Abstract
Cholangiocarcinoma (CCA) is a cancer that originates within the bile ducts. Traditionally considered to be a rare neoplasm, increased awareness of CCA alongside advancements in diagnosis and the rising prevalence of certain risk factors have contributed to a global increase in incidence and mortality. CCAs are highly heterogeneous from the clinical, histomorphological and molecular perspectives but commonly share a poor prognosis. These tumours usually develop and progress silently; by the time they are detected, it is often too late for curative surgical intervention. In such cases, current therapeutic approaches offer modest survival improvements and are generally considered palliative. Although well-known risk factors predispose individuals to developing CCA, the majority of cases are considered sporadic, occurring without any identifiable underlying condition. Over the past decade, substantial collaborative efforts have been made to improve our understanding of the aetiopathogenesis of these tumours, aiming to identify novel biomarkers and therapeutic targets to develop more effective treatments. The ultimate goal is to improve patient outcomes and overall well-being. However, there are significant gaps in our understanding of the molecular mechanisms that drive cholangiocarcinogenesis. In this international Consensus Statement, which is endorsed by the European Network for the Study of Cholangiocarcinoma, we provide a critical overview of the latest advancements in the field of CCA. We highlight the key aspects of CCA aetiopathogenesis and clinical management and provide insights into promising new treatments. Finally, we provide a set of consensus recommendations and future research priorities for CCA based on a Delphi panel questionnaire involving international experts.
Key points
-
Surgery is currently the only potentially curative option for cholangiocarcinoma (CCA), and surgical resection can be offered to 20–30% of newly diagnosed patients with CCA.
-
Adjuvant chemotherapy with capecitabine for 6 months after surgical resection with curative intent is recommended for CCA.
-
Liver transplantation is a potentially curative option for small intrahepatic CCAs and selected cases of perihilar CCAs.
-
Combining cisplatin, gemcitabine and immune-checkpoint inhibitors is the standard of care for patients with advanced CCA.
-
FOLFOX is recommended as the second-line standard-of-care chemotherapy in CCA without druggable molecular alterations.
-
Molecular profiling of CCA tumours is highly recommended to guide access to targeted therapies that are particularly relevant in CCA such as FGFR2 inhibitors, IDH1 inhibitors, BRAF inhibitors and HER2-targeting therapies.
Similar content being viewed by others
Introduction
Biliary tract cancers encompass a heterogeneous group of malignancies, including cholangiocarcinoma (CCA), gallbladder cancer and ampullary cancer, which show differences in aetiology, presentation, management and outcomes. Specifically, CCA is a heterogeneous and aggressive cancer originating from bile duct epithelial cells1,2. It can arise anywhere in the biliary system between the intrahepatic and common bile ducts. Although CCA is a rare neoplasm, unlike many other cancers, the incidence and mortality of CCA have been increasing worldwide in the past two decades1,3. For example, data on primary liver cancers in England, UK, suggest that the prevalence of intrahepatic CCA (iCCA) has now equalled that of hepatocellular carcinoma (HCC), imposing a significant social, health and economic burden4. Furthermore, CCA is considered to be responsible for 2% of all cancer-related deaths1,2. However, these statistics should be interpreted with caution as they are likely to be an underestimation of the real values due to global errors in cancer coding and data retrieval.
In 2022, after identifying potential dissimilarities in aetiopathogenesis, risk factors, incidence and prognosis of distinct types of CCAs, the WHO approved a new classification for these tumours based on their anatomical origin (International Classification of Diseases 11th revision (ICD-11)). This classification distinguishes between iCCA, perihilar CCA (pCCA) and distal CCA (dCCA) (Fig. 1). iCCA originates proximal to the second-order bile ducts, whereas the cystic duct delimits pCCA from dCCA in the extrahepatic forms of CCA (eCCA)1,2. Although previous studies have reported distinct prevalences for the CCA subtypes, these overall percentages require further validation using the new ICD-11 coding system.
Taking into consideration the anatomical site of origin of cholangiocarcinoma (CCA), the new International Classification of Diseases 11th revision (ICD-11) classifies these tumours into intrahepatic CCA (iCCA), perihilar CCA (pCCA) and distal CCA (dCCA). iCCAs usually arise in the intrahepatic biliary tree, proximal to the second-order bile ducts, and two distinct histological subtypes exist: small bile duct (SBD) and large bile duct (LBD) iCCAs; pCCAs develop in the right and/or left hepatic ducts and common hepatic duct whereas the cystic duct delimits these tumours from dCCAs, which develop distal to this in the common bile duct. Each CCA anatomical and/or histological subtype displays different genetic landscapes, and the most frequent genetic mutations and/or alterations are highlighted for each of them. The prevalence (%) of the genetic mutations in each CCA subtype was obtained from previous reports194,376,377,378,379,380. Representative histological staining (haematoxylin and eosin (H&E), cytokeratin 7 (CK7) or periodic acid–Schiff (PAS)) for each CCA subtype is presented.
From the clinical, histomorphological and molecular perspectives, CCA is highly heterogeneous; however, the different subtypes of CCA share a poor prognosis, as they often grow silently and, when detected, patients are frequently no longer eligible for curative intent options. In this setting, current systemic anticancer treatments for patients with advanced CCA exhibit limited efficacy and are considered palliative, highlighting the need for better therapeutic options1,2.
In this Consensus Statement, we aim to provide a comprehensive and critical perspective on the current understanding of CCA, as well as unmet needs and priorities. This focus extends to epidemiology, risk factors, clinical presentation, diagnosis, genetic and molecular landscapes, therapies, patient perspectives, and future directions and recommendations.
Methods
This international panel of multidisciplinary experts in the management and/or study of CCA, including physicians and clinical researchers (oncologists, surgeons, hepatologists, gastroenterologists and pathologists), basic and translational scientists, and patient representatives, is affiliated with the European Network for the Study of Cholangiocarcinoma (ENS-CCA) and is part of the European COST Actions Euro-Cholangio-Net (2019–2023) and Precision-BTC-Network (2023–2027). This pan-European network is dedicated to enhancing the understanding and treatment of CCA through the establishment of collaborative projects and to provide education and awareness of this cancer globally. The main objective of this multidisciplinary Consensus Statement is to offer a comprehensive and critical assessment of the current knowledge generated in the field in recent years by multiple research groups and patient associations worldwide, and to outline future directions and research priorities, along with expert recommendations generated from a Delphi panel questionnaire involving international experts.
To generate this statement, J. M. Banales, P. M. Rodrigues and C. Braconi identified the areas of interest, organized the Consensus Statement into sections, and assigned each section to various expert members. To draft the manuscript, a systematic PubMed search was conducted, combining the term “cholangiocarcinoma” with key terms such as “epidemiology”, “risk factors”, “classification”, “diagnosis”, “staging”, “genetics”, “signalling pathways”, “metabolism”, “stemness”, “cancer stem cells”, “tumour microenvironment”, “immunology”, “biomarkers”, “endoscopy”, “surgery”, “liver transplantation”, “therapies” and “clinical trials”. No specific search dates were applied; however, priority was given to publications from 2020 to 2025 to ensure the inclusion of the most up-to-date content in the field. All sections were merged into an initial draft by P. M. Rodrigues and J. M. Banales. Subsequently, the manuscript underwent extensive revisions, involving all authors for further corrections, improvements, discussions and approval.
Expert consensus
Panel of experts
A core group constituted by J. M. Banales, P. M. Rodrigues and C. Braconi, in close collaboration with all the authors of the manuscript, who have significantly contributed to the field of CCA (including founders and representatives from patient charities Cholangiocarcinoma Foundation and AMMF), created a Delphi study to identify scientific and clinical gaps and barriers, research priorities, and future directions, leading to the establishment of a set of recommendations for CCA. In parallel, the core group identified 176 additional international experts based on their experience in the field and existing collaborations, interactions, or personal connections with ENS-CCA members. These experts were invited to participate in the Delphi questionnaire, with 108 actually enrolling (61.4% participation). These experts provided their critical opinion but were not involved in writing the recommendations. Overall, the panel of experts consisted of 147 individual experts from 35 countries/regions across Europe (n = 19), North America (n = 2), Latin America (n = 7), Asia (n = 5), Africa (n = 1) and Oceania (n = 1). Of note, as most participants in the Delphi questionnaire were from Europe, establishing collaborative connections with Asia, Latin America and Africa is warranted to increase participation from these regions in the future. Supplementary Table 1 summarizes the names, affiliations and demographics of the panel of experts.
Questionnaire and consensus building
We used a modified Delphi method to reach a consensus, comprising one round of questions. The core team created the questionnaire using an online Google Form before distributing it to all the experts. The questionnaire encompassed 59 questions divided into 8 sections: Epidemiology and Risk Factors (section 1); Clinical Presentation: Diagnosis and Staging (section 2); Genetics (section 3); Tumour Biology (section 4); Biomarkers (section 5); Treatment (section 6); Supportive Care (section 7); and Future Research (section 8). Experts had the opportunity to provide comments on selected questions when asked. The questionnaire and summary of the results are provided in Supplementary Data. The classification of the consensus was conducted as follows: unanimous (U; 100% agreement); A (90–99% agreement); B (70–89% agreement); C (50–69% agreement); and D (no consensus, <50% agreement). By engaging the consensus of experts in the field, we hereby propose statements and recommendations (Table 1) and future research priorities (Table 2).
Mortality
Data presented in Fig. 2 were obtained by calculating the mortality for CCA in selected countries in 2021 (unless otherwise specified). Mortality data for CCA was obtained from the WHO Mortality Database using the International Classification of Diseases 10th revision (ICD-10) codes for iCCA (C22.1) and eCCA (C24.0)5. The total number of deaths for men and women, and iCCA and eCCA, for 38 countries were calculated for the years 2011, 2013, 2015, 2017, 2019 and 2021. Age-standardized mortality rates (ASMRs) were calculated for each year and country using population data from 5-yearly age strata (0–4, 5–9, 10–14, 15–19, 20–24, 25–29, 30–34, 35–39, 40–44, 45–49, 50–54, 55–59, 60–64, 65–69, 70–74, 75–79, 80–84, 85+) obtained from the Global Burden of Disease Study 2021 (ref. 6). ASMR per 100,000 inhabitants was calculated using the WHO standard population7. The average annual percentage change of ASMR was calculated for each country. Data are displayed in Supplementary Table 2 and Fig. 2.
Worldwide age-standardized mortality rates (ASMRs) for cholangiocarcinoma (CCA) per 100,000 inhabitants with the average annual percentage change (AAPC) between 2011 and 2021. Mortality data for 2021 are shown unless otherwise stated. Mortality data for CCA were obtained from the WHO Mortality Database using the International Classification of Diseases 10th revision (ICD-10) codes for intrahepatic (C22.1) and extrahepatic (C24.0) CCA5. Yellow indicates countries/regions with low mortality (<1 death per 100,000 inhabitants), orange indicates countries/regions with mortality between 1 and 2 deaths per 100,000 inhabitants, and red indicates countries/regions with high mortality (>2 deaths per 100,000 inhabitants).
Epidemiology and risk factors
-
Statements and recommendations: 1.1–1.5
-
Research priorities: I
Incidence of CCA appears to be increasing globally, and CCA represents ~10–15% of all primary liver tumours and 3% of gastrointestinal cancers1,3. Nevertheless, we believe these data might be underestimated due to systematic errors in data retrieval, diagnosis and coding. Numerous studies spanning the past two decades consistently report ongoing increases in the incidence and mortality for iCCA, while rates for eCCA remain stable or decline in most countries examined8,9,10,11. Of note, the reported worldwide increase in iCCA incidence may be linked to an increased awareness of cancers previously misclassified as ‘cancer of unknown primary’ during recent years. Nevertheless, an increase in the mortality of CCA in the last decade (2011–2021) was identified in almost all of the countries analysed (Fig. 2 and Supplementary Table 2). Importantly, in Europe, CCA mortality is particularly high (>2 per 100,000 inhabitants). The increasing CCA rates are likely a result of the interplay between the geographical distribution of associated risk factors and host-specific factors, including underlying liver conditions and genetic background12. Notably, the highest CCA rates in the world are observed in northeastern Thailand and neighbouring regions, where the primary risk factor is chronic liver fluke infection associated with the consumption of raw fish10. Conversely, in the Western world, where liver flukes are not a concern and CCA incidence is much lower, the most well-recognized risk factor for CCA is the chronic inflammatory bile duct disease primary sclerosing cholangitis (PSC)1. An important caveat when analysing CCA epidemiological studies is the lack of information regarding the incidence and mortality for pCCA. This is a result of the previous lack of a specific code for pCCA in historic ICD coding systems. Consequently, some studies suggest that pCCA was likely (mis)coded primarily as iCCA and to a lesser extent as eCCA in the past13,14. While the latest version of ICD coding (ICD-11) will rectify this issue by introducing specific and separate codes for iCCA, pCCA and dCCA, countries adopt new ICD classifications at different times, and it may take several years for uniform data collection to become established internationally. Furthermore, the new ICD classifications will not assist in understanding historical rates of pCCA as distinct from iCCA and dCCA.
In Western countries, the majority of CCA cases are currently classified as sporadic, with no identifiable risk factors10. Certain low-prevalence factors pose a significant risk for CCA development, often associated with chronic inflammation of the biliary epithelium and/or biliary stasis. These factors include the presence of choledochal cysts, gallstones, cirrhosis, biliary diseases (such as Caroli disease and PSC), viral infections (such as hepatitis B virus and hepatitis C virus), infestation with liver flukes (Opisthorchis viverrini and Clonorchis sinensis, particularly in Asia), and germline mutations in tumour suppressor genes (that is, BRCA1/2, ATM, BAP1). Conversely, some factors confer a moderate risk of CCA development and are highly prevalent in high-income countries. These factors include alcohol-related liver disease, type 2 diabetes mellitus, tobacco use, metabolic dysfunction-associated steatotic liver disease (MASLD) and ageing. Of note, some risk factors are shared between iCCA and eCCA (pCCA + dCCA), while others are presumed to be more specific to one CCA subtype (Fig. 3). Regarding genetic predisposition, polymorphisms in host genes involved in functions, such as xenobiotic detoxification, DNA repair, multidrug resistance, immune response and folate metabolism, may predispose individuals to cholangiocarcinogenesis1,10,15. However, it is essential to note that, to date, no genome-wide association studies have been published on CCA.
Relationship between worldwide prevalence of risk factors and the odds of cholangiocarcinoma (CCA) development. Although the majority of CCA cases are currently classified as sporadic, with no identifiable risk factors, there are certain low/medium-prevalence factors that pose a significant risk for CCA development such as biliary diseases (such as Caroli disease and primary sclerosing cholangitis (PSC)), the presence of choledochal cysts, biliary gallstones, chronic pancreatitis, cirrhosis, liver flukes (Opisthorchis viverrini and Clonorchis sinensis), or viral infections (such as hepatitis B virus (HBV) and hepatitis C virus (HCV)). On the other hand, there are factors that confer a moderate/low risk of CCA development but are highly prevalent in high-income countries, including alcohol-related liver disease, type 2 diabetes mellitus (T2DM), tobacco use and metabolic dysfunction-associated steatotic liver disease (MASLD). Almost all risk factors are shared between both intrahepatic CCA (iCCA) and extrahepatic CCA (eCCA; perihilar and distal CCA), while only MASLD is presumed to be more associated with iCCA development. The odds ratio (OR) values for each one of the risk factors were obtained from previous published data381,382,383. IBD, inflammatory bowel disease. aThe prevalence of liver flukes was considered for the Asian countries where they are endemic.
Although still pending prospective validation, aspirin and statin use have been associated with reduced risk of CCA development. In a systematic review and meta-analysis including 9 studies (12,535 patients with CCA and 92,970,450 controls), aspirin use was associated with a 30% reduction of CCA risk (OR 0.69), particularly for iCCA (OR 0.33 versus 0.56 for eCCA)16. However, the association between low-dose aspirin use and CCA risk was not confirmed in a Swedish population-based cohort study17. In patients with advanced CCA, aspirin use was associated with improved overall survival18. The preventive effect of aspirin use for the development of CCA and to improve the outcomes in patients with PSC is now being evaluated in a prospective trial (Asp-PSC; ISRCTN12358813). Similarly, statin use was associated with a decreased risk of both iCCA (OR 0.69) and eCCA (OR 0.54)17. Two additional case–control studies from Taiwan19 and the UK20 confirmed this association and reported that the effect is independent of the type of statin used and is more pronounced among long-term users. Although the current results suggest a protective effect of aspirin and/or statins for CCA development in the general population, none of them should be currently recommended due to the lack of strong evidence, and further research is required.
Clinical presentation
-
Statements and recommendations: 2.1–2.3
Diagnosis
-
Research priorities: II
The clinical presentation of CCA is highly diverse and varies depending on aetiology, anatomical location and pathological characteristics21,22. Frequently, individuals with CCA remain asymptomatic until advanced stages. At the time of diagnosis, a significant proportion of patients have either locally advanced disease or metastatic disease, rendering curative-intent surgery not feasible21. Patients with CCA may experience non-specific symptoms such as asthenia, weight loss, abdominal pain and fatigue. A national survey conducted in Italy highlighted that jaundice is more prevalent in patients with pCCA and dCCA (74%) as a result of tumour-induced bile duct obstruction, whilst in patients with iCCA (29%), jaundice is mainly associated with advanced disease22. Elevated serum levels of ALT, GGT, ALP and carbohydrate antigen 19-9 (CA19-9) are frequently observed during diagnosis, particularly in patients with pCCA or dCCA. Nevertheless, CA19-9 exhibits low sensitivity for early-stage disease and is more commonly associated with advanced disease21. CCA predominantly occurs in older people, with the average age at diagnosis around 66 years21. However, CCA can also manifest in younger individuals, especially in those with underlying chronic liver and/or biliary diseases such as PSC21, with increasing incidence trends now being experienced in this specific population23.
Intrahepatic CCA
-
Statements and recommendations: 1.6
In 25% of cases, iCCA presents without any symptoms and diagnostic evaluation is initiated due to incidental findings in imaging procedures such as ultrasound22. In fact, radiological imaging has a pivotal role in establishing the diagnosis24,25,26. Suggestive radiological findings in computed tomography (CT) and magnetic resonance imaging (MRI), such as capsular retraction and a homogeneous mass with early rim-like enhancement and progressive contrast uptake, raise strong suspicions of iCCA24,25,26. However, distinguishing iCCA from HCC, particularly in patients with small tumours and underlying liver disease, remains challenging. Current international guidelines, such as those from EASL-ILCA (European Association for the Study of the Liver and the International Liver Cancer Association) and ESMO (European Society For Medical Oncology), recommend liver MRI with contrast as the primary diagnostic tool for iCCA, specifically in patients with potentially resectable disease27,28. In patients with liver cirrhosis, assessing washout in the portal phase can help to avoid misclassification as HCC27. Moreover, it has been recently suggested that, even in cases of radiological findings typical of HCC, a biopsy should be performed as false diagnosis of HCC can occur in more than 10% of cases29,30. In cases where patients do not have chronic liver diseases or when individuals with liver cirrhosis present with indeterminate liver nodules on MRI, a definitive diagnosis of iCCA should be established through the exclusion of extrahepatic cancer27. For systemic treatment, a tumour biopsy is required for all patients. In patients eligible for surgical resection, a biopsy is rarely required because of typical appearances on radiological imaging.
Histologically, iCCAs are mainly ductal adenocarcinomas, with two main histological subtypes, namely small duct and large duct iCCAs, according to the ICD-O-3.2 (5th edition of the WHO International Classification of Diseases for Oncology). Reporting the tumour subtype is recommended by EASL-ILCA as it can provide valuable information regarding prognosis and tumour molecular profiles27. Differentiation between iCCA and HCC is based on histological features, while in some cases, immunohistochemistry profiles help to establish a definite diagnosis. The distinction between iCCA and liver metastasis from a primary upper gastrointestinal, pancreatic or extrahepatic biliary source should not be made based only on morphology and immunohistochemical profiling but should also include clinical and radiological information27. Additionally, for differentiating iCCA from cancers of unknown primary, although more data are needed to recommend specific diagnostic modalities, upper and/or lower gastrointestinal endoscopy may be considered based on the patient’s symptomatology.
Perihilar and distal CCA
Diagnosing patients with pCCA and dCCA can be challenging due to the difficulty of obtaining sufficient tissue for cytology or histology. The combined use of magnetic resonance cholangiopancreatography (MRCP) and contrast-enhanced MRI may help to differentiate between malignant and benign bile duct strictures31,32. However, at least 10% of bile duct strictures initially diagnosed as malignant were subsequently found to be benign upon pathological examination of resected specimens22. Various approaches are available to establish cytological or histological diagnoses based on the location and extension of the bile duct stricture and the volume of the solid mass22,33. In patients requiring biliary drainage, bile duct brush specimens and intraductal biopsy samples should be obtained if feasible. However, the sensitivity of brush cytology for CCA diagnosis is less than 75% in most studies. Meta-analyses indicate that endoscopic ultrasound (EUS)-guided fine-needle aspiration or biopsy offers higher accuracy for cytological and histological diagnosis compared to an endoscopic retrograde cholangiopancreatography (ERCP)-based approach34,35,36,37, and single-operator cholangioscopy38 or peroral cholangioscopy39,40 may also be helpful for the identification of malignant strictures and to guide targeted intraductal biopsies, alone or in combination with other procedures.
In patients with PSC, the 20-year cumulative incidence of CCA is approximately 15%, with a median age at diagnosis typically between 40 and 50 years41,42. In these patients, CCA frequently develops within the first 2 years following a diagnosis of PSC41,43. Diagnosing CCA in patients with PSC can be challenging as they often display overlapping symptoms and imaging features of benign and malignant strictures43. It is recommended to monitor patients with PSC every 6–12 months using MRI/MRCP44,45. If a high-grade stricture and/or elevated serum CA19-9 levels are observed, ERCP with brush cytology and biopsy should be performed to establish the diagnosis. Testing for polysomy using fluorescent in situ hybridization (FISH) is used in some centres, specifically in potential candidates for liver transplantation44,45. A Swedish nationwide multicentre prospective study demonstrated that a surveillance strategy with yearly CA19-9 and MRI/MRCP followed by diagnostic ERCP in the event of severe or progressive bile duct changes fails to detect CCA early enough for long-term survival in an unselected cohort of patients with PSC46. According to a meta-analysis of 21 studies in patients with PSC, single-operator cholangioscopy plus biopsies is the most cost-effective strategy for CCA diagnosis in PSC47. Similar to patients with CCA without PSC, patients with PSC-CCA have a poor prognosis, with a median overall survival of 5–12 months in unresectable cases, making CCA the leading cause of PSC-associated mortality (24–58%)41,43.
Staging
According to the current consensus, the 8th edition of the American Joint Committee on Cancer (AJCC v.8) staging system should be employed for staging27. While the TNM system offers a clinically meaningful classification system that aligns with prognosis, it faces limitations in discriminating the outcome between T2 and T3 tumours. This is particularly pertinent in the context of iCCA staging, which warrants specific considerations.
Approximately 20% of patients with iCCA have multiple liver lesions but without extrahepatic metastasis (‘liver-only’) at diagnosis48,49. The current AJCC v.8 classifies iCCA with multiple liver lesions (liver metastases), in the absence of lymph nodes or extrahepatic spread, as early stage (stage II if T2N0M0 or stage III if T2N1M0). However, a study49 from the ENS-CCA Registry demonstrated that the presence of liver metastases significantly correlates with a worse prognosis (HR 2.53, 95% CI 1.18–5.42) compared to earlier stages. These results were validated internally and in the external Surveillance, Epidemiology, and End Results Program series50. Consequently, a modification of AJCC v.8 (mAJCC v.8) has been suggested, including “liver metastases: multiple liver lesions, with or without vascular invasion” as an “M1a stage”, and is waiting for revision and approval49.
A retrospective multicentre study, using current imaging modalities51 to assess the key staging tools for iCCA, revealed that MRI outperformed CT for the clinical staging of T1B, T2, and even T3 and T4 tumours. In line with recent systematic reviews and meta-analyses52, the latest guidelines27,28 mention the option of using 18F-fluorodeoxyglucose positron emission tomography (18FDG-PET) to assess lymph node status (sensitivity 88.4% and specificity 69.1%; AUC 0.852) and detect distant metastases (sensitivity 85.4% and specificity 89.7%; AUC 0.925), although its effect on the outcome is still unknown. This may be important, particularly when the identification of hidden disease sites could impact clinical decisions (for example, surgery or local therapies), as the performance of 18FDG-PET in these scenarios led to management changes in 15% of patients, with the majority (78%) due to disease upstaging. A meta-analysis52 also suggests using 18FDG-PET to confirm disease recurrence or when the diagnosis of relapse remains unclear following standard-of-care imaging (sensitivity 90.1% and specificity 83.5%; AUC 0.959).
The role of EUS in identifying lymph node metastases through fine-needle aspiration was explored in only a single retrospective study, but no prospective validation has been conducted so far53. Current guidelines recommend lymph node sampling by EUS-guided fine-needle aspiration (typically involving sampling three accessible lymph nodes) in patients who are considered for surgery. Patients with positive nodes are very rarely cured with surgery. They may still live longer, but this should be balanced with the risk of surgery that can vary from <1% to >20% depending on the extent of resection and the patient’s comorbidities27.
In a comprehensive study on the natural course of CCA using data from the ENS-CCA Registry Database21, elevated serum levels of CA19-9 and CEA at the time of diagnosis were associated with an increased risk of presenting with disseminated disease, locally advanced disease with lymph node invasion and particularly metastatic disease. It is worth noting that the elevation of both biomarkers was linked to a higher risk of presenting with metastatic disease when compared to the elevation of either of these two biomarkers individually and these may therefore be considered biomarkers of disease dissemination.
Genetics
-
Statements and recommendations: 3.1–3.7
Multiple sequencing efforts have been made in the past decade to decode the genetic landscape of CCA, underscoring the broad genetic heterogeneity of biliary tumours and the involvement of distinct oncogenic pathways1,2. Genetic profiles of CCA vary depending on the malignancy’s anatomical location; therefore, identifying specific mutational profiles can aid in accurately classifying tumours into their anatomical subtypes and are often associated with underlying risk factors54. In particular, iCCA shows high rates of targetable genomic alterations, including IDH1/2 mutations (13–20%) and FGFR2 fusion/rearrangements (11–14%) (Fig. 1), which are almost exclusively found in this CCA subtype and often co-occur with alterations in epigenetic modifiers (ARID1A and BAP1)55. Of note, the prevalence of these mutations may differ according to iCCA histological subtypes (Supplementary Table 3). Considering the most recurrent driver mutations in iCCA, a recent meta-analysis including 1,481 tumours revealed three genetic clusters with distinct histopathological features and prognosis54. iCCA tumours with IDH1 mutations or FGFR2 fusions, both co-associated with BAP1 mutations, display a small bile duct phenotype, decreased CA19-9 levels, and better prognosis when compared with tumours with KRAS, TP53, and SMAD4 mutations or with tumours without any of these alterations. Although not frequent, iCCA tumours with high microsatellite instability (MSI-high; 1.3%) and high tumour mutational burden (TMB-high; 3.7%) were shown to be enriched in genetic mutations affecting the WNT pathway55. Mutations in KRAS (predominantly KRASG12D/G12V variants, with a prevalence of 56%), one of the most common oncogenic drivers in biliary cancers, are more frequently identified in eCCA (28.6% in pCCA and 36.1% in dCCA versus 11.8% in iCCA)55,56. These KRAS mutations have been causally linked to the pro-tumour inflammatory milieu, affecting alternative mRNA splicing of specific genes (that is, IL1RN) and altering cell recruitment in the myeloid compartment57,58. Similarly, alterations in ERBB2/3 are more frequently detected in eCCA tumours (15%) compared to iCCA tumours (2–7%). TP53 mutations are often equally detected in iCCA (27–40%)55 and eCCA tumours (40% for pCCA and for dCCA)56.
Distinct oncogenic drivers are associated with different co-mutational patterns55, indicating driver-defined mutational landscapes and supporting the presence of distinct regulatory networks promoting cholangiocarcinogenesis59. For instance, higher-than-expected rates of BAP1 alterations are found in patients harbouring FGFR2 fusions (31% versus 12% in the non-FGFR2-rearranged population), or the predominance of TP53 alterations is observed in ERBB2-amplified iCCA as compared to BRAFV600E-mutant iCCA (75% versus 19%)55. Sex-associated differences in the frequency of common oncogenic drivers exist, with a prevalence of FGFR2 alterations and IDH1 mutations in female patients with iCCA, while KRAS mutations are enriched in male patients with iCCA55. Moreover, an analysis of genome-wide data suggests an association of ethnicity with the mutational landscape, showing a high-to-intermediate TMB and prevalent KRAS mutations in Asian populations compared to low TMB and mutant IDH as well as FGFR2 alterations in white populations60. Nevertheless, other reports have shown KRAS mutation prevalences in Asian populations61,62 comparable to those observed in Western countries. Future studies including larger patient cohorts are warranted. Additionally, different mutational landscapes were also described according to the underlying aetiology of the tumour63. Clinical-grade next-generation sequencing (NGS), such as targeted panel sequencing, is crucial for guiding patient care and treatment decisions64. It is important to note that NGS assays can profile both DNA and RNA alterations to capture the most therapeutically relevant genetic alterations that may guide clinical management65. Future studies should explore the role of NGS in liquid biopsies for monitoring post-progression and detecting minimal residual disease after surgery.
Higher intratumour molecular heterogeneity is a pivotal feature of carcinogenesis that was previously associated with poor prognosis and lack of response to therapies. Phylogenetic analysis demonstrates strong evidence of functionality-based tumour branching evolution, linking functional diversity and genetic heterogeneity. Interestingly, patients with iCCA generally exhibit high clonality, and specifically SPP1, whose stromal expression was identified as an independent predictor of poor prognosis in iCCA66, is closely associated with tumour cell evolution and microenvironmental reprogramming. CCA subtyping has benefited from single-cell RNA sequencing, allowing, for instance, the definition of perihilar large duct-type and peripheral small duct-type iCCA and their associated biomarkers, S100P and SPP1, respectively67. Additionally, gene expression analyses at spatial and single-cell resolution contribute to our understanding of cholangiocarcinogenesis by highlighting the functional heterogeneity and the single-cell transcriptomic architecture of CCA tumours. In fact, multiregional single-cell analysis has revealed that variation in the tumour cell composition and intercellular crosstalk within the tumour ecosystem are smaller within each tumour than between tumours from different patients, regardless of tumour size68. Notably, a single-cell atlas of tumour cell evolution in response to therapy has been reported in HCC and iCCA69,70 and, for instance, patients with tumours exhibiting a higher cluster number (and therefore, higher transcriptomic diversity) have shorter survival compared to those with a lower cluster number.
Beyond the genome, epigenetic alterations have also been reported in CCA71. A genome-wide analysis of aberrant promoter methylation in human CCA tumours identified hypermethylation of genes involved in Wnt, TGFβ and PI3K signalling pathways, including WNT3A, DKK2, SFRP1, SFRP2, SFRP4 and SOX17 (ref. 72). Notably, SOX17 expression was found downregulated in human CCA tumours, correlating negatively with promotor hypermethylation and associated with poorer prognosis due to its tumour suppressor role in CCA73. Additionally, a large multiomics analysis (that is, whole-genome and targeted/exome sequencing, copy number, gene expression and DNA methylation) of 489 patients with CCA from 10 countries identified specific patient subsets (clusters 1 and 4) characterized by distinct DNA hypermethylation patterns (CpG islands or shores), independent of liver fluke infection74. In a subset of liver fluke-positive CCAs (cluster 1), global hypermethylation was linked to ARID1A and BRCA1/2 mutations and increased gene promoter alterations related to histone 3 lysine 27 trimethylation, predicted to affect transcription factor binding and, consequently, gene expression74. Furthermore, recent integrative multiomics enhancer activity profiling revealed that a subgroup of patients, mostly with fluke-positive CCAs (many of cluster 1 tumours), exhibited increased oestrogen signalling activation and greater sensitivity to mTOR inhibitors. Two other subgroups were characterized by increased oxidative phosphorylation (cluster 4) or activation of immune-related pathways, respectively75. Additionally, dysregulation of small non-coding RNAs has also been implicated in cholangiocarcinogenesis71. A recent microRNAs (miRNA) sequencing study of 119 resected CCAs, 63 surrounding liver tissues, and 22 normal livers identified miR-27a-3p as a key mediator of cholangiocarcinogenesis through targeting of the FoxO signalling pathway76.
Tumour microenvironment and immunobiology
-
Statements and recommendations: 4.1
-
Research priorities: III–IV
The CCA tumour microenvironment (TME) exhibits a highly desmoplastic stroma and a complex tumour immune microenvironment (TIME). Recent integrative genomics, molecular and immune characterizations of the TIME have revealed unique patient classifications into immune desert, immune activated, myeloid enriched and mesenchymal-like subsets56,77,78,79, directly related to inflamed or non-inflamed profiles80,81 and with prognostic implications, underscoring the heterogeneity of the CCA TME.
Cancer-associated fibroblasts
Cancer-associated fibroblasts (CAFs) stand out as one of the most abundant cell types in the CCA TME, and they have a role in cholangiocarcinogenesis82,83. Traditionally defined as αSMA-positive fibroblasts, their presence has been described to correlate with a poor prognosis1,84. With the advent of single-cell RNA sequencing technologies, CAFs have recently emerged as multifaceted entities78,85,86. Originating mainly from hepatic stellate cells in both human and mouse models82,85, CAFs have an overall tumour-promoting role. Transcriptomically distinct CAF subtypes have been identified in iCCA tumours, categorizing them mainly into myofibroblastic CAFs enriched in extracellular matrix (ECM)-related genes78,85,86, and vascular or inflammatory CAFs enriched in cytokine and growth factor transcripts (such as hepatocyte growth factor (HGF), vascular endothelial growth factor (VEGF), HB-EGF, IL-6 and CXCL12), with CD146+ vascular CAFs being the most abundant subtype described in human iCCA86. Within the TME of CCA, CAFs interact with tumour, endothelial and immune cells85,86, thereby influencing various biological processes, including tumour growth, metabolism, stiffness, lymphangiogenesis, chemoresistance, immunosuppression, and metastasis83,84,87. Indeed, the crosstalk between CD146+ vascular CAFs and tumour cells, which can enhance iCCA malignancy through the IL-6–IL-6R axis, has been demonstrated86. Given their overall tumour-promoting role85,88 and potential correlation with survival1,83, targeting CAFs in combination with chemotherapy or immunotherapy may emerge as a promising therapeutic approach in CCA82.
Role of the ECM
The ECM in CCA undergoes extensive remodelling, involving both the degradation of native components within the TME and the deposition of newly synthesised proteins (for example, periostin, tenascin C, osteopontin)89. Overexpression of these proteins correlates with increased tumour size and lymph node metastasis90,91,92. Periostin, through interaction with integrin α5 expressed by CCA cells, activates PI3K–AKT signalling, sustaining tumour cell proliferation and invasion93,94. Similarly, the secretion of periostin by iCCA cells recruits tumour-associated macrophages (TAMs)95. Tenascin C, highly expressed in embryonic tissues, has effects on tumour cells, stimulating cell proliferation by activating Wnt–MAPK signalling, and on fibroblasts, promoting the secretion of angiogenic factors96. Osteopontin, acting both as a cytokine and matricellular protein, decreases TAM cytotoxicity against neoplastic cells and interacts with integrins to activate Wnt–β-catenin, sustaining iCCA growth and metastasis97. Excess collagen I and III and perturbed assembly, resulting from cross-linking and tight packaging due to MMP and LOX enzyme activities in the ECM, induce increased stiffness in CCA98. This, in turn, stimulates the activity of intracellular mechanosensors (YAP and TAZ), which are relevant for tumour initiation and progression99 (Fig. 4).
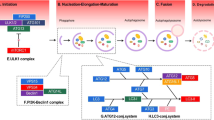
Cholangiocarcinoma (CCA) development and progression involve a series of intricate and varied processes that are dependent on the interaction of extracellular ligands present in the tumour microenvironment such as pro-inflammatory cytokines, growth factors and bile acids. This interaction, along with the upregulation or abnormal activation of cell-surface receptors, leads to the disruption of intracellular signalling pathways, resulting in increased cell proliferation, survival, and migration or invasion. Key genes that are commonly mutated or amplified in CCA tumours, leading to the overactivation of these pathways, include KRAS, BRAF, ARID1, BAP1, IDH1/2 and TP53, and are highlighted in the figure. The activation of these signalling pathways can also be triggered by the interaction between the malignant epithelial cells and the immune cells and/or reactive stroma within the tumour. Cancer-associated fibroblasts (CAFs) are recruited and continuously activated by CCA malignant epithelial cells in response to signals such as PDGFD, FGF and TGFβ1, which are also released by tumour-associated macrophages (TAMs). In turn, CAFs promote the proliferation and invasiveness of CCA cells, either directly or by modulating the behaviour of other cells within the tumour microenvironment. Vascular CAFs contribute to tumour-associated lymphangiogenesis by stimulating lymphatic endothelial cells. Inflammatory CAFs support the M2 polarization of TAMs, and activate regulatory T (Treg) cells, while suppressing the activity of CD8+ T cells, natural killer (NK) cells, and dendritic cells. Additionally, myofibroblastic CAFs induce extensive remodelling of the extracellular matrix, which becomes stiffer and influences the mechanotransduction of CCA cells, leading to the activation of intracellular pathways such as YAP–TAZ. Current targeted therapies against specific upregulated and/or overactivated pathways that are being used for the treatment of CCA are highlighted in light purple boxes. αKG, α-ketoglutarate; 2-HG, 2-hydroxyglutarate; FAO, fatty acid oxidation; GPCRs, G-protein-coupled receptors; M-CSF, macrophage colony-stimulating factor; MDSCs, myeloid-derived suppressor cells; PDGFD, platelet-derived growth factor D; RTKs, receptor tyrosine kinases; TANs, tumour-associated neutrophils; TCA, tricarboxylic acid; VEGFR, vascular endothelial growth factor receptor.
Immunobiology
The TIME of CCA harbours innate and adaptive immune cells, having a dual role in both promoting and inhibiting tumorigenesis100,101. Innate immune cells in the TIME include TAMs, tumour-associated neutrophils, myeloid-derived suppressor cells (MDSCs), dendritic cells and natural killer cells, among others100. TAMs, being heterogeneous and plastic immune cells, are recruited and activated in the CCA TIME, correlating with poor prognosis. However, the role of TAMs should be carefully evaluated based on tumour staging and anatomical location102. An elevated neutrophil-to-lymphocyte ratio before treatment has been associated with a worse prognosis in CCA103, though there is limited evidence supporting a tumour-promoting role for tumour-associated neutrophils104. The presence of MDSCs, with immunosuppressive effects facilitating tumour growth in mouse models of CCA105,106, is supported by increased circulating monocytic-MDSCs in patients107. Myeloid cells also have a major role in driving poor prognosis in advanced iCCA under chemotherapy58. Dendritic cells have a key role in activating the adaptive immune response, and an increased number of classical dendritic cells has been linked to better outcomes108,109,110. Dendritic cell-based immunotherapies show therapeutic potential by enhancing antitumour T cell responses100,104. Natural killer cells, inhibiting tumour growth in preclinical CCA mouse models111 and described to be altered in natural killer immunoglobulin-like receptors and HLA gene loci in patients112, hold promise in CCA cell-based therapy.
Although the role of the adaptive immune system in CCA remains largely unexplored, tumour progression seems to be associated with a reduction of tumour-infiltrating lymphocytes113, and patients with higher infiltration of CD8+ and CD4+ T cells show a better overall survival113,114,115. In addition, a decreased number of cytotoxic immune cells and extensive tumour infiltration of FOXP3+ CD4+ regulatory T cells overexpressing co-inhibitory receptors, such as PD1 and CTLA4, in human CCA tumours indicate an immunosuppressive TIME86,116,117,118. Accordingly, deconvolution analysis of transcriptomic data from ~900 iCCAs provided a novel classification system of these tumours considering the TIME78. Inflamed tumours (‘immune hot’; constituting 35% of iCCAs) are characterized by increased infiltration of exhausted CD8+ T cells and extensive activated inflammatory stroma, and are associated with KRAS (inflammatory stroma) and TP53 (immune classical) mutations. On the other hand, non-inflamed tumours (‘immune cold’; 65% of iCCAs) are enriched in immunosuppressive M2-like macrophages and hyperactivated CD4+ regulatory T cells that induce a suppressive local immune milieu119, which is associated with FGFR2 fusions, mutations in IDH1/2 and BAP1 and co-occurrence of KRAS/TP53 mutations. Although the overall positivity of PD1 and PDL1 is low in both classes, inflamed tumours present higher levels, and may therefore benefit the most from anti-PD1 and anti-PDL1 therapies78. Finally, while B cells represent a minor proportion of tumour-infiltrating lymphocytes in CCA, their presence has been associated with a better prognosis113,120,121. In conclusion, the TIME in CCA is poorly immunogenic, heterogeneous and varies among subtypes. Additionally, given the high global prevalence of MASLD, an increase in MASLD-associated CCA is alarmingly expected, making the study of specific alterations in TIME critically important. Although no information is yet available for CCA, patients with advanced metabolic dysfunction-associated steatohepatitis-associated HCC have shown reduced responsiveness to immunotherapy and worsened overall survival compared to patients with other aetiologies. This is likely due to metabolic dysfunction-associated steatohepatitis-related aberrant T cell activation, which causes tissue damage and impairs immune surveillance122. These findings highlight the urgent need for detailed investigation of the TIME in MASLD-associated CCA. Overall, strategies aiming to modulate the CCA TIME must shift the balance from immunosuppressive factors in favour of cytotoxic elements to enhance the antitumour response.
Signalling and molecular networks
-
Research priorities: V
CCA tumours may arise in the context of prolonged biliary or liver injury driven by a broad range of aetiologies1. Chronic disease and injury stimulate a complex regenerative response in the liver. This environment comprises cellular and acellular components rich in toxic bile acids, pro-inflammatory cytokines, growth factors, and mitogenic signals2, all of which have been shown to promote the proliferation of both non-transformed and malignant cholangiocytes. It is widely considered that the regenerative microenvironment directly contributes to the formation of the cancerous stroma, providing pro-proliferative signals. These signals enable the further accumulation of mutations, uncontrolled growth and evolution, resistance to apoptosis, and impact on immune detection. Concurrently, they promote angiogenesis, tumour invasion and metastasis (Fig. 4).
Signalling within the cancer cells
Numerous molecular pathways affecting malignant cholangiocytes have been identified, leading to the sub-classification of CCA into ‘inflammatory’ or ‘proliferative’ classes characterized by the induction of pro-inflammatory signals and the activation of oncogenic cascades1,80,123. Inflammatory mediators, such as IL-6–STAT3, TNF and COX2, induced by biliary stasis and chronic inflammation, promote the formation of nitric oxide and oxidative species. These factors result in DNA damage and mutations, genomic instability, inhibition of DNA repair, and enhanced cell survival124,125,126, thus priming biliary cells for carcinogenesis1,80,123,127,128.
Among the critical oncogenic cascades activated in malignant cells are the PI3K–AKT–mTOR, RAS–RAF–MEK–ERK, HER2, EGFR, FGFR, JAK–STAT, Notch, YAP–TAZ–Hippo, TGFβ, pRb–CDK4–CDK6, Hedgehog, and WNT–β-catenin pathways1,80,123,127,128,129,130,131,132,133. These signalling modules are involved in complex, interrelated circuits that often reinforce each other and are responsible for various cellular processes underlying tumour development and progression. These processes include proliferation, survival, invasion, metastasis, lineage commitment, metabolism and chemoresistance1,80,123,127,128,129,130,131,132,133. Among these pathways, the Notch cascade, which is physiologically implicated in the differentiation of bipotent hepatoblasts into cholangiocytes, and therefore in biliary development, stands out as a critical player in lineage commitment towards the malignant cholangiocellular phenotype134,135. Indeed, overexpression of Notch pathway members in the mouse liver, alone or in combination, drives CCA development136,137,138,139. Conversely, inhibition of this cascade delayed tumorigenesis and suppressed the cholangiocellular phenotype induced by the Hippo pathway effectors YAP and TAZ140,141.
The relevance of these pathways is further underscored by accumulating evidence in experimental models and clinical practice, where FGFR2 inhibitors are being used for the personalized treatment of patients with FGFR2 fusions142. FGFR translocations, commonly observed in intrahepatic disease, activate various CCA-associated signals, including PI3K–AKT143,144, MEK–ERK145 and PKC146. While clinical stratification of patients based on FGFR ‘status’ is an inclusion criterion for treatment, patients with iCCA lacking an FGFR rearrangement could still be susceptible to inhibitors of downstream signalling, regardless of their FGFR status. Indeed, lessons from other cancers indicate that targeting FGFR rearrangements and the MEK pathway improve patient outcomes147. The signalling processes that drive pCCA and dCCA are distinct from intrahepatic tumours, with a greater reliance on RAS or EGFR signalling. Enhanced signalling through RAS–AKT–NOTCH may directly cause eCCA148,149, suggesting that pharmacological inhibition of PI3K–AKT and Notch signalling has the potential to limit extrahepatic tumour growth. While there are common signalling features across CCAs from diverse anatomical subtypes, there is selectivity in signalling across these sites. For example, eCCA cells are sensitive to IL-33-dependent proliferation, whereas intrahepatic cells are not150. Consequently, it will be necessary to define the commonalities and differences between CCA at different anatomical sites150.
Post-translational modifications, which are reversible chemical changes occurring after protein biosynthesis that affect their function and localization, mediate CCA development and progression. High levels of NEDDylation pathway enzymes, and consequently NEDDylated proteins, have been observed in both human premalignant (biliary intra-epithelial neoplasia) and malignant tissues, correlating with poor prognosis151,152. Genetic or pharmacological inhibition of this process substantially reduces the survival and growth of CCA in vitro and halts tumorigenesis in experimental mouse models151,152,153. Similar observations were recently reported for protein sumoylation, with the selective targeting of this process, either genetically or pharmacologically, not only impacting CCA cells but also disturbing the TME154. Additionally, increased protein fucosylation in human iCCA tissues contributes to tumour cell proliferation and migration through activation of the NOTCH, EGFR–NF-κB and EGFR–AKT–ERK pathways155,156. Furthermore, increased terminal fucosylation has been associated with cholangiocarcinogenesis in a hamster model of CCA155. Elevated O-GlcNAcylation levels in human CCA tissues were also correlated with an unfavourable prognosis157. Indeed, augmented O-GlcNAcylation promotes vimentin stabilization and enhances the nuclear translocation of proteins that induce the expression of downstream genes involved in epithelial–mesenchymal transition and in CCA cell migration and invasion158,159. Notably, high glucose levels boost the aggressiveness of metastatic CCA cells through O-GlcNAcylation160. On the other hand, a decrease in histone H3K9me3 modifications, with concomitant overexpression of H3K9me3-modulated genes from the WNT and PI3K–AKT pathways, promotes iCCA growth161. Furthermore, post-translational modifications can modulate cancer-stromal cell interactions151 and affect microenvironment components156 within CCA, thus supporting tumour progression.
Extrinsic signals that support tumour growth
The desmoplasia and hypovascularity of CCA tumours, often defined as the tumour-responsive stroma, promote tumour growth, dissemination and immune evasion with the help of various extrinsic factors101,162,163,164. CAFs are recruited by platelet-derived growth factor D (PDGFD) secretion from CCA cells and are a key cell type within the tumour microenvironment that supports tumour survival in multiple ways165. VEGFA and VEGFC secretion by CAFs promotes tumour lymphangiogenesis, and PDGFB secretion has been suggested to protect tumour cells from TNF-mediated apoptosis87,166. CD146+ vascular CAFs express a strong microvascular signature and induce epigenetic changes in CCA cells through IL-6 secretion, leading to the upregulation of enhancer of zeste homologue 2, which increases malignancy86. Recently, distinct subpopulations of CAFs have been identified that specifically induce tumour growth by different mechanisms: inflammatory and growth factor-enriched CAFs via HGF (interacting with tumour-expressed MET) and myofibroblastic CAFs via hyaluronan synthase 2 (ref. 85). CAFs are also involved in tumour immune evasion through interactions with MDSCs, which exert immunosuppressive functions and affect T cell proliferation, and with dendritic cells, leading to the downregulation of their HLA molecules167,168,169.
Another cell type of interest within the tumour-responsive stroma are the TAMs. They provide PDL1 in murine and human CCA, thereby facilitating tumour progression via T cell suppression, a process mediated by tumour-derived exosomal miR-183-5p through the miR-183-5p–PTEN–AKT–PDL1 pathway170. Interestingly, selective blockade of TAMs results in the compensatory emergence of an ApoE-expressing subset of granulocyte-MDSCs that impairs T cell responses, suggesting the need for dual blockade of TAMs and granulocyte-MDSCs to improve anti-PD1 therapy in CCA105. In addition, TAMs directly affect CCA growth via Wnt signalling and neovascularization through the secretion of VEGFs and angiopoietins131,171.
Several mechanisms of programmed cell death are involved in the development and progression of CCA172. The Fas–FasL pathway is activated in CCA cells (Fas downregulated, FasL upregulated), leading to cell survival and immune evasion by inducing apoptosis in T cells and natural killer cells173,174. In addition, the apoptosis inhibitor cFLIP is upregulated in CCA cells, protecting them from apoptotic signals secreted by inflammatory cells174. Moreover, it has been shown that receptor-interacting serine–threonine-protein kinase 3 — a key mediator of necroptosis — is much more highly expressed in cholangiocytes than in hepatocytes, which is associated with a switch from sublethal necrosome activation in hepatocytes to lethal necroptosis in cholangiocytes175. It is of particular interest that different forms of programmed cell death may control lineage commitment during early liver cancer development. In particular, a necroptosis-dominated microenvironment promotes CCA development, whereas apoptosis is more closely associated with HCC176. Accordingly, the necroptotic environment is created by inflammatory cytokines (such as C-C motif ligand 6) released by damage-associated molecular pattern-activated immune cells176.
Tumour metabolism and stemness
-
Statements and recommendations: 4.2–4.3
-
Research priorities: VI
To meet the anabolic demands, malignant cells and cellular components of the microenvironment tend to rewire their metabolic pathways. Although different types of malignant cells share this phenomenon, these metabolic patterns display significant intracellular variability. In CCA, there is a high metabolic heterogeneity, influenced by factors such as anatomical location, driver oncogenes, epigenetic modifications and microenvironmental adaptations. This metabolic rewiring represents potential therapeutic target options. Therefore, understanding the specific pathways involved in different CCA subtypes is crucial for developing effective therapies. Evidence indicates an increased nutrient uptake in CCA, similar to other cancer types. Glucose demand177,178 and glutamine utilization179 are elevated, supporting active glycolysis and fuelling the tricarboxylic acid cycle intermediates, respectively. In terms of lipids, crucial for energy and new cell membrane synthesis in proliferating cells, highly proliferative CCA cells show heightened uptake of free fatty acids and lipoproteins180. These cells also exhibit increased levels of proteins involved in the uptake (FABP5, CD36) and intracellular metabolism (ACSL5, ACADM) of lipids. In addition, the accumulation of fatty acids into triglycerides and consequent increased consumption fuel the highly proliferative CCAs and, importantly, the inhibition of mitochondrial fatty acid oxidation with etomoxir in different experimental CCA models suppresses tumour growth180.
The metabolic reprogramming in CCA extends to cancer stem cells (CSCs), a cell type with embryonic characteristics and higher resistance to chemotherapeutic agents than that of the tumour mass181,182. The metabolic regulation of stemness is crucial in controlling stem cell fate183,184. CCA stemness maintenance was shown to depend on mitochondrial oxidative phosphorylation metabolism rather than on glycolytic pathway activity183. Related, the mitochondrion’s importance in regulating CCA stemness and activating epithelial–mesenchymal transition has previously been shown185, with BEX2 protein being crucial in the maintenance of dormant CSCs by suppressing mitochondrial activity186. Furthermore, insulin receptor substrate 1 promotes the transcription of tumour stem cell marker SALL4, implicating it in CCA stemness properties, and its increased levels in human CCA tumours are associated with worse survival187. A reduced redox state is essential for CSC self-renewal and, therefore, CCA CSCs were shown to rely on powerful antioxidant networks to detoxify mitochondrial reactive oxygen species and maintain stemness in oxidative CSCs. In addition, glutathione metabolism is critical for this process. Indeed, CD44+ iCCA cells resistant to chemotherapy display reduced reactive oxygen species levels through enhanced glutathione synthesis188. The CSC niche also influences CCA stem-like metabolism. CAFs activate 5-lipoxygenase metabolism in CD33+ MDSCs, promoting CCA stemness and chemotherapy refractoriness by producing leukotriene B4 and its receptor BLT2 activation189. Overall, based on this body of evidence, developing compounds that target key players of mitochondrial pathways may be promising in CCA.
Interestingly, metabolic alterations associated with CCA generation, progression and recurrence may be reflected in serum, suggesting the potential of metabolomics as a tool for predicting disease course and improving diagnosis. In line with this, metabolomics could be a useful tool to perform an accurate diagnosis of CCA190,191 and even to predict CCA progression or recurrence192. In fact, patients with CCA refractory to cisplatin plus gemcitabine (CisGem) exhibit distinct metabolic profiles, including higher tissue αD-glucose levels and lower amino acid content, correlating with increased expression of CSC markers193.
Biomarkers
-
Statements and recommendations: 5.1–5.5
-
Research priorities: VII–X
The identification of accurate non-invasive biomarkers for the diagnosis of CCA is still a pending challenge. CA19-9 and/or CEA are currently the only circulating non-invasive biomarkers used in the clinical setting; however, they are not recommended for surveillance and early detection due to their low sensitivity and specificity. An important limitation of CA19-9 in diagnosis is its absence in about 10% of individuals who lack the Lewis antigen due to an inactivating mutation in FUT3 (ref. 194). Additionally, elevated levels of these proteins may also be detected in individuals with biliary obstruction or other benign biliary diseases, potentially leading to a considerable rate of false positive results33,195,196,197. Rather than being used as a diagnostic biomarker, elevated CA19-9 levels may be useful in predicting prognosis: increased serum CA19-9 levels are associated with an increased risk of metastatic disease and are an independent prognostic biomarker, along with disease stage and the ECOG performance status21. In addition, a decline in CA19-9 levels during chemotherapy may be indicative of radiological response to treatment in patients with inoperable CCA198. Overall, considering the lack of accurate non-invasive biomarkers for CCA, numerous studies have looked for alternative circulating biomarkers for diagnosis and prognosis33.
Circulating non-coding RNAs have emerged as valuable tools for cancer diagnosis. Several single-centre pilot studies have identified some miRNAs and circular RNAs in biological fluids (that is, serum, plasma, urine and bile) with diagnostic and/or prognostic potential for CCA199,200,201,202,203,204,205,206,207,208,209,210,211,212. More recently, a Danish study involving 218 patients with biliary tract cancer conducted whole-blood miRNA profiling and identified a novel miRNA signature (comprising four circulating miRNAs: let-7a-3p, miR-92b-5p, miR-145-3p, miR-582-3p). Combined with CA19-9 in an index, this miRNA signature showed an AUC of 0.93 in diagnostic performance across discovery and validation cohorts. Notably, this miRNA index was shown to be upregulated in human CCA tissue and within specific cell types, potentially influencing distinct pathobiological and immune features213.
Cell-free DNA (cfDNA) analysis may also prove valuable for cancer genomic profiling, assisting in both diagnosis and therapeutic decision-making. Blood cfDNA profiling in patients with biliary tract cancer revealed genetic alterations in 84% of cases, with high concordance rates between cfDNA and tumour tissue mutations for specific genes, such as IDH1 or BRAF, though with significantly lower sensitivity for FGFR2 fusions, albeit depending on the respective assays employed214. Mutational analysis of bile cfDNA with suspicious biliary strictures may help to establish the differential diagnosis of benign and malignant biliary strictures215. Additionally, cfDNA methylation changes have been explored as diagnostic tools for malignant biliary strictures. A panel of three genes (TWIST1, HOXA1, VSTM2B) provided 73% sensitivity and 93% specificity for the identification of these strictures, being superior to cytology and FISH analysis216.
Extracellular vesicles (membrane-bound spheres measuring between 40 and 1,000 nm) contain distinct biomolecules and have become a useful tool in the search for disease biomarkers217. In an initial discovery study, serum extracellular vesicles were shown to contain distinct protein biomarkers useful for the early, sensitive and specific diagnosis of PSC, CCA and HCC218. Similarly, both serum and urine extracellular vesicles harboured RNA candidate biomarkers that allowed for CCA diagnosis by reflecting tumour tissue expression, representing promising liquid biopsy biomarkers219. More recently, serum extracellular vesicle proteins were shown to enable the prediction, early diagnosis and prognosis estimation of patients with PSC-associated CCA220. While some newly identified biomarkers are specific to PSC-associated CCA, most serum extracellular vesicle proteins showed strong diagnostic performance in identifying CCA, regardless of disease aetiology, and are thus termed pan-CCA biomarkers. Notably, these pan-CCA biomarkers can be detected using total serum. For instance, a combination of three serum proteins (CRP, fibrinogen and FRIL) enabled the specific diagnosis of CCA in patients with PSC, particularly those with early-stage disease, and, when combined with CA19-9, outperformed the diagnostic capacity of CA19-9 alone. Moreover, individual CRP, fibrinogen, FRIL or PIGR levels may identify patients with PSC who developed CCA during follow-up, well before clinical evidence of malignancy. Besides diagnostic biomarkers, independent prognostic biomarker panels have also been proposed220. In line with this, IL-6 has been identified as an independent prognostic biomarker, and anti-IL-6R (tocilizumab) has been suggested as a potential treatment option to enhance chemotherapeutic response221.
Altered metabolism is a hallmark of cancer cells, including CCA cells222,223, leading to significant changes in metabolites due to disruptions in cellular energetics, oxidative stress and inflammatory processes. Consequently, changes in circulating metabolites (molecules smaller than 1.5 kDa) may be of diagnostic and prognostic interest. Several studies have reported altered metabolic profiles in various biological fluids from patients with CCA compared to healthy individuals and those with hepatobiliary pathologies. Elevated serum levels of 2-hydroxyglutarate have been detected in patients with iCCA carrying IDH1/2 mutations, correlating with tumour burden224. Alterations in total bile acid levels and the proportions of bile acid species or families in serum, bile and urine have also been observed190,191,225,226,227, although these findings require validation, as bile acid homeostasis is also disrupted in cases of cholestasis and other liver diseases228,229. Changes in serum lipid and amino acid levels have also been detected in patients with iCCA compared to those with other hepatobiliary diseases190, and metabolite panels have been identified that can distinguish patients with iCCA from patients with HCC, even at early stages, in both discovery and validation cohorts. Some of these metabolites may be particularly interesting for early detection in high-risk groups such as patients with PSC. Differences in serum metabolic profiles have also proven useful for differentiating malignant and benign lesions in the pancreatic head as well as for distinguishing dCCA from pancreatic ductal adenocarcinoma191. To date, no single metabolic biomarker has demonstrated absolute specificity for CCA, suggesting the need for a combination of clinical information and other biomarker analyses. This will likely require the implementation of artificial intelligence tools.
Clinical management
Biliary drainage
ERCP is pivotal in the treatment of patients with CCA, serving three key purposes: acquiring tissue for cytological or histological diagnosis; facilitating biliary drainage in selected patients; and providing a valuable palliative tool in cases deemed inoperable. In fact, ERCP proves beneficial for patients with pCCA or dCCA experiencing biliary tract obstruction. Before ERCP, patients should undergo adequate imaging to facilitate staging and procedural planning230. In patients with jaundice and suspected dCCA, ERCP-directed cytological brushings and biopsy, along with EUS-guided fine-needle aspiration or biopsy including local lymph nodes, are recommended230,231. Cytology brushings demonstrate a sensitivity of 45–55% and a specificity exceeding 95%232. Complementary techniques, such as FISH (to detect polysomy and/or trisomy/tetrasomy) or fluoroscopy-directed biopsies, enhance sensitivity by 80% while maintaining the same specificity233. Therefore, employing multiple techniques is advisable when sampling a suspected malignant biliary stricture231,233,234. In selected cases, peroral cholangioscopy aids in histological diagnosis through targeted tissue biopsies, with a sensitivity of 65% and a specificity of 98%38,234. For patients with jaundice and a suspected perihilar stricture due to pCCA, intraductal sampling via ERCP is preferred over EUS-guided fine-needle aspiration or biopsy or percutaneous biopsy of the primary lesion (perihilar stricture or mass) due to the risk of peritoneal seeding230,231.
For patients with a resectable dCCA, routine preoperative biliary drainage is not mandatory. However, it is recommended in patients with acute cholangitis, intractable pruritus, serum bilirubin levels exceeding 14.6 mg/dl and those facing surgery delays beyond 4 weeks230,231. ERCP is utilized to place a fully covered self-expandable metal stent for dCCA or one or more plastic stents for pCCA. In cases where a transpapillary stent cannot be placed, consideration for EUS-guided biliary drainage is warranted231. Percutaneous transhepatic cholangiography and drainage is reserved for patients in whom EUS biliary drainage is not technically feasible or available.
In patients with resectable pCCA experiencing biliary obstruction, the need for preoperative drainage should be critically discussed with the multidisciplinary team. If drainage is deemed necessary, assessing the affected lobe should precede the procedure230. Unilateral drainage in the remnant lobe is considered before surgery. Bilateral stenting is typically reserved for patients with type IV Bismuth–Corlette strictures, those unresponsive to unilateral stenting. Antibiotic administration is recommended when there is incomplete drainage of contrast from opacified bile ducts. For patients with a malignant perihilar stricture, placement of either plastic stents or uncovered self-expandable metallic stents is an option231. Patients with unresectable pCCA with biliary obstruction should be considered for palliative stenting through either ERCP or percutaneous transhepatic cholangiography and drainage.
Surgery
-
Research priorities: XI
Only a minority of patients with CCA qualify for surgical resection, primarily because the disease is typically metastatic or locally advanced (that is, unresectable) at initial presentation. The primary aim of surgical resection is to enhance life expectancy, as achieving a cure (that is, 10-year survival) is uncommon (approximately 10%) due to the high recurrence rate235,236. Surgical procedures for iCCA and pCCA typically involve major liver resection, with postoperative mortality reaching up to 10% even in experienced centres237,238. Shared decision-making entails balancing this short-term mortality risk with the potential long-term survival benefit239. Staging laparoscopy is recommended for all patients with CCA due to the high risk of occult peritoneal and liver metastases240,241,242,243. For iCCA and pCCA staging, a minimum of six regional lymph nodes should be harvested, though it remains uncertain whether an adequate lymphadenectomy improves survival244,245. iCCA tumours are typically large, necessitating major liver resection. Around 15% of patients may also require resection of the extrahepatic bile duct due to invasion of the liver hilum. A biopsy is rarely necessary before surgery, as the diagnosis is typically made based on the characteristic radiological enhancement pattern. Patients with multifocal disease (especially if more than three lesions) or nodal disease (especially if extraregional) are prone to early recurrence post-surgery, suggesting a limited benefit from the procedure246,247,248.
The primary surgical challenge in pCCA lies in its ductal spread and early involvement of the hepatic artery and portal vein, both adjacent to the biliary confluence. Although pCCA tumours are small at presentation, complete margin-negative resection of the biliary confluence and affected liver sections requires major liver resection. Post-resection reconstruction involves a hepatico-jejunostomy to the liver remnant’s bile ducts. Fewer than 10% of patients with pCCA have a tumour below the confluence of the left and right duct (that is, Bismuth I), allowing for complete resection without partial hepatectomy249,250. Bilateral involvement of the second-order bile ducts was previously considered a contraindication for resection, but recent studies have found acceptable postoperative outcomes251. Vascular reconstruction is increasingly performed, although involvement exceeding 180 degrees of the main portal vein or proper hepatic artery is associated with poor survival252.
For both iCCA and pCCA, postoperative mortality is largely attributed to liver failure. The risk of liver failure is particularly high in patients with a liver remnant of less than 40% or pre-existing liver disease (for example, PSC). Other risk factors include preoperative cholangitis and advanced age. European consensus guidelines recommend volumetrics in preoperative liver assessment253. Functional MRI and liver scintigraphy have the theoretical benefit of measuring the function of the liver remnant. If the liver remnant volume is less than 40% in patients with pCCA, portal vein embolization of the resected liver can be performed to induce hypertrophy of the future liver remnant254. Hepatic vein embolization might be considered as an additional technique if volume remains inadequate after portal vein embolization255.
The diagnostic work-up and surgical resection of dCCA are similar to tumours of the pancreatic head. dCCA can rarely be managed solely with extrahepatic bile duct resection, and only in cases confined to the extrahepatic bile duct. However, in most cases, patients require a pancreatoduodenectomy.
Liver transplantation
-
Statements and recommendations: 6.1
The consideration for liver transplantation in CCA involves three distinct populations without extrahepatic disease and lymph node metastases: unresectable locally advanced pCCA or resectable pCCA with underlying liver disease (for example, PSC) less than 3 cm with or without chemo-(radio)therapy; patients with decompensated cirrhosis who have a single liver mass compatible with iCCA following biopsy analyses; and patients with unresectable locally advanced iCCA without liver disease after responding to induction therapy.
In the first scenario, the Mayo Clinic introduced a concept of neoadjuvant chemoradiation combined with liver transplantation in 2000 (ref. 256), with evolving protocols and indications. The current protocol257 employs a reduced radiation dose in external beam radiation therapy (45 Gy in 30 fractions) with continuous 5-fluorouracil infusion over 3 weeks as well as brachytherapy to mitigate the risk of radiation-induced complications. Subsequently, the patient receives oral capecitabine maintenance until transplantation. The comprehensive sequential series258, spanning from 1993 to 2018 and encompassing 211 patients who underwent liver transplantation, has revealed an overall survival after transplantation for patients with PSC-associated pCCA (n = 138) of 92%, 76% and 70% at 1, 5 and 10 years, respectively, compared to 90%, 58% and 49% for patients with sporadic or de novo pCCA (n = 73). Other studies have shown excellent results with different regimens259,260,261, such as CisGem with or without radiotherapy, aiming to circumvent the perioperative morbidity associated with neoadjuvant radiation262. However, a comprehensive study revealed that only about 5% of all patients presenting with pCCA may be eligible for liver transplantation263.
For iCCA, the experience is more limited and it remains a formal contraindication for liver transplantation outside clinical trials in most situations. In the case of early iCCA in cirrhosis, retrospective studies show promising survival rates (100%, 73% and 73% at 1, 3 and 5 years) for small solitary tumours (≤2 cm), while larger or multifocal lesions have poorer outcomes264,265. In addition, comparable results were observed in patients with lesions ≤5 cm (90%, 76% and 67% at 1, 3 and 5 years)266. Nevertheless, this is a rare procedure in clinical practice.
The third scenario involves unresectable locally advanced iCCA in healthy livers without extrahepatic disease and sustained response to systemic therapy with or without locoregional therapies (LRTs). Prospective studies267,268 included patients demonstrating disease stability for 6 months (some cases under gemcitabine-based chemotherapy and LRT), with a significant proportion undergoing successful transplantation and promising survival rates. It is important to mention that the proportion of patients with genetic alterations in FGFR2 or DNA damage repair pathways observed in this cohort was higher than the expected incidence. In the coming years, molecular profiling and mutational analyses of circulating tumour DNA may guide personalized approaches for liver transplantation in CCA. Currently, while liver transplantation for pCCA is established in several centres, for iCCA, it should be considered only in clinical trials.
Locoregional therapies
-
Research priorities: XII
For unresectable CCA, various LRTs have been proposed for patients with liver-only, unresectable CCA either as downsizing therapy before surgical resection or transplantation or as rescue therapy following systemic treatment failure269,270. The evaluated LRTs for CCA involve thermal ablation, including radiofrequency ablation and microwave ablation, external beam radiotherapy, or transarterial treatments such as transarterial chemoembolization (TACE), selective internal radiation therapy (SIRT; also known as transarterial radioembolization), and hepatic arterial infusion (HAI) of chemotherapy. Unfortunately, the efficacy of LRTs in CCA has never been demonstrated in randomized controlled trials. The evidence supporting the recommendation of LRTs relies mostly on retrospective studies, most of which are monocentric and involve a limited number of patients with heterogeneous inclusion criteria. Their use depends primarily on tumour and patient characteristics as well as local expertise.
Thermal ablation (radiofrequency ablation and microwave ablation)
Evidence supporting the recommendation of these treatments is scarce271,272. Overall, these studies suggest adequate local tumour control in small iCCA lesions categorized as unresectable (either due to inadequate localization, previous major liver resection, underlying advanced chronic liver disease and/or comorbidities). However, their efficacy in terms of disease-free survival and overall survival is inferior to that obtained in HCC273,274. A recent systematic review and meta-analysis, which included 15 studies involving 645 patients, reported that thermal ablation achieved an overall complete response rate of 93.9% and a median overall survival of 30.2 months275. Notably, in a proportion of studies, thermal ablation was applied after post-resection recurrence, hindering any direct comparison with front-line resection. Additionally, thermal ablation was shown to be a reliable therapeutic approach for individuals with pre-existing cirrhosis, with the best results observed for patients with a small single iCCA tumour (<2 cm), achieving similar survival trends to those in patients with HCC and comparable with those reported after surgical resection276.
External beam radiotherapy
The role of external beam radiotherapy in the treatment of CCA is also uncertain. There is high heterogeneity concerning dose, schedules or techniques (some use proton therapy, while others use conformal radiotherapy or SBRT). In most cases, it is offered with palliative intention, and a systematic review and meta-analysis involving 541 patients reported a 2-year local control rate of 69.1%, a median progression-free survival (PFS) of 15.6 months and a median overall survival of 18.9 months275. A phase II multicentre randomized trial (ABC-07) included patients with locally advanced CCA (46% pCCA, 39% dCCA, 14% iCCA)277. Patients who did not progress after four cycles of CisGem were randomized into two groups: those receiving two cycles of chemotherapy followed by SBRT, or a control arm group receiving four cycles of chemotherapy. The trial yielded negative results, showing no improvement in PFS and no trend in overall survival. These findings suggest that SBRT is not effective in the treatment of pCCA and dCCA, although the role of iCCA remains unclear due to the limited number of patients in this subgroup.
Transarterial therapies
The most common transarterial therapy evaluated in CCA is SIRT278,279,280,281,282,283,284,285. In line with this, a recent systematic review and meta-analysis that included a total of 921 patients from 21 studies reported an overall disease control rate of 82.3% and median PFS and overall survival of 7.8 months and 12.7 months for SIRT, respectively; of note 11% of patients subsequently underwent surgical resection286. Furthermore, in a phase II trial assessing the therapeutic efficacy of SIRT combined with CisGem in 41 treatment-naive patients, the response and disease control rates by RECIST were 41% and 98%. After a median follow-up of 36 months, the median PFS was 14 months and the median overall survival was 22 months, with 9 (22%) patients being successfully downstaged to surgery, of whom 8 underwent R0 resection287. In liver-only iCCA, a comparison was conducted of 41 patients treated with SIRT and systemic chemotherapy versus 73 patients treated with chemotherapy alone, with data extracted from prospective trials and analysed with emulated target trial paradigm and inverse probability of treatment weighting methods to minimize biases. In this analysis, the overall survival was significantly higher in patients treated with SIRT, with a median of 21.7 months versus 15.9 months (HR 0.59, 95% CI 0.34–0.99), which suggests that SIRT combined with chemotherapy might improve outcomes over chemotherapy alone288. A systematic review and pooled analysis of 22 studies including 1,145 patients reported that TACE achieved an overall response rate of 23.4%, with a mean PFS of 15 months and an overall survival of 15.9 months275. Moreover, a randomized controlled trial including 48 patients evaluated the addition of TACE with irinotecan-loaded drug-eluting microspheres to CisGem compared with CisGem alone. This combination demonstrated a significant increase in downsizing for resection (25% versus 8%) as well as improved PFS and overall survival (33.7 versus 12.6 months), while maintaining an acceptable safety profile289. Larger confirmatory trials are still needed before this strategy can be recommended in routine practice.
Hepatic arterial infusion
A systematic review of 16 studies including 331 patients, of whom a large proportion had bilobar disease (75%), multifocal tumours (66%) and a high incidence of macrovascular invasion (~40%), found that HAI achieved a pooled response rate of 41.3%, with a median PFS of 10 months and a median overall survival of 21.3 months275. A recent phase II trial from the Netherlands found a 3-year overall survival of 28.6% after HAI with floxuridine for patients with advanced iCCA confined to the liver. This overall survival compares favourably to a 3-year overall survival of less than 5%, which is mostly observed for patients receiving systemic therapy alone290.
Systemic therapies
-
Research priorities: XIII–XIV
Adjuvant systemic treatment
-
Research priorities: XV
After curative-intent resection, relapse rates are high, reaching approximately 70% at 5 years235,236. In this context, adjuvant systemic treatment has been evaluated. Two phase III trials conducted in France and Japan did not demonstrate any benefit from adjuvant gemcitabine-based regimens (gemcitabine-oxaliplatin combination and gemcitabine monotherapy, respectively). A combination analysis of these trials confirmed the lack of benefit of these therapies291,292,293. Conversely, the BILCAP trial294,295 randomized 447 patients with resected biliary tract cancer to receive either surveillance or adjuvant capecitabine. In the updated analysis, the adjusted HR for overall survival comparing capecitabine to surveillance was 0.84 (95% CI 0.67–1.06) in the intention-to-treat analysis, which did not reach statistical significance. However, in a protocol-specified sensitivity analysis, the HR improved significantly to 0.74 (95% CI 0.59–0.94) after adjusting for minimization factors, nodal status, grade and sex. Furthermore, in the intention-to-treat analysis, the median relapse-free survival was 24.3 months (95% CI 18.6–34.6) for capecitabine and 17.4 months (95% CI 11.8–23.0) for surveillance, with an adjusted HR of 0.81 (95% CI 0.65–1.01). Additionally, results from the STAMP randomized trial showed no benefit of adjuvant CisGem in patients with resected lymph node-positive eCCA compared to capecitabine alone296, leaving capecitabine as the only currently recommended adjuvant chemotherapy. The value of adjuvant fluoropyrimidines is further supported by the positive results of the ASCOT trial, conducted in Japan, which tested adjuvant S-1 chemotherapy versus surveillance297. This trial randomized 440 patients and demonstrated an improvement in overall survival for the S-1 group (adjusted HR 0.69, 95% CI 0.51–0.94) in the primary analysis. However, S-1 has not been studied in Western populations in this context and further clinical assessment should be conducted to allow evaluation by European regulatory bodies. Despite the statistical limitations of the BILCAP trial (intent-to-treat analysis not statistically significant), capecitabine is now considered as the standard adjuvant treatment in European guidelines due to its favourable safety profile and the absence of alternative treatments28. To date, no neoadjuvant therapy has been approved in the management of CCA. However, two phase III clinical trials (NCT04669496, jRCTs031200388) are currently evaluating the combination of toripalimab (anti-PD1) with lenvatinib and gemcitabine-oxaliplatin or CisGem plus S-1, respectively, as neoadjuvant treatments prior to surgery.
First-line and second-line chemotherapeutic regimens
-
Statements and recommendations: 6.2–6.4
The management of advanced CCA relies on the administration of systemic therapies or supportive care. First-line chemotherapy with CisGem was established in 2010 as the standard chemotherapy option for patients with advanced disease, based on the ABC-02 clinical trial data. In this study, the CisGem arm demonstrated an improved median overall survival and PFS of 11.7 months (versus 8.1 months; HR 0.64, 95% CI 0.52–0.80) and 8.0 months (versus 5.0 months; HR 0.63, 95% CI 0.51–0.77), respectively298, with an overall response rate (ORR) of 26.1% (versus 15.5%). Triple combinations of CisGem plus MET inhibitors (merestinib)299, VEGFR inhibitors (ramucirumab)299, EGFR inhibitors300,301,302 or a triple option of modified FOLFIRINOX (5-fluorouracil, irinotecan and oxaliplatin)303 and nab-paclitaxel with CisGem304 failed to show superiority to CisGem. A moderate survival benefit was observed when S-1 was added to CisGem in a Japanese population, which has not yet been evaluated in those of European descent305. Thus, CisGem remains the backbone chemotherapy of choice. Of note, alternative options, such as the combination of gemcitabine and oxaliplatin, may be considered for patients with contraindications to cisplatin306. The adequate duration of CisGem chemotherapy has been a matter of controversy. The ABC-02 study utilized a 6-month (eight-cycle) schedule of CisGem298. While some physicians consider the option of administering maintenance gemcitabine or to continue with the double until disease progression, more recent real-world evidence suggests that continuation of chemotherapy is not associated with a survival benefit307.
The addition of immune-checkpoint inhibitors to CisGem has modestly improved overall survival and is now considered the first-line standard of care for patients with advanced disease. The randomized phase III TOPAZ-1 clinical trial explored the addition of durvalumab or placebo to CisGem308 and observed an improved overall survival (12.9 months versus 11.3 months; HR 0.76, 95% CI 0.64–0.91), PFS (7.2 months versus 5.7 months; HR 0.75, 95% CI 0.63–0.89) and a higher ORR (27% versus 19%) in favour of CisGem combined with durvalumab over placebo308,309. A second randomized phase III clinical trial, KEYNOTE-966, also supports the addition of immune-checkpoint inhibitors to CisGem310. In the KEYNOTE-966 trial, pembrolizumab or placebo was added to CisGem311. The study also showed an improved overall survival (12.7 months versus 10.9 months; HR 0.83, 95% CI 0.72–0.95) in the pembrolizumab arm, while the impact on PFS (6.5 months versus 5.6 months; HR 0.86, 95% CI 0.75–1.00) did not reach pre-specified statistical significance310; ORR was similar in both arms (28.7%). There is currently no comparative study between durvalumab and pembrolizumab. Durvalumab or pembrolizumab combined with CisGem is already approved by the FDA and EMA in this indication and both are recommended by most national and international guidelines as first-line treatment in biliary tract cancer, especially taking into account that the 24-month survival rates in both studies are comparable (23.6%308 versus 25%310, respectively). Importantly, maintenance therapy with durvalumab or pembrolizumab in these patients may be continued for up to 2 years, or until disease progression or intolerability occurs.
Following progression to first-line chemotherapy, the randomized phase III ABC-06 clinical trial established FOLFOX as the chemotherapy of choice312. Benefit from FOLFOX (over active symptom control alone) remains modest, with a median overall survival of 6.2 months (versus 5.3 months; HR 0.69, 95% CI 0.50–0.97), PFS of 4 months and ORR of 5%. The therapy is generally recommended to fit patients in the second line, especially to ease symptoms or as a bridge to targeted treatments within or outside of clinical trials. The combination of liposomal irinotecan and 5-fluorouracil has been studied in two studies, with contradictory results. The phase II randomized NIFTY study313 identified a significant improvement in overall survival (8.6 months versus 5.3 months; HR 0.68, 95% CI 0.48–0.95), PFS (4.2 months versus 1.7 months; HR 0.61, 95% CI 0.44–0.86) and ORR (19.3% versus 2.3%) in favour of liposomal irinotecan and 5-fluorouracil over 5-fluorouracil alone. In contrast, the NALIRICC study314 was not able to show a significant improvement with that same combination over 5-fluorouracil alone. Thus, the role of this combination remains controversial, and even though it could be considered as a treatment option, its real benefit is unclear. A combined per-patient analysis of both trials confirmed the survival benefit for the double chemotherapy315,316. There is so far limited evidence for the use of FOLFIRI in biliary tract cancer317. Despite the limited evidence, irinotecan-based therapies are frequently used in many countries as an option after failure of first-line treatment.
There are significant unmet needs in terms of first-line and second-line chemotherapy options in CCA, constituting a major gap, especially considering that most patients are not able to receive targeted therapies as subsequent treatment due to the limited availability of broad molecular testing and access to drugs310,318. Firstly, despite the addition of immunotherapy to CisGem, median overall survival remains at around 12 months308,310, and only about 50% of patients are fit enough to receive second-line therapy. Secondly, there are no reliable biomarkers to identify patients that respond to immunotherapy in biliary tract cancer (beyond MSI-high and mismatch repair deficiency). In fact, patients with MSI-high may greatly benefit from pembrolizumab treatment, as an ORR of 73% was reported in individuals with MSI-high biliary tract cancers, significantly higher than the response rates observed in patients with pancreatic cancer (ORR 30%)319. Higher PDL1 expression may correlate with slightly better outcomes but this measure is not suitable to select patients for immune-checkpoint inhibitor treatment based on the data from TOPAZ-1 or KEYNOTE-966 (refs. 310,318). This is of significant importance, as even though some patients may derive long-term benefits from immunotherapy, these patients cannot be identified upfront due to the lack of predictive biomarkers. Therefore, once identified, a biomarker-based approach to select patients likely to benefit from immunotherapy may be a more prudent strategy than treating all patients with this regimen. In terms of chemotherapy, there is also a lack of biomarkers for tailoring chemotherapy choices. The potential role of DNA damage repair as a predictive marker to platinum-based chemotherapy in biliary tract cancer has not shown consistent results320,321.
Targeted therapies
-
Statements and recommendations: 6.5–6.6
In many retrospective analyses, about 30–40% of patients with biliary tumours have genetic alterations for which targeted therapies are available today. The IDH1/2 mutations and FGFR2 fusions are the most common drug-treatable genetic alterations; others include ERBB2, BRAF, BRCA1/2 and KRAS mutations as well as MDM2 amplifications55. IDH1 mutations and FGFR2 amplifications are considered to occur only in patients with iCCAs.
The phase III ClarIDHy trial demonstrated a significantly improved primary end point PFS with ivosidenib, an IDH1 inhibitor, versus placebo in patients with IDH1-mutant CCA322. Overall survival was improved after adjustment for crossover from placebo to ivosidenib in 70% of patients55. These results led to ivosidenib approval by the FDA and EMA. Multiple phase II trials have consistently demonstrated the efficacy of FGFR inhibitors, such as pemigatinib, infigratinib, erdafitinib, Debio 1347, derazantinib and futibatinib, for patients harbouring FGFR2 genomic rearrangements323,324,325,326,327,328. Response rates ranged from 21% to 42%, with a disease control rate of over 80%. The median PFS and median overall survival ranged from 7 to 9 and 12 to 22 months, respectively, in the study populations, which have led to the approval of pemigatinib and futibatinib by the FDA and EMA, significantly outperforming second-line chemotherapy results in cross-study comparisons. Three phase III trials were initiated to evaluate the efficacy of pemigatinib, futibatinib and infigratinib in the first-line setting against chemotherapy, but all trials closed prematurely due to poor accrual.
The rapid development of FGFR inhibitors provides a first indication of how precision oncology ‘sequence concepts’ might look in the future: due to different binding properties, a sequential use of suitable FGFR ‘inhibitor classes’ is already possible in individual patients even if therapy-induced ‘on-target’ resistances have already occurred. Repeated tumour genetic monitoring, for example, liquid biopsy, allows the detection of the respective resistance-inducing changes and enables the informed use of further targeted therapy lines. New generations of FGFR inhibitors, such as the FGFR2 inhibitor lirafugratinib and tinengotinib, have been and are being evaluated as second-line FGFR inhibitor therapy to overcome resistance to acquired FGFR2 mutations329.
Regarding HER2-directed therapies targeting HER2-overexpressing tumours, either chemotherapy-free combinations of antibodies or combination of targeted therapy and chemotherapy are under study. For purely molecular therapies, the efficacy of dual antibody-based therapy using trastuzumab and pertuzumab was demonstrated in biliary tumours in intensively pretreated patients in both the MyPathway and TAPUR studies330,331. A similar mode of action is also present with zanidatamab, a bi-specific antibody targeting the extracellular juxtamembrane domain and the dimerization domain of HER2. Positive data from the phase IIb HERIZON-BTC-01 trial were announced for zanidatamab with response rates above 40% in pretreated patients332. A current phase III clinical trial (NCT06282575) is now ongoing to confirm the therapeutic potential of zanidatamab. Promising data from the phase II SGNTUC-019 study of tucatinib and trastuzumab with a response rate of 47% have been presented333. For the combination of targeted therapy and chemotherapy, results from the phase II DESTINY-PanTumour02 were presented for the antibody–drug conjugate trastuzumab-deruxtecan, with response rates of 56% in pretreated patients with immunohistochemistry score 3+ HER2-positive biliary tract cancer334. Based on the DESTINY study, trastuzumab-deruxtecan has been approved as tumour-agnostic therapy for patients with HER2 overexpression or amplification, including for biliary tract cancer.
In BRAFV600E-mutant CCA, positive data have been reported from the ROAR basket trial (combination of trametinib (MEK inhibitor) and dabrafenib (BRAF inhibitor)) as well as from the TAPUR trial (combination of cobimetinib (MEK inhibitor) and vemurafenib (BRAF inhibitor)), with response rates of over 40% and disease control rates of over 80%335,336,337. These findings led to the FDA approval of dabrafenib-trametinib. For class 2 and 3 BRAF mutations, convincing studies are not yet available, so it is imperative to present the cases to the molecular tumour board or to enrol patients in clinical trials such as TAPISTRY (NCT04589845) or SORATRAM (CT no. 2024-512887-77- 00).
Rare but therapy-relevant alterations include other fusion proteins, such as NTRK fusions, for which there is already an entity-independent EMA approval for the inhibitors larotrectinib and entrectinib338,339. RET fusions are known from lung carcinoma, among others, and — like NRG1 fusions — also have a role in rare cases as putative driver mutations in biliary tumours. Patients with CCA were included in basket studies of RET inhibitors (selpercatinib and pralsetinib) as well as of NRG1 fusions (seribantumab and zenocutuzumab), and the tumour-agnostic approval by FDA also includes CCA340,341,342,343. In this regard, the ESCAT scale on targeted therapies for CCAs was recently created to guide the use of targeted therapies based on the scientific and clinical evidence344.
Overall, the clinical decisions and management of CCA are complex and should be conducted upon discussion in multidisciplinary teams. Based on the current formal guidelines27,28,345, Fig. 5 contains a decision tree for the clinical management of CCA.
Next-generation therapies: what’s in the pipeline?
-
Statements and recommendations: 6.7–6.10
-
Research priorities: XVI–XVIII
The pace of drug development, especially in connection with targetable alterations, is rapidly increasing in the field of biliary tract cancer, with therapies directed towards specific genetic alterations under development. Genetic alterations of interest in current clinical trials include MDM2 amplifications, KRASG12C mutations, Claudin 18.2 expression, core HRD mutations, such as BRCA1/2 and PALB2, and MTAP deletions.
The normal function of an intact wild-type p53 in cell cycle control may be compromised through abnormal interactions with its regulators. Overexpression of MDM2, an E3 ubiquitin ligase, in malignancy, primarily through amplification, inhibits wild-type p53, leading to a pro-mitotic state. Inhibition of MDM2 restores the pro-apoptotic function of p53 (ref. 346). The prevalence of MDM2 amplification in bile duct cancer is 5%, with a higher prevalence in gallbladder cancer (11%)347. The small-molecule inhibitors milademetan (RAIN-32)348 and brigimadlin (BI 907828)349 are being evaluated in patients with wild-type TP53, MDM2-amplified biliary tract cancer in the MANTRA-2 (NCT05012397) and BRIGHTLINE-2 (NCT05512377) basket studies, respectively.
The emergence of KRASG12C inhibitors has raised hope that KRAS, previously considered ‘undruggable’, can now be targeted. The phase I study of sotorasib included one patient with advanced biliary tract cancer who achieved stable disease at the full dose (960 mg once daily)350. A larger cohort of 12 patients with previously treated advanced biliary tract cancer was included in the adagrasib (KRYSTAL-1) phase II study, with five responses documented (response rate 41.7%) with a median PFS of 8.6 months and median overall survival of 15.1 months351. In the phase I study of another inhibitor, divarasib, a response was seen in one patient with CCA, with stable disease in another four352. New inhibitors, such as LY3537982 (NCT04956640), are under investigation. However, unlike lung cancer, where KRASG12C mutations are relatively common (42%), the prevalence in biliary tract cancer is much lower (8%), with KRASG12D as the most common KRAS mutation353. Several clinical trials (mainly phase I) are currently ongoing to test new drugs targeting KRASG12D, including studies involving patients with CCA.
Claudin 18.2 is a cell-surface protein that, in healthy cells, is restricted to membrane epithelial cells. In malignancy, there is abnormal overexpression, with a study showing Claudin 18.2 positivity in 6.3% of patients with biliary tract tumours as well as in 16.7% of patients with pancreatic cancer and in 14.1% with gastric cancer354. This overexpression is open to therapeutic targeting, primarily studied in gastro-oesophageal cancer. Studies in biliary tract cancer include monotherapy studies of TST001 (a recombinant humanized anti-Claudin 18.2 IgG1 monoclonal antibody; NCT05190575) as well as BNT141 (a novel RNA-based therapeutic which, after intravenous injection, is translated into anti-Claudin 18.2 antibodies; NCT04683939). A combination study is also under way of LM-302 (Claudin 18.2 antibody–drug conjugate) with candonilimab (bi-specific antibody for PD1 and CTLA4) in previously treated Claudin 18.2-positive biliary tract tumours (NCT05994001)355.
Within cancer cells, methylthioadenosine (MTA) may accumulate due to the homozygous deletion of the gene encoding MTA phosphorylase (MTAP) in approximately 10–15% of all cancers. MTA then forms a complex with PRMT5, inhibiting its function in controlling the cell cycle. MRTX1719 binds the MTA-PRMT5 complex and demonstrates antitumour activity in preclinical models and early clinical trials (NCT05245500)356,357,358.
Biliary tract tumours are moderately sensitive to DNA damage repair agents298,312. Based on studies of other tumour types, such as pancreatic cancer, there is a growing interest in evaluating the role of core HRD mutations in CCA359,360. While the efficacy of PARP inhibitors has been evaluated in several studies in pancreatic cancer, there are hardly any reported in CCA and data from two first-line umbrella studies using veliparib (NCT01282333) and rucaparib (NCT03337087) in combination with chemotherapy are awaited. The combination of immunotherapy and PARP inhibitors is also being investigated361 (NCT03639935), so far with modest benefit. Additionally, a novel family of next-generation chemotherapeutic agents called Aurkines, characterized by unique polyelectrophilic properties that induce a high frequency of double-strand DNA breaks, has shown promise in treating both treatment-naive and cisplatin-resistant CCAs362. In various preclinical models, Aurkines selectively caused severe DNA damage to cancer cells, bypassing DNA repair mechanisms and triggering cancer cell death, thereby inhibiting tumour growth362. The therapeutic potential of Aurkines, alone or in combination with immunotherapy, in the clinical setting is now eagerly awaited.
Developing drugs for small subgroups of a rare cancer poses challenges. Ongoing evaluations of many discussed agents, among others, are being conducted through efficient platform studies, where different agents are tested within a single protocol. Examples include the SAFIR-ABC10 umbrella study (NCT05615818), which assesses the maintenance molecular targeted therapies after induction chemotherapy with or without immunotherapy in advanced biliary tract cancer. The TAPISTRY platform study (NCT04589845) selects patients based on molecular alterations, regardless of the primary tumour site. Additionally, a pragmatic, real-world evaluation is being conducted in the DETERMINE (Determining Extended Therapeutic Indications for Existing Drugs in Rare Molecularly Defined Indications Using a National Evaluation Platform Trial) study (NCT05722886).
Perspectives from patient representatives
-
Statements and recommendations: 7.1
CCA is the second most prevalent primary liver cancer globally. Its incidence is on the rise, contributing to significant mortality due to its inherent aggressiveness and often late-stage diagnosis. Several nations, including England, the USA and Canada, have reported a continual increase in both incidence and mortality. Intriguingly, this oncological trend is no longer confined to individuals over 60 years old, impacting younger cohorts4,363,364. These aspects underscore the growing necessity for patient advocacy.
Many patients with CCA and their families encounter substantial challenges, from numerous initial visits to primary care practitioners before being referred for further investigations, to the multitude of tests they undergo, often with inconclusive results, adding confusion and fear to their journey. Additionally, several disparities in patient access to high-cost drugs across European countries have been identified. These issues need to be addressed in the future to ensure equitable access to appropriate therapeutic options worldwide65,365. Patients and their loved ones frequently find themselves as their own best advocates, seeking information and exploring the best treatment paths. Across the globe, there is a growing number of advocacy organizations, such as the Cholangiocarcinoma Foundation (USA), AMMF — The Cholangiocarcinoma Charity (UK), ATUVIBI (Spain), APiC (Italy), The Cholangiocarcinoma Foundation of Thailand, and groups in Japan, Australia and Canada. These organizations operate at various levels in each region, working to improve the situation for those with CCA. In addition to day-to-day support, these advocacy groups have played a pivotal part in:
-
Highlighting the rising incidence of CCA and its parallel mortality366, assessing regional disparities in diagnosis and treatment367, engaging with the WHO at committee level for the correct recoding of CCA in ICD-11 across its three anatomical sites, and characterizing CCA in the young23. These findings support campaigns for better recognition and access to CCA treatment.
-
Enhancing patient care by raising awareness of CCA in the community (for example, the UK and Thailand)368, developing guidelines for multidisciplinary teamwork, translating patient information into non-English languages369, detailing the patient experience370, addressing financial toxicity issues371,372, and promoting patient engagement in cancer genome sequencing373.
-
Actively contributing to the development of clinical guidelines in the USA, the UK and Europe (for example, NCCN guidelines, BSG guidelines230, Italian guidelines32,374, ASCO guidelines for adjuvant therapy375).
-
Providing the patient’s perspective in clinical trial design.
Despite the efforts of CCA advocacy organizations worldwide, there remains limited awareness of the public health significance of this cancer across all levels. Patient advocacy is crucial in ensuring that the patient’s voice is an active partner rather than a passive recipient in developments across these organizations, including the community, non-specialist clinical teams, the pharmaceutical industry, regulatory agencies, policymakers and governments.
Future directions, recommendations and conclusions
Despite several advancements having been achieved over the past decade, the prognosis of patients with CCA continues to be dismal, with a 5-year overall survival rate of less than 20%21. Curative intent options are reserved for patients who are diagnosed at an early stage, sometimes occurring as a result of an incidental diagnosis of CCA. With a mortality that almost equals its incidence, CCA is predicted to become one of the major health issues in Europe in the next few decades. Indeed, although it is still considered a rare cancer, its incidence is increasing worldwide, partly because of the rising incidence of risk factors and partly because of increased awareness and more appropriate diagnostic pathways in the clinical community.
The increase in incidence has matched a growing understanding of the heterogeneity of CCA, related to the surge of diverse risk factors in the worldwide population, larger implementation of genomic profiling in the clinic, and expanded exposure to various treatments that boost temporal and spatial tumour evolution. In this scenario, the need to steer clinical commitment and research interests within the worldwide community drives them in different directions. On the one hand, it becomes necessary to increase the understanding and awareness of CCA to identify the reversible risk factors that are associated with CCA development, implement strategies for the elimination of these risk factors, understand its pathobiology to tackle the emergence of CCA both in the presence or absence of predisposing conditions, and to develop novel biomarkers for early diagnosis. In the presence of such heterogeneity, data from larger populations are needed, with an increased value of real-world data and the investigation of non-invasive methodologies (such as liquid biopsy) that are widely applicable. On the other hand, a shared effort must be steered towards improving the current clinical strategies to affect the lives of patients being diagnosed with CCA, with the ultimate goal of providing personalized treatment. Overall, through critical revision of the literature and by conducting a Delphi questionnaire, the group of experts herein proposes a set of statements and recommendations for CCA management (Table 1), unmet needs (Box 1) and research priorities in the field (Table 2).
We have witnessed the success of a paired approach between science and the clinic, represented, among others, by the improvement of clinical outcomes achieved through the implementation of targeted therapies that have followed the deep investigation of the molecular landscape of CCA. In an attempt to continue this rewarding approach, the following steps should include investment in understanding the mechanisms of tumour evolution and the heterogeneity of drug responses. As novel forms of systemic therapies have entered clinical practice, new limitations have come to light, including the unmet needs of understanding how to optimize the benefit of personalized therapies by overcoming primary and secondary drug resistance, understanding the immunobiology of CCA to implement biomarkers for the selection of patients who would benefit from immune-checkpoint inhibitors, and refining immunotherapeutic strategies through the targeting of diversified immune populations and developing more personalized vaccine strategies.
As we acknowledge that more diversified chemotherapy drugs have proven efficacious in CCA, we need to consider novel strategies to optimize and personalize their use, to improve their delivery within a complex TME, and to identify the appropriate scheduling in terms of combinations and sequences. In future years, it will be important to focus on refining the activity and efficacy of systemic anticancer therapies not only to improve the life expectancy for patients with advanced CCA but also to pave the way to a neoadjuvant or downstaging approach that optimizes the curative rate of surgery and widens the opportunities for local therapies. Additional challenges in clinical study designs remain critical, as new effective targeted therapies are increasingly used as first-line treatments in real-world practice despite limited supporting evidence from trials that were prematurely closed. To overcome this, future trials should move beyond commercial sponsors and individual group strategies. Importantly, ongoing global collaborative efforts will be invaluable, enabling faster and more efficient patient accrual across multiple countries, in contrast to older trials that enrolled patients from only one country over extended periods.
With the need to identify novel therapeutic targets and biomarkers of activity, discovery science will benefit from the generation of ameliorated in vivo and patient-centric CCA models that better mimic human CCA and from the integration of a forward and reverse translational approach, boosting the adoption of novel strategies in the clinic. In this context, it is becoming increasingly more important to establish collaborative networks with multi-stakeholders, such as the ENS-CCA, to accelerate scientific knowledge and clinical development. In this regard, the COST-Action Precision-BTC-Network aims to coordinate a pan-European interdisciplinary effort to promote personalized approaches for patients with CCA, covering prevention, diagnosis, prognosis and therapy. Based on integrating retrospective and prospective pan-European studies and creating a life science ecosystem, the network supports interaction between basic scientists, clinicians, regulatory authorities, advocacy groups, small-sized and medium-sized enterprises, and pharmaceutical industry partners. Precision-BTC-Network provides the critical mass of expertise, connections and integration to address the innovation needed to implement personalized management in CCA and train the next generation of clinicians and scientists focusing on CCA. The multidisciplinary nature of ENS-CCA and Precision-BTC lies on a patient-centric approach and relies on the active engagement of patients and public representatives who support the need for patient education, with the overall goal of making the patients active players in their decisions and not passive receivers of their clinician’s choice. The partnership between the academic community and industry, regulatory bodies, and funding agencies must be the pillar for future work because no effect on patients’ lives will be delivered if the scientific advancement is not coupled with the implementation of international policies for the harmonization of clinical management and reduction in world disparities.
References
Banales, J. M. et al. Cholangiocarcinoma 2020: the next horizon in mechanisms and management. Nat. Rev. Gastroenterol. Hepatol. 17, 557–588 (2020).
Rodrigues, P. M. et al. Pathogenesis of cholangiocarcinoma. Annu. Rev. Pathol. 16, 433–463 (2021).
Banales, J. M. et al. Expert consensus document: cholangiocarcinoma: current knowledge and future perspectives consensus statement from the European Network for the Study of Cholangiocarcinoma (ENS-CCA). Nat. Rev. Gastroenterol. Hepatol. 13, 261–280 (2016).
Tataru, D. et al. Cholangiocarcinoma across England: temporal changes in incidence, survival and routes to diagnosis by region and level of socioeconomic deprivation. JHEP Rep. 6, 100983 (2024).
International Agency for Research on Cancer. WHO Mortality Database http://www-dep.iarc.fr/WHOdb/WHOdb.htm (World Health Organization, 2024).
GBD 2021 Diseases and Injuries Collaborators. Global incidence, prevalence, years lived with disability (YLDs), disability-adjusted life-years (DALYs), and healthy life expectancy (HALE) for 371 diseases and injuries in 204 countries and territories and 811 subnational locations, 1990-2021: a systematic analysis for the Global Burden of Disease Study 2021. Lancet 403, 2133–2161 (2024).
Ahmad, O. B. et al. Age standardization of rates: a new WHO standard. GPE Discussion Pap. Ser. 31, 3 (2001).
Taylor-Robinson, S. D. et al. Increase in mortality rates from intrahepatic cholangiocarcinoma in England and Wales 1968-1998. Gut 48, 816–820 (2001).
Khan, S. A. et al. Changing international trends in mortality rates for liver, biliary and pancreatic tumours. J. Hepatol. 37, 806–813 (2002).
Khan, S. A., Tavolari, S. & Brandi, G. Cholangiocarcinoma: epidemiology and risk factors. Liver Int. 39 (Suppl. 1), 19–31 (2019).
Vithayathil, M. & Khan, S. A. Current epidemiology of cholangiocarcinoma in Western countries. J. Hepatol. 77, 1690–1698 (2022).
Brindley, P. J. et al. Cholangiocarcinoma. Nat. Rev. Dis. Primers 7, 65 (2021).
Selvadurai, S. et al. Cholangiocarcinoma miscoding in hepatobiliary centres. Eur. J. Surg. Oncol. 47, 635–639 (2021).
Khan, S. A. et al. Rising trends in cholangiocarcinoma: is the ICD classification system misleading us? J. Hepatol. 56, 848–854 (2012).
Khan, S. A., Thomas, H. C., Davidson, B. R. & Taylor-Robinson, S. D. Cholangiocarcinoma. Lancet 366, 1303–1314 (2005).
Xiong, J. et al. Aspirin use is associated with a reduced risk of cholangiocarcinoma: a systematic review and meta-analysis. Cancer Manag. Res. 10, 4095–4104 (2018).
Marcano-Bonilla, L. et al. Aspirin, statins, non-aspirin NSAIDs, metformin, and the risk of biliary cancer: a Swedish population-based cohort study. Cancer Epidemiol. Biomark. Prev. 31, 804–810 (2022).
Jackson, S. S. et al. Association between aspirin use and biliary tract cancer survival. JAMA Oncol. 5, 1802–1804 (2019).
Peng, Y. C. et al. Statins are associated with a reduced risk of cholangiocarcinoma: a population-based case-control study. Br. J. Clin. Pharmacol. 80, 755–761 (2015).
Liu, Z. et al. Statin use and reduced risk of biliary tract cancers in the UK Clinical Practice Research Datalink. Gut 68, 1458–1464 (2019).
Izquierdo-Sanchez, L. et al. Cholangiocarcinoma landscape in Europe: diagnostic, prognostic and therapeutic insights from the ENSCCA Registry. J. Hepatol. 76, 1109–1121 (2022).
Alvaro, D. et al. Cholangiocarcinoma in Italy: a national survey on clinical characteristics, diagnostic modalities and treatment. Results from the “Cholangiocarcinoma” committee of the Italian Association for the Study of Liver Disease. Dig. Liver Dis. 43, 60–65 (2011).
Pappas, L. et al. The Cholangiocarcinoma in the Young (CITY) study: tumor biology, treatment patterns, and survival outcomes in adolescent young adults with cholangiocarcinoma. JCO Precis. Oncol. 7, e2200594 (2023).
Forner, A. et al. Clinical presentation, diagnosis and staging of cholangiocarcinoma. Liver Int. 39 (Suppl. 1), 98–107 (2019).
Iavarone, M. et al. Contrast enhanced CT-scan to diagnose intrahepatic cholangiocarcinoma in patients with cirrhosis. J. Hepatol. 58, 1188–1193 (2013).
Rimola, J. et al. Cholangiocarcinoma in cirrhosis: absence of contrast washout in delayed phases by magnetic resonance imaging avoids misdiagnosis of hepatocellular carcinoma. Hepatology 50, 791–798 (2009).
European Association for the Study of the Liver.EASL-ILCA clinical practice guidelines on the management of intrahepatic cholangiocarcinoma. J. Hepatol. 79, 181–208 (2023).
Vogel, A. et al. Biliary tract cancer: ESMO clinical practice guideline for diagnosis, treatment and follow-up. Ann. Oncol. 34, 127–140 (2023).
Childs, A. et al. Biopsy for advanced hepatocellular carcinoma: results of a multicentre UK audit. Br. J. Cancer 125, 1350–1355 (2021).
Brusset, B. et al. Radiological diagnosis of hepatocellular carcinoma does not preclude biopsy before treatment. JHEP Rep. 6, 100957 (2024).
Park, M. S. et al. Differentiation of extrahepatic bile duct cholangiocarcinoma from benign stricture: findings at MRCP versus ERCP. Radiology 233, 234–240 (2004).
Cholangiocarcinoma Working Group. Italian clinical practice guidelines on cholangiocarcinoma — part I: classification, diagnosis and staging. Dig. Liver Dis. 52, 1282–1293 (2020).
Macias, R. I. R. et al. Clinical relevance of biomarkers in cholangiocarcinoma: critical revision and future directions. Gut 71, 1669–1683 (2022).
Navaneethan, U., Njei, B., Venkatesh, P. G., Lourdusamy, V. & Sanaka, M. R. Endoscopic ultrasound in the diagnosis of cholangiocarcinoma as the etiology of biliary strictures: a systematic review and meta-analysis. Gastroenterol. Rep. 3, 209–215 (2015).
Sadeghi, A. et al. Diagnostic yield of EUS-guided FNA for malignant biliary stricture: a systematic review and meta-analysis. Gastrointest. Endosc. 83, 290–298.e1 (2016).
De Moura, D. T. H. et al. Endoscopic retrograde cholangiopancreatography versus endoscopic ultrasound for tissue diagnosis of malignant biliary stricture: systematic review and meta-analysis. Endosc. Ultrasound 7, 10–19 (2018).
de Moura, D. T. H. et al. Endoscopic ultrasound-guided fine needle aspiration and endoscopic retrograde cholangiopancreatography-based tissue sampling in suspected malignant biliary strictures: a meta-analysis of same-session procedures. Clin. Endosc. 53, 417–428 (2020).
Navaneethan, U. et al. Single-operator cholangioscopy and targeted biopsies in the diagnosis of indeterminate biliary strictures: a systematic review. Gastrointest. Endosc. 82, 608–614.e2 (2015).
Mohamed, R. et al. Per-oral cholangioscopy in patients with primary sclerosing cholangitis: a 12-month follow-up study. Endosc. Int. Open 12, E237–E244 (2024).
Pereira, P. et al. Role of peroral cholangioscopy for diagnosis and staging of biliary tumors. Dig. Dis. 38, 431–440 (2020).
Weismuller, T. J. et al. Patient age, sex, and inflammatory bowel disease phenotype associate with course of primary sclerosing cholangitis. Gastroenterology 152, 1975–1984.e8 (2017).
Boonstra, K. et al. Population-based epidemiology, malignancy risk, and outcome of primary sclerosing cholangitis. Hepatology 58, 2045–2055 (2013).
Grimsrud, M. M. & Folseraas, T. Pathogenesis, diagnosis and treatment of premalignant and malignant stages of cholangiocarcinoma in primary sclerosing cholangitis. Liver Int. 39, 2230–2237 (2019).
European Society of Gastrointestinal Endoscopy; European Association for the Study of the Liver. Role of endoscopy in primary sclerosing cholangitis: European Society of Gastrointestinal Endoscopy (ESGE) and European Association for the Study of the Liver (EASL) clinical guideline. J. Hepatol. 66, 1265–1281 (2017).
European Association for the Study of the Liver. EASL clinical practice guidelines on sclerosing cholangitis. J. Hepatol. 77, 761–806 (2022).
Villard, C. et al. Prospective surveillance for cholangiocarcinoma in unselected individuals with primary sclerosing cholangitis. J. Hepatol. 78, 604–613 (2023).
Njei, B., McCarty, T. R., Varadarajulu, S. & Navaneethan, U. Systematic review with meta-analysis endoscopic retrograde cholangiopancreatography-based modalities for the diagnosis of cholangiocarcinoma in primary sclerosing cholangitis. Aliment. Pharmacol. Ther. 44, 1139–1151 (2016).
Lamarca, A. et al. Advanced intrahepatic cholangiocarcinoma: post hoc analysis of the ABC-01, -02, and -03 clinical trials. J. Natl Cancer Inst. 112, 200–210 (2020).
Lamarca, A. et al. Liver metastases of intrahepatic cholangiocarcinoma: implications for an updated staging system. Hepatology 73, 2311–2325 (2021).
National Cancer Institute. Surveillance, Epidemiology, and End Results Program. Monographs https://seer.cancer.gov/publications/monographs.html (2025).
Kim, Y. Y. et al. Clinical staging of mass-forming intrahepatic cholangiocarcinoma: computed tomography versus magnetic resonance imaging. Hepatol. Commun. 5, 2009–2018 (2021).
Lamarca, A. et al. 18F-fluorodeoxyglucose positron emission tomography (18FDG-PET) for patients with biliary tract cancer: systematic review and meta-analysis. J. Hepatol. 71, 115–129 (2019).
Malikowski, T. et al. Endoscopic ultrasound/fine needle aspiration is effective for lymph node staging in patients with cholangiocarcinoma. Hepatology 72, 940–948 (2020).
Wang, X. Y. et al. Driver mutations of intrahepatic cholangiocarcinoma shape clinically relevant genomic clusters with distinct molecular features and therapeutic vulnerabilities. Theranostics 12, 260–276 (2022).
Kendre, G. et al. Charting co-mutation patterns associated with actionable drivers in intrahepatic cholangiocarcinoma. J. Hepatol. 78, 614–626 (2023).
Montal, R. et al. Molecular classification and therapeutic targets in extrahepatic cholangiocarcinoma. J. Hepatol. 73, 315–327 (2020).
Zhang, M. et al. An inflammatory checkpoint generated by IL1RN splicing offers therapeutic opportunity for KRAS-mutant intrahepatic cholangiocarcinoma. Cancer Discov. 13, 2248–2269 (2023).
O’Rourke, C. J. et al. Molecular portraits of patients with intrahepatic cholangiocarcinoma who diverge as rapid progressors or long survivors on chemotherapy. Gut 73, 496–508 (2024).
Nepal, C. et al. Genomic perturbations reveal distinct regulatory networks in intrahepatic cholangiocarcinoma. Hepatology 68, 949–963 (2018).
Cao, J. et al. Intrahepatic cholangiocarcinoma: genomic heterogeneity between Eastern and Western Patients. JCO Precis. Oncol. 4, PO.18.00414 (2020).
Nakamura, H. et al. Genomic spectra of biliary tract cancer. Nat. Genet. 47, 1003–1010 (2015).
Zhou, S. L. et al. Association of KRAS variant subtypes with survival and recurrence in patients with surgically treated intrahepatic cholangiocarcinoma. JAMA Surg. 157, 59–65 (2022).
Tavolari, S. & Brandi, G. Mutational landscape of cholangiocarcinoma according to different etiologies: a review. Cells 12, 1216 (2023).
Harding, J. J., Khalil, D. N., Fabris, L. & Abou-Alfa, G. K. Rational development of combination therapies for biliary tract cancers. J. Hepatol. 78, 217–228 (2023).
Rimassa, L. et al. New systemic treatment paradigms in advanced biliary tract cancer and variations in patient access across Europe. Lancet Reg. Health Eur. 50, 101170 (2025).
Sulpice, L. et al. Molecular profiling of stroma identifies osteopontin as an independent predictor of poor prognosis in intrahepatic cholangiocarcinoma. Hepatology 58, 1992–2000 (2013).
Song, G. et al. Single-cell transcriptomic analysis suggests two molecularly subtypes of intrahepatic cholangiocarcinoma. Nat. Commun. 13, 1642 (2022).
Ma, L. et al. Multiregional single-cell dissection of tumor and immune cells reveals stable lock-and-key features in liver cancer. Nat. Commun. 13, 7533 (2022).
Ma, L. et al. Single-cell atlas of tumor cell evolution in response to therapy in hepatocellular carcinoma and intrahepatic cholangiocarcinoma. J. Hepatol. 75, 1397–1408 (2021).
Ma, L. et al. Tumor Cell biodiversity drives microenvironmental reprogramming in liver cancer. Cancer Cell 36, 418–430.e6 (2019).
Putatunda, V., Jusakul, A., Roberts, L. & Wang, X. W. Genetic, epigenetic, and microenvironmental drivers of cholangiocarcinoma. Am. J. Pathol. 195, 362–377 (2025).
Goeppert, B. et al. Global alterations of DNA methylation in cholangiocarcinoma target the Wnt signaling pathway. Hepatology 59, 544–554 (2014).
Merino-Azpitarte, M. et al. SOX17 regulates cholangiocyte differentiation and acts as a tumor suppressor in cholangiocarcinoma. J. Hepatol. 67, 72–83 (2017).
Jusakul, A. et al. Whole-genome and epigenomic landscapes of etiologically distinct subtypes of cholangiocarcinoma. Cancer Discov. 7, 1116–1135 (2017).
Hong, J. H. et al. Integrative multiomics enhancer activity profiling identifies therapeutic vulnerabilities in cholangiocarcinoma of different etiologies. Gut 73, 966–984 (2024).
Duwe, L. et al. MicroRNA-27a-3p targets FoxO signalling to induce tumour-like phenotypes in bile duct cells. J. Hepatol. 78, 364–375 (2023).
Job, S. et al. Identification of four immune subtypes characterized by distinct composition and functions of tumor microenvironment in intrahepatic cholangiocarcinoma. Hepatology 72, 965–981 (2020).
Martin-Serrano, M. A. et al. Novel microenvironment-based classification of intrahepatic cholangiocarcinoma with therapeutic implications. Gut https://doi.org/10.1136/gutjnl-2021-326514 (2022).
Bramel, E. R. & Sia, D. Novel insights into molecular and immune subtypes of biliary tract cancers. Adv. Cancer Res. 156, 167–199 (2022).
Sia, D. et al. Integrative molecular analysis of intrahepatic cholangiocarcinoma reveals 2 classes that have different outcomes. Gastroenterology 144, 829–840 (2013).
Andersen, J. B. et al. Genomic and genetic characterization of cholangiocarcinoma identifies therapeutic targets for tyrosine kinase inhibitors. Gastroenterology 142, 1021–1031.e15 (2012).
Affo, S., Filliol, A., Gores, G. J. & Schwabe, R. F. Fibroblasts in liver cancer: functions and therapeutic translation. Lancet Gastroenterol. Hepatol. 8, 748–759 (2023).
Ilyas, S. I. et al. Cholangiocarcinoma — novel biological insights and therapeutic strategies. Nat. Rev. Clin. Oncol. 20, 470–486 (2023).
Cantallops Vila, P., Ravichandra, A., Agirre Lizaso, A., Perugorria, M. J. & Affo, S. Heterogeneity, crosstalk, and targeting of cancer-associated fibroblasts in cholangiocarcinoma. Hepatology 79, 941–958 (2024).
Affo, S. et al. Promotion of cholangiocarcinoma growth by diverse cancer-associated fibroblast subpopulations. Cancer Cell 39, 866–882.e11 (2021).
Zhang, M. et al. Single-cell transcriptomic architecture and intercellular crosstalk of human intrahepatic cholangiocarcinoma. J. Hepatol. 73, 1118–1130 (2020).
Cadamuro, M. et al. Platelet-derived growth factor-D enables liver myofibroblasts to promote tumor lymphangiogenesis in cholangiocarcinoma. J. Hepatol. 70, 700–709 (2019).
Mertens, J. C. et al. Therapeutic effects of deleting cancer-associated fibroblasts in cholangiocarcinoma. Cancer Res. 73, 897–907 (2013).
Fabris, L., Cadamuro, M., Cagnin, S., Strazzabosco, M. & Gores, G. J. Liver matrix in benign and malignant biliary tract disease. Semin. Liver Dis. 40, 282–297 (2020).
Aishima, S. et al. Tenascin expression at the invasive front is associated with poor prognosis in intrahepatic cholangiocarcinoma. Mod. Pathol. 16, 1019–1027 (2003).
Utispan, K. et al. Gene expression profiling of cholangiocarcinoma-derived fibroblast reveals alterations related to tumor progression and indicates periostin as a poor prognostic marker. Mol. Cancer 9, 13 (2010).
Sirica, A. E., Almenara, J. A. & Li, C. Periostin in intrahepatic cholangiocarcinoma: pathobiological insights and clinical implications. Exp. Mol. Pathol. 97, 515–524 (2014).
Utispan, K. et al. Periostin activates integrin alpha5beta1 through a PI3K/AKT-dependent pathway in invasion of cholangiocarcinoma. Int. J. Oncol. 41, 1110–1118 (2012).
Mino, M. et al. Periostin promotes malignant potential by induction of epithelial-mesenchymal transition in intrahepatic cholangiocarcinoma. Hepatol. Commun. 1, 1099–1109 (2017).
Zeng, J. et al. Tumor-associated macrophages recruited by periostin in intrahepatic cholangiocarcinoma stem cells. Oncol. Lett. 15, 8681–8686 (2018).
Lowy, C. M. & Oskarsson, T. Tenascin C in metastasis: a view from the invasive front. Cell Adh. Migr. 9, 112–124 (2015).
Zheng, Y. et al. Osteopontin promotes metastasis of intrahepatic cholangiocarcinoma through recruiting MAPK1 and mediating Ser675 phosphorylation of β-catenin. Cell Death Dis. 9, 179 (2018).
Nissen, N. I., Karsdal, M. & Willumsen, N. Collagens and cancer associated fibroblasts in the reactive stroma and its relation to cancer biology. J. Exp. Clin. Cancer Res. 38, 115 (2019).
Chang, L. et al. The SWI/SNF complex is a mechanoregulated inhibitor of YAP and TAZ. Nature 563, 265–269 (2018).
Loeuillard, E., Conboy, C. B., Gores, G. J. & Rizvi, S. Immunobiology of cholangiocarcinoma. JHEP Rep. 1, 297–311 (2019).
Fabris, L. et al. The tumour microenvironment and immune milieu of cholangiocarcinoma. Liver Int. 39 (Suppl. 1), 63–78 (2019).
Zhou, M. et al. Tumor-associated macrophages in cholangiocarcinoma: complex interplay and potential therapeutic target. EBioMedicine 67, 103375 (2021).
Liu, D. et al. The prognostic value of neutrophil-to-lymphocyte ratio in cholangiocarcinoma: a systematic review and meta-analysis. Sci. Rep. 12, 12691 (2022).
Cao, H. et al. Tumor microenvironment and its implications for antitumor immunity in cholangiocarcinoma: future perspectives for novel therapies. Int. J. Biol. Sci. 18, 5369–5390 (2022).
Loeuillard, E. et al. Targeting tumor-associated macrophages and granulocytic myeloid-derived suppressor cells augments PD-1 blockade in cholangiocarcinoma. J. Clin. Invest. 130, 5380–5396 (2020).
Zhang, Q. et al. Gut microbiome directs hepatocytes to recruit MDSCs and promote cholangiocarcinoma. Cancer Discov. 11, 1248–1267 (2021).
Xu, X. D. et al. Circulating myeloid-derived suppressor cells in patients with pancreatic cancer. Hepatobiliary Pancreat. Dis. Int. 15, 99–105 (2016).
Takagi, S. et al. Dendritic cells, T-cell infiltration, and Grp94 expression in cholangiocellular carcinoma. Hum. Pathol. 35, 881–886 (2004).
Diggs, L. P. et al. CD40-mediated immune cell activation enhances response to anti-PD-1 in murine intrahepatic cholangiocarcinoma. J. Hepatol. 74, 1145–1154 (2021).
Shimizu, K. et al. Clinical utilization of postoperative dendritic cell vaccine plus activated T-cell transfer in patients with intrahepatic cholangiocarcinoma. J. Hepatobiliary Pancreat. Sci. 19, 171–178 (2012).
Jung, I. H. et al. In vivo study of natural killer (NK) cell cytotoxicity against cholangiocarcinoma in a nude mouse model. In Vivo 32, 771–781 (2018).
Cornillet, M. et al. Imbalance of genes encoding natural killer immunoglobulin-like receptors and human leukocyte antigen in patients with biliary cancer. Gastroenterology 157, 1067–1080.e9 (2019).
Goeppert, B. et al. Prognostic impact of tumour-infiltrating immune cells on biliary tract cancer. Br. J. Cancer 109, 2665–2674 (2013).
Carapeto, F. et al. The immunogenomic landscape of resected intrahepatic cholangiocarcinoma. Hepatology 75, 297–308 (2022).
Vigano, L. et al. Tumor-infiltrating lymphocytes and macrophages in intrahepatic cholangiocellular carcinoma. impact on prognosis after complete surgery. J. Gastrointest. Surg. 23, 2216–2224 (2019).
Zhou, G. et al. Reduction of immunosuppressive tumor microenvironment in cholangiocarcinoma by ex vivo targeting immune checkpoint molecules. J. Hepatol. 71, 753–762 (2019).
Alvisi, G. et al. Multimodal single-cell profiling of intrahepatic cholangiocarcinoma defines hyperactivated Tregs as a potential therapeutic target. J. Hepatol. 77, 1359–1372 (2022).
Li, H. et al. Single-cell transcriptomic architecture unraveling the complexity of tumor heterogeneity in distal cholangiocarcinoma. Cell Mol. Gastroenterol. Hepatol. 13, 1592–1609.e9 (2022).
Konishi, D. et al. Regulatory T cells induce a suppressive immune milieu and promote lymph node metastasis in intrahepatic cholangiocarcinoma. Br. J. Cancer 127, 757–765 (2022).
Chen, Z. et al. PNOC expressed by B cells in cholangiocarcinoma was survival related and LAIR2 could be a T cell exhaustion biomarker in tumor microenvironment: characterization of immune microenvironment combining single-cell and bulk sequencing technology. Front. Immunol. 12, 647209 (2021).
Milardi, G. & Lleo, A. Tumor-infiltrating B lymphocytes: promising immunotherapeutic targets for primary liver cancer treatment. Cancers 15, 2182 (2023).
Pfister, D. et al. NASH limits anti-tumour surveillance in immunotherapy-treated HCC. Nature 592, 450–456 (2021).
Labib, P. L., Goodchild, G. & Pereira, S. P. Molecular pathogenesis of cholangiocarcinoma. BMC Cancer 19, 185 (2019).
Jaiswal, M., LaRusso, N. F., Burgart, L. J. & Gores, G. J. Inflammatory cytokines induce DNA damage and inhibit DNA repair in cholangiocarcinoma cells by a nitric oxide-dependent mechanism. Cancer Res. 60, 184–190 (2000).
Jaiswal, M., LaRusso, N. F., Shapiro, R. A., Billiar, T. R. & Gores, G. J. Nitric oxide-mediated inhibition of DNA repair potentiates oxidative DNA damage in cholangiocytes. Gastroenterology 120, 190–199 (2001).
Aishima, S. et al. Different roles of inducible nitric oxide synthase and cyclooxygenase-2 in carcinogenesis and metastasis of intrahepatic cholangiocarcinoma. Hum. Pathol. 44, 1031–1037 (2013).
Braconi, C. et al. Molecular perturbations in cholangiocarcinoma: is it time for precision medicine? Liver Int. 39 (Suppl. 1), 32–42 (2019).
Moeini, A., Sia, D., Bardeesy, N., Mazzaferro, V. & Llovet, J. M. Molecular pathogenesis and targeted therapies for intrahepatic cholangiocarcinoma. Clin. Cancer Res. 22, 291–300 (2016).
Song, X. et al. Combined CDK4/6 and Pan-mTOR Inhibition is synergistic against intrahepatic cholangiocarcinoma. Clin. Cancer Res. 25, 403–413 (2019).
Anichini, G., Carrassa, L., Stecca, B., Marra, F. & Raggi, C. The role of the hedgehog pathway in cholangiocarcinoma. Cancers 13, 4774 (2021).
Boulter, L. et al. WNT signaling drives cholangiocarcinoma growth and can be pharmacologically inhibited. J. Clin. Invest. 125, 1269–1285 (2015).
Carotenuto, P. et al. Wnt signalling modulates transcribed-ultraconserved regions in hepatobiliary cancers. Gut 66, 1268–1277 (2017).
Zhang, Y. et al. β-Catenin sustains and is required for YES-associated protein oncogenic activity in cholangiocarcinoma. Gastroenterology 163, 481–494 (2022).
Geisler, F. & Strazzabosco, M. Emerging roles of Notch signaling in liver disease. Hepatology 61, 382–392 (2015).
Martinez Lyons, A. & Boulter, L. NOTCH signalling — a core regulator of bile duct disease? Dis. Model. Mech. 16, dmm.050231 (2023).
Fan, B. et al. Cholangiocarcinomas can originate from hepatocytes in mice. J. Clin. Invest. 122, 2911–2915 (2012).
Sekiya, S. & Suzuki, A. Intrahepatic cholangiocarcinoma can arise from Notch-mediated conversion of hepatocytes. J. Clin. Invest. 122, 3914–3918 (2012).
Zender, S. et al. A critical role for notch signaling in the formation of cholangiocellular carcinomas. Cancer Cell 30, 353–356 (2016).
Guest, R. V. et al. Notch3 drives development and progression of cholangiocarcinoma. Proc. Natl Acad. Sci. USA 113, 12250–12255 (2016).
Zhang, S. et al. Pan-mTOR inhibitor MLN0128 is effective against intrahepatic cholangiocarcinoma in mice. J. Hepatol. 67, 1194–1203 (2017).
Cigliano, A. et al. The Hippo pathway effector TAZ induces intrahepatic cholangiocarcinoma in mice and is ubiquitously activated in the human disease. J. Exp. Clin. Cancer Res. 41, 192 (2022).
Amadeo, E. et al. Past, present, and future of FGFR inhibitors in cholangiocarcinoma: from biological mechanisms to clinical applications. Expert Rev. Clin. Pharmacol. 16, 631–642 (2023).
Younger, N. T. et al. In vivo modeling of patient genetic heterogeneity identifies new ways to target cholangiocarcinoma. Cancer Res. 82, 1548–1559 (2022).
Datta, J. et al. Akt activation mediates acquired resistance to fibroblast growth factor receptor inhibitor BGJ398. Mol. Cancer Ther. 16, 614–624 (2017).
Wu, Q. et al. EGFR inhibition potentiates FGFR inhibitor therapy and overcomes resistance in FGFR2 fusion-positive cholangiocarcinoma. Cancer Discov. 12, 1378–1395 (2022).
Francis, H. et al. H3 histamine receptor-mediated activation of protein kinase Cα inhibits the growth of cholangiocarcinoma in vitro and in vivo. Mol. Cancer Res. 7, 1704–1713 (2009).
Bockorny, B. et al. RAS-MAPK reactivation facilitates acquired resistance in FGFR1-amplified lung cancer and underlies a rationale for upfront FGFR-MEK blockade. Mol. Cancer Ther. 17, 1526–1539 (2018).
Namikawa, M. et al. Simultaneous activation of Kras-Akt and Notch pathways induces extrahepatic biliary cancer via the mTORC1 pathway. J. Pathol. 260, 478–492 (2023).
Quinn, L. M. et al. Genomic profiling of idiopathic peri-hilar cholangiocarcinoma reveals new targets and mutational pathways. Sci. Rep. 13, 6681 (2023).
Li, J. et al. Biliary repair and carcinogenesis are mediated by IL-33-dependent cholangiocyte proliferation. J. Clin. Invest. 124, 3241–3251 (2014).
Olaizola, P. et al. Targeting NAE1-mediated protein hyper-NEDDylation halts cholangiocarcinogenesis and impacts on tumor-stroma crosstalk in experimental models. J. Hepatol. 77, 177–190 (2022).
Gao, Q. et al. Neddylation pathway is up-regulated in human intrahepatic cholangiocarcinoma and serves as a potential therapeutic target. Oncotarget 5, 7820–7832 (2014).
Zhao, B. et al. Knockdown of Nedd8-conjugating enzyme UBE2M suppresses the proliferation and induces the apoptosis of intrahepatic cholangiocarcinoma cells. Oncol. Rep. 42, 2670–2679 (2019).
Olaizola, P. et al. Targeting protein hyperSUMOylation halts cholangiocarcinoma progression by impairing cancer cell viability and tumor-stroma crosstalk. Hepatology https://doi.org/10.1097/HEP.0000000000001259 (2025).
Indramanee, S. et al. Terminal fucose mediates progression of human cholangiocarcinoma through EGF/EGFR activation and the Akt/Erk signaling pathway. Sci. Rep. 9, 17266 (2019).
Ament, C. E. et al. Aberrant fucosylation sustains the NOTCH and EGFR/NF-κB pathways and has a prognostic value in human intrahepatic cholangiocarcinoma. Hepatology https://doi.org/10.1097/HEP.0000000000000322 (2023).
Phoomak, C. et al. Overexpression of O-GlcNAc-transferase associates with aggressiveness of mass-forming cholangiocarcinoma. Asian Pac. J. Cancer Prev. 13 (Suppl.), 101–105 (2012).
Phoomak, C. et al. Mechanistic insights of O-GlcNAcylation that promote progression of cholangiocarcinoma cells via nuclear translocation of NF-κB. Sci. Rep. 6, 27853 (2016).
Phoomak, C. et al. O-GlcNAc-induced nuclear translocation of hnRNP-K is associated with progression and metastasis of cholangiocarcinoma. Mol. Oncol. 13, 338–357 (2019).
Phoomak, C. et al. High glucose levels boost the aggressiveness of highly metastatic cholangiocarcinoma cells via O-GlcNAcylation. Sci. Rep. 7, 43842 (2017).
Hu, S. et al. Differential enrichment of H3K9me3 in intrahepatic cholangiocarcinoma. BMC Med. Genomics 15, 185 (2022).
Chen, F. et al. New horizons in tumor microenvironment biology: challenges and opportunities. BMC Med. 13, 45 (2015).
Sirica, A. E. & Gores, G. J. Desmoplastic stroma and cholangiocarcinoma clinical implications and therapeutic targeting. Hepatology 59, 2397–2402 (2014).
Cadamuro, M., Morton, S. D., Strazzabosco, M. & Fabris, L. Unveiling the role of tumor reactive stroma in cholangiocarcinoma: an opportunity for new therapeutic strategies. Transl. Gastrointest. Cancer 2, 130–144 (2013).
Cadamuro, M. et al. Platelet-derived growth factor-D and Rho GTPases regulate recruitment of cancer-associated fibroblasts in cholangiocarcinoma. Hepatology 58, 1042–1053 (2013).
Fingas, C. D. et al. Myofibroblast-derived PDGF-BB promotes Hedgehog survival signaling in cholangiocarcinoma cells. Hepatology 54, 2076–2088 (2011).
Veglia, F., Perego, M. & Gabrilovich, D. Myeloid-derived suppressor cells coming of age. Nat. Immunol. 19, 108–119 (2018).
Yang, X. et al. FAP promotes immunosuppression by cancer-associated fibroblasts in the tumor microenvironment via STAT3-CCL2 signaling. Cancer Res. 76, 4124–4135 (2016).
Cheng, J. T. et al. Hepatic carcinoma-associated fibroblasts induce IDO-producing regulatory dendritic cells through IL-6-mediated STAT3 activation. Oncogenesis 5, e198 (2016).
Luo, C. et al. Tumor-derived exosomes induce immunosuppressive macrophages to foster intrahepatic cholangiocarcinoma progression. Hepatology 76, 982–999 (2022).
Roy, S., Glaser, S. & Chakraborty, S. Inflammation and progression of cholangiocarcinoma: role of angiogenic and lymphangiogenic mechanisms. Front. Med. 6, 293 (2019).
Scimeca, M. et al. Programmed cell death pathways in cholangiocarcinoma: opportunities for targeted therapy. Cancers 15, 3638 (2023).
Pan, G. et al. Reciprocal co-expression of Fas and Fas ligand in human cholangiocarcinoma. Int. J. Oncol. 31, 843–850 (2007).
Carnevale, G. et al. Activation of Fas/FasL pathway and the role of c-FLIP in primary culture of human cholangiocarcinoma cells. Sci. Rep. 7, 14419 (2017).
Vucur, M. et al. Sublethal necroptosis signaling promotes inflammation and liver cancer. Immunity 56, 1578–1595.e8 (2023).
Seehawer, M. et al. Necroptosis microenvironment directs lineage commitment in liver cancer. Nature 562, 69–75 (2018).
Kubo, Y. et al. Different expression of glucose transporters in the progression of intrahepatic cholangiocarcinoma. Hum. Pathol. 45, 1610–1617 (2014).
Suzuki, H. et al. Relationship between 18-F-fluoro-deoxy-D-glucose uptake and expression of glucose transporter 1 and pyruvate kinase M2 in intrahepatic cholangiocarcinoma. Dig. Liver Dis. 47, 590–596 (2015).
Wappler, J. et al. Glutamine deprivation counteracts hypoxia-induced chemoresistance. Neoplasia 22, 22–32 (2020).
Ruiz de Gauna, M. et al. Cholangiocarcinoma progression depends on the uptake and metabolization of extracellular lipids. Hepatology 76, 1617–1633 (2022).
Raggi, C. et al. Cholangiocarcinoma stem-like subset shapes tumor-initiating niche by educating associated macrophages. J. Hepatol. 66, 102–115 (2017).
Raggi, C. et al. Epigenetic reprogramming modulates malignant properties of human liver cancer. Hepatology 59, 2251–2262 (2014).
Raggi, C. et al. Mitochondrial oxidative metabolism contributes to a cancer stem cell phenotype in cholangiocarcinoma. J. Hepatol. 74, 1373–1385 (2021).
Raggi, C., Taddei, M. L., Rae, C., Braconi, C. & Marra, F. Metabolic reprogramming in cholangiocarcinoma. J. Hepatol. 77, 849–864 (2022).
Di Matteo, S. et al. Metformin exerts anti-cancerogenic effects and reverses epithelial-to-mesenchymal transition trait in primary human intrahepatic cholangiocarcinoma cells. Sci. Rep. 11, 2557 (2021).
Tamai, K. et al. BEX2 suppresses mitochondrial activity and is required for dormant cancer stem cell maintenance in intrahepatic cholangiocarcinoma. Sci. Rep. 10, 21592 (2020).
Kaewlert, W. et al. Overexpression of insulin receptor substrate 1 (IRS1) relates to poor prognosis and promotes proliferation, stemness, migration, and oxidative stress resistance in cholangiocarcinoma. Int. J. Mol. Sci. 24, 2428 (2023).
Thanee, M. et al. CD44 variant-dependent redox status regulation in liver fluke-associated cholangiocarcinoma: a target for cholangiocarcinoma treatment. Cancer Sci. 107, 991–1000 (2016).
Lin, Y. et al. CAFs shape myeloid-derived suppressor cells to promote stemness of intrahepatic cholangiocarcinoma through 5-lipoxygenase. Hepatology 75, 28–42 (2022).
Banales, J. M. et al. Serum metabolites as diagnostic biomarkers for cholangiocarcinoma, hepatocellular carcinoma, and primary sclerosing cholangitis. Hepatology 70, 547–562 (2019).
Macias, R. I. R. et al. A novel serum metabolomic profile for the differential diagnosis of distal cholangiocarcinoma and pancreatic ductal adenocarcinoma. Cancers 12, 1433 (2020).
Padthaisong, S. et al. Integration of global metabolomics and lipidomics approaches reveals the molecular mechanisms and the potential biomarkers for postoperative recurrence in early-stage cholangiocarcinoma. Cancer Metab. 9, 30 (2021).
Suksawat, M. et al. Metabolic phenotyping predicts gemcitabine and cisplatin chemosensitivity in patients with cholangiocarcinoma. Front. Public Health 10, 766023 (2022).
Valle, J. W., Kelley, R. K., Nervi, B., Oh, D. Y. & Zhu, A. X. Biliary tract cancer. Lancet 397, 428–444 (2021).
Macias, R. I. R. et al. The search for risk, diagnostic, and prognostic biomarkers of cholangiocarcinoma and their biological and clinicopathologic significance. Am. J. Pathol. 195, 422–436 (2025).
Macias, R. I. R. et al. Diagnostic and prognostic biomarkers in cholangiocarcinoma. Liver Int. 39 (Suppl. 1), 108–122 (2019).
Rodrigues, P. M. et al. Next-generation biomarkers for cholangiocarcinoma. Cancers 13, 3222 (2021).
Grunnet, M. et al. Decline in CA19-9 during chemotherapy predicts survival in four independent cohorts of patients with inoperable bile duct cancer. Eur. J. Cancer 51, 1381–1388 (2015).
Han, J. Y. et al. Circulating microRNAs as biomarkers in bile-derived exosomes of cholangiocarcinoma. Ann. Surg. Treat. Res. 101, 140–150 (2021).
Hu, J. et al. A high-accuracy model based on plasma miRNAs diagnoses intrahepatic cholangiocarcinoma: a single center with 1001 samples. Diagnostics 11, 610 (2021).
Salem, P. E. S., Ghazala, R. A., El Gendi, A. M., Emara, D. M. & Ahmed, N. M. The association between circulating microRNA-150 level and cholangiocarcinoma. J. Clin. Lab. Anal. 34, e23397 (2020).
Han, H. S. et al. Bile-derived circulating extracellular miR-30d-5p and miR-92a-3p as potential biomarkers for cholangiocarcinoma. Hepatobiliary Pancreat. Dis. Int. 19, 41–50 (2020).
Meijer, L. L. et al. Unravelling the diagnostic dilemma: a microRNA panel of circulating MiR-16 and MiR-877 as a diagnostic classifier for distal bile duct tumors. Cancers 11, 1181 (2019).
Loosen, S. H. et al. Serum levels of miR-29, miR-122, miR-155 and miR-192 are elevated in patients with cholangiocarcinoma. PLoS ONE 14, e0210944 (2019).
Liu, C. H. et al. Circulating microRNA-21 as a prognostic, biological marker in cholangiocarcinoma. J. Cancer Res. Ther. 14, 220–225 (2018).
Correa-Gallego, C. et al. Circulating plasma levels of microRNA-21 and microRNA-221 are potential diagnostic markers for primary intrahepatic cholangiocarcinoma. PLoS ONE 11, e0163699 (2016).
Wu, X. et al. Profiling of downregulated blood-circulating miR-150-5p as a novel tumor marker for cholangiocarcinoma. Tumour Biol. 37, 15019–15029 (2016).
Cheng, Q. et al. Circulating miR-106a is a novel prognostic and lymph node metastasis indicator for cholangiocarcinoma. Sci. Rep. 5, 16103 (2015).
Plieskatt, J. et al. A microRNA profile associated with Opisthorchis viverrini-induced cholangiocarcinoma in tissue and plasma. BMC Cancer 15, 309 (2015).
Wang, S. et al. Upregulated circulating miR-150 is associated with the risk of intrahepatic cholangiocarcinoma. Oncol. Rep. 33, 819–825 (2015).
Silakit, R. et al. Circulating miR-192 in liver fluke-associated cholangiocarcinoma patients: a prospective prognostic indicator. J. Hepatobiliary Pancreat. Sci. 21, 864–872 (2014).
Louis, C. et al. TGFβ-induced circLTBP2 predicts a poor prognosis in intrahepatic cholangiocarcinoma and mediates gemcitabine resistance by sponging miR-338-3p. JHEP Rep. 5, 100900 (2023).
Hogdall, D. et al. Whole blood microRNAs capture systemic reprogramming and have diagnostic potential in patients with biliary tract cancer. J. Hepatol. 77, 1047–1058 (2022).
Berchuck, J. E. et al. The clinical landscape of cell-free DNA alterations in 1671 patients with advanced biliary tract cancer. Ann. Oncol. 33, 1269–1283 (2022).
Arechederra, M. et al. Next-generation sequencing of bile cell-free DNA for the early detection of patients with malignant biliary strictures. Gut 71, 1141–1151 (2022).
Cooley, M. A. et al. Utility of methylated DNA markers for the diagnosis of malignant biliary strictures. Hepatology https://doi.org/10.1097/HEP.0000000000000970 (2024).
Lapitz, A. et al. Extracellular vesicles in hepatobiliary malignancies. Front. Immunol. 9, 2270 (2018).
Arbelaiz, A. et al. Serum extracellular vesicles contain protein biomarkers for primary sclerosing cholangitis and cholangiocarcinoma. Hepatology 66, 1125–1143 (2017).
Lapitz, A. et al. Patients with cholangiocarcinoma present specific RNA profiles in serum and urine extracellular vesicles mirroring the tumor expression: novel liquid biopsy biomarkers for disease diagnosis. Cells 9, 721 (2020).
Lapitz, A. et al. Liquid biopsy-based protein biomarkers for risk prediction, early diagnosis and prognostication of cholangiocarcinoma. J. Hepatol. https://doi.org/10.1016/j.jhep.2023.02.027 (2023).
Hogdall, D. et al. Serum IL6 as a prognostic biomarker and IL6R as a therapeutic target in biliary tract cancers. Clin. Cancer Res. 26, 5655–5667 (2020).
Hanahan, D. Hallmarks of cancer: new dimensions. Cancer Discov. 12, 31–46 (2022).
Satriano, L., Lewinska, M., Rodrigues, P. M., Banales, J. M. & Andersen, J. B. Metabolic rearrangements in primary liver cancers: cause and consequences. Nat. Rev. Gastroenterol. Hepatol. 16, 748–766 (2019).
Borger, D. R. et al. Circulating oncometabolite 2-hydroxyglutarate is a potential surrogate biomarker in patients with isocitrate dehydrogenase-mutant intrahepatic cholangiocarcinoma. Clin. Cancer Res. 20, 1884–1890 (2014).
Albiin, N. et al. Detection of cholangiocarcinoma with magnetic resonance spectroscopy of bile in patients with and without primary sclerosing cholangitis. Acta Radiol. 49, 855–862 (2008).
Song, W. S. et al. Discovery of glycocholic acid and taurochenodeoxycholic acid as phenotypic biomarkers in cholangiocarcinoma. Sci. Rep. 8, 11088 (2018).
Urman, J. M. et al. Pilot multi-omic analysis of human bile from benign and malignant biliary strictures: a machine-learning approach. Cancers 12, 1644 (2020).
Marin, J. J., Macias, R. I., Briz, O., Banales, J. M. & Monte, M. J. Bile acids in physiology, pathology and pharmacology. Curr. Drug Metab. 17, 4–29 (2015).
Gallego-Duran, R. et al. Metabolic-associated fatty liver disease: from simple steatosis toward liver cirrhosis and potential complications. Proceedings of the third translational hepatology meeting, organized by the Spanish Association for the Study of the Liver (AEEH). Gastroenterol. Hepatol. 45, 724–734 (2022).
Rushbrook, S. M. et al. British Society of Gastroenterology guidelines for the diagnosis and management of cholangiocarcinoma. Gut 73, 16–46 (2023).
Elmunzer, B. J. et al. ACG clinical guideline: diagnosis and management of biliary strictures. Am. J. Gastroenterol. 118, 405–426 (2023).
Wang, J. et al. More endoscopy-based brushing passes improve the detection of malignant biliary strictures: a multicenter randomized controlled trial. Am. J. Gastroenterol. 117, 733–739 (2022).
Baroud, S. et al. Impact of trimodality sampling on detection of malignant biliary strictures compared with patients with primary sclerosing cholangitis. Gastrointest. Endosc. 95, 884–892 (2022).
Fujii-Lau, L. L. et al. American Society for Gastrointestinal Endoscopy guideline on role of endoscopy in the diagnosis of malignancy in biliary strictures of undetermined etiology: methodology and review of evidence. Gastrointest. Endosc. 98, 694–712.e8 (2023).
Spolverato, G. et al. Can hepatic resection provide a long-term cure for patients with intrahepatic cholangiocarcinoma? Cancer 121, 3998–4006 (2015).
van Keulen, A. M. et al. Actual 10-year survival after resection of perihilar cholangiocarcinoma: what factors preclude a chance for cure? Cancers 13, 6260 (2021).
van Keulen, A. M. et al. Major complications and mortality after resection of intrahepatic cholangiocarcinoma: a systematic review and meta-analysis. Surgery 173, 973–982 (2023).
Franken, L. C. et al. Morbidity and mortality after major liver resection in patients with perihilar cholangiocarcinoma: a systematic review and meta-analysis. Surgery 165, 918–928 (2019).
van Keulen, A. M. et al. Multivariable prediction model for both 90-day mortality and long-term survival for individual patients with perihilar cholangiocarcinoma: does the predicted survival justify the surgical risk? Br. J. Surg. 110, 599–605 (2023).
Ruys, A. T., Busch, O. R., Gouma, D. J. & van Gulik, T. M. Staging laparoscopy for hilar cholangiocarcinoma: is it still worthwhile? Ann. Surg. Oncol. 18, 2647–2653 (2011).
Bird, N. et al. Role of staging laparoscopy in the stratification of patients with perihilar cholangiocarcinoma. Br. J. Surg. 104, 418–425 (2017).
Coelen, R. J. et al. Development of a risk score to predict detection of metastasized or locally advanced perihilar cholangiocarcinoma at staging laparoscopy. Ann. Surg. Oncol. 23, 904–910 (2016).
Davidson, J. T. T. et al. Staging laparoscopy among three subtypes of extra-hepatic biliary malignancy: a 15-year experience from 10 institutions. J. Surg. Oncol. 119, 288–294 (2019).
Sposito, C. et al. Survival benefit of adequate lymphadenectomy in patients undergoing liver resection for clinically node-negative intrahepatic cholangiocarcinoma. J. Hepatol. 78, 356–363 (2023).
Koerkamp, B. G., Clavien, P. A. & Polak, W. G. Surgical resection for intrahepatic cholangiocarcinoma — can we really improve survival by resecting more lymph nodes? J. Hepatol. 78, 235–237 (2023).
Franssen, S. et al. Comparison of hepatic arterial infusion pump chemotherapy vs resection for patients with multifocal intrahepatic cholangiocarcinoma. JAMA Surg. 157, 590–596 (2022).
Turner, K. M. et al. A national assessment of T2 staging for intrahepatic cholangiocarcinoma and the poor prognosis associated with multifocality. Ann. Surg. Oncol. 29, 5094–5102 (2022).
Wright, G. P. et al. Surgical resection does not improve survival in multifocal intrahepatic cholangiocarcinoma: a comparison of surgical resection with intra-arterial therapies. Ann. Surg. Oncol. 25, 83–90 (2018).
Keulen, A. V. et al. Success, complication, and mortality rates of initial biliary drainage in patients with unresectable perihilar cholangiocarcinoma. Surgery 172, 1606–1613 (2022).
Nooijen, L. E. et al. Long-term follow-up of a randomized trial of biliary drainage in perihilar cholangiocarcinoma. HPB 25, 210–217 (2023).
Ebata, T. et al. Surgical resection for Bismuth type IV perihilar cholangiocarcinoma. Br. J. Surg. 105, 829–838 (2018).
van Vugt, J. L. A. et al. The prognostic value of portal vein and hepatic artery involvement in patients with perihilar cholangiocarcinoma. HPB 20, 83–92 (2018).
Primavesi, F. et al. E-AHPBA-ESSO-ESSR Innsbruck consensus guidelines for preoperative liver function assessment before hepatectomy. Br. J. Surg. 110, 1331–1347 (2023).
Nagino, M. et al. Two hundred forty consecutive portal vein embolizations before extended hepatectomy for biliary cancer: surgical outcome and long-term follow-up. Ann. Surg. 243, 364–372 (2006).
Heil, J. et al. Preoperative portal vein or portal and hepatic vein embolization: DRAGON collaborative group analysis. Br. J. Surg. 108, 834–842 (2021).
De Vreede, I. et al. Prolonged disease-free survival after orthotopic liver transplantation plus adjuvant chemoirradiation for cholangiocarcinoma. Liver Transpl. 6, 309–316 (2000).
Rosen, C. B., Heimbach, J. K. & Gores, G. J. Liver transplantation for cholangiocarcinoma. Transpl. Int. 23, 692–697 (2010).
Tan, E. K., Taner, T., Heimbach, J. K., Gores, G. J. & Rosen, C. B. Liver transplantation for peri-hilar cholangiocarcinoma. J. Gastrointest. Surg. 24, 2679–2685 (2020).
Ito, T. et al. A 3-decade, single-center experience of liver transplantation for cholangiocarcinoma: impact of era, tumor size, location, and neoadjuvant therapy. Liver Transpl. 28, 386–396 (2022).
Loveday, B. P. T. et al. Neoadjuvant hyperfractionated chemoradiation and liver transplantation for unresectable perihilar cholangiocarcinoma in Canada. J. Surg. Oncol. 117, 213–219 (2018).
Abdelrahim, M. et al. Gemcitabine and cisplatin as neo-adjuvant for cholangiocarcinoma patients prior to liver transplantation: case-series. Curr. Oncol. 29, 3585–3594 (2022).
Hoogwater, F. J. H. et al. Role of neoadjuvant chemoradiotherapy in liver transplantation for unresectable perihilar cholangiocarcinoma: multicentre, retrospective cohort study. BJS Open 7, zrad025 (2023).
Vugts, J. J. A. et al. Eligibility for liver transplantation in patients with perihilar cholangiocarcinoma. Ann. Surg. Oncol. 28, 1483–1492 (2021).
Sapisochin, G. et al. “Very early” intrahepatic cholangiocarcinoma in cirrhotic patients: should liver transplantation be reconsidered in these patients? Am. J. Transpl. 14, 660–667 (2014).
Sapisochin, G. et al. Liver transplantation for “very early” intrahepatic cholangiocarcinoma: international retrospective study supporting a prospective assessment. Hepatology 64, 1178–1188 (2016).
De Martin, E. et al. Analysis of liver resection versus liver transplantation on outcome of small intrahepatic cholangiocarcinoma and combined hepatocellular-cholangiocarcinoma in the setting of cirrhosis. Liver Transpl. 26, 785–798 (2020).
Lunsford, K. E. et al. Liver transplantation for locally advanced intrahepatic cholangiocarcinoma treated with neoadjuvant therapy: a prospective case-series. Lancet Gastroenterol. Hepatol. 3, 337–348 (2018).
McMillan, R. R. et al. Survival following liver transplantation for locally advanced, unresectable intrahepatic cholangiocarcinoma. Am. J. Transpl. 22, 823–832 (2022).
Bourien, H. et al. Locoregional treatment in intrahepatic cholangiocarcinoma: which treatment for which patient? Cancers 15, 4217 (2023).
Mauro, E. et al. New challenges in the management of cholangiocarcinoma: the role of liver transplantation, locoregional therapies, and systemic therapy. Cancers 15, 1244 (2023).
Adeva, J. et al. Medical treatment for cholangiocarcinoma. Liver Int. 39 (Suppl. 1), 123–142 (2019).
Valle, J. W. et al. Biliary cancer: ESMO clinical practice guidelines for diagnosis, treatment and follow-up. Ann. Oncol. 27, v28–v37 (2016).
Kim, J. H., Won, H. J., Shin, Y. M., Kim, K. A. & Kim, P. N. Radiofrequency ablation for the treatment of primary intrahepatic cholangiocarcinoma. AJR Am. J. Roentgenol. 196, W205–W209 (2011).
Fu, Y. et al. Radiofrequency ablation in the management of unresectable intrahepatic cholangiocarcinoma. J. Vasc. Interv. Radiol. 23, 642–649 (2012).
Edeline, J. et al. Locoregional therapies in patients with intrahepatic cholangiocarcinoma: a systematic review and pooled analysis. Cancer Treat. Rev. 99, 102258 (2021).
Diaz-Gonzalez, A. et al. Thermal ablation for intrahepatic cholangiocarcinoma in cirrhosis: safety and efficacy in non-surgical patients. J. Vasc. Interv. Radiol. 31, 710–719 (2020).
Hawkins, M. A. V. et al. Addition of stereotactic body radiotherapy (SBRT) to systemic chemotherapy in locally advanced cholangiocarcinoma (CC) (ABC-07): results from a randomized phase II trial. J. Clin. Oncol. 42, 4006 (2024).
Paprottka, K. J. et al. Outcome and safety after 103 radioembolizations with Yttrium-90 resin microspheres in 73 patients with unresectable intrahepatic cholangiocarcinoma — an evaluation of predictors. Cancers 13, 5399 (2021).
Gangi, A. et al. Intrahepatic cholangiocarcinoma treated with transarterial Yttrium-90 glass microsphere radioembolization: results of a single institution retrospective study. J. Vasc. Interv. Radiol. 29, 1101–1108 (2018).
Buettner, S. et al. Yttrium-90 radioembolization in intrahepatic cholangiocarcinoma: a multicenter retrospective analysis. J. Vasc. Interv. Radiol. 31, 1035–1043.e2 (2020).
Mouli, S. et al. Yttrium-90 radioembolization for intrahepatic cholangiocarcinoma: safety, response, and survival analysis. J. Vasc. Interv. Radiol. 24, 1227–1234 (2013).
Levillain, H. et al. Personalised radioembolization improves outcomes in refractory intra-hepatic cholangiocarcinoma: a multicenter study. Eur. J. Nucl. Med. Mol. Imaging 46, 2270–2279 (2019).
Bargellini, I. et al. Yttrium-90 radioembolization in unresectable intrahepatic cholangiocarcinoma: results of a multicenter retrospective study. Cardiovasc. Interv. Radiol. 43, 1305–1314 (2020).
White, J. et al. Yttrium-90 transarterial radioembolization for chemotherapy-refractory intrahepatic cholangiocarcinoma: a prospective, observational study. J. Vasc. Interv. Radiol. 30, 1185–1192 (2019).
Kohler, M. et al. Prognostic factors for overall survival in advanced intrahepatic cholangiocarcinoma treated with Yttrium-90 radioembolization. J. Clin. Med. 9, 56 (2019).
Schartz, D. A. et al. Transarterial Yttrium-90 radioembolization for unresectable intrahepatic cholangiocarcinoma: a systematic review and meta-analysis. J. Vasc. Interv. Radiol. 33, 679–686 (2022).
Edeline, J. et al. Radioembolization plus chemotherapy for first-line treatment of locally advanced intrahepatic cholangiocarcinoma: a phase 2 clinical trial. JAMA Oncol. 6, 51–59 (2020).
Edeline, J. et al. Chemotherapy with or without selective internal radiation therapy for intrahepatic cholangiocarcinoma: data from clinical trials. Hepatology 79, 96–106 (2024).
Martin, R. C. G. 2nd et al. Drug-eluting bead, irinotecan therapy of unresectable intrahepatic cholangiocarcinoma (DELTIC) with concomitant systemic gemcitabine and cisplatin. Ann. Surg. Oncol. 29, 5462–5473 (2022).
Franssen, S. et al. Hepatic arterial infusion pump chemotherapy in patients with advanced intrahepatic cholangiocarcinoma confined to the liver: a multicenter phase II trial. J. Clin. Oncol. https://doi.org/10.1200/JCO-25-00923 (2025).
Edeline, J. et al. Gemcitabine and oxaliplatin chemotherapy or surveillance in resected biliary tract cancer (PRODIGE 12-ACCORD 18-UNICANCER GI): a randomized phase III study. J. Clin. Oncol. 37, 658–667 (2019).
Ebata, T. et al. Randomized clinical trial of adjuvant gemcitabine chemotherapy versus observation in resected bile duct cancer. Br. J. Surg. 105, 192–202 (2018).
Edeline, J. et al. Individual patient data meta-analysis of adjuvant gemcitabine-based chemotherapy for biliary tract cancer: combined analysis of the BCAT and PRODIGE-12 studies. Eur. J. Cancer 164, 80–87 (2022).
Primrose, J. N. et al. Capecitabine compared with observation in resected biliary tract cancer (BILCAP): a randomised, controlled, multicentre, phase 3 study. Lancet Oncol. 20, 663–673 (2019).
Bridgewater, J. et al. Long-term outcomes and exploratory analyses of the randomized phase III BILCAP study. J. Clin. Oncol. 40, 2048–2057 (2022).
Jeong, H. et al. Adjuvant gemcitabine plus cisplatin versus capecitabine in node-positive extrahepatic cholangiocarcinoma: the STAMP randomized trial. Hepatology 77, 1540–1549 (2023).
Nakachi, K. et al. Adjuvant S-1 compared with observation in resected biliary tract cancer (JCOG1202, ASCOT): a multicentre, open-label, randomised, controlled, phase 3 trial. Lancet 401, 195–203 (2023).
Valle, J. et al. Cisplatin plus gemcitabine versus gemcitabine for biliary tract cancer. N. Engl. J. Med. 362, 1273–1281 (2010).
Valle, J. W. et al. Addition of ramucirumab or merestinib to standard first-line chemotherapy for locally advanced or metastatic biliary tract cancer: a randomised, double-blind, multicentre, phase 2 study. Lancet Oncol. 22, 1468–1482 (2021).
Chen, J. S. et al. A KRAS mutation status-stratified randomized phase II trial of gemcitabine and oxaliplatin alone or in combination with cetuximab in advanced biliary tract cancer. Ann. Oncol. 26, 943–949 (2015).
Malka, D. et al. Gemcitabine and oxaliplatin with or without cetuximab in advanced biliary-tract cancer (BINGO): a randomised, open-label, non-comparative phase 2 trial. Lancet Oncol. 15, 819–828 (2014).
Lee, J. et al. Gemcitabine and oxaliplatin with or without erlotinib in advanced biliary-tract cancer: a multicentre, open-label, randomised, phase 3 study. Lancet Oncol. 13, 181–188 (2012).
Phelip, J. M. et al. Modified FOLFIRINOX versus CISGEM chemotherapy for patients with advanced biliary tract cancer (PRODIGE 38 AMEBICA): a randomized phase II study. J. Clin. Oncol. 40, 262–271 (2022).
Shroff, R. T. G. et al. SWOG 1815 A phase III randomized trial of gemcitabine, cisplatin, and nab-paclitaxel versus gemcitabine and cisplatin in newly diagnosed, advanced biliary tract cancers. J. Clin. Oncol. 43, 536–544 (2025).
Ioka, T. et al. Randomized phase III study of gemcitabine, cisplatin plus S-1 versus gemcitabine, cisplatin for advanced biliary tract cancer (KHBO1401- MITSUBA). J. Hepatobiliary Pancreat. Sci. 30, 102–110 (2023).
Kim, S. T. et al. Capecitabine plus oxaliplatin versus gemcitabine plus oxaliplatin as first-line therapy for advanced biliary tract cancers: a multicenter, open-label, randomized, phase III, noninferiority trial. Ann. Oncol. 30, 788–795 (2019).
Hyung, J. et al. Clinical benefit of maintenance therapy for advanced biliary tract cancer patients showing no progression after first-line gemcitabine plus cisplatin. Cancer Res. Treat. 51, 901–909 (2019).
Oh, D. Y. et al. Durvalumab plus gemcitabine and cisplatin in advanced biliary tract cancer. NEJM Evid. 1, EVIDoa2200015 (2022).
Oh, D. Y. et al. Durvalumab or placebo plus gemcitabine and cisplatin in participants with advanced biliary tract cancer (TOPAZ-1): updated overall survival from a randomised phase 3 study. Lancet Gastroenterol. Hepatol. 9, 694–704 (2024).
Kelley, R. K. et al. Pembrolizumab in combination with gemcitabine and cisplatin compared with gemcitabine and cisplatin alone for patients with advanced biliary tract cancer (KEYNOTE-966): a randomised, double-blind, placebo-controlled, phase 3 trial. Lancet 401, 1853–1865 (2023).
Finn, R. S. et al. Three-year follow-up KEYNOTE-966: pembrolizumab (pembro) plus gemcitabine and cisplatin (gem/cis) compared with gem/cis alone for patients (pts) with advanced biliary tract cancer (BTC). J. Clin. Oncol. 42 (Suppl. 16), 4093 (2024).
Lamarca, A. et al. Second-line FOLFOX chemotherapy versus active symptom control for advanced biliary tract cancer (ABC-06): a phase 3, open-label, randomised, controlled trial. Lancet Oncol. 22, 690–701 (2021).
Hyung, J. et al. Treatment with liposomal irinotecan plus fluorouracil and leucovorin for patients with previously treated metastatic biliary tract cancer: the phase 2b NIFTY randomized clinical trial. JAMA Oncol. 9, 692–699 (2023).
Vogel, A. et al. 53MO Nal-IRI and 5-FU/LV compared to 5-FU/LV in patients with cholangio- and gallbladder carcinoma previously treated with gemcitabine-based therapies (NALIRICC – AIO-HEP-0116). Ann. Oncol. 33, S19–S26 (2022).
Delaye, M. et al. Real-life data on biliary tract cancers in France: the nested Amber study from the French ACABi GERCOR PRONOBIL retro-prospective, observational cohort. Dig. Liver Dis. https://doi.org/10.1016/j.dld.2024.06.032 (2024).
Yoo, C. et al. 58P Efficacy and safety of liposomal irinotecan (nal-IRI) plus fluorouracil (5-FU) and leucovorin (LV) versus 5-FU/LV alone for advanced biliary tract cancer (BTC) after progression on gemcitabine-based therapy: a pooled analysis of the NIFTY and NALIRICC trials. Ann. Oncol. 35, S235 (2024).
Choi, I. S. et al. A randomised phase II study of oxaliplatin/5-FU (mFOLFOX) versus irinotecan/5-FU (mFOLFIRI) chemotherapy in locally advanced or metastatic biliary tract cancer refractory to first-line gemcitabine/cisplatin chemotherapy. Eur. J. Cancer 154, 288–295 (2021).
Lamarca, A. et al. Molecular profiling in daily clinical practice: practicalities in advanced cholangiocarcinoma and other biliary tract cancers. J. Clin. Med. 9, 2854 (2020).
Maruki, Y. et al. Clinical characteristics and the course of hepatobiliary and pancreatic malignant tumors with high microsatellite instability after pembrolizumab administration: a multicenter observational study. J. Clin. Oncol. 42 (Suppl. 3), 459 (2024).
Chae, H. et al. Therapeutic relevance of targeted sequencing in management of patients with advanced biliary tract cancer: DNA damage repair gene mutations as a predictive biomarker. Eur. J. Cancer 120, 31–39 (2019).
Lamarca, A. P. et al. Significance of alterations in DNA damage repair (DDR) genes in advanced biliary cancers (ABCs) treated with second-line active-symptom-control (ASC) alone or ASC with oxaliplatin/5-FU chemotherapy (ASC + FOLFOX) in the randomised phase III, multicentre, open-label ABC-06 trial. J. Clin. Oncol. 41, 593 (2023).
Zhu, A. X. et al. Final overall survival efficacy results of ivosidenib for patients with advanced cholangiocarcinoma with IDH1 mutation: the phase 3 randomized clinical ClarIDHy trial. JAMA Oncol. 7, 1669–1677 (2021).
Javle, M. et al. Infigratinib (BGJ398) in previously treated patients with advanced or metastatic cholangiocarcinoma with FGFR2 fusions or rearrangements: mature results from a multicentre, open-label, single-arm, phase 2 study. Lancet Gastroenterol. Hepatol. 6, 803–815 (2021).
Goyal, L. et al. Futibatinib for FGFR2-rearranged intrahepatic cholangiocarcinoma. N. Engl. J. Med. 388, 228–239 (2023).
Pant, S. et al. Erdafitinib in patients with advanced solid tumours with FGFR alterations (RAGNAR): an international, single-arm, phase 2 study. Lancet Oncol. 24, 925–935 (2023).
Javle, M. M. et al. Efficacy of derazantinib in intrahepatic cholangiocarcinoma patients with FGFR2 mutations or amplifications: interim results from the phase 2 study FIDES-01. J. Clin. Oncol. 40, 427 (2022).
Park, J. O. F. et al. Updated results of a phase IIa study to evaluate the clinical efficacy and safety of erdafitinib in Asian advanced cholangiocarcinoma (CCA) patients with FGFR alterations. J. Clin. Oncol. 37, 4117 (2019).
Abou-Alfa, G. K. et al. Pemigatinib for previously treated, locally advanced or metastatic cholangiocarcinoma: a multicentre, open-label, phase 2 study. Lancet Oncol. 21, 671–684 (2020).
Subbiah, V. et al. RLY-4008, the first highly selective FGFR2 inhibitor with activity across FGFR2 alterations and resistance mutations. Cancer Discov. 13, 2012–2031 (2023).
Javle, M. et al. Pertuzumab and trastuzumab for HER2-positive, metastatic biliary tract cancer (MyPathway): a multicentre, open-label, phase 2a, multiple basket study. Lancet Oncol. 22, 1290–1300 (2021).
Cannon, T. L. et al. Pertuzumab plus trastuzumab in patients with biliary tract cancer with ERBB2/3 alterations: results from the targeted agent and profiling utilization registry study. J. Clin. Oncol. 42, 3228–3237 (2024).
Harding, J. J. et al. Zanidatamab for HER2-amplified, unresectable, locally advanced or metastatic biliary tract cancer (HERIZON-BTC-01): a multicentre, single-arm, phase 2b study. Lancet Oncol. 24, 772–782 (2023).
Nakamura, Y. et al. Tucatinib and trastuzumab for previously treated human epidermal growth factor receptor 2-positive metastatic biliary tract cancer (SGNTUC-019): a phase II basket study. J. Clin. Oncol. 41, 5569–5578 (2023).
Meric-Bernstam, F. et al. Efficacy and safety of trastuzumab deruxtecan in patients with HER2-expressing solid tumors: primary results from the DESTINY-PanTumor02 phase II trial. J. Clin. Oncol. 42, 47–58 (2024).
Subbiah, V. et al. Dabrafenib plus trametinib in patients with BRAF(V600E)-mutated biliary tract cancer (ROAR): a phase 2, open-label, single-arm, multicentre basket trial. Lancet Oncol. 21, 1234–1243 (2020).
Subbiah, V. et al. Dabrafenib plus trametinib in BRAFV600E-mutated rare cancers: the phase 2 ROAR trial. Nat. Med. 29, 1103–1112 (2023).
Meric-Bernstam, F. et al. Cobimetinib plus vemurafenib in patients with solid tumors with BRAF mutations: results from the targeted agent and profiling utilization registry study. JCO Precis. Oncol. 7, e2300385 (2023).
Drilon, A. et al. Efficacy of larotrectinib in TRK fusion-positive cancers in adults and children. N. Engl. J. Med. 378, 731–739 (2018).
Doebele, R. C. et al. Entrectinib in patients with advanced or metastatic NTRK fusion-positive solid tumours integrated analysis of three phase 1-2 trials. Lancet Oncol. 21, 271–282 (2020).
Subbiah, V. et al. Pan-cancer efficacy of pralsetinib in patients with RET fusion-positive solid tumors from the phase 1/2 ARROW trial. Nat. Med. 28, 1640–1645 (2022).
Subbiah, V. et al. Tumour-agnostic efficacy and safety of selpercatinib in patients with RET fusion-positive solid tumours other than lung or thyroid tumours (LIBRETTO-001): a phase 1/2, open-label, basket trial. Lancet Oncol. 23, 1261–1273 (2022).
Carrizosa, D. R. et al. CRESTONE: initial efficacy and safety of seribantumab in solid tumors harboring NRG1 fusions. J. Clin. Oncol. 40, 3006 (2022).
Schram, A. M. et al. Efficacy and safety of zenocutuzumab, a HER2 x HER3 bispecific antibody, across advanced NRG1 fusion (NRG1+) cancers. J. Clin. Oncol. 40, 105 (2022).
Verdaguer, H. et al. ESMO scale for clinical actionability of molecular targets driving targeted treatment in patients with cholangiocarcinoma. Clin. Cancer Res. 28, 1662–1671 (2022).
European Association for the Study of the Liver. EASL clinical practice guidelines on the management of extrahepatic cholangiocarcinoma. J. Hepatol. 83, 211–238 (2025).
Chene, P. Inhibiting the p53-MDM2 interaction: an important target for cancer therapy. Nat. Rev. Cancer 3, 102–109 (2003).
Kato, S. et al. Analysis of MDM2 amplification: next-generation sequencing of patients with diverse malignancies. JCO Precis. Oncol. 2018, PO.17.00235 (2018).
Gounder, M. M. et al. A first-in-human phase I study of milademetan, an MDM2 inhibitor, in patients with advanced liposarcoma, solid tumors, or lymphomas. J. Clin. Oncol. 41, 1714–1724 (2023).
LoRusso, P. et al. The MDM2-p53 antagonist brigimadlin (BI 907828) in patients with advanced or metastatic solid tumors: results of a phase Ia, first-in-human, dose-escalation study. Cancer Discov. 13, 1802–1813 (2023).
Hong, D. S. et al. KRAS(G12C) inhibition with sotorasib in advanced solid tumors. N. Engl. J. Med. 383, 1207–1217 (2020).
Bekaii-Saab, T. S. et al. Adagrasib in advanced solid tumors harboring a KRAS(G12C) mutation. J. Clin. Oncol. 41, 4097–4106 (2023).
Sacher, A. et al. Single-agent divarasib (GDC-6036) in solid tumors with a KRAS G12C mutation. N. Engl. J. Med. 389, 710–721 (2023).
Uprety, D. & Adjei, A. A. KRAS: from undruggable to a druggable cancer target. Cancer Treat. Rev. 89, 102070 (2020).
Hong, J. Y. et al. Claudin 18.2 expression in various tumor types and its role as a potential target in advanced gastric cancer. Transl. Cancer Res. 9, 3367–3374 (2020).
Papadopoulos, K. P. T. et al. A phase I/II dose escalation and expansion trial to evaluate safety and preliminary efficacy of BNT141 in patients with claudin-18.2-positive solid tumors. J. Clin. Oncol. 41, TPS2670 (2023).
Engstrom, L. D. et al. MRTX1719 is an MTA-cooperative PRMT5 inhibitor that exhibits synthetic lethality in preclinical models and patients with MTAP-deleted cancer. Cancer Discov. 13, 2412–2431 (2023).
Rodon, J. et al. First-in-human study of AMG 193, an MTA-cooperative PRMT5 inhibitor, in patients with MTAP-deleted solid tumors: results from phase I dose exploration. Ann. Oncol. https://doi.org/10.1016/j.annonc.2024.08.2339 (2024).
Sacher, A. G. et al. 604O Phase I dose escalation and initial dose expansion results of AMG 193: A MTA-cooperative PRMT5 inhibitor, in patients (pts) with MTAP-deleted solid tumors. Ann. Oncol. 35 (Suppl. 2), S484–S485 (2024).
Breast Cancer Linkage Consortium. Cancer risks in BRCA2 mutation carriers. J. Natl Cancer Inst. 91, 1310–1316 (1999).
Spizzo, G. et al. Molecular profile of BRCA-mutated biliary tract cancers. ESMO Open 5, e000682 (2020).
O’Bryan, J. Y. et al. Phase II clinical trial of olaparib plus pembrolizumab in the treatment of patients with advanced biliary tract cancer. J. Clin. Oncol. 41, 4087 (2023).
Olaizola, I. et al. New platinum derivatives selectively cause double-strand DNA breaks and death in naive and cisplatin-resistant cholangiocarcinomas. J. Hepatol. https://doi.org/10.1016/j.jhep.2025.04.034 (2025).
Koh, B. et al. Patterns in cancer incidence among people younger than 50 years in the US, 2010 to 2019. JAMA Netw. Open 6, e2328171 (2023).
Rahman, R. et al. Age trends in biliary tract cancer incidence by anatomical subtype: a Swedish cohort study. Eur. J. Cancer 175, 291–298 (2022).
Rimassa, L. et al. Mapping the landscape of biliary tract cancer in Europe: challenges and controversies. Lancet Reg. Health Eur. 50, 101171 (2025).
Khan, S. A. et al. Global trends in mortality from intrahepatic and extrahepatic cholangiocarcinoma. J. Hepatol. 71, 1261–1262 (2019).
Zalin-Miller, A. et al. Regional variation in routes to diagnosis of cholangiocarcinoma in England from 2006 to 2017. World J. Gastroenterol. 29, 3825–3842 (2023).
Morement, H. & Khuntikeo, N. Community awareness and education: in the West and Southeast Asia. Recent Results Cancer Res. 219, 349–359 (2023).
Casolino, R., Braconi, C. & BABEL working group of the COST Action 18122 (EURO-CHOLANGIO-NET), part of the European Network for the Study of Cholangiocarcinoma. Bridging the equity gap in patient education the biliary tract cancer BABEL project. Lancet Oncol. 23, 568–570 (2022).
Patel, N. et al. Understanding patient experience in biliary tract cancer: a qualitative patient interview study. Oncol. Ther. 9, 557–573 (2021).
Keilson, J. M. et al. Patient perspective: financial toxicity is a barrier to clinical trials and personalized therapy in cholangiocarcinoma. J. Surg. Oncol. 126, 1011 (2022).
Keilson, J. M. et al. Patient reported outcomes: financial toxicity is a barrier to clinical trials and personalized therapy in cholangiocarcinoma. J. Surg. Oncol. 126, 1003–1010 (2022).
Schuster, A. L. et al. Priorities to promote participant engagement in the participant engagement and cancer genome sequencing (PE-CGS) network. Cancer Epidemiol. Biomark. Prev. 32, 487–495 (2023).
Cholangiocarcinoma Working Group. Italian clinical practice guidelines on cholangiocarcinoma — Part II: treatment. Dig. Liver Dis. 52, 1430–1442 (2020).
Shroff, R. T. et al. Adjuvant therapy for resected biliary tract cancer: ASCO clinical practice guideline. J. Clin. Oncol. 37, 1015–1027 (2019).
Moffat, G. T. et al. KRAS allelic variants in biliary tract cancers. JAMA Netw. Open 7, e249840 (2024).
Mody, K. et al. Clinical, genomic, and transcriptomic data profiling of biliary tract cancer reveals subtype-specific immune signatures. JCO Precis. Oncol. 6, e2100510 (2022).
Zheng, Y. et al. Specific genomic alterations and prognostic analysis of perihilar cholangiocarcinoma and distal cholangiocarcinoma. J. Gastrointest. Oncol. 12, 2631–2642 (2021).
Valle, J. W., Lamarca, A., Goyal, L., Barriuso, J. & Zhu, A. X. New horizons for precision medicine in biliary tract cancers. Cancer Discov. 7, 943–962 (2017).
Carotenuto, M., Sacco, A., Forgione, L. & Normanno, N. Genomic alterations in cholangiocarcinoma: clinical significance and relevance to therapy. Explor. Target. Antitumor Ther. 3, 200–223 (2022).
Clements, O., Eliahoo, J., Kim, J. U., Taylor-Robinson, S. D. & Khan, S. A. Risk factors for intrahepatic and extrahepatic cholangiocarcinoma: a systematic review and meta-analysis. J. Hepatol. 72, 95–103 (2020).
Petrick, J. L. et al. Risk factors for intrahepatic and extrahepatic cholangiocarcinoma in the United States: a population-based study in SEER-Medicare. PLoS ONE 12, e0186643 (2017).
Shin, H. R. et al. Epidemiology of cholangiocarcinoma: an update focusing on risk factors. Cancer Sci. 101, 579–585 (2010).
Calvisi, D. F. et al. Criteria for preclinical models of cholangiocarcinoma: scientific and medical relevance. Nat. Rev. Gastroenterol. Hepatol. 20, 462–480 (2023).
Acknowledgements
The authors acknowledge the Spanish Carlos III Health Institute (ISCIII) (J.M.B. (FIS PI21/00922, PI24/00148 and Miguel Servet Program CPII19/00008); M.J.P. (FIS PI20/00186; PI23/01565); R.I.R.M. (FIS PI20/00189, PI23/00681); A.C. (PI19/00752); P.M.R. (Miguel Servet Program CP22/00073; FIS PI23/01850)) cofinanced by Fondo Europeo de Desarrollo Regional (FEDER); CIBERehd (ISCIII) to J.M.B., P.M.R., P.A., A.C., A.F., R.I.R.M., P.O. and M.J.P.; IKERBASQUE, Basque foundation for Science (to J.M.B. and P.M.R.); Spanish Ministry of Science, Innovation and Universities (CNS2024-154885 to P.M.R.); Diputación Foral Gipuzkoa (2023-CIEN-000008-01 to P.M.R.; 2023-CIEN-000045-01 to M.J.P.); Department of Health of the Basque Country (2019111024 and 2023111017 to M.J.P.; 2022111070 to P.M.R.; 2017111010, 2020111077 to J.M.B.), Euskadi RIS3 (2019222054, 2020333010, 2022333032 to J.M.B.; 2022333041 to M.J.P.); BIOEF (Basque Foundation for Innovation and Health Research EiTB Maratoia BIO15/CA/016/BD to J.M.B.); La Caixa Scientific Foundation (HR17-00601 to J.M.B.); Fundación Científica de la Asociación Española Contra el Cáncer (AECC Lab call 2023; LABAE235286RODR to P.M.R.); PSC Partners US (to J.M.B.) and PSC Support UK (to J.M.B. 06119JB; to J.M.B. and P.M.R. JMBPROJ23); European Union’s Horizon 2020 Research and Innovation Program (grant number 825510, ESCALON to J.M.B.); and AMMF — The Cholangiocarcinoma Charity (EU/2019/AMMFt/001 to J.M.B. and P.M.R.). M.J.P. was funded by the Spanish Ministry of Economy and Competitiveness (MINECO Ramón y Cajal Program RYC-2015-17755). C.C. acknowledges funding from the ERA-NET TRANSCAN-3 program (INCa_18688), French Ministry of Health and National Cancer Institute (PRT-K20-136). J.M.B., P.M.R. and A. Lleo received funding from the TRANSCAN-3 Joint Transnational Call (JTC 2024) under the European Union’s Horizon 2020 research and innovation programme (TRANSCAN2024-2930-061_TITAN). S.A. is funded by European Union grant agreement 101077312, the Ramón y Cajal Program RYC2022-036321-I, and PID2021-124694OA-I00 (MCIN/AEI/ 10.13039/501100011033 and by “FEDER Una manera de hacer Europa”). M.H. was supported by the Rainer-Hoenig Stiftung. C.R. was funded by the Associazione Italiana per la Ricerca sul Cancro (AIRC) (IG23117). A.S. is supported by the German Cancer Aid (70114101) and the Deutsche Forschungsgemeinschaft (DFG, German Research Foundation) project number 493345156. G.C. was supported by PNRR M4C2-Investimento 1.4-CN00000041, funded by the European Union – NextGenerationEU and by Project PNC 0000001 D3 4 Health, CUP B53C22006120001, The National Plan for Complementary Investments to the NRRP. A. Lleo received funding from the Associazione Italiana per la Ricerca sul Cancro (AIRC IG-2019-23408) and from the Italian Ministry of Health (NET-2019-12370049). A. Lamarca received funding from the Spanish Society of Medical Oncology (SEOM) Fellowship Programme (Return Fellowship) and the European Union’s Horizon 2020 Research. L.F. was funded by PRIN 2022AHM4AA. V.C. received funding from Next Generation Europe Grant National Center 3 — Spoke 2 CN 312184522E9D9A, Piano Nazionale Complementare Salute (PNC1221852F49EDDD). Some of the authors of this article are members of the European Reference Network (ERN) RARE-LIVER and/or the European Network for the Study of Cholangiocarcinoma (ENS-CCA). This publication is based upon work from the ENS-CCA and the COST-Action Precision-BTC-Network CA22125, supported by COST (European Cooperation in Science and Technology). The funding sources had no involvement in study design, data collection and analysis, decision to publish, or preparation of the article. Finally, we would like to thank those who completed the Delphi process, who are listed in Supplementary Table 1.
Author information
Authors and Affiliations
Consortia
Contributions
J.M.B., P.M.R. and C.B. researched data for the article, made a substantial contribution to discussion of content, wrote the article, and reviewed/edited the manuscript before submission. All other authors researched data for the article and wrote the article.
Corresponding authors
Ethics declarations
Competing interests
J.M.B. declares research grants from Incyte, Albireo-Ipsen, Servier, Taiho and Cymabay-Gilead, personal fees for lectures from Servier, AstraZeneca, Eisai, Advanz and Ipsen, and consulting fees for Ipsen, Cymabay, AstraZeneca, Servier, Rubió Metabolomics and Jazz Pharmaceuticals. J.B.A. declares research grant support from Incyte and Adcendo and consultancies from Flagship Pioneering, AstraZeneca and QED. A.C. declares speaker and advisory honoraria from Boston Scientific and has received grant support from Boston Scientific. V.C. declares travel support from Advanz Pharma, speaker honoraria from Ipsen and Mirum, and grant support from Ipsen. J.E. declares consulting fees from MSD, Eisai, BMS, AstraZeneca, Bayer, Roche, Ipsen, Basilea, Merck Serono, Incyte, Servier, Beigene, Taiho, Boston Scientific and Guerbet, travel expenses from Amgen, and research funding (institutional) from BMS, Beigene, Boston Scientific, Exeliom Biosciences and SUMMIT. A.F. declares speaker honoraria from AstraZeneca, Roche and Boston Scientific and advisory honoraria from AstraZeneca, Incyte, Boston Scientific and Taiho. A. Lamarca declares travel and educational support from Ipsen, Pfizer, Bayer, AAA, SirtEx, Novartis, Mylan, Delcath Advanz Pharma and Roche, speaker honoraria from Merck, Pfizer, Ipsen, Incyte, AAA/Novartis, QED, Servier, AstraZeneca, EISAI, Roche, Advanz Pharma and MSD, advisory and consultancy honoraria from EISAI, Nutricia, Ipsen, QED, Roche, Servier, Boston Scientific, Albireo Pharma, AstraZeneca, Boehringer Ingelheim, GENFIT, TransThera Biosciences, Taiho and MSD, and principal investigator-associated Institutional Funding from QED, Merck, Boehringer Ingelheim, Servier, AstraZeneca, GenFit, Albireo Pharma, Taiho, TransThera and Roche; she is also a member of the Knowledge Network and NETConnect Initiatives funded by Ipsen. A. Lleo declares travel support from Ipsen, principal investigator-associated institutional funding form Mirum, GSK, Ipsen, Dr. Falk and Gilead, consulting fees from Advanz Pharma, GSK, Ipsen, Gilead, Dr. Falk, AlfaSigma, Takeda, and Albireo Pharma, and speaker fees from Gilead, Abbvie, MSD, Advanz Pharma, AlfaSigma, GSK and Incyte. L.R. declares consulting fees from AbbVie, AstraZeneca, Basilea, Bayer, BMS, Boehringer Ingelheim, Eisai, Elevar Therapeutics, Exelixis, Genenta, Guerbet, Hengrui, Incyte, Ipsen, Jazz Pharmaceuticals, MSD, Nerviano Medical Sciences, Roche, Servier, Taiho Oncology and Zymeworks, lecture fees from AstraZeneca, Bayer, Biologix, BMS, Eisai, Guerbet, Incyte, Ipsen, Roche and Servier, travel expenses from AstraZeneca, Servier and research grants (to institution) from Abbvie, AstraZeneca, BeiGene, Exelixis, Fibrogen, Incyte, Ipsen, Jazz Pharmaceuticals, MSD, Nerviano Medical Sciences, Roche, Servier, Taiho Oncology, TransThera Sciences and Zymeworks. A.S. received honoraria as a speaker from BMS, Roche, Servier, Ipsen, Lilly, AstraZeneca, MSD and Eisai, travel support from Ipsen, Servier, Pierre-Fabre, MSD and Eisai, and consulting fees from Eisai, MSD, Roche, Incyte and BMS. J.W.V. declares personal fees for consulting/advisory role from AstraZeneca, Incyte, Taiho Oncology, Servier, and Jazz Pharmaceuticals and fees to the institution for consulting/advisory role from RedX Pharma, Cogent Biosciences, Owkin, Jazz Pharmaceuticals and Oncosil.
Peer review
Peer review information
Nature Reviews Gastroenterology & Hepatology thanks Milind Javle and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Related links
AMMF: https//ammf.org.uk/
Cholangiocarcinoma Foundation: https//cholangiocarcinoma.org/
COST: http://www.cost.eu/
Euro-Cholangio-Net: https://eurocholangionet.eu/
European Network for the Study of Cholangiocarcinoma: https//precision-btc.eu/enscca
Precision-BTC-Network: https://precision-btc.eu
Supplementary information
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Banales, J.M., Rodrigues, P.M., Affò, S. et al. Cholangiocarcinoma 2026: status quo, unmet needs and priorities. Nat Rev Gastroenterol Hepatol 23, 65–96 (2026). https://doi.org/10.1038/s41575-025-01153-w
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41575-025-01153-w
This article is cited by
-
Yttrium-90 Transarterial Radioembolization for Intrahepatic Cholangiocarcinoma: A Comprehensive Review
Journal of Gastrointestinal Cancer (2026)
-
Hypoalbuminemia as a prognostic marker for survival in biliary tract cancer: associations with tumor type, treatment and sex
Journal of Cancer Research and Clinical Oncology (2026)