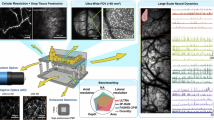
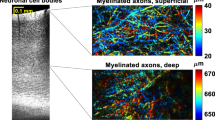
Abstract
Understanding brain function and pathology requires observation of cellular dynamics within intact neural circuits. Although two-photon microscopy revolutionized mammalian in vivo brain imaging, its limitation to upper cortical layers has restricted access to many important brain regions. Three-photon microscopy overcomes this constraint, enabling minimally invasive yet high-resolution visualization of the deep cortical and subcortical structures that are crucial for higher-order brain functions. This emerging technology opens new avenues for investigating fundamental aspects of neuroscience, from circuit dynamics to disease mechanisms. Here, we examine how three-photon microscopy has started to transform our ability to investigate neural circuits, glial biology, and oncological and neuroimmune interactions in previously inaccessible brain regions, primarily in the mouse, but also in other model organisms. We discuss current technical challenges, recent innovations and future applications that promise to bring us greater understanding of the living brain.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$32.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Wilt, B. A. et al. Advances in light microscopy for neuroscience. Annu. Rev. Neurosci. 32, 435–506 (2009).
Svoboda, K., Denk, W., Kleinfeld, D. & Tank, D. W. In vivo dendritic calcium dynamics in neocortical pyramidal neurons. Nature 385, 161–165 (1997).
Waters, J., Larkum, M., Sakmann, B. & Helmchen, F. Supralinear Ca2+ influx into dendritic tufts of layer 2/3 neocortical pyramidal neurons in vitro and in vivo. J. Neurosci. 23, 8558–8567 (2003).
Chen, X., Leischner, U., Rochefort, N. L., Nelken, I. & Konnerth, A. Functional mapping of single spines in cortical neurons in vivo. Nature 475, 501–505 (2011).
Trachtenberg, J. T. et al. Long-term in vivo imaging of experience-dependent synaptic plasticity in adult cortex. Nature 420, 788–794 (2002).
Grutzendler, J., Kasthuri, N. & Gan, W. B. Long-term dendritic spine stability in the adult cortex. Nature 420, 812–816 (2002).
Zuo, Y., Yang, G., Kwon, E. & Gan, W.-B. Long-term sensory deprivation prevents dendritic spine loss in primary somatosensory cortex. Nature 436, 261–265 (2005).
Holtmaat, A., Wilbrecht, L., Knott, G. W., Welker, E. & Svoboda, K. Experience-dependent and cell-type-specific spine growth in the neocortex. Nature 441, 979–983 (2006).
Lendvai, B., Stern, E. A., Chen, B. & Svoboda, K. Experience-dependent plasticity of dendritic spines in the developing rat barrel cortex in vivo. Nature 404, 876–881 (2000).
Tian, L. et al. Imaging neural activity in worms, flies and mice with improved GCaMP calcium indicators. Nat. Methods 6, 875–881 (2009).
Chen, J. L. et al. Pathway-specific reorganization of projection neurons in somatosensory cortex during learning. Nat. Neurosci. 18, 1101–1108 (2015).
Condylis, C. et al. Dense functional and molecular readout of a circuit hub in sensory cortex. Science 375, eabl5981 (2022).
O’Toole, S. M., Oyibo, H. K. & Keller, G. B. Molecularly targetable cell types in mouse visual cortex have distinguishable prediction error responses. Neuron 111, 2918–2928.e8 (2023).
Zuend, M. et al. Arousal-induced cortical activity triggers lactate release from astrocytes. Nat. Metab. 2, 179–191 (2020).
Helmchen, F. & Denk, W. Deep tissue two-photon microscopy. Nat. Methods 2, 932–940 (2005).
Denk, W., Strickler, J. H. & Webb, W. W. Two-photon laser scanning fluorescence microscopy. Science 248, 73–76 (1990).
Kerr, J. N. D. & Denk, W. Imaging in vivo: watching the brain in action. Nat. Rev. Neurosci. 9, 195–205 (2008).
Svoboda, K. & Yasuda, R. Principles of two-photon excitation microscopy and its applications to neuroscience. Neuron 50, 823–839 (2006).
Pologruto, T. A., Yasuda, R. & Svoboda, K. Monitoring neural activity and [Ca2+] with genetically encoded Ca2+ indicators. J. Neurosci. 24, 9572–9579 (2004).
Abdelfattah, A. S. et al. Bright and photostable chemigenetic indicators for extended in vivo voltage imaging. Science 365, 699–704 (2019).
Zhang, Y. et al. Fast and sensitive GCaMP calcium indicators for imaging neural populations. Nature 615, 884–891 (2023).
Lee, S. et al. Improving positively tuned voltage indicators for brightness and kinetics. Preprint at bioRxiv https://doi.org/10.1101/2024.06.21.599617 (2024).
Dombeck, D. A., Harvey, C. D., Tian, L., Looger, L. L. & Tank, D. W. Functional imaging of hippocampal place cells at cellular resolution during virtual navigation. Nat. Neurosci. 13, 1433–1440 (2010).
Barretto, R. P. J., Messerschmidt, B. & Schnitzer, M. J. In vivo fluorescence imaging with high-resolution microlenses. Nat. Methods 6, 511–512 (2009).
Horton, N. G. et al. In vivo three-photon microscopy of subcortical structures within an intact mouse brain. Nat. Photon. 7, 205–209 (2013). This paper three-photon microscopy for in vivo imaging of subcortical structures in an intact mouse brain, establishing the potential of the technique for deep tissue imaging.
Wang, T. & Xu, C. Three-photon neuronal imaging in deep mouse brain. Optica 7, 947 (2020).
Squier, J. A., Müller, M., Brakenhoff, G. J. & Wilson, K. R. Third harmonic generation microscopy. Opt. Express 3, 315 (1998).
Weigelin, B., Bakker, G.-J. & Friedl, P. Third harmonic generation microscopy of cells and tissue organization. J. Cell Sci. 129, 245–255 (2016).
Theer, P., Hasan, M. T. & Denk, W. Two-photon imaging to a depth of 1000 µm in living brains by use of a Ti:Al2O3 regenerative amplifier. Opt. Lett. 28, 1022–1024 (2003).
Akbari, N., Rebec, M. R., Xia, F. & Xu, C. Imaging deeper than the transport mean free path with multiphoton microscopy. Biomed. Opt. Express 13, 452 (2022).
Bakker, G. J. et al. Intravital deep-tumor single-beam 3-photon, 4-photon, and harmonic microscopy. eLife 11, e63776 (2022).
Wang, M. et al. Comparing the effective attenuation lengths for long wavelength in vivo imaging of the mouse brain. Biomed. Opt. Express 9, 3534 (2018).
Wang, M., Kim, M., Xia, F. & Xu, C. Impact of the emission wavelengths on in vivo multiphoton imaging of mouse brains. Biomed. Opt. Express 10, 1905 (2019).
Cheng, L.-C., Horton, N. G., Wang, K., Chen, S.-J. & Xu, C. Measurements of multiphoton action cross sections for multiphoton microscopy. Biomed. Opt. Express 5, 3427–3433 (2014).
Podgorski, K. & Ranganathan, G. Brain heating induced by near-infrared lasers during multiphoton microscopy. J. Neurophysiol. 116, 1012–1023 (2016).
Yildirim, M., Sugihara, H., So, P. T. C. & Sur, M. Functional imaging of visual cortical layers and subplate in awake mice with optimized three-photon microscopy. Nat. Commun. 10, 177 (2019). This work highlights how three-photon microscopy across different layers enabled the discovery of functional specialization of neurons across different brain regions, and it defines pulse parameters and safety limits for damage-free three-photon imaging.
Wang, T. et al. Quantitative analysis of 1300-nm three-photon calcium imaging in the mouse brain. eLife 9, e53205 (2020). This paper establishes a quantitative framework for three-photon neuronal imaging in the mouse, identifying the depth at which three-photon imaging offers superior recording fidelity compared to two-photon microscopy, while validating safe excitation parameters.
Wokosin, D. L., Centonze, V. E., Crittenden, S. & White, J. Three-photon excitation fluorescence imaging of biological specimens using an all-solid-state laser. Bioimaging 4, 208–214 (1996).
Hell, S. W. Three-photon excitation in fluorescence microscopy. J. Biomed. Opt. 1, 71 (1996). This work proves the feasibility of three-photon excitation in fluorescence microscopy.
Xu, C., Zipfel, W., Shear, J. B., Williams, R. M. & Webb, W. W. Multiphoton fluorescence excitation: new spectral windows for biological nonlinear microscopy. Proc. Natl Acad. Sci. USA. 93, 10763–10768 (1996). This study characterizes the two-photon and three-photon excitation spectra of 25 fluorophores, highlighting their suitability for near-infrared nonlinear microscopy.
Ouzounov, D. G. et al. In vivo three-photon imaging of activity of GCaMP6-labeled neurons deep in intact mouse brain. Nat. Methods 14, 388–390 (2017). This paper reports three-photon imaging of GCaMP6-labelled neuronal activity from ~150 neurons in the hippocampal stratum pyramidale at ~1-mm depth within an intact mouse brain.
Chen, X. et al. In vivo three-photon microscopy of mouse brain excited at the 2200 nm window. ACS Photon. 8, 2898–2903 (2021).
Wang, Y., Wang, K., Wen, W., Qiu, P. & Wang, K. Comparison of signal detection of GaAsP and GaAs PMTs for multiphoton microscopy at the 1700-nm window. IEEE Photon. J. 8, 1–6 (2016).
Li, B., Wu, C., Wang, M., Charan, K. & Xu, C. An adaptive excitation source for high-speed multiphoton microscopy. Nat. Methods 17, 163–166 (2020).
Hampson, K. M. et al. Adaptive optics for high-resolution imaging. Nat. Rev. Methods Prim. 1, 68 (2021).
Streich, L. et al. High-resolution structural and functional deep brain imaging using adaptive optics three-photon microscopy. Nat. Methods 18, 1253–1258 (2021). This paper combines adaptive optics with three-photon microscopy to achieve near-diffraction-limited imaging of unprecedented deep brain structures and the functional activity of astrocytes.
Rodríguez, C. et al. An adaptive optics module for deep tissue multiphoton imaging in vivo. Nat. Methods 18, 1259–1264 (2021).
Qin, Z. et al. Deep tissue multi-photon imaging using adaptive optics with direct focus sensing and shaping. Nat. Biotechnol. 40, 1663–1671 (2022).
Berlage, C. et al. Deep tissue scattering compensation with three-photon F-SHARP. Optica 8, 1613 (2021).
Chen, B. et al. Rapid volumetric imaging with Bessel-beam three-photon microscopy. Biomed. Opt. Express 9, 1992–2000 (2018).
Rodríguez, C., Liang, Y., Lu, R. & Ji, N. Three-photon fluorescence microscopy with an axially elongated Bessel focus. Opt. Lett. 43, 1914 (2018).
Takasaki, K., Abbasi-Asl, R. & Waters, J. Superficial bound of the depth limit of two-photon imaging in mouse brain. eNeuro 7, ENEURO.0255-19.2019 (2020).
Takasaki, K. T., Tsyboulski, D. & Waters, J. Dual-plane 3-photon microscopy with remote focusing. Biomed. Opt. Express 10, 5585 (2019).
Weisenburger, S. et al. Volumetric Ca2+ imaging in the mouse brain using hybrid multiplexed sculpted light microscopy. Cell 177, 1050–1066.e14 (2019).
Hontani, Y., Xia, F. & Xu, C. Multicolor three-photon fluorescence imaging with single-wavelength excitation deep in mouse brain. Sci. Adv. 7, eabf3531 (2021). This work reports a new three-photon excitation scheme for multicolour three-photon fluorescence imaging using excitation with a single wavelength, which broadens the possible deep tissue imaging applications of 3PM.
Li, D. et al. AIE-nanoparticle assisted ultra-deep three-photon microscopy in the in vivo mouse brain under 1300 nm excitation. Mater. Chem. Front. 5, 3201–3208 (2021).
Schubert, M. C. et al. Deep intravital brain tumor imaging enabled by tailored three-photon microscopy and analysis. Nat. Commun. 15, 7383 (2024). This study tailored three-photon microscopy, adaptive optics and AI analysis workflows for intravital brain tumour imaging inside the corpus callosum, revealing distinct tumour invasion routes and cellular migration mechanisms.
Buzsáki, G. Rhythms of the Brain (Oxford Univ. Press, 2006).
Harris, K. D. & Shepherd, G. M. G. The neocortical circuit: themes and variations. Nat. Neurosci. 18, 170–181 (2015).
Ohki, K., Chung, S., Ch’ng, Y. H., Kara, P. & Reid, R. C. Functional imaging with cellular resolution reveals precise micro-architecture in visual cortex. Nature 433, 597–603 (2005).
Stamenkovic, S. et al. Impaired drainage through capillary-venous networks contributes to age-related white matter loss. Preprint at bioRxiv https://doi.org/10.1101/2024.02.11.579849 (2024). This study uses three-photon microscopy to reveal that age-related cognitive decline is linked to selective microvascular deterioration in deep brain tissues, highlighting impaired capillary–venular drainage as a critical factor in cerebral ageing.
Rupprecht, P. et al. Centripetal integration of past events in hippocampal astrocytes regulated by locus coeruleus. Nat. Neurosci. 27, 927–939 (2024).
Fan, L. Z. et al. All-optical physiology resolves a synaptic basis for behavioral timescale plasticity. Cell 186, 543–559.e19 (2023).
Mok, A. T. et al. A large field-of-view, single-cell-resolution two- and three-photon microscope for deep and wide imaging. eLight 4, 20 (2024).
Zhao, C. et al. Miniature three-photon microscopy maximized for scattered fluorescence collection. Nat. Methods 20, 617–622 (2023).
Klioutchnikov, A. et al. A three-photon head-mounted microscope for imaging all layers of visual cortex in freely moving mice. Nat. Methods 20, 610–616 (2023). This paper introduces a three-photon head-mounted microscope lightweight enough for mice, which also contained a z-drive allowing imaging across all layers of the visual cortex in freely moving mice, even in a fully lit environment.
Wallace, D. J. et al. Rats maintain an overhead binocular field at the expense of constant fusion. Nature 498, 65–69 (2013).
Michaiel, A. M., Abe, E. T. & Niell, C. M. Dynamics of gaze control during prey capture in freely moving mice. eLife 9, e57458 (2020).
Cheng, Y.-T., Lett, K. M., Xu, C. & Schaffer, C. B. Three-photon excited fluorescence microscopy enables imaging of blood flow, neural structure and inflammatory response deep into mouse spinal cord in vivo. eLife 13, RP95804 (2024).
Zhang, H. et al. Large-depth three-photon fluorescence microscopy imaging of cortical microvasculature on nonhuman primates with bright AIE probe in vivo. Biomaterials 289, 121809 (2022).
Tatarsky, R. L., Akbari, N., Wang, K., Xu, C. & Bass, A. H. Label-free multiphoton imaging reveals volumetric shifts across development in sensory-related brain regions of a miniature transparent vertebrate. J. Comp. Neurol. 533, e70048 (2025).
Akbari, N. et al. Label‐free, whole‐brain in vivo mapping in an adult vertebrate with third harmonic generation microscopy. J. Comp. Neurol. 532, e25614 (2024).
Ferrer Ortas, J. et al. Label-free imaging of red blood cells and oxygenation with color third-order sum-frequency generation microscopy. Light Sci. Appl. 12, 29 (2023).
Aragon, M. J. et al. Multiphoton imaging of neural structure and activity in Drosophila through the intact cuticle. eLife 11, e69094 (2022).
Hsu, K.-J., Lin, Y.-Y., Chiang, A.-S. & Chu, S.-W. Optical properties of adult Drosophila brains in one-, two-, and three-photon microscopy. Biomed. Opt. Express 10, 1627 (2019).
Tao, X. et al. Transcutical imaging with cellular and subcellular resolution. Biomed. Opt. Express 8, 1277 (2017).
Chow, D. M. et al. Deep three-photon imaging of the brain in intact adult zebrafish. Nat. Methods 17, 605–608 (2020).
Yildirim, M. et al. Label-free three-photon imaging of intact human cerebral organoids for tracking early events in brain development and deficits in Rett syndrome. eLife 11, e78079 (2022).
Deng, P. et al. Long-working-distance high-collection-efficiency three-photon microscopy for in vivo long-term imaging of zebrafish and organoids. iScience 27, 110554 (2024).
Cheng, H. et al. In vivo deep-brain imaging of microglia enabled by three-photon fluorescence microscopy. Opt. Lett. 45, 5271 (2020).
Hill, R. A. & Nishiyama, A. NG2 cells (polydendrocytes): listeners to the neural network with diverse properties. Glia 62, 1195–1210 (2014).
Boshans, L. L., Sherafat, A. & Nishiyama, A. The effects of developmental and current niches on oligodendrocyte precursor dynamics and fate. Neurosci. Lett. 715, 134593 (2020).
Thornton, M. A. et al. Long-term in vivo three-photon imaging reveals region-specific differences in healthy and regenerative oligodendrogenesis. Nat. Neurosci. 27, 846–861 (2024). This study establishes longitudinal in vivo three-photon microscopy to reveal distinct regional dynamics of oligodendrocyte generation in the adult mouse brain, highlighting the role of local microenvironments in regulating myelination and repair.
Araque, A., Parpura, V., Sanzgiri, R. P. & Haydon, P. G. Tripartite synapses: glia, the unacknowledged partner. Trends Neurosci. 22, 208–215 (1999).
Bezzi, P., Domercq, M., Vesce, S. & Volterra, A. Neuron-astrocyte cross-talk during synaptic transmission: physiological and neuropathological implications. Prog. Brain Res. 132, 255–265 (2001).
Semyanov, A., Henneberger, C. & Agarwal, A. Making sense of astrocytic calcium signals — from acquisition to interpretation. Nat. Rev. Neurosci. 21, 551–564 (2020).
Lanjakornsiripan, D. et al. Layer-specific morphological and molecular differences in neocortical astrocytes and their dependence on neuronal layers. Nat. Commun. 9, 1623 (2018).
Takata, N. & Hirase, H. Cortical layer 1 and layer 2/3 astrocytes exhibit distinct calcium dynamics in vivo. PLoS ONE 3, e2525 (2008).
Agulhon, C. et al. What is the role of astrocyte calcium in neurophysiology? Neuron 59, 932–946 (2008).
Pasti, L., Volterra, A., Pozzan, T. & Carmignoto, G. Intracellular calcium oscillations in astrocytes: a highly plastic, bidirectional form of communication between neurons and astrocytes in situ. J. Neurosci. 17, 7817–7830 (1997).
Man, J. H. K. et al. Region-specific and age-related differences in astrocytes in the human brain. Neurobiol. Aging 140, 102–115 (2024).
Sofroniew, M. V. & Vinters, H. V. Astrocytes: biology and pathology. Acta Neuropathol. 119, 7–35 (2010).
Aggarwal, A. et al. Glutamate indicators with improved activation kinetics and localization for imaging synaptic transmission. Nat. Methods 20, 925–934 (2023).
Marvin, J. S. et al. A genetically encoded fluorescent sensor for in vivo imaging of GABA. Nat. Methods 16, 763–770 (2019).
Deng, F. et al. Improved green and red GRAB sensors for monitoring spatiotemporal serotonin release in vivo. Nat. Methods 21, 692–702 (2024).
Marvin, J. S. et al. An optimized fluorescent probe for visualizing glutamate neurotransmission. Nat. Methods 10, 162–170 (2013).
Saab, A. S. & Nave, K.-A. Myelin dynamics: protecting and shaping neuronal functions. Curr. Opin. Neurobiol. 47, 104–112 (2017).
Fuhrmann, F. et al. Three-photon in vivo imaging of neurons and glia in the medial prefrontal cortex with sub-cellular resolution. Commun. Biol. 8, 795 (2025). This study applies three-photon microscopy to measure neuronal and astrocytic Ca2+ dynamics in the medial prefrontal cortex of awake head-fixed mice up to a depth of 1,700 µm, opening new possibilities for studying cognitive and emotional brain functions in this region.
Nebeling, F. C. et al. Dynamics of microglia-glioblastoma crosstalk at the far infiltration zone. Preprint at bioRxiv https://doi.org/10.1101/2024.09.23.614016 (2024).
Scherer, H. J. Structural development in gliomas. Am. J. Cancer 34, 333–351 (1938).
Drumm, M. R. et al. Extensive brainstem infiltration, not mass effect, is a common feature of end-stage cerebral glioblastomas. Neuro Oncol. 22, 470–479 (2020).
Venkataramani, V. et al. Glutamatergic synaptic input to glioma cells drives brain tumour progression. Nature 573, 532–538 (2019).
Venkataramani, V. et al. Glioblastoma hijacks neuronal mechanisms for brain invasion. Cell 185, 2899–2917 (2022).
Tetzlaff, S. K. et al. Characterizing and targeting glioblastoma neuron-tumor networks with retrograde tracing. Cell 188, 390–411.e36 (2025).
Karimian-Jazi, K. et al. Monitoring innate immune cell dynamics in the glioma microenvironment by magnetic resonance imaging and multiphoton microscopy (MR-MPM). Theranostics 10, 1873–1883 (2020).
Breckwoldt, M. O. et al. Correlated MRI and ultramicroscopy (MR-UM) of brain tumors reveals vast heterogeneity of tumor infiltration and neoangiogenesis in preclinical models and human disease. Front. Neurosci. 12, 1004 (2018).
Schregel, K. et al. A cellular ground truth to develop MRI signatures in glioma models by correlative light sheet microscopy and atlas-based coregistration. J. Neurosci. 43, 5574–5587 (2023).
Marchetti, L. & Engelhardt, B. Immune cell trafficking across the blood-brain barrier in the absence and presence of neuroinflammation. Vasc. Biol. 2, H1–H18 (2020).
Caravagna, C. et al. Diversity of innate immune cell subsets across spatial and temporal scales in an EAE mouse model. Sci. Rep. 8, 5146 (2018).
Xu, C., Nedergaard, M., Fowell, D. J., Friedl, P. & Ji, N. Multiphoton fluorescence microscopy for in vivo imaging. Cell 187, 4458–4487 (2024).
Choe, K. et al. Intravital three-photon microscopy allows visualization over the entire depth of mouse lymph nodes. Nat. Immunol. 23, 330–340 (2022). This work showcases intravital three-photon microscopy as a powerful tool for imaging the full depth of mouse lymph nodes (up to 900 μm), enabling detailed and non-invasive visualization of vasculature and T cell motility and, thereby, extending immune cell imaging beyond the limits of conventional techniques.
Mary, A., Mancuso, R. & Heneka, M. T. Immune activation in Alzheimer disease. Annu. Rev. Immunol. 42, 585–613 (2024).
Ahn, S. J. et al. Label-free assessment of hemodynamics in individual cortical brain vessels using third harmonic generation microscopy. Biomed. Opt. Express 11, 2665 (2020).
Farrar, M. J., Wise, F. W., Fetcho, J. R. & Schaffer, C. B. In vivo imaging of myelin in the vertebrate central nervous system using third harmonic generation microscopy. Biophys. J. 100, 1362–1371 (2011).
Sinefeld, D., Paudel, H. P., Ouzounov, D. G., Bifano, T. G. & Xu, C. Adaptive optics in multiphoton microscopy: comparison of two, three and four photon fluorescence. Opt. Express 23, 31472–31483 (2015).
Sinefeld, D. et al. Three-photon adaptive optics for mouse brain imaging. Front. Neurosci. 16, 880859 (2022).
Rodríguez, C. et al. Adaptive optical third-harmonic generation microscopy for in vivo imaging of tissues. Biomed. Opt. Express 15, 4513 (2024).
Hong, G., Antaris, A. L. & Dai, H. Near-infrared fluorophores for biomedical imaging. Nat. Biomed. Eng. 1, 0010 (2017).
Liu, R., Li, Z., Marvin, J. S. & Kleinfeld, D. Direct wavefront sensing enables functional imaging of infragranular axons and spines. Nat. Methods 16, 615–618 (2019).
Gigan, S. Optical microscopy aims deep. Nat. Photon. 11, 14–16 (2017).
Gigan, S. et al. Roadmap on wavefront shaping and deep imaging in complex media. J. Phys. Photon. 4, 042501 (2022).
Rauer, B., de Aguiar, H. B., Bourdieu, L. & Gigan, S. Scattering correcting wavefront shaping for three-photon microscopy. Opt. Lett. 47, 6233 (2022).
Nadella, K. M. N. S. et al. Random-access scanning microscopy for 3D imaging in awake behaving animals. Nat. Methods 13, 1001–1004 (2016).
Villette, V. et al. Ultrafast two-photon imaging of a high-gain voltage indicator in awake behaving mice. Cell 179, 1590–1608.e23 (2019).
Frostig, H. et al. Three-photon population imaging of subcortical brain regions. Preprint at bioRxiv https://doi.org/10.1101/2025.03.21.644611 (2025).
Yu, C. H. et al. The Cousa objective: a long-working distance air objective for multiphoton imaging in vivo. Nat. Methods 21, 132–141 (2024).
Tsai, P. S. et al. Ultra-large field-of-view two-photon microscopy. Opt. Express 23, 13833 (2015).
Sofroniew, N. J., Flickinger, D., King, J. & Svoboda, K. A large field of view two-photon mesoscope with subcellular resolution for in vivo imaging. eLife 5, e14472 (2016).
Stirman, J. N., Smith, I. T., Kudenov, M. W. & Smith, S. L. Wide field-of-view, multi-region, two-photon imaging of neuronal activity in the mammalian brain. Nat. Biotechnol. 34, 857–862 (2016).
Ota, K. et al. Fast, cell-resolution, contiguous-wide two-photon imaging to reveal functional network architectures across multi-modal cortical areas. Neuron 109, 1810–1824.e9 (2021).
Yu, C.-H., Stirman, J. N., Yu, Y., Hira, R. & Smith, S. L. Diesel2p mesoscope with dual independent scan engines for flexible capture of dynamics in distributed neural circuitry. Nat. Commun. 12, 6639 (2021).
Tamura, R. et al. Genetically encoded fluorescent sensors for visualizing polyamine levels, uptake, and distribution. Preprint at bioRxiv https://doi.org/10.1101/2024.08.21.609037 (2024).
Xiao, Y., Deng, P., Zhao, Y., Yang, S. & Li, B. Three-photon excited fluorescence imaging in neuroscience: from principles to applications. Front. Neurosci. 17, 1085682 (2023).
Ashesh, Krull, A., Di Sante, M., Pasqualini, F. S. & Jug, F. μSplit: efficient image decomposition for microscopy data. Preprint at https://doi.org/10.48550/arXiv.2211.12872 (2022).
Marshel, J. H. et al. Cortical layer-specific critical dynamics triggering perception. Science 365, eaaw5202 (2019).
Chen, F., Tillberg, P. W. & Boyden, E. S. Expansion microscopy. Science 347, 543–548 (2015).
M’Saad, O. & Bewersdorf, J. Light microscopy of proteins in their ultrastructural context. Nat. Commun. 11, 3850 (2020).
Tønnesen, J., Inavalli, V. V. G. K. & Nägerl, U. V. Super-resolution imaging of the extracellular space in living brain tissue. Cell 172, 1108–1121.e15 (2018).
Tavakoli, M. R. et al. Light-microscopy based connectomic reconstruction of mammalian brain tissue. Nature 642, 398–410 (2025).
Kuan, A. T. et al. Dense neuronal reconstruction through X-ray holographic nano-tomography. Nat. Neurosci. 23, 1637–1643 (2020).
Bosch, C. et al. Functional and multiscale 3D structural investigation of brain tissue through correlative in vivo physiology, synchrotron microtomography and volume electron microscopy. Nat. Commun. 13, 2923 (2022).
Weigert, M. et al. Content-aware image restoration: pushing the limits of fluorescence microscopy. Nat. Methods 15, 1090–1097 (2018).
Krull, A., Buchholz, T.-O. & Jug, F. Noise2Void — learning denoising from single noisy images. In 2019 IEEE/CVF Conference on Computer Vision and Pattern Recognition (CVPR) 2124–2132 (IEEE, 2019).
Hu, Q. et al. Universal adaptive optics for microscopy through embedded neural network control. Light Sci. Appl. 12, 270 (2023).
Mahecic, D. et al. Event-driven acquisition for content-enriched microscopy. Nat. Methods 19, 1262–1267 (2022).
Carpenter, A. E., Cimini, B. A. & Eliceiri, K. W. Smart microscopes of the future. Nat. Methods 20, 962–964 (2023).
Morgado, L., Gómez‐de‐Mariscal, E., Heil, H. S. & Henriques, R. The rise of data‐driven microscopy powered by machine learning. J. Microsc. 295, 85–92 (2024).
Ou, Z. et al. Achieving optical transparency in live animals with absorbing molecules. Science 385, eadm6869 (2024).
Franzesi, G. T. et al. In vivo optical clearing of mammalian brain. Preprint at bioRxiv https://doi.org/10.1101/2024.09.05.611421 (2024).
Inagaki, S. et al. Isotonic and minimally invasive optical clearing media for live cell imaging ex vivo and in vivo. Preprint at bioRxiv https://doi.org/10.1101/2024.09.13.612584 (2024).
Wang, T. et al. Three-photon imaging of mouse brain structure and function through the intact skull. Nat. Methods 15, 789–792 (2018).
Höhne, J. et al. Intraoperative imaging of brain tumors with fluorescein: confocal laser endomicroscopy in neurosurgery. Clinical and user experience. Neurosurg. Focus 50, E19 (2021).
Fürtjes, G. et al. Intraoperative microscopic autofluorescence detection and characterization in brain tumors using stimulated Raman histology and two-photon fluorescence. Front. Oncol. 13, 1146031 (2023).
Neidert, N. et al. Stimulated Raman histology in the neurosurgical workflow of a major European neurosurgical center — part A. Neurosurg. Rev. 45, 1731–1739 (2022).
Restelli, F. et al. Confocal laser imaging in neurosurgery: a comprehensive review of sodium fluorescein-based CONVIVO preclinical and clinical applications. Front. Oncol. 12, 998384 (2022).
Zipfel, W. R., Williams, R. M. & Webb, W. W. Nonlinear magic: multiphoton microscopy in the biosciences. Nat. Biotechnol. 21, 1369–1377 (2003).
Drexler, W. & Fujimoto, J. G. (eds) Optical Coherence Tomography (Springer, 2015).
Arridge, S. R. Methods in diffuse optical imaging. Philos. Trans. R. Soc. A 369, 4558–4576 (2011).
Yamada, Y. & Okawa, S. Diffuse optical tomography: present status and its future. Opt. Rev. 21, 185–205 (2014).
Weber, J., Beard, P. C. & Bohndiek, S. E. Contrast agents for molecular photoacoustic imaging. Nat. Methods 13, 639–650 (2016).
Wang, L. V. & Yao, J. A practical guide to photoacoustic tomography in the life sciences. Nat. Methods 13, 627–638 (2016).
Ntziachristos, V. Going deeper than microscopy: the optical imaging frontier in biology. Nat. Methods 7, 603–614 (2010).
Wang, K., Liang, R. & Qiu, P. Fluorescence signal generation optimization by optimal filling of the high numerical aperture objective lens for high-order deep-tissue multiphoton fluorescence microscopy. IEEE Photon. J. 7, 1–8 (2015).
Aharoni, D., Khakh, B. S., Silva, A. J. & Golshani, P. All the light that we can see: a new era in miniaturized microscopy. Nat. Methods 16, 11–13 (2019).
Ghosh, K. K. et al. Miniaturized integration of a fluorescence microscope. Nat. Methods 8, 871–878 (2011).
Cai, D. J. et al. A shared neural ensemble links distinct contextual memories encoded close in time. Nature 534, 115–118 (2016).
Helmchen, F., Fee, M. S., Tank, D. W. & Denk, W. A miniature head-mounted two-photon microscope. Neuron 31, 903–912 (2001).
Sawinski, J. et al. Visually evoked activity in cortical cells imaged in freely moving animals. Proc. Natl Acad. Sci. USA 106, 19557–19562 (2009).
Zong, W. et al. Fast high-resolution miniature two-photon microscopy for brain imaging in freely behaving mice. Nat. Methods 14, 713–719 (2017).
Wallace, D. J. & Kerr, J. N. D. Circuit interrogation in freely moving animals. Nat. Methods 16, 9–11 (2019).
Asinof, S. K. & Card, G. M. Neural control of naturalistic behavior choices. Annu. Rev. Neurosci. 47, 369–388 (2024).
Mittmann, W. et al. Two-photon calcium imaging of evoked activity from L5 somatosensory neurons in vivo. Nat. Neurosci. 14, 1089–1093 (2011).
Klioutchnikov, A. et al. Three-photon head-mounted microscope for imaging deep cortical layers in freely moving rats. Nat. Methods 17, 509–513 (2020).
Russell, P. Photonic crystal fibers. Science 299, 358–362 (2003).
Beaulieu, D. R., Davison, I. G., Kılıç, K., Bifano, T. G. & Mertz, J. Simultaneous multiplane imaging with reverberation two-photon microscopy. Nat. Methods 17, 283–286 (2020).
Zong, W. et al. Large-scale two-photon calcium imaging in freely moving mice. Cell 185, 1240–1256.e30 (2022).
Accanto, N. et al. A flexible two-photon fiberscope for fast activity imaging and precise optogenetic photostimulation of neurons in freely moving mice. Neuron 111, 176–189.e6 (2023).
Acknowledgements
We thank P. Rupprecht and F. Xia for the valuable feedback and critical reading of this manuscript. This work was supported by the European Molecular Biology Laboratory (J.F.O. and R.P.). R.P. acknowledges support from the European Commission (ERC Consolidator Grant 864027, Brillouin4Life) and the Chan Zuckerberg Initiative DAF, an advised fund of Silicon Valley Community Foundation (projects 2020-225346 and 2024-337799). J.F.O. has received funding from the European Union’s Horizon 2020 research and innovation programme under the Marie Skłodowska-Curie grant agreement number 945405. M.B. acknowledges funding from the Emmy Noether Program of the German Research Foundation (DFG; Grant BR 6153/1-1) and the Else Kröner Fresenius Stiftung (2024_EKCS.06). V.V. and M.O.B. were supported by the Collaborative Research Center (CRC) 1389 (UNITE Glioblastoma). J.W. received funding from the National Institutes of Health (NS115585, NS121766). V.V. was supported by the DFG (project number VE1373/2-1516). V.V. received financial support from Else Kröner–Fresenius–Stiftung (2020-EKEA.135), the European Center for Neurooncology, the Health + Life Science Alliance Heidelberg Mannheim (together with R.P.), Heidelberg University, the Schwiete-Stiftung, the Wilhelm-Sander-Stiftung, the Stiftung Sibylle Assmus and Research Seed Capital from the Ministry of Science, Research and the Arts Baden Württemberg.
Author information
Authors and Affiliations
Contributions
R.P., J.F.O., J.W., S.J.S. and V.V. researched data for the article. R.P., J.N.D.K., J.W., M.M. and V.V. contributed substantially to the discussion of the content. R.P., J.F.O., J.N.D.K., J.W., M.O.B., B.D., S.J.S. and V.V. wrote the article and reviewed and/or edited the manuscript before submission. R.P. and V.V. coordinated and led the writing of the article.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Reviews Neuroscience thanks Ethan Hughes; Cristina Rodríguez, who co-reviewed with Malika Datta; and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Glossary
- Absorption cross section
-
A measure of the probability that a photon will be absorbed to excite a (fluorescence) molecule, determining the efficiency of fluorescence signal generation.
- Adaptive optics
-
A technology that corrects for wavefront distortions caused by the sample or imaging system, thereby improving image resolution and signal in deep and/or scattering biological tissues.
- Attenuation coefficient
-
A parameter describing the reduction in light intensity as it propagates through a medium, owing to scattering and absorption.
- Confocal microscopy
-
An imaging technique that uses a spatial pinhole to block out-of-focus light, thereby producing high-contrast optical sections of thick specimens.
- Dual-plane imaging
-
A microscopy method that simultaneously captures images from two different focal planes, enabling faster acquisition of three-dimensional data.
- Guidestars
-
Artificial or natural reference points emitting (fluorescence) signals, used in adaptive optics systems to measure and correct wavefront distortions for improved resolution and image quality.
- Multiphoton microscopy
-
An advanced imaging method wherein two or more low-energy photons are absorbed simultaneously to excite a fluorophore, enabling deep tissue imaging with high resolution and minimal photodamage.
- Optical sectioning
-
The ability of an imaging system to isolate thin slices of a sample along the axial direction, improving resolution and contrast in three-dimensional imaging.
- Optical wavefront shaping
-
The manipulation of light wavefronts using devices such as spatial light modulators to control and correct for aberrations introduced by complex, highly scattering media.
- Remote focusing
-
A technique to adjust the focal plane of a microscope rapidly and precisely by manipulating the optical path without moving the primary objective lens.
- Third-order harmonic signal
-
A nonlinear optical signal generated when three photons combine to produce a single photon with triple the energy (or one-third the wavelength), enabling visualization of intrinsic sample structures without fluorescent labels.
- Volumetric imaging
-
A method for capturing 3D data sets by acquiring images across multiple depths, enabling detailed reconstruction of structures in three dimensions.
- Wavefront distortions
-
Aberrations in the light wavefront caused by refractive index variations in a sample, degrading image quality in microscopy.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Prevedel, R., Ferrer Ortas, J., Kerr, J.N.D. et al. Three-photon microscopy: an emerging technique for deep intravital brain imaging. Nat. Rev. Neurosci. 26, 521–537 (2025). https://doi.org/10.1038/s41583-025-00937-y
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41583-025-00937-y