Abstract
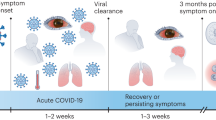
Post-COVID-19 condition (PCC), also known as long COVID, is a heterogeneous condition marked by persistent symptoms following acute SARS-CoV-2 infection. As approximately 6% of people who have experienced acute COVID-19 are estimated to develop PCC, the potential population is vast. Many of the key symptoms of PCC reflect involvement of the nervous system, ranging from cognitive impairment (‘brain fog’), headaches and fatigue to anxiety and depression. This Review summarizes the spectrum of neurological and psychological symptoms that occur following acute SARS-CoV-2 infection, with a particular focus on the international consensus-based core outcome set for PCC. We also explore the proposed underlying mechanisms, including evidence for immune system dysregulation, microvascular dysfunction and volumetric changes on neuroimaging. In addition, we review ongoing and completed large-scale treatment trials. Growing evidence suggests a bidirectional interaction between symptoms traditionally considered neurobiological in origin, such as cognitive deficits and headache, and those within the purview of psychiatry, such as anxiety and depression. PCC represents an opportunity to better understand the long-term consequences of acute infection and improve management strategies and outcomes, not only for people with the condition but also for those with other post-viral syndromes that affect brain health.
Key points
-
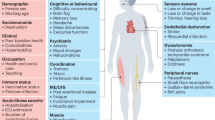
Post-COVID-19 condition (PCC) encompasses persistent symptoms including headache, cognitive impairment, fatigue, sleep disturbances, dysautonomia, pain and psychiatric disorders.
-
Systematic reviews and cohort studies highlight variable prevalence of PCC symptoms, and cognitive, sleep and fatigue symptoms often persist for 12–24 months or longer.
-
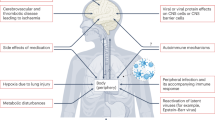
The proposed pathophysiology of PCC includes viral persistence, immune dysregulation, neuroinflammation, latent herpesvirus reactivation, microvascular injury, gut–brain axis disruption, and structural and functional brain changes.
-
Neurological and psychiatric symptoms substantially impair daily living, employment and health-related quality of life, with some deficits persisting years after infection.
-
Current approaches to PCC management are largely symptomatic and multidisciplinary, including rehabilitation, pacing strategies, cognitive behavioural therapy, pharmacological management for pain and dysautonomia, and sleep interventions. Graded exercise therapy is not recommended.
-
Completed and ongoing trials are exploring pharmacological, immunological and microbiome-targeted interventions, and vaccination remains a key modifiable factor in the reducing risk and severity of PCC.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$32.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$189.00 per year
only $15.75 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout

Similar content being viewed by others
References
World Health Organization. WHO Coronavirus (COVID-19) Dashboard (WHO, 2025); https://data.who.int/dashboards/covid19/cases?n=c2025.
Ray, S. T. J. et al. Neurological manifestations of SARS-CoV-2 infection in hospitalised children and adolescents in the UK: a prospective national cohort study. Lancet Child Adolesc. Health 5, 631–641 (2021).
Ross Russell AL et al. Spectrum, risk factors and outcomes of neurological and psychiatric complications of COVID-19: a UK-wide cross-sectional surveillance study. Brain Commun. 3, fcab168 (2021).
Varatharaj, A. et al. Neurological and neuropsychiatric complications of COVID-19 in 153 patients: a UK-wide surveillance study. Lancet Psychiatry 7, 875–882 (2020).
Chou, S. H. et al. Global incidence of neurological manifestations among patients hospitalized with COVID-19 — a report for the GCS–NeuroCOVID Consortium and the ENERGY Consortium. JAMA Netw. Open 4, e2112131 (2021).
Shi, J. et al. Prevalence of and factors associated with long COVID among US adults: a nationwide survey. BMC Public Health 25, 1758 (2025).
World Health Organization. Post COVID-19 condition (long COVID). who.int https://www.who.int/news-room/fact-sheets/detail/post-covid-19-condition-%28long-covid%29 (accessed 25 June 2025).
Soriano, J. B., Murthy, S., Marshall, J. C., Relan, P. & Diaz, J. V. A clinical case definition of post-COVID-19 condition by a Delphi consensus. Lancet Infect. Dis. 22, e102–e107 (2022).
Munblit, D. et al. A core outcome set for post-COVID-19 condition in adults for use in clinical practice and research: an international Delphi consensus study. Lancet Respir. Med. 10, 715–724 (2022).
Adjaye-Gbewonyo D., Vahratian A., Perrine C. G. & Bertolli J. Long COVID in adults: United States, 2022. NCHS Data Brief. 480, 1–8 (2023).
Hou, Y. et al. Global prevalence of long COVID, its subtypes, and risk factors: an updated systematic review and meta-analysis. Open Forum Infect. Dis. 12, ofaf533 (2025).
Elboraay, T. et al. Long-term neurological and cognitive impact of COVID-19: a systematic review and meta-analysis in over 4 million patients. BMC Neurol. 25, 250 (2025).
Subramanian, A. et al. Symptoms and risk factors for long COVID in non-hospitalized adults. Nat. Med. 28, 1706–1714 (2022).
Sudre, C. H. et al. Attributes and predictors of long COVID. Nat. Med. 27, 626–631 (2021).
Thompson, E. J. et al. Long COVID burden and risk factors in 10 UK longitudinal studies and electronic health records. Nat. Commun. 13, 3528 (2022).
Burn, E. et al. Venous or arterial thrombosis and deaths among COVID-19 cases: a European network cohort study. Lancet Infect. Dis. 22, 1142–1152 (2022).
Qureshi, A. I. et al. Acute ischemic stroke and COVID-19: an analysis of 27,676 patients. Stroke 52, 905–912 (2021).
Nannoni, S., de Groot, R., Bell, S. & Markus, H. S. Stroke in COVID-19: a systematic review and meta-analysis. Int. J. Stroke 16, 137–149 (2021).
Zarifkar, P., Peinkhofer, C., Benros, M. E. & Kondziella, D. Frequency of neurological diseases after COVID-19, influenza A/B and bacterial pneumonia. Front. Neurol. 13, 904796 (2022).
Garg, R. K., Paliwal, V. K. & Gupta, A. Encephalopathy in patients with COVID-19: a review. J. Med. Virol. 93, 206–222 (2021).
Legriel, S. et al. Outcomes in patients with COVID-19 with acute encephalopathy and coma: an international prospective study. Neurology 100, e2247–e2258 (2023).
Liotta, E. M. et al. Frequent neurologic manifestations and encephalopathy-associated morbidity in COVID-19 patients. Ann. Clin. Transl. Neurol. 7, 2221–2230 (2020).
O’Mahoney, L. L. et al. The prevalence and long-term health effects of long COVID among hospitalised and non-hospitalised populations: a systematic review and meta-analysis. EClinicalMedicine 55, 101762 (2023).
Fernández-de-Las-Peñas, C. et al. Headache as an acute and post-COVID-19 symptom in COVID-19 survivors: a meta-analysis of the current literature. Eur. J. Neurol. 28, 3820–3825 (2021).
Tana, C. et al. Long COVID headache. J. Headache Pain. 23, 93 (2022).
Silva, L. et al. Long-term persistent headache after SARS-CoV-2 infection: a follow-up population-based study. Eur. J. Neurol. 32, e70130 (2025).
Uygun, O. et al. Headache characteristics in COVID-19 pandemic — a survey study. J. Headache Pain 21, 121 (2020).
Caronna, E. et al. Headache: a striking prodromal and persistent symptom, predictive of COVID-19 clinical evolution. Cephalalgia 40, 1410–1421 (2020).
Trigo, J. et al. Factors associated with the presence of headache in hospitalized COVID-19 patients and impact on prognosis: a retrospective cohort study. J. Headache Pain 21, 94 (2020).
Karadaş, Ö et al. Latent class cluster analysis identified hidden headache phenotypes in COVID-19: impact of pulmonary infiltration and IL-6. Neurol. Sci. 42, 1665–1673 (2021).
Davis, H. E. et al. Characterizing long COVID in an international cohort: 7 months of symptoms and their impact. eClinicalMedicine 38, 101019 (2021).
Larsen, N. W. et al. Characterization of autonomic symptom burden in long COVID: a global survey of 2,314 adults. Front. Neurol. 13, 1012668 (2022).
Novak, P. et al. Multisystem involvement in post-acute sequelae of coronavirus disease 19. Ann. Neurol. 91, 367–379 (2022).
Larsen, N. W., Stiles, L. E. & Miglis, M. G. Preparing for the long-haul: autonomic complications of COVID-19. Auton. Neurosci. 235, 102841 (2021).
Sletten, D. M., Suarez, G. A., Low, P. A., Mandrekar, J. & Singer, W. COMPASS 31: a refined and abbreviated Composite Autonomic Symptom Score. Mayo Clin. Proc. 87, 1196–1201 (2012).
Eastin, E. et al. Evaluating long-term autonomic dysfunction and functional impacts of long COVID: a follow-up study (P1-7.004). Neurology 104, 4290 (2025).
Dani, M. et al. Autonomic dysfunction in ‘long COVID’: rationale, physiology and management strategies. Clin. Med. 21, e63–e67 (2021).
Huang, C. et al. 6-Month consequences of COVID-19 in patients discharged from hospital: a cohort study. Lancet 401, e21–e33 (2023).
Merikanto, I. et al. Sleep symptoms are essential features of long-COVID — comparing healthy controls with COVID-19 cases of different severity in the International COVID Sleep Study (ICOSS-II). J. Sleep Res. 32, e13754 (2023).
Moura, A. E. F. et al. Central hypersomnia and chronic insomnia: expanding the spectrum of sleep disorders in long COVID syndrome — a prospective cohort study. BMC Neurol. 22, 417 (2022).
Henríquez-Beltrán, M. et al. Sleep and circadian health of critical survivors: a 12-month follow-up study. Crit. Care Med. 52, 1206–1217 (2024).
Chinvararak, C. & Chalder, T. Prevalence of sleep disturbances in patients with long COVID assessed by standardised questionnaires and diagnostic criteria: a systematic review and meta-analysis. J. Psychosom. Res. 175, 111535 (2023).
Linh, T. T. D. et al. Global prevalence of post-COVID-19 sleep disturbances in adults at different follow-up time points: a systematic review and meta-analysis. Sleep Med. Rev. 71, 101833 (2023).
Zhou, J. et al. Pre-existing sleep disturbances and risk of COVID-19: a meta-analysis. eClinicalMedicine 74, 102719 (2024).
Punj, M., Schwartz, M., Morris, A., Cheung, J. & Munipalli, B. A survey of sleep quality from a post-COVID clinic. J. Prim. Care Commun. Health 15, 21501319241233205 (2024).
Goldstein, C. A. et al. The possibility of circadian rhythm disruption in long COVID. Brain Behav. Immun. Health 23, 100476 (2022).
Global Burden of Disease Long COVID Collaborators. Estimated global proportions of individuals with persistent fatigue, cognitive, and respiratory symptom clusters following symptomatic COVID-19 in 2020 and 2021. JAMA 328, 1604–1615 (2022).
Thaweethai, T. et al. Development of a definition of postacute sequelae of SARS-CoV-2 infection. JAMA 329, 1934–1946 (2023).
Peter, R. S. et al. Post-acute sequelae of COVID-19 six to 12 months after infection: population based study. Br. Med. J. 379, e071050 (2022).
Joli, J., Buck, P., Zipfel, S. & Stengel, A. Post-COVID-19 fatigue: a systematic review. Front. Psychiatry 13, 947973 (2022).
Kedor, C. et al. A prospective observational study of post-COVID-19 chronic fatigue syndrome following the first pandemic wave in Germany and biomarkers associated with symptom severity. Nat. Commun. 13, 5104 (2022).
Appelman, B. et al. Muscle abnormalities worsen after post-exertional malaise in long COVID. Nat. Commun. 15, 17 (2024).
Saito, S. et al. Metabolomic and immune alterations in long COVID patients with chronic fatigue syndrome. Front. Immunol. 15, 1341843 (2024).
Jason, L. A., Yoo, S. & Bhatia, S. Patient perceptions of infectious illnesses preceding myalgic encephalomyelitis/chronic fatigue syndrome. Chronic Illn. 18, 901–910 (2022).
Nakatomi, Y. et al. Neuroinflammation in patients with chronic fatigue syndrome/myalgic encephalomyelitis: an (1)(1)C-(R)-PK11195 PET study. J. Nucl. Med. 55, 945–950 (2014).
Komaroff, A. L. & Lipkin, W. I. ME/CFS and long COVID share similar symptoms and biological abnormalities: road map to the literature. Front. Med. 10, 1187163 (2023).
McWhirter, L. et al. What is brain fog? J. Neurol. Neurosurg. Psychiatry 94, 321–325 (2023).
Xu, E., Xie, Y. & Al-Aly, Z. Long-term neurologic outcomes of COVID-19. Nat. Med. 28, 2406–2415 (2022).
Al-Aly, Z. et al. Long COVID science, research and policy. Nat. Med. 30, 2148–2164 (2024).
Al-Aly, Z. & Rosen, C. J. Long COVID and impaired cognition — more evidence and more work to do. N. Engl. J. Med. 390, 858–860 (2024).
Hampshire, A. et al. Cognition and memory after COVID-19 in a large community sample. N. Engl. J. Med. 390, 806–818 (2024).
Wood, G. K. et al. Posthospitalization COVID-19 cognitive deficits at 1 year are global and associated with elevated brain injury markers and gray matter volume reduction. Nat. Med. 31, 245–257 (2025).
Hosp, J. A. et al. Cognitive impairment and altered cerebral glucose metabolism in the subacute stage of COVID-19. Brain 144, 1263–1276 (2021).
Serrano del Pueblo, V. M. et al. Brain and cognitive changes in patients with long COVID compared with infection-recovered control subjects. Brain 147, 3611–3623 (2024).
Becker, J. H. et al. Assessment of cognitive function in patients after COVID-19 infection. JAMA Netw. Open 4, e2130645 (2021).
Fanshawe, J. B. et al. Cognitive domains affected post-COVID-19; a systematic review and meta-analysis. Eur. J. Neurol. 32, e16181 (2025).
Fernandez-Castaneda, A. et al. Mild respiratory COVID can cause multi-lineage neural cell and myelin dysregulation. Cell 185, 2452–68 e16 (2022).
Pretorius, E. et al. Prevalence of symptoms, comorbidities, fibrin amyloid microclots and platelet pathology in individuals with long COVID/post-acute sequelae of COVID-19 (PASC). Cardiovasc. Diabetol. 21, 148 (2022).
Rensma, S. P. et al. Microvascular dysfunction is associated with worse cognitive performance: the Maastricht Study. Hypertension 75, 237–245 (2020).
Douaud, G. et al. SARS-CoV-2 is associated with changes in brain structure in UK Biobank. Nature 604, 697–707 (2022).
Abrams, R. M. C., Zhou, L. & Shin, S. C. Persistent post-COVID-19 neuromuscular symptoms. Muscle Nerve 68, 350–355 (2023).
Premraj, L. et al. Mid and long-term neurological and neuropsychiatric manifestations of post-COVID-19 syndrome: a meta-analysis. J. Neurol. Sci. 434, 120162 (2022).
Taga, A. & Lauria, G. COVID-19 and the peripheral nervous system. A 2-year review from the pandemic to the vaccine era. J. Peripher. Nerv. Syst. 27, 4–30 (2022).
Oaklander, A. L. et al. Peripheral neuropathy evaluations of patients with prolonged long COVID. Neurol. Neuroimmunol. Neuroinflamm. 9, e1146 (2022).
Falco, P. et al. Small fibre neuropathy frequently underlies the painful long-COVID syndrome. Pain 165, 2002–2010 (2024).
Drobinska, N. et al. Small fiber neuropathy in long COVID: a cohort study with multimodal assessment and follow-up. Eur. Neurol. 88, 52–63 (2025).
van der Feltz-Cornelis, C. et al. Prevalence of mental health conditions and brain fog in people with long COVID: a systematic review and meta-analysis. Gen. Hosp. Psychiatry 88, 10–22 (2024).
Marchi, M. et al. Psychiatric symptoms in long-COVID patients: a systematic review. Front. Psychiatry 14, 1138389 (2023).
Yuan, K. et al. A systematic review and meta-analysis on prevalence of and risk factors associated with depression, anxiety and insomnia in infectious diseases, including COVID-19: a call to action. Mol. Psychiatry 27, 3214–3222 (2022).
Taquet, M. et al. Neurological and psychiatric risk trajectories after SARS-CoV-2 infection: an analysis of 2-year retrospective cohort studies including 1,284,437 patients. Lancet Psychiatry 9, 815–827 (2022).
Taquet, M. et al. Cognitive and psychiatric symptom trajectories 2–3 years after hospital admission for COVID-19: a longitudinal, prospective cohort study in the UK. Lancet Psychiatry 11, 696–708 (2024).
Yin, K. et al. Long COVID manifests with T cell dysregulation, inflammation and an uncoordinated adaptive immune response to SARS-CoV-2. Nat. Immunol. 25, 218–225 (2024).
Zakia, H., Pradana, K. & Iskandar, S. Risk factors for psychiatric symptoms in patients with long COVID: a systematic review. PLoS ONE 18, e0284075 (2023).
Kubota, T., Kuroda, N. & Sone, D. Neuropsychiatric aspects of long COVID: a comprehensive review. Psychiatry Clin. Neurosci. 77, 84–93 (2023).
Zeng, N. et al. A systematic review and meta-analysis of long term physical and mental sequelae of COVID-19 pandemic: call for research priority and action. Mol. Psychiatry 28, 423–433 (2023).
Wen, J. et al. Trajectories of symptoms in the acute phase of COVID-19 infection and their association with anxiety and depression 2 years after infection. BMC Psychiatry 26, 29 (2025).
Huarcaya-Victoria, J. et al. Mental health in COVID-2019 survivors from a general hospital in Peru: sociodemographic, clinical, and inflammatory variable associations. Int. J. Ment. Health Addict. 21, 1264–1285 (2023).
Ahmed, G. K. et al. Long term impact of COVID-19 infection on sleep and mental health: a cross-sectional study. Psychiatry Res. 305, 114243 (2021).
Kappelmann, N., Dantzer, R. & Khandaker, G. M. Interleukin-6 as potential mediator of long-term neuropsychiatric symptoms of COVID-19. Psychoneuroendocrinology 131, 105295 (2021).
Couto Amendola, F. et al. A two-year cohort study examining the impact of cytokines and chemokines on cognitive and psychiatric outcomes in long-COVID-19 patients. Brain Behav. Immun. 124, 218–225 (2025).
Teodoro, T., Chen, J., Gelauff, J. & Edwards, M. J. Functional neurological disorder in people with long COVID: a systematic review. Eur. J. Neurol. 30, 1505–1514 (2023).
Davenport, T. E. et al. Long COVID is not a functional neurologic disorder. J. Pers. Med. 14, 799 (2024).
Van der Feltz-Cornelis, C. M., Moriarty, A. S. & Strain, W. D. Neurological dysfunction in long COVID should not be labelled as functional neurological disorder. Viruses 15, 783 (2023).
Davis, H. E., McCorkell, L., Vogel, J. M. & Topol, E. J. Long COVID: major findings, mechanisms and recommendations. Nat. Rev. Microbiol. 21, 133–146 (2023).
Stein, S. R. et al. SARS-CoV-2 infection and persistence in the human body and brain at autopsy. Nature 612, 758–763 (2022).
Zuo, W. et al. The persistence of SARS-CoV-2 in tissues and its association with long COVID symptoms: a cross-sectional cohort study in China. Lancet Infect. Dis. 24, 845–855 (2024).
Martínez-Mármol, R. et al. SARS-CoV-2 infection and viral fusogens cause neuronal and glial fusion that compromises neuronal activity. Sci. Adv. 9, eadg2248 (2023).
Khan, M. et al. Anatomical barriers against SARS-CoV-2 neuroinvasion at vulnerable interfaces visualized in deceased COVID-19 patients. Neuron 110, 3919–35.e6 (2022).
Meinhardt, J. et al. Olfactory transmucosal SARS-CoV-2 invasion as a port of central nervous system entry in individuals with COVID-19. Nat. Neurosci. 24, 168–175 (2021).
Wong, A. C. et al. Serotonin reduction in post-acute sequelae of viral infection. Cell 186, 4851–67.e20 (2023).
Greenhalgh, T., Sivan, M., Perlowski, A. & Nikolich, J. Ž Long COVID: a clinical update. Lancet 404, 707–724 (2024).
Klein, J. et al. Distinguishing features of long COVID identified through immune profiling. Nature 623, 139–148 (2023).
Dunai, C. et al. Pulmonary SARS-CoV-2 infection leads to para-infectious immune activation in the brain. Front. Immunol. 15, 1440324 (2024).
Michael, B. D. et al. Para-infectious brain injury in COVID-19 persists at follow-up despite attenuated cytokine and autoantibody responses. Nat. Commun. 14, 8487 (2023).
Etter, M. M. et al. Severe neuro-COVID is associated with peripheral immune signatures, autoimmunity and neurodegeneration: a prospective cross-sectional study. Nat. Commun. 13, 6777 (2022).
Needham, E. J. et al. Brain injury in COVID-19 is associated with dysregulated innate and adaptive immune responses. Brain 145, 4097–4107 (2022).
Gold, J. E., Okyay, R. A., Licht, W. E. & Hurley, D. J. Investigation of long COVID prevalence and its relationship to Epstein–Barr virus reactivation. Pathogens 10, 763 (2021).
Su, Y. et al. Multiple early factors anticipate post-acute COVID-19 sequelae. Cell 185, 881–95.e20 (2022).
Ruiz-Pablos, M., Paiva, B., Montero-Mateo, R., Garcia, N. & Zabaleta, A. Epstein–Barr virus and the origin of myalgic encephalomyelitis or chronic fatigue syndrome. Front. Immunol. 12, 656797 (2021).
Monje, M. & Iwasaki, A. The neurobiology of long COVID. Neuron 110, 3484–3496 (2022).
Bjornevik, K., Münz, C., Cohen, J. I. & Ascherio, A. Epstein–Barr virus as a leading cause of multiple sclerosis: mechanisms and implications. Nat. Rev. Neurol. 19, 160–171 (2023).
Thakur, K. T. et al. COVID-19 neuropathology at Columbia University Irving Medical Center/New York Presbyterian Hospital. Brain 144, 2696–2708 (2021).
Varga, Z. et al. Endothelial cell infection and endotheliitis in COVID-19. Lancet 395, 1417–1418 (2020).
Leng, A. et al. Pathogenesis underlying neurological manifestations of long COVID syndrome and potential therapeutics. Cells 12, 816 (2023).
Fogarty, H. et al. Persistent endotheliopathy in the pathogenesis of long COVID syndrome. J. Thromb. Haemost. 19, 2546–2553 (2021).
Pretorius, E. et al. Persistent clotting protein pathology in long COVID/post-acute sequelae of COVID-19 (PASC) is accompanied by increased levels of antiplasmin. Cardiovasc. Diabetol. 20, 172 (2021).
Greene, C. et al. Blood–brain barrier disruption and sustained systemic inflammation in individuals with long COVID-associated cognitive impairment. Nat. Neurosci. 27, 421–432 (2024).
Zuo, Y. et al. Prothrombotic autoantibodies in serum from patients hospitalized with COVID-19. Sci. Transl. Med. 12, eabd3876 (2020).
Tung, M. L., Tan, B., Cherian, R. & Chandra, B. Anti-phospholipid syndrome and COVID-19 thrombosis: connecting the dots. Rheumatol. Adv. Pract. 5, rkaa081 (2021).
Altmann, D. M., Whettlock, E. M., Liu, S., Arachchillage, D. J. & Boyton, R. J. The immunology of long COVID. Nat. Rev. Immunol. 23, 618–634 (2023).
Liu, Q. et al. Gut microbiota dynamics in a prospective cohort of patients with post-acute COVID-19 syndrome. Gut 71, 544–552 (2022).
Lau, R. I. et al. A synbiotic preparation (SIM01) for post-acute COVID-19 syndrome in Hong Kong (RECOVERY): a randomised, double-blind, placebo-controlled trial. Lancet Infect. Dis. 24, 256–265 (2024).
Ancona, G. et al. Gut and airway microbiota dysbiosis and their role in COVID-19 and long-COVID. Front. Immunol. 14, 1080043 (2023).
Cecchetti, G. et al. Cognitive, EEG, and MRI features of COVID-19 survivors: a 10-month study. J. Neurol. 269, 3400–3412 (2022).
Petersen, M. et al. Brain imaging and neuropsychological assessment of individuals recovered from a mild to moderate SARS-CoV-2 infection. Proc. Natl Acad. Sci. USA 120, e2217232120 (2023).
Samanci, B. et al. Persistent neurocognitive deficits in long COVID: evidence of structural changes and network abnormalities following mild infection. Cortex 187, 98–110 (2025).
Troll, M. et al. Altered corticostriatal connectivity in long-COVID patients is associated with cognitive impairment. Psychol. Med. 55, e49 (2025).
Jaywant, A. et al. Cognitive symptoms of post-COVID-19 condition and daily functioning. JAMA Netw. Open 7, e2356098–e2356098 (2024).
Cai, M., Xie, Y., Topol, E. J. & Al-Aly, Z. Three-year outcomes of post-acute sequelae of COVID-19. Nat. Med. 30, 1564–1573 (2024).
Shil, R. S. K. et al. Patients with neurological or psychiatric complications of COVID-19 have worse long-term functional outcomes: COVID-CNS — a multicentre case–control study. Sci. Rep. 15, 3443 (2025).
Kim, Y., Bae, S., Chang, H. H. & Kim, S. W. Long COVID prevalence and impact on quality of life 2 years after acute COVID-19. Sci. Rep. 13, 11207 (2023).
Rodrigues, A. N. et al. Effect of long COVID-19 syndrome on health-related quality of life: a cross-sectional study. Front. Psychol. 15, 1394068 (2024).
World Health Organization. WHO Guidelines Approved by the Guidelines Review Committee. Clinical Management of COVID-19: Living Guideline (WHO, 2022).
Kuut, T. A. et al. Efficacy of cognitive-behavioral therapy targeting severe fatigue following coronavirus disease 2019: results of a randomized controlled trial. Clin. Infect. Dis. 77, 687–695 (2023).
Berry, C. et al. Resistance exercise therapy after COVID-19 infection: a randomized clinical trial. JAMA Netw. Open 8, e2534304 (2025).
Mooren, J. M. et al. Medical rehabilitation of patients with post-COVID-19 syndrome — a comparison of aerobic interval and continuous training. J. Clin. Med. 12, 6739 (2023).
Blitshteyn, S. et al. Multi-disciplinary collaborative consensus guidance statement on the assessment and treatment of autonomic dysfunction in patients with post-acute sequelae of SARS-CoV-2 infection (PASC). PM R 14, 1270–1291 (2022).
Rinaldi, L. et al. Long-COVID autonomic syndrome in working age and work ability impairment. Sci. Rep. 14, 11835 (2024).
Herring, T. E. et al. Post traumatic stress and sleep disorders in long COVID: patient management and treatment. Life Sci. 357, 123081 (2024).
Shil, R. S. K. et al. A clinical approach to the investigation and management of long COVID associated neuropathic pain. Eur. Arch. Psychiatry Clin. Neurosci. 274, 1787–1795 (2024).
National Institute for Health and Care Excellence. Neuropathic pain in adults: pharmacological management in non-specialist settings. nice.org.uk https://www.nice.org.uk/guidance/cg173/chapter/Recommendations (2020).
Guttuso, T. Jr., Zhu, J. & Wilding, G. E. Lithium aspartate for long COVID fatigue and cognitive dysfunction: a randomized clinical trial. JAMA Netw. Open 7, e2436874 (2024).
McIntyre, R. S. et al. Vortioxetine for the treatment of post-COVID-19 condition: a randomized controlled trial. Brain 147, 849–857 (2024).
Geng, L. N. et al. Nirmatrelvir–ritonavir and symptoms in adults with postacute sequelae of SARS-CoV-2 infection: the STOP-PASC randomized clinical trial. JAMA Intern. Med. 184, 1024–1034 (2024).
Gaylis, N. B. et al. Reduced cell surface levels of C-C chemokine receptor 5 and immunosuppression in long coronavirus disease 2019 syndrome. Clin. Infect. Dis. 75, 1232–1234 (2022).
Tanashyan, M. M., Raskurazhev, A. A., Kuznetsova, P. I., Bely, P. A. & Zaslavskaya, K. I. Prospects and possibilities for the treatment of patients with long COVID-19 syndrome. Ter. Arkh. 94, 1285–1293 (2022).
Putilina, M. V., Teplova, N. V., Bairova, K. I., Petrikeeva, A. E. & Shabalina, N. I. The result of prospective randomized study CITADEL — the efficacy and safety of drug cytoflavin in postcovid rehabilitation. Zh. Nevrol. Psikhiatr. Im. S. S. Korsakova 121, 45–51 (2021).
Tosato, M. et al. Effects of l-arginine plus vitamin c supplementation on physical performance, endothelial function, and persistent fatigue in adults with long COVID: a single-blind randomized controlled trial. Nutrients 14, 4984 (2022).
Achleitner, M. et al. Clinical improvement of long-COVID is associated with reduction in autoantibodies, lipids, and inflammation following therapeutic apheresis. Mol. Psychiatry 28, 2872–2877 (2023).
Novak, P. Post COVID-19 syndrome associated with orthostatic cerebral hypoperfusion syndrome, small fiber neuropathy and benefit of immunotherapy: a case report. eNeurologicalSci 21, 100276 (2020).
Bonilla, H. et al. Therapeutic trials for long COVID-19: a call to action from the interventions taskforce of the RECOVER initiative. Front. Immunol. 14, 1129459 (2023).
Preview : Journal Type Where are the long COVID trials? Lancet Infect. Dis. 23, 879 (2023).
Ledford, H. Long-COVID treatments: why the world is still waiting. Nature 608, 258–260 (2022).
Notarte, K. I. et al. Impact of COVID-19 vaccination on the risk of developing long-COVID and on existing long-COVID symptoms: a systematic review. EClinicalMedicine 53, 101624 (2022).
Ayoubkhani, D. et al. Trajectory of long covid symptoms after COVID-19 vaccination: community based cohort study. Br. Med. J. 377, e069676 (2022).
Mizrahi, B. et al. Long covid outcomes at one year after mild SARS-CoV-2 infection: nationwide cohort study. Br. Med. J. 380, e072529 (2023).
Ellul, M. A. et al. Neurological associations of COVID-19. Lancet Neurol. 19, 767–783 (2020).
Taquet, M. et al. Incidence, co-occurrence, and evolution of long-COVID features: a 6-month retrospective cohort study of 273,618 survivors of COVID-19. PLoS Med. 18, e1003773 (2021).
Romero-Duarte, Á et al. Sequelae, persistent symptomatology and outcomes after COVID-19 hospitalization: the ANCOHVID multicentre 6-month follow-up study. BMC Med. 19, 129 (2021).
Huang, L. et al. Health outcomes in people 2 years after surviving hospitalisation with COVID-19: a longitudinal cohort study. Lancet Respir. Med. 10, 863–876 (2022).
Pazukhina, E. et al. Prevalence and risk factors of post-COVID-19 condition in adults and children at 6 and 12 months after hospital discharge: a prospective, cohort study in Moscow (StopCOVID). BMC Med. 20, 244 (2022).
Perlis, R. H. et al. Prevalence and Correlates of Long COVID Symptoms Among US Adults. JAMA Netw. Open 5, e2238804 (2022).
Whitaker, M. et al. Persistent COVID-19 symptoms in a community study of 606,434 people in England. Nat. Commun. 13, 1957 (2022).
Zhao, Y. et al. The phenotype and prediction of long-term physical, mental and cognitive COVID-19 sequelae 20 months after recovery, a community-based cohort study in China. Mol. Psychiatry 28, 1793–1801 (2023).
Tok, P. S. K. et al. Post COVID-19 condition among adults in Malaysia following the Omicron wave: a prospective cohort study. PLoS ONE 19, e0296488 (2024).
Cai, J. et al. A one-year follow-up study of systematic impact of long COVID symptoms among patients post SARS-CoV-2 omicron variants infection in Shanghai, China. Emerg. Microbes Infect. 12, 2220578 (2023).
Leisman, D. E. et al. Cytokine elevation in severe and critical COVID-19: a rapid systematic review, meta-analysis, and comparison with other inflammatory syndromes. Lancet Respir. Med. 8, 1233–1244 (2020).
Verde, F. et al. Serum neurofilament light chain levels in Covid-19 patients without major neurological manifestations. J. Neurol. 269, 5691–5701 (2022).
Author information
Authors and Affiliations
Contributions
R.M., A.A. and B.D.M. researched data for the article. All authors contributed substantially to discussion of the content. R.M. and A.A. wrote the article. E.B. and B.D.M. reviewed and/or edited the manuscript before submission.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Reviews Neurology thanks Mar Ariza and Judith Bellmann-Strobl for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Matthews, R., Alam, A., Bullmore, E. et al. Understanding the long-term neurological effects of SARS-CoV-2 infection. Nat Rev Neurol (2026). https://doi.org/10.1038/s41582-026-01205-y
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41582-026-01205-y