitoring disease progression and managing medications, enabling limited resources to be more effectively utilized. Collaboration across various sectors of chronic inflammatory disease care is essential to ensure the effective delivery of care in these areas.
Regarding technology, steps should be taken to ensure that historically neglected populations are included in artificial intelligence (AI) models to prevent exacerbating existing health inequities (6.5). The environmental effects of AI technologies should also be addressed58. Statement 6.5 was the only one in this domain assigned grade B, reflecting concerns about the practical challenges of achieving effective inclusion of marginalized populations in AI models. This grade highlights the potential difficulties in operationalizing equity within AI frameworks and ensuring that their outcomes benefit all population groups equitably. Finally, strengthening the role of patient organizations and advocacy groups in research leadership (6.6, ranked second in the domain) will be key to ensuring that under-represented communities have a voice in shaping future IBD research, clinical trials and health-care policies59. By promoting inclusive leadership and policymaking, the broader goal of achieving health equity in IBD can be advanced.
Study strengths and limitations
The Delphi method is an appropriate and strong approach for consensus-building; however, it has inherent challenges60. The study was designed to facilitate expert consensus rather than achieve strict statistical representation. We employed a combination of purposive, snowball and targeted sampling methods to engage key stakeholders while balancing feasibility and inclusivity in participant recruitment. In this global health initiative, we provide clear, actionable recommendations that stakeholders worldwide can easily implement. Our goal was to develop a concise yet impactful framework to enhance IBD care across diverse health-care systems. By focusing on well-defined priorities, we aim to facilitate widespread adoption and meaningful change, particularly in regions with limited resources and infrastructure. All statements in this paper achieved significant agreement with an overall combined agreement of >85% among participants, underscoring their relevance as priorities. We emphasize that these areas should be addressed in parallel, ensuring that the most pressing needs across different regions and health-care systems are met. Despite efforts to include a diverse group of contributors actively shaping IBD care, this study had limitations. Notably, there was an under-representation of participants from Africa and patient advocacy groups in the survey respondents and in the core author group, which could have affected the generalizability of certain recommendations. We acknowledge that the panel’s composition across all respondents, predominantly from high-income countries and the academic sector, might have influenced the study outcomes. Additionally, the survey was only available in English, which might have limited the willingness of participants to contribute and access full understanding of every statement. Offering translations could have enhanced engagement across a broader range of regions by reducing language barriers and ensuring more inclusive global participation.
Future outlook
The future of IBD care and research calls for a holistic, integrated approach that involves multiple stakeholders, each playing a critical role in shaping the next phase of management and treatment. Clinicians and researchers must prioritize collaboration across disciplines, combining expertise in genetics, environmental factors, epidemiology and health-care systems to drive more precise, personalized care. Researchers should focus on uncovering the SDOH and the effect of governance models on IBD outcomes, ensuring that their work reflects the diverse needs of populations worldwide. Pharmaceutical companies must innovate in developing therapies that are tailored to individual patient profiles, integrating both pharmacological interventions and public health and social measures, whilst also considering the environmental effects of their products. Meanwhile, health-care policies should be tailored to promote equitable access to care, with a particular emphasis on resource-limited settings and the increasing demand for mental health support within IBD management. Patient organizations and advocacy groups are vital in raising awareness, influencing policy changes and driving research agendas, especially in under-served regions. Governments and Non-Governmental Organizations are essential in prioritizing health equity by funding initiatives that enhance treatment access and education while promoting adaptable health-care models. Reimbursement policies should reflect the full spectrum of IBD care, including preventive and personalized interventions, while ensuring that financial barriers do not limit access to effective treatments. Collaboration across these sectors is essential to fostering a patient-centred, globally accessible approach to IBD care, ensuring better outcomes and a more supportive health-care environment for those affected by the disease.
Conclusions
Approximately 7 million people worldwide live with IBD. Despite notable advances in scientific knowledge over the past 30 years, the prevalence of IBD persists, and increasing inequities underscore the need for accelerated efforts to develop prevention, management and treatment strategies. Through a collaborative global consensus process involving a multidisciplinary group, research priorities have been identified. The research priorities focus on epidemiology, defining care models, treatment, education, patient perspectives and leadership for health equity in IBD. Emphasis is placed on quantifying IBD burdens, addressing disparities, validating care models, exploring novel treatments, advancing education and awareness, engaging patients and advocating for health equity policies (Fig. 1). The comprehensive approach aims to enhance understanding, optimize care models, promote patient engagement and ensure equitable access to quality health care. Future steps should focus on a fundamental transformation in the IBD field, emphasizing a more inclusive and comprehensive approach. This progress will require collaboration among a broad range of stakeholders beyond gastroenterologists, such as paediatricians, IMID specialists, surgeons, primary care providers, public health experts, social scientists, policymakers, governments, pharmaceutical companies, patient advocates and, crucially, patients themselves.
Consensus statements and recommendations across six priority domains. IBD, inflammatory bowel disease.
References
Ng, S. C. et al. Worldwide incidence and prevalence of inflammatory bowel disease in the 21st century: a systematic review of population-based studies. Lancet 390, 2769–2778 (2017).
GBD 2017 Inflammatory Bowel Disease Collaborators. The global, regional, and national burden of inflammatory bowel disease in 195 countries and territories, 1990–2017: a systematic analysis for the Global Burden of Disease Study 2017. Lancet Gastroenterol. Hepatol. 5, 17–30 (2020).
Caron, B., Honap, S. & Peyrin-Biroulet, L. Epidemiology of inflammatory bowel disease across the ages in the era of advanced therapies. J. Crohns Colitis 18, ii3–ii15 (2024).
Burisch, J. et al. The cost of inflammatory bowel disease care – how to make it sustainable. Clin. Gastroenterol. Hepatol. 23, 386–395 (2024).
Burisch, J. et al. The cost of inflammatory bowel disease in high-income settings: a Lancet Gastroenterology & Hepatology Commission. Lancet Gastroenterol. Hepatol. 8, 458–492 (2023).
Gearry, R. B. et al. What are the most challenging aspects of inflammatory bowel disease? An international survey of gastroenterologists comparing developed and developing countries. Inflamm. Intest. Dis. 6, 78–86 (2021).
Baingana, F. et al. Global research challenges and opportunities for mental health and substance-use disorders. Nature 527, S172–S177 (2015).
Chapman, N. et al. A roadmap of strategies to support cardiovascular researchers: from policy to practice. Nat. Rev. Cardiol. 19, 765–777 (2022).
Lazarus, J. V. et al. A global action agenda for turning the tide on fatty liver disease. Hepatology 79, 502–523 (2024).
Francis, A. et al. Chronic kidney disease and the global public health agenda: an international consensus. Nat. Rev. Nephrol. 20, 473–485 (2024).
Kaplan, G. G. & Ng, S. C. Understanding and preventing the global increase of inflammatory bowel disease. Gastroenterology 152, 313–321.e2 (2017).
Kaplan, G. G. & Windsor, J. W. The four epidemiological stages in the global evolution of inflammatory bowel disease. Nat. Rev. Gastroenterol. Hepatol. 18, 56–66 (2021).
Molodecky, N. A. et al. Increasing incidence and prevalence of the inflammatory bowel diseases with time, based on systematic review. Gastroenterology 142, 46–54 (2012).
Mak, J. W. Y. et al. Development of the global inflammatory bowel disease visualization of epidemiology studies in the 21s) century (GIVES-21). BMC Med. Res. Methodol. 23, 129 (2023).
Chaparro, M. et al. EpidemIBD: rationale and design of a large-scale epidemiological study of inflammatory bowel disease in Spain. Ther. Adv. Gastroenterol. 12, 1756284819847034 (2019).
Chen, X. et al. Global burden of inflammatory bowel disease among children and adolescents: a comprehensive analysis (1990–2019). Int. J. Public. Health 69, 1607440 (2024).
Zheng, H. B., de la Morena, M. T. & Suskind, D. L. The growing need to understand very early onset inflammatory bowel disease. Front. Immunol. 12, 675186 (2021).
Granot, M. et al. Differences in disease characteristics and treatment exposures between paediatric and adult-onset inflammatory bowel disease using a registry-based cohort. Aliment. Pharmacol. Ther. 60, 1435–1446 (2024).
Torres, J. et al. Serum biomarkers identify patients who will develop inflammatory bowel diseases up to 5 years before diagnosis. Gastroenterology 159, 96–104 (2020).
Gaifem, J. et al. A unique serum IgG glycosylation signature predicts development of Crohn’s disease and is associated with pathogenic antibodies to mannose glycan. Nat. Immunol. 25, 1692–1703 (2024).
Misra, R. et al. Epidemiology of inflammatory bowel disease in racial and ethnic migrant groups. World J. Gastroenterol. 24, 424–437 (2018).
Spekhorst, L. M. et al. The impact of ethnicity and country of birth on inflammatory bowel disease phenotype: a prospective cohort study. J. Crohns Colitis 11, 1463–1470 (2017).
Agrawal, M. et al. Multiomics to elucidate inflammatory bowel disease risk factors and pathways. Nat. Rev. Gastroenterol. Hepatol. 19, 399–409 (2022).
Hawkins, R. L. et al. Inequalities in healthcare access, experience and outcomes in adults with inflammatory bowel disease: a scoping review. Inflamm. Bowel Dis. 30, 2486–2499 (2024).
Shah, S. et al. Racial and ethnic disparities in patients with inflammatory bowel disease: an online survey. Inflamm. Bowel Dis. 30, 1467–1474 (2024).
Sempere, L. et al. Gender biases and diagnostic delay in inflammatory bowel disease: multicenter observational study. Inflamm. Bowel Dis. 29, 1886–1894 (2023).
Klein, S. L. & Flanagan, K. L. Sex differences in immune responses. Nat. Rev. Immunol. 16, 626–638 (2016).
Siegel, C. A. Refocusing IBD patient management: personalized, proactive, and patient-centered care. Am. J. Gastroenterol. 113, 1440–1443 (2018).
Heisler, C. et al. Patient-centered access to IBD care: a qualitative study. Crohns Colitis 5, otac045 (2023).
Morar, P. et al. Defining the optimal design of the inflammatory bowel disease multidisciplinary team: results from a multicentre qualitative expert-based study. Frontline Gastroenterol. 6, 290–297 (2015).
Schoenfeld, R., Nguyen, G. C. & Bernstein, C. N. Integrated care models: optimizing adult ambulatory care in inflammatory bowel disease. J. Can. Assoc. Gastroenterol. 3, 44–53 (2020).
Habashi, P., Bouchard, S. & Nguyen, G. C. Transforming access to specialist care for inflammatory bowel disease: the PACE telemedicine program. J. Can. Assoc. Gastroenterol. 2, 186–194 (2019).
Click, B., Cross, R. K., Regueiro, M. & Keefer, L. The IBD clinic of tomorrow: holistic, patient-centric, and value-based care. Clin. Gastroenterol. Hepatol. 23, 419–427.e3 (2025).
Danese, S. et al. Catching the therapeutic window of opportunity in early Crohn’s disease. Curr. Drug. Targets 15, 1056–1063 (2014).
Choung, R. S. et al. Preclinical serological signatures are associated with complicated Crohn’s disease phenotype at diagnosis. Clin. Gastroenterol. Hepatol. 21, 2928–2937.e12 (2023).
Danese, S. et al. The future of drug development for inflammatory bowel disease: the need to ACT (advanced combination treatment). Gut 71, 2380–2387 (2022).
Stalgis, C. et al. Rational combination therapy to overcome the plateau of drug efficacy in inflammatory bowel disease. Gastroenterology 161, 394–399 (2021).
Godny, L. et al. Mechanistic implications of the Mediterranean diet in patients with newly diagnosed Crohn’s disease: multi-omic results from a prospective cohort. Gastroenterology https://doi.org/10.1053/j.gastro.2024.12.031 (2025).
Milo, F. et al. Short-term psychodynamic psychotherapy in addition to standard medical therapy increases clinical remission in adolescents and young adults with inflammatory bowel disease: a randomised controlled trial. J. Crohns Colitis 18, 256–263 (2024).
Singh, S., Picardo, S. & Seow, C. H. Management of inflammatory bowel diseases in special populations: obese, old, or obstetric. Clin. Gastroenterol. Hepatol. 18, 1367–1380 (2020).
Newman, K. L., Chedid, V. G. & Boden, E. K. A systematic review of inflammatory bowel disease epidemiology and health outcomes in sexual and gender minority individuals. Clin. Gastroenterol. Hepatol. 21, 1127–1133 (2023).
Jena, A., Sharma, V. & Sebastian, S. Reducing disparities in training in inflammatory bowel disease. Lancet Gastroenterol. Hepatol. 8, 692–693 (2023).
Fiorindi, C. et al. Inadequate food literacy is related to the worst health status and limitations in daily life in subjects with inflammatory bowel disease. Clin. Nutr. ESPEN 52, 151–157 (2022).
Barreiro-de Acosta, M. et al. Recommendations of the Spanish Working Group on Crohn’s Disease and Ulcerative Colitis (GETECCU) and the association of Crohn’s disease and ulcerative colitis patients (ACCU) in the management of psychological problems in Inflammatory bowel disease patients. Gastroenterol. Hepatol. 41, 118–127 (2018).
Kochar, B. et al. Depression is associated with more aggressive inflammatory bowel disease. Am. J. Gastroenterol. 113, 80–85 (2018).
Sheehan, J. L. et al. Are depression and anxiety underdiagnosed in socially vulnerable patients with inflammatory bowel disease? Inflamm. Bowel Dis. 30, 1696–1706 (2024).
Zeisler, B. & Hyams, J. S. Transition of management in adolescents with IBD. Nat. Rev. Gastroenterol. Hepatol. 11, 109–115 (2014).
McDermott, E. et al. Patient education in inflammatory bowel disease: a patient-centred, mixed methodology study. J. Crohns Colitis 12, 419–424 (2018).
Le Berre, C. et al. Evolving short- and long-term goals of management of inflammatory bowel diseases: getting it right, making it last. Gastroenterology 162, 1424–1438 (2022).
Temido, M. J. et al. Overcoming the challenges of overtreating and undertreating inflammatory bowel disease. Lancet Gastroenterol. Hepatol. 10, 462–474 (2025).
Yu, N. et al. Outcomes of a comprehensive specialist inflammatory bowel disease nursing service. Inflamm. Bowel Dis. 30, 960–969 (2024).
Graffigna, G. et al. Promoting psycho-social wellbeing for engaging inflammatory bowel disease patients in their care: an Italian consensus statement. BMC Psychol. 9, 186 (2021).
IBD UK. The State of IBD Care in the UK (IBD UK, 2024).
Deepak, P., Barnes, E. L. & Shaukat, A. Health disparities in inflammatory bowel disease care driven by rural versus urban residence: challenges and potential solutions. Gastroenterology 165, 11–15 (2023).
Fierens, L. et al. A core outcome set for inflammatory bowel diseases: development and recommendations for implementation in clinical practice through an international multi-stakeholder consensus process. J. Crohns Colitis 18, 1583–1595 (2024).
Anyane-Yeboa, A. et al. The impact of the social determinants of health on disparities in inflammatory bowel disease. Clin. Gastroenterol. Hepatol. 20, 2427–2434 (2022).
Nguyen, N. H. et al. Prevalence and effects of food insecurity and social support on financial toxicity in and healthcare use by patients with inflammatory bowel diseases. Clin. Gastroenterol. Hepatol. 19, 1377–1386.e5 (2021).
Vinuesa, R. et al. The role of artificial intelligence in achieving the sustainable development goals. Nat. Commun. 11, 233 (2020).
Chedid, V. et al. Culturally sensitive and inclusive IBD care. Clin. Gastroenterol. Hepatol. 23, 440–453 (2024).
Boulkedid, R. et al. Using and reporting the Delphi method for selecting healthcare quality indicators: a systematic review. PLoS ONE 6, e20476 (2011).
Acknowledgements
The authors are grateful to the following experts for their invaluable contributions to the development of the R1 statements: E. Benchimol (University of Toronto, Canada), J. Burisch (University of Copenhagen, Denmark), E. Loftus (Mayo Clinic, USA), J. Lewis (University of Pennsylvania, USA), A. Ananthakrishnan (Massachusetts General Hospital, USA), A. Armuzzi (Humanitas University, IRCCS Humanitas Research Hospital, Italy), G. Fiorino (San Camillo-Forlanini Hospital, Italy), J. Halfvarson (Örebro University, Sweden), F. Rieder (Cleveland Clinic, USA), P. Kotze (Pontifical Catholic Univeristy of Paraná, Brazil), B. Feagan (Alimentiv, Western University, Canada), J. Panés (Hospital Clínic Barcelona, Spain), R. Leong (University of Sydney, Australia), B. Shah (Icahn School of Medicine at Mount Sinai Hospital, USA), J. Torres (Hospital Beatriz Ângelo, Portugal), R. Cohen (University of Chicago, USA), S. Coderre (University of Calgary, Canada), A. Spinelli (Humanitas University, IRCCS Humanitas Research Hospital, Italy), P. Munkholm (University of Copenhagen, Denmark), E. Louis (University Hospital Liège, Belgium), V. Ahuja (All India Institute of Medical Sciences, India), L. Plung (Crohn’s & Colitis Foundation, USA), S. Leone (National Association for Chronic Inflammatory Bowel Diseases, Italy), A. Lobo (Royal Hallamshire Hospital, UK), J. Andrews (University of Adelaide, Australia), M. Wong (Chinese University of Hong Kong, China) and S. Sebastian (Hull University Teaching Hospitals NHS Trust, UK). Their expertise and support were crucial to the development of these statements, and the authors sincerely thank them for their contributions. The project would not have been possible without the valuable support of organizations, including the core project team as well as members of the IOIBD (International Organization for the Study of Inflammatory Bowel Disease) mailing list. We also received significant support from the IOIBD Secretariat (M. Konings), the EFCCA (European Federation of Crohn’s and Colitis Associations), represented by S. Leone, the EUPHA (European Public Health Association), represented by S. Cuschieri and S. Stranges, and the IGIBD (Italian Group for the Study of Inflammatory Bowel Disease).
Author information
Authors and Affiliations
Contributions
The authors contributed equally to all aspects of the article.
Corresponding author
Ethics declarations
Competing interests
V.S. has received speaker fees from Pfizer. C.N.B. is supported by the Bingham Chair in Gastroenterology. C.N.B. has served on advisory boards for AbbVie Canada, Amgen Canada, Bristol Myers Squibb Canada, Eli Lilly Canada, JAMP Pharmaceuticals, Janssen Canada, Pendopharm Canada, Sandoz Canada, Takeda Canada and Pfizer Canada; has received educational grants from Abbvie Canada, Bristol Myers Squibb Canada, Ferring Canada, Pfizer Canada, Takeda Canada, Janssen Canada, Organon Canada, Eli Lilly Canada, Amgen Canada and Medtronic; is a member of the speaker’s panel for Abbvie Canada, Eli Lilly, Janssen Canada, Pfizer Canada and Takeda Canada; and has received research funding from Abbvie Canada, Amgen Canada, Sandoz Canada, Takeda Canada and Pfizer Canada. I.D. has received consultancy fees or honoraria from Abbott, Abbvie, Athos, Arena, Altman Research, Cambridge Healthcare, Celltrion, Celgene/B.M.S., Eli Lilly, Ferring, Falk Pharma, Food Industries Organization, Gilead, Galapagos, Iterative Scopes, Integra Holdings, Janssen, Neopharm, Pfzer, Rafa laboratories, Roche/Genentech, Sangamo, Sublimity, Sandoz, Takeda and Wildbio; and grants from Altman Research, B.M.S. and Pfzer. A.D. has received fees for participation in clinical trials, review activities such as data monitoring boards, statistical analysis and end point committees from Abivax, AbbVie, Bristol Myers Squibb, Dr Falk Pharma, Galapagos, Gilead, Janssen and Pfizer; consultancy fees from AbbVie, Amgen, Biogen, Boehringer Ingelheim, Bristol Myers Squibb, Celltrion, Dr Falk Foundation, Ferring Pharmaceuticals, Fresenius Kabi, Galapagos, J&J, Lilly, MSD, Pfizer, Pharmacosmos, Sandoz, Stada, Takeda, Tillotts and Vifor Pharma; payments for lectures including service on speakers bureaus from AbbVie, Biogen, CED Service, Celltrion, Falk Foundation, Ferring, Galapagos, Gilead, High5MD, Janssen, Materia Prima, MedToday, MSD, Pfizer, Streamed-Up, Takeda, Tillotts and Vifor Pharma; and payments for manuscript preparation from Falk Foundation, Takeda, Thieme and UniMed Verlag. M.C.D. has received consultancy fees from Abbvie, Arena, BMS, Eli Lilly, Janssen, Pfizer, Prometheus Biosciences and Takeda. R.B.G. has received research grants from Janssen, Zespri, Comvita; and is a member of advisory boards: AbbVie, Janssen, Celltrion, Zespri and Comvita. A.H. has received consultancy, advisory board member or speaker’s fees from AbbVie, Arena, Atlantic, Bristol-Myers Squibb, Celgene, Celltrion, Falk, Galapogos, Lilly, Janssen, MSD, Napp Pharmaceuticals, Pfizer, Pharmacosmos, Shire and Takeda. G.G.K. has received honoraria for speaking or consultancy from AbbVie, Amgen, Janssen, Pfizer and Takeda; grants for research from Ferring and for educational activities from AbbVie, Bristol Myers Squibb, Ferring, Fresenius-Kabi, Janssen, Pfizer, Takeda; and shares ownership of a patent (Use of mirtazapine in the treatment of inflammatory disorders, autoimmune disease and PBC; patent WO2019046959A1). C.M. has received consultancy fees from AbbVie, Alimentiv, Amgen, AVIR Pharma, Bristol Myers Squibb, Celltrion, Eli Lilly, Ferring, Forte Biosciences, Fresenius Kabi, Gilead, Janssen, McKesson, Mirador Therapeutics, Mylan, Pendopharm, Pfizer, Prometheus Biosciences, Roche, Sanofi, Takeda, Tillotts and Pharma; speaker’s fees from AbbVie, Amgen, AVIR Pharma, Alimentiv, Bristol Myers Squibb, Eli Lilly, Ferring, Fresenius Kabi, Janssen, Merck, Organon, Pendopharm, Pfizer, Sanofi, Takeda and Tillotts Pharma; royalties from Springer Publishing; and research support from AbbVie, Eli Lilly, Ferring and Pfizer. F.M. has received fees from AbbVie, Amgen, Arena, Celgene/ Bristol Myers Squibb, Falk, Ferring, Fresenius Kabi, Galapagos, Gilead, high5MD, Janssen, Johnson & Johnson, Lilly, Materia Prima, Merck Sharp & Dohme, Pharmacosmos, Pfizer, Sandoz/Hexal, Takeda, Tillotts and Vifor. S.C.N. has served as an advisory board member for Pfizer, Ferring, Janssen and Abbvie; has received honoraria as a speaker from Ferring, Tillotts, Menarini, Janssen, Abbvie and Takeda; has received research grants through her affiliated institutions from Olympus, Ferring and Abbvie; is a founder member, non-executive director, non-executive scientific adviser and shareholder of GenieBiome; receives patent royalties through her affiliated institutions; and is a named inventor on patent applications held by the CUHK and MagIC that cover the therapeutic and diagnostic use of microbiome. R.P. has received consultancy fees from Abbott, AbbVie, Abbivax, Alimentiv (formerly Robarts), Amgen, AnaptysBio, Arena Pharmaceuticals, AstraZeneca, Biogen, Boehringer Ingelheim, Bristol-Myers Squibb, Celgene, Celltrion, Cosmos Pharmaceuticals, Eisai, Elan, Eli Lilly, Ferring, Galapagos, Fresenius Kabi, Genentech, Gilead Sciences, Glaxo-Smith Kline, JAMP Bio, Janssen, Merck, Mylan, Novartis, Oppilan Pharma, Organon, Pandion Pharma, Pendopharm, Pfizer, Progenity, Prometheus Biosciences, Protagonist Therapeutics, Roche, Sandoz, Satisfai Health, Shire, Sublimity Therapeutics, Spyre Therapeutics, Takeda Pharmaceuticals, Theravance Biopharma, Trellus, Union Biopharma, Viatris, Ventyx and UCB; speaker’s fees from AbbVie, Amgen, Arena Pharmaceuticals, Bristol-Myers Squibb, Celgene, Eli Lilly, Ferring, Fresenius Kabi, Gilead Sciences, Janssen, Merck, Organon, Pfizer, Roche, Sandoz, Shire and Takeda Pharmaceuticals; and is a member of advisory boards for AbbVie, Alimentiv (formerly Robarts), Amgen, Arena Pharmaceuticals, AstraZeneca, Biogen, Boehringer Ingelheim, Bristol-Myers Squibb, Celgene, Eli Lilly, Ferring, Fresenius Kabi, Genentech, Gilead Sciences, Glaxo-Smith Kline, JAMP Bio, Janssen, Merck, Mylan, Novartis, Oppilan Pharma, Organon, Pandion Pharma, Pfizer, Progenity, Protagonist Therapeutics, Roche, SandozShire, Sublimity Therapeutics, Takeda Pharmaceuticals and Ventyx. D.T.R. has received grant support from Takeda; and has served as a consultant for Abbvie, Altrubio, Bellatrix Pharmaceuticals, Boehringer Ingelheim, Bristol-Myers Squibb, Syneos, Dizal Pharmaceuticals, Galapagos, Ichnos Sciences, InDex Pharmaceuticals, Iterative Health, Janssen Pharmaceuticals, Lilly, Pfizer, Prometheus Biosciences, Reistone and Takeda. C.A.S. has served as a consultant for Abbvie, BMS, Boomerang, Buhlmann, Lilly, Janssen, Napo Pharmaceuticals, Pfizer, ProciseDx, Prometheus Biosciences, Prometheus Labs, Roivant, Takeda and Trellus Health; has served as a speaker for CME activities for Abbvie, Janssen, Pfizer, Sandoz and Takeda; and has received grant support from Abbvie, BMS, Janssen, Lilly, Pfizer and Takeda. V.J. has received consultancy/advisory board fees from AbbVie, Alimentiv, Arena pharmaceuticals, Asahi Kasei Pharma, Asieris, Astra Zeneca, Bristol Myers Squibb, Celltrion, Eli Lilly, Ferring, Flagship Pioneering, Fresenius Kabi, Galapagos, GlaxoSmithKline, Genentech, Gilead, Janssen, Merck, Mylan, Pandion, Pendopharm, Pfizer, Protagonist, Reistone Biopharma, Roche, Sandoz, Second Genome, Takeda, Teva, Topivert, Ventyx and Vividion; and speaker’s fees from, Abbvie, Ferring, Galapagos, Janssen Pfizer Shire, Takeda and Fresenius Kabi. L.P.-B. has received consultancy fees from Abbvie, Abivax, Adacyte, Alimentiv, Amgen, Applied Molecular Transport, Arena, Banook, Biogen, BMS, Celltrion, Connect Biopharm, Cytoki Pharma, Enthera, Ferring, Fresenius Kabi, Galapagos, Genentech, Gilead, Gossamer Bio, GSK, IAC Image Analysis, Index Pharmaceuticals, Inotrem, Janssen, Lilly, Medac, Mopac, Morphic, MSD, Nordic Pharma, Novartis, Oncodesign Precision Medicine, ONO Pharma, OSE Immunotherapeuthics, Pandion Therapeuthics, Par’ Immune, Pfizer, Prometheus, Protagonist, Roche, Samsung, Sandoz, Sanofi, Satisfay, Takeda, Telavant, Theravance, Thermo Fischer, Tigenix, Tillots, Viatris, Vectivbio, Ventyx and Ysopia; grants from: Celltrion, Fresenius Kabi, Medac, MSD and Takeda; lecture fees from Abbvie, Amgen, Arena, Biogen, Celltrion, Ferring, Galapagos, Genentech, Gilead, Janssen, Lilly, Medac, MSD, Nordic Pharma, Pfizer, Sandoz, Takeda, Tillots and Viatris. S.D. has received consultancy fees from AbbVie, Alimentiv, Allergan, Amgen, AstraZeneca, Athos Therapeutics, Biogen, Boehringer Ingelheim, Bristol Myers Squibb, Celgene, Celltrion, Dr Falk Pharma, Eli Lilly, Enthera, Ferring Pharmaceuticals, Gilead, Hospira, Inotrem, Janssen, Johnson & Johnson, MSD, Mundipharma, Mylan, Pfizer, Roche, Sandoz, Sublimity Therapeutics, Takeda, TiGenixa, UCB and Vifor; and lecture fees from AbbVie, Amgen, Ferring Pharmaceuticals, Gilead, Janssen, Mylan, Pfizer and Takeda. R.D., J.W.Y.M. and S.R. declare no competing interests.
Peer review
Peer review information
Nature Reviews Gastroenterology & Hepatology thanks Sunanda Kane, Leolin Katsidzira and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Related links
International Organization for the Study of Inflammatory Bowel Disease: https://ioibd.org/
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Solitano, V., Bernstein, C.N., Dotan, I. et al. Shaping the future of inflammatory bowel disease: a global research agenda for better management and public health response. Nat Rev Gastroenterol Hepatol 22, 438–452 (2025). https://doi.org/10.1038/s41575-025-01063-x
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41575-025-01063-x
This article is cited by
-
The global burden of inflammatory bowel disease: from 2025 to 2045
Nature Reviews Gastroenterology & Hepatology (2025)
-
Maternal mid-pregnancy dietary patterns and inflammatory bowel disease in offspring from a prospective cohort study
Communications Medicine (2025)
-
Global Training in Inflammatory Bowel Disease: Addressing Growing Challenges in IBD
Digestive Diseases and Sciences (2025)
-
Intravenous iron in ulcerative colitis—Raising the (iron) bar
Indian Journal of Gastroenterology (2025)
-
Curcumin for inflammatory bowel disease therapy: advances in mechanisms, clinical applications, and drug delivery
Archives of Pharmacal Research (2025)