Abstract
Tuberculosis (TB) is the most common cause of death from infectious disease worldwide. A substantial proportion of patients presenting with extrapulmonary TB have urogenital TB (UG-TB), which can easily be overlooked owing to non-specific symptoms, chronic and cryptic protean clinical manifestations, and lack of clinician awareness of the possibility of TB. Delay in diagnosis results in disease progression, irreversible tissue and organ damage and chronic renal failure. UG-TB can manifest with acute or chronic inflammation of the urinary or genital tract, abdominal pain, abdominal mass, obstructive uropathy, infertility, menstrual irregularities and abnormal renal function tests. Advanced UG-TB can cause renal scarring, distortion of renal calyces and pelvic, ureteric strictures, stenosis, urinary outflow tract obstruction, hydroureter, hydronephrosis, renal failure and reduced bladder capacity. The specific diagnosis of UG-TB is achieved by culturing Mycobacterium tuberculosis from an appropriate clinical sample or by DNA identification. Imaging can aid in localizing site, extent and effect of the disease, obtaining tissue samples for diagnosis, planning medical or surgical management, and monitoring response to treatment. Drug-sensitive TB requires 6–9 months of WHO-recommended standard treatment regimens. Drug-resistant TB requires 12–24 months of therapy with toxic drugs with close monitoring. Surgical intervention as an adjunct to medical drug treatment is required in certain circumstances. Current challenges in UG-TB management include making an early diagnosis, raising clinical awareness, developing rapid and sensitive TB diagnostics tests, and improving treatment outcomes.
Key points
-
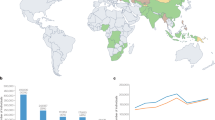
Between 15% and 40% of the 10 million new patients diagnosed with tuberculosis (TB) annually present with extrapulmonary TB (EPTB), of which a considerable proportion have urogenital TB (UG-TB). Patients who have had a renal transplant, have HIV infection, receive immunosuppressive therapies, have diabetes, have COPD and those undergoing dialysis often experience reactivation of latent TB infection.
-
UG-TB is often missed clinically or is diagnosed late, owing to the lack of awareness among clinicians, its insidious onset, chronic non-specific symptoms, and cryptic and protean clinical manifestations, resulting in disease progression.
-
Specific diagnosis of TB is made by identification of Mycobacterium tuberculosis (Mtb) in clinical samples, by microscopy and culture, or by identification of Mtb DNA. Imaging can aid in identifying disease sites and obstructive lesions, guiding biopsies and surgical interventions
-
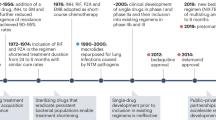
Treatment of drug-sensitive TB requires 6–9 months of the WHO-recommended standard treatment regimen, but longer therapy is needed for severe disease or in patients in whom immunosuppression is an underlying risk factor. Multidrug-resistant TB requires between 12 and 24 months of therapy with toxic drugs and careful monitoring.
-
Surgery is indicated for complications of UG-TB. Nephrectomy is required for severely damaged kidneys, and reconstruction procedures include pyeloureteral anastomosis, ureterocalyceal anastomosis, caliceal reconstruction, uretero-ureteral anastomosis and ureter substitution by ileum.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$32.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$189.00 per year
only $15.75 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout









Similar content being viewed by others
References
WHO. The top 10 causes of death. WHO https://www.who.int/news-room/fact-sheets/detail/the-top-10-causes-of-death (2019).
WHO. WHO Global Tuberculosis report 2018. WHO http://who.int/tb/publications/global_report/en/ (2018).
Floyd, K., Glaziou, P., Zumla, A. & Raviglione, M. The global tuberculosis epidemic and progress in care, prevention, and research: an overview in year 3 of the End TB era. Lancet Respir. Med. 6, 299–314 (2018).
WHO. Guidelines for the treatment of drug-susceptible tuberculosis and patient care. 2017 update. WHO http://apps.who.int/iris/bitstream/10665/255052/1/9789241550000-eng.pdf?ua=1 (2017).
Lawn, S. D. & Zumla, A. I. Tuberculosis (Seminar). Lancet 378, 57–72 (2011).
Furin, J., Cox, H. & Pai, M. Tuberculosis. Lancet 393, 1642–1656 (2019).
Kulchavenya, E. Extrapulmonary tuberculosis: are statistical reports accurate? Ther. Adv. Infect. Dis. 2, 61–70 (2014).
Porter, M. F. III Uro-genital tuberculosis in the male. Ann. Surg. 20, 396–405 (1894).
Wildbolz, H. Ueber urogenical tuberkulose. Schweiz. Med. Wochenschr. 67, 1125 (1937).
Kulchavenya, E., Naber, K. & Bjerklund Johansen, T. E. Urogenital tuberculosis: classification, diagnosis, and treatment. Eur. Urol. 15, 112–121 (2016).
Adhikari, S. & Basnyat, B. Extrapulmonary tuberculosis: a debilitating and often neglected public health problem. BMJ Case Rep. 11, e226098 (2018).
Abbara, A. & Davidson, R. N. Etiology and management of genitourinary tuberculosis. Nat. Rev. Urol. 8, 678–688 (2011).
Figueiredo, A. A. & Lucon, A. Urogenital tuberculosis: update and review of 8961 cases from the world literature. Rev. Urol. 10, 207–217 (2008).
Gow, J. & Barbosa, S. Genitourinary tuberculosis: a study of 1117 cases over a period of 34 years. Br. J. Urol. 56, 449–455 (1984).
Ishibashi, Y., Takeda, T., Nishimura, R. & Ohshima, H. A clinical observation of genitourinary tract tuberculosis during the last decade. HinyokikaKiy 31, 107–112 (1985).
Chtourou, M. et al. Management of genito-urinary tuberculosis. A report of 225 cases. J. Urol. 161, 9 (1999).
Gokce, G. et al. Genitourinary tuberculosis: review of 174 cases. Scand. J. Infect. Dis. 34, 338–340 (2002).
Ferrie, B. G. & Rundle, J. S. H. Genito-urinary tuberculosis in Glasgow 1970 to 1979: a review of 230 patients. Scott. Med. J. 30, 30–34 (1985).
Krishnamoorthy, S. et al. Aspects of evolving genito urinary tuberculosis – a profile of genito urinary tuberculosis (GUTB) in 110 patients. J. Clin. Diagn. Res. 11, PC01–PC05 (2017).
Christensen, W. I. Genitourinary tuberculosis. Review of 102 cases. Medicine 53, 377–390 (1974).
Soriano-Rosas, J. et al. AIDS-associated nephropathy: 5-year retrospective morphologic analysis of 87 cases. Pathol. Res. Pract. 194, 567–570 (1998).
Hsieh, H. C. et al. Uro-genital tuberculosis in a medical center in southern Taiwan: an eleven-year experience. J. Microbiol. Immunol. Infect. 39, 408–413 (2006).
Lessnau, K. D. et al. Tuberculosis of the genito-urinary system overview of GUTB. Medscape http://emedicine.medscape.com/article/450651-overview#aw2aab6b4.(2015).
Ye, Y. et al. Clinical features and drug-resistance profile of urinary tuberculosis in south-western China: a cross-sectional study. Medicine 95, e3537 (2016).
Garcia-Rodríguez, J. Á., García Sanchez, J. E. & Muñoz Bellido, J. L. Genitourinary tuberculosis in Spain: review of 81 cases. Clin. Infect. Dis. 18, 557–561 (1994).
Nogales-Ortiz, F., Tarancón, I. & Nogales, F. F. Jr. The pathology of female genital tuberculosis. A 31-year study of 1436 cases. Obstet. Gynecol. 53, 422 (1979).
Grange, J. M. in Tuberculosis — A Comprehensive Clinical Reference. 44–59 (eds Schaff, S. & Alimuddin Zumla, A.) (Saunders Elsevier, 2009).
Riojas, M. A., McGough, K. J., Rider-Riojas, C. J., Rastogi, N. & Hazbón, M. H. Phylogenomic analysis of the species of the Mycobacterium tuberculosis complex demonstrates that Mycobacterium africanum, Mycobacterium bovis, Mycobacterium caprae, Mycobacterium microti and Mycobacterium pinnipedii are later heterotypic synonyms of Mycobacterium tuberculosis. Int. J. Syst. Evol. Microbiol. 68, 324–332 (2018).
Watt, C. J., Hosseini, S. M., Lonnroth, K., Williams, B. G. & Dye, C. in Tuberculosis — a comprehensive clinical reference. 37 (eds Schaff, S. & Alimuddin Zumla, A.) (Saunders Elsevier, 2009).
Silva, M. R. et al. Risk factors for human Mycobacterium bovis infections in an urban area of Brazil. Mem. Inst. Oswaldo Cruz 113, e170445 (2018).
Zachoval, R. et al. The incidence of subclinical forms of urogenital tuberculosis in patients with pulmonary tuberculosis. J. Infect. Publ. Health 11, 243–245 (2018).
Figueiredo, A. A., Lucon, A. M. & Junior, R. F. Epidemiology of urogenital tuberculosis worldwide. Int. J. Urol. 15, 827–832 (2008).
Yadav, S., Singh, P., Hemal, A. & Kumar, R. Genital tuberculosis: current status of diagnosis and management. Transl. Androl. Urol. 6, 222–233 (2017).
Schubert, G. E., Haltaufderheide, T. & Golz, R. Frequency of urogenital tuberculosis in an unselected autopsy series from 1928 to 1949 and 1976 to 1989. Eur. Urol. 21, 216–223 (1992).
Medlar, E., Spain, D. & Holliday, R. Post mortem compared with clinical diagnosis of genitourinary tuberculosis in adult males. J. Urol. 61, 1078–1088 (1949).
Vithalani, N. & Udani, P. M. A study of 292 autopsies proved cases of tuberculosis. Indian J. Tuber. 29, 93–97 (1982).
Lanjewar, D. N., Ansari, M. A., Shetty, C. R., Maheshwary, M. B. & Jain, P. Renal lesions associated with AIDS — an autopsy study. Indian J. Pathol. Microbiol. 42, 63–68 (1999).
Perez, S., Andrade, M., Bergel, P., Bracho, Y. & de Waard, J. H. A simple algorithm for the diagnosis of AIDS-associated genitourinary tuberculosis. Clin. Infect. Dis. 42, 1807–1808 (2006).
Grace, G. A., Devaleenal, D. B. & Natrajan, M. Genital tuberculosis in females. Indian J. Med. Res. 145, 425–436 (2017).
Parvez, R. et al. Prevalence of female genital tuberculosis, its risk factors and associated clinical features among the women of Andaman Islands, India: a community-based study. Publ. Health 148, 56–62 (2017).
Naing, C., Mak, J. W., Maung, M., Wong, S. F. & Kassim, A. I. Meta-analysis: the association between HIV infection and extrapulmonary tuberculosis. Lung 191, 27–34 (2013).
Fanosie, A. et al. Mycobacterium tuberculosis complex and HIV co-infection among xxtrapulmonary tuberculosis suspected cases at the University of Gondar Hospital, Northwestern Ethiopia. PLOS ONE 11, e0150646 (2016).
Tubach, F. et al. Risk of tuberculosis is higher with anti-tumor necrosis factor monoclonal antibody therapy than with soluble tumor necrosis factor receptor therapthehe three-year prospective French Research Axed on Tolerance of Biotherapies registry. Arthritis Rheum. 60, 1884–1894 (2009).
Ferrara, G. et al. Risk factors associated with pulmonary tuberculosis: smoking, diabetes and anti-TNFα drugs. Curr. Opin. Pulm. Med. 18, 233–240 (2012).
Chattopadhyay, A. et al. Genitourinary tuberculosis in pediatric surgical practice. J. Pediatr. Surg. 32, 1283–1286 (1997).
Narayana, A. S. Overview of renal tuberculosis. Urology 19, 231–237 (1982).
Rutkowski, B., Gillow, A. S., Kustosz, J., Liberek, T. & Zdrojewski, Z. Increasing incidence of tuberculosis in hemodialysis patients. Dial. Transplant. 26, 21–25 (1997).
Ulubay, G. et al. 10-year experience of tuberculosis in solid-organ transplant recipients. Exp. Clin. Transplant. 13, 214–218 (2015).
Gras, J. et al. Rapid diagnosis of tuberculosis through the detection of mycobacterial DNA in urine by nucleic acid amplification methods. Lancet Infect. Dis. 9, 505–511 (2009).
Higuita, L. M. et al. Tuberculosis in renal transplant patients: the experience of a single center in Medellín-Colombia, 2005–2013. J. Bras. Nefrol. 36, 512–518 (2014).
Reis-Santos, B., Gomes, T., Horta, B. L. & Maciel, E. L. Tuberculosis prevalence in renal transplant recipients: systematic review and meta-analysis. J. Bras. Nefrol. 35, 206–213 (2013).
Anand, M., Nayyar, E., Concepcion, B., Salani, M. & Schaefer, H. Tuberculosis in kidney transplant recipients: a case series. World J. Transplant. 7, 213–221 (2017).
Dharmadhikari, A. S. & Nardell, E. A. in Tuberculosis — A Comprehensive Clinical Reference 8–16 (eds Schaff, S. & Alimuddin Zumla, A.) (Saunders Elsevier, 2009).
Chang, C. W. et al. Congenital tuberculosis: case report and review of the literature. Paediatr. Int. Child Health. 19, 1–4 (2017).
Dewan, P., Gomber, S. & Das, S. Congenital tuberculosis: a rare manifestation of a common diseases. Paediatr. Int. Child Health 34, 60–62 (2014).
Newberry, D. M. & Robertson-Bell, T. Congenital tuberculosis: a new concern in neonatal intensive care units. Adv. Neonatal Care 18, 341–349 (2018).
Lhadon, T. & Jullien, S. Congenital multidrug-resistant tuberculosis in a neonate: a case report. J. Trop. Pediatr. 65, 188–191 (2018).
Aldana-Aguirre, J. C., El-Hakim, H., Phillipos, E. & Landry, M. A. Congenital tuberculosis presenting as otorrhoea in a preterm infant. BMJ Case Rep. 2018, bcr-2017-221797 (2018).
Fang, X., Mai, R., Guo, J. & Lin, N. A pre-term infant of 32 weeks gestation with congenital tuberculosis treated successfully with antituberculosis chemotherapy. Paediatr. Int. Child Health 14, 1–3 (2017).
Raj, P. & Sarin, Y. K. Congential tuberculosis in a neonate: a diagnostic dilemma. J. Neonatal Surg. 3, 49 (2014).
Angus, B. J., Yates, M., Conlon, C. & Byren, I. Cutaneous tuberculosis of the penis and sexual transmission of tuberculosis confirmed by molecular typing. Clin. Infect. Dis. 33, E132–E134 (2001).
Veenema, R. J. & Lattimer, J. K. Genital tuberculosis in the male: clinical pathology and effect on fertility. J. Urol. 78, 65–77 (1957).
Lattimer, J. Renal tuberculosis. N. Engl. J. Med. 273, 208–211 (1965).
Venyo, A. K. Tuberculosis of the penis: a review of the literature. Scientifica 2015, 601–624 (2015).
Hesseling, A. C. et al. Consensus statement on the revised World Health Organization recommendations for BCG vaccination in HIV-infected infants. Int. J. Tuberc. Lung Dis. 12, 1376–1379 (2008).
Von Reyn, C. F. et al. Disseminated tuberculosis in human immunodeficiency virus infection: ineffective immunity, polyclonal disease and high mortality. Int. J. Tuberc. Lung Dis. 8, 1087–1089 (2011).
Sihra, N., Diasuke, N., Thurairaja, R., Khan, M. S. & Malde, S. Renal tuberculosis following intravesical bacillus Calmette-Guérin for high-grade non-muscle-invasive bladder cancer. Urology 107, e3–e4 (2017).
Lamm, D. Efficacy and safety of bacille Calmette Guerin immunotherapy in superficial bladder cancer. Clin. Infect. Dis. 31, 86–90 (2000).
Falkensammer, C. et al. Late occurrence of bilateral tuberculous-like epididymo-orchitis after intravesical bacille Calmette-Guerin therapy for superficial bladder carcinoma. Urology 65, 175 (2005).
Colomba, C. et al. Case of epididymo-orchitis after intravesical bacille Calmette-Guérin therapy for superficial bladder carcinoma in a patient with latent tuberculosis infection. Infect. Agent. Cancer 11, 25 (2016).
Lee, S. Y. & Choi, S. H. Treatment experience for incidentally diagnosed asymptomatic prostate tuberculosis in a patient with history of BCG intravesical therapy. Urol. Case Rep. 17, 39–41 (2017).
Klebanov, N. & Raghavan, A. Tuberculous orchitis following intravesical Bacille Calmette-Guérin (BCG) therapy. Cureus 10, e2703 (2018).
Hunter, R. L. Tuberculosis as a three-act play: A new paradigm for the pathogenesis of pulmonary tuberculosis. Tuberculosis 97, 8–17 (2016).
Rao, M. et al. Latent TB Infection (LTBI) — Mycobacterium tuberculosis pathogenesis and the dynamics of the granuloma battleground. Int. J. Infect. Dis. 80S, S58–S61 (2019).
Bezuidenbhout, J. & Schneider, J. W. in Tuberculosis — A Comprehensive Clinical Reference 117–128 (eds Schaff, S. & Alimuddin Zumla, A.) (Saunders Elsevier, 2009).
Menzies, N. A. et al. Progression from latent infection to active disease in dynamic tuberculosis transmission models: a systematic review of the validity of modelling assumptions. Lancet Infect. Dis. 18, e228–e238 (2018).
Salvatore, P. P., Proaño, A., Kendall, E. A., Gilman, R. H. & Dowdy, D. W. Linking Individual natural history to population outcomes in tuberculosis. J. Infect. Dis. 217, 112–121 (2017).
Vynnycky, E. & Fine, P. E. The natural history of tuberculosis: the implications of age-dependent risks of disease and the role of reinfection. Epidemiol. Infect. 119, 183–201 (1997).
Simmons, J. D. et al. Immunological mechanisms of human resistance to persistent Mycobacterium tuberculosis infection. Nat. Rev. Immunol. 18, 575–589 (2018).
Lamba, H., Byrne, M., Goldin, R. & Jenkins, C. Tuberculosis of the cervix: case presentation and a review of the literature. Sex. Transm. Infect. 78, 62–63 (2002).
Aphonin, A. B., Perezmanas, E. O., Toporkova, E. E. & Khodakovsky, E. P. Tuberculous infection as sexually transmitted infection. Vestn. Poslediplomnogo Obrazovaniya 3, 69–71 (2006).
Regmi, S. K., Singh, U. B., Sharma, J. B. & Kumar, R. Relevance of semen polymerase chain reaction positive for tuberculosis in asymptomatic men undergoing infertility evaluation. J. Hum. Reprod. Sci. 8, 165–169 (2015).
Barmon, D., Kataki, A. C., Sharma, J. D. & Gharpholia, D. A case of cervical tuberculosis mimicking cervical carcinoma. J. Obstet. Gynaecol. India. 63, 285–287 (2013).
Holt, L. E. Tuberculosis acquired through ritual circumcision. J. Am. Med. Associ. 61, 99–102 (1913).
Zumla, A. & James, D. G. Granulomatous infections: etiology and classification. Clin. Infect. Dis. 23, 146–158 (1996).
Houben, R. M. & Dodd, P. J. The global burden of latent tuberculosis infection: a re-estimation using mathematical modelling. PLOS Med. 13, e1002152 (2016).
Butler, M. R. & O’Flynn, D. Reactivation of genitourinary tuberculosis. Eur. Urol. 1, 14–17 (1975).
Arora, N., Saha, A. & Kaur, M. Tuberculous pyelonephritis in children: three case reports. Paediatr. Int. Child Health. 37, 292–297 (2017).
Kretschmer, H. L. Tuberculosis of the kidney, a critical review based on a series of 221 cases. N. Engl. J. Med. 202, 660–671 (1930).
Ross, J. C. Renal tuberculosis. Br. J. Urol. 25, 277–292 (1953).
Eastwood, J. B., Corbishley, C. M. & Grange, J. M. Tuberculosis and the kidney. J. Am. Soc. Nephrol. 12, 1307–1314 (2001).
Qiu, S. P. et al. A clinical study of 281 cases of renal tuberculosis. Chin. J. Urol. 23, 398–400 (2002).
Daher, E. F., Bezerra da Silva, G. & Guardao Barros, E. J. Renal tuberculosis in the modern era. Am. Trop. Med. Hyg. 88, 54–64 (2013).
Medlar, E. M. Cases of renal infection in pulmonary tuberculosis: evidence of healed tuberculous lesions. Am. J. Pathol. 2, 401–411 (1926).
Medlar, E. M. & Sasano, K. T. Experimental renal tuberculosis, with special reference to excretory bacilluria. Am. Rev. Tuberc. 10, 370–377 (1924).
Marques, L. P., Rioja, L. S., Oliveira, C. A. & Santos, O. D. AIDS-associated renal tuberculosis. Nephron 74, 701–704 (1996).
Shah, S., Carter-Monroe, N. & Atta, M. G. Granulomatous interstitial nephritis. Clin. Kidney J. 5, 516–523 (2015).
Prakash, J., Goel, A. & Sankhwar, Singh, B. Extensive renal and ureteral calcification due to tuberculosis: rare images for an uncommon condition. BMJ Case Rep. 2013, bcr2012008508 (2013).
Patil, S. B., Desai, A. S., Biradar, A. N. & Kundargi, V. S. Extensive nephroureteric calcification presenting with renal failure: a rare case report. Urol. Ann. 7, 375–377 (2015).
Kulchavenya, E. V., Shevchenko, S. Y. & Cherednichenko, A. G. Diagnosis and treatment of cystitis: more questions than answers? Urologiia 5, 37–42 (2016).
Lima, N. A. et al. Review of uro-genital tuberculosis with focus on end-stage renal disease. Rev. Inst. Med. Trop. Sao Paulo 54, 57–60 (2012).
Mallinson, W. J., Fuller, R. W., Levoson, D. A., Baker, L. R. & Cattel, W. R. Diffuse interstitial renal tuberculosis an unusual cause of renal failure. Q. J. Med. 50, 137–148 (1981).
Eastwood, J. B., Zaidi, M., Maxwell, J. D., Wing, A. J. & Pazianas, M. Tuberculosis as primary renal diagnosis in end-stage uraemia. J. Nephrol. 7, 290–293 (1994).
Oliverira, J. L., Silva Junior, G. B. & Daher, E. F. Tuberculosis associated chronic kidney disease. Am. J. Trop. Med. Hyg. 84, 843–844 (2011).
Lee, K. S. et al. Laparoscopic nephrectomy for tuberculous non-functioning kidney: comparison with laparoscopic simple nephrectomy for other diseases. Urology 60, 411–414 (2002).
Hemal, A. K., Gupta, N. P. & Rajeev, K. Comparison of retroperitoneoscopic nephrectomy with open surgery for tuberculous non-functioning kidneys. J. Urol. 164, 32–35 (2000).
Merchant, S., Bharati, A. & Merchant, N. Tuberculosis of the uro-genital system — urinary tract tuberculosis: renal tuberculosis. I. Indian J. Radiol. Imaging 23, 46–63 (2013).
Merchant, S., Bharati, A. & Merchant, N. Tuberculosis of the uro-genital system — urinary tract tuberculosis: renal tuberculosis. II. Indian J. Radiol. Imaging 23, 64–77 (2013).
Wei, H. L. et al. Renal tuberculosis and iliopsoas abscess: two case reports. Exp. Ther. Med. 7, 1718–1720 (2014).
Puigvert, A. The ureter in renal tuberculosis. Br. J. Urol. 27, 258–262 (1955).
Friedenberg, R. M., Ney, C. & Stachenfeld, R. A. Roentgenographic manifestations of tuberculosis of ureter. J. Urol. 99, 25–29 (1968).
Gibson, M. S., Puckett, M. L. & Shelly, M. E. Renal tuberculosis. Radiographics 24, 1 (2004).
Roylance, J., Penry, B., Rhys Davies, E. & Roberts, M. Radiology in the management of urinary tract tuberculosis. Br. J. Urol. 42, 679–687 (1970).
Johnstone, A. S. Tuberculous cystitis; notes on three cases. Br. J. Radiol. 20, 61–62 (1947).
Kulchavenya, E. & Cherednichenko, A. Urogenital tuberculosis, the cause of ineffective antibacterial therapy for urinary tract infections. Ther. Adv. Urol. 10, 95–101 (2017).
Shah, H. U., Sannananja, B., Baheti, A. D., Udare, A. S. & Badhe, P. V. Hysterosalpingography and ultrasonography findings of female genital tuberculosis. Diagn. Interv. Radiol. 21, 10–15 (2015).
Singal, A., Pandhi, D., Kataria, V. & Arora, V. K. Tuberculosis of the glans penis: an important differential diagnosis of genital ulcer disease. Int. J. STD AIDS 28, 1453–1455 (2017).
Kulchavenya, E., Brizhatyuk, E. & Khomyakov, V. Diagnosis and therapy for prostate tuberculosis. Ther. Adv. Urol. 6, 129–134 (2014).
Miller, E. & Lustok, M. Genital tuberculosis. JAMA 113, 1388–1394 (1939).
López Barón, E., Gómez-Arbeláez, D. & Díaz-Pérez, J. Primary prostatic tuberculosis. Case report and bibliographic review. Arch. Esp. Urol. 62, 309–313 (2009).
Sporer, A. & Oppenheimer, G. Tuberculosis of the prostate and seminal vesicles. J. Urol. 78, 278–286 (1957).
Stillwell, T., Engen, D. & Farrow, G. The clinical spectrum of granulomatous prostatitis: a report of 200 cases. J. Urol. 138, 320–323 (1987).
Gupta, N., Mandal, A. K. & Singh, S. K. Tuberculosis of the prostate and urethra: a review. Indian J. Urol. 24, 388–391 (2008).
Bouchikhi, A. A., Khallouk, A., El Fassi, M. J. & Farih, M. H. Atypical isolated urethral tuberculosis associated with inflammatory stenosis and fistulas. Urol. Ann. 6, 270–271 (2014).
Hosamirudsari, H. & Mohammadizia, F. Unilateral tuberculous epididymo-orchitis with scrotal fistula: a case report. Iran. J. Pathol. 10, 165–168 (2015).
Joneja, U., Short, W. R. & Roberts, A. L. Disseminated tuberculosis with prostatic abscesses in an immunocompromised patient — a case report and review of literature. IDCases 5, 15–20 (2016).
Kostakopoulos, A. et al. Tuberculosis of the prostate. Int. Urol. Nephrol. 30, 153–157 (1998).
Bour, L. et al. Multiparametric MRI features of granulomatous prostatitis and tubercular prostate abscess. Diagn. Interv. Imaging 94, 84–90 (2013).
Lee, I. K., Yang, W. C. & Liu, J. W. Scrotal tuberculosis in adult patients: a 10-year clinical experience. Am. J. Trop. Med. Hyg. 77, 714–718 (2007).
Borthwick, W. The pathogenesis of tuberculous epididymitis. Edin. Med. J. 53, 55–70 (1946).
Riehle, R. A. & Jayraman, K. Tuberculosis of testis. Urology 1, 43–46 (1982).
Ross, J. C., Gow, J. G. & St. Hill, C. A. Tuberculous epididymitis. A review of 170 patients. Br. J. Surg. 48, 663–666 (1961).
Das, A., Batabyal, S., Bhattacharjee, S. & Sengupta, A. A rare case of isolated testicular tuberculosis and review of literature. J. Family Med. Prim. Care. 5, 468–470 (2016).
Jacob, J. T., Nguyen, T. M. & Ray, S. M. Male genital tuberculosis. Lancet Infect. Dis. 8, 335–342 (2008).
Madeb, R., Marshal, J., Nativ, O. & Erturk, E. Epididymal tuberculosis: case report and review of the literature. Urology 65, 798 (2005).
Viswaroop, B. S., Kekre, N. & Gopalakrishnan, G. Isolated tuberculous epididymitis: a review of forty cases. J. Postgrad. Med. 51, 109–111 (2005).
Hadadi, A., Pourmand, G. & Mehdipour-Aghabagher, B. Unilateral testicular tuberculosis: case report. Andrology 44, 70–72 (2012).
Kretschmer, H. Tuberculosis of the epididymis: a review of 170 patients. Surg. Gynecol. Obstet. 47, 652–659 (1928).
Heaton, N. D., Hogan, B., Michell, M., Thompson, P. & Yates-Bell, A. J. Tuberculous epididymo-orchitis: clinical and ultrasound observations. Br. J. Urol. 64, 305–309 (1989).
Benjelloun, A., Elktaibi, A., Elharrech, Y., Touiti, D. & Ghoundale, O. Tuberculosis of the spermatic cord: case report. Urol. Case Rep. 2, 176–177 (2014).
Premkumar, A. & Newhouse, J. Seminal vesicle tuberculosis: CT appearance. J. Comput. Assist. Tomogr 12, 676–677 (1988).
Kar, J. & Kar, M. Primary tuberculosis of the glans penis. J. Assoc. Physicians India 60, 52–53 (2012).
Khan, D., Choudhary, A., Dutta, A. & Khan, I. Tuberculosis of the glans penis mimicking as carcinoma. Int. J. Mycobacteriol. 5, 341–342 (2016).
Symes, J. M. & Blandy, J. P. Tuberculosis of the male urethra. Br. J. Urol. 45, 432–436 (1973).
Indudhara, R., Vaidyanathan, S. & Radotra, B. D. Urethral tuberculosis. Urol. Int. 48, 436–438 (1992).
Paul, J., Krishnamoorthy, S., Teresa, M. & Kumar, S. Isolated tuberculous orchitis: a mimicker of testicular malignancy. Indian J. Urol. 26, 284–286 (2010).
Gangalakshmi, C. & Sankaramahalingam, A. Tuberculosis of the glans penis. J. Clin. Diagn. Res. 12, PD05–PD06 (2016).
Chowdhury, A. & Dey, R. Penile tuberculosis following intravesical Bacille Calmette-Guérin immunotherapy. Indian J. Urol. 29, 64–66 (2013).
Mayilvaganan, K. R., Naren Satya Srinivas, M., Reddy, V. N. & Singh, R. K. Tuberculosis penis with ‘Watering can penis’ appearance: report of a rare case with retrograde urethrography and voiding cystourethrography findings. Pol. J. Radiol. 81, 454–457 (2016).
Buppasiri, P., Temtanakitpaisan, T. & Somboonporn, W. Tuberculosis at vulva and vagina. J. Med. Assoc. Thai. 93, 613–615 (2010).
Nemati, E., Taheri, S., Nourbala, M. H. & Einollahi, B. Vaginal tuberculosis in an elderly kidney transplant recipient. Saudi J. Kidney Dis. Transpl. 20, 465–467 (2009).
Sharma, C., Shekhar, S., Sharma, V., Sharma, M. & Aggarwal, T. Paucibacillary tubercular vulval ulcer in a sexually inactive pubertal girl: role of therapeutic trial. J. Pediatr. Adolesc. Gynecol. 25, e123–e124 (2012).
Tiwari, P., Pal, D. K., Moulik, D. & Choudhury, M. K. Hypertrophic tuberculosis of vulva — a rare presentation of tuberculosis. Indian J. Tuberc 57, 95–97 (2010).
Mondal, S. K. Histopathologic analysis of female genital tuberculosis: a fifteen-year retrospective study of 110 cases in eastern India. Turk Patoloji Derg. 29, 41–45 (2013).
Kulchavenya, E. & Dubrovina, S. Typical and unusual cases of female genital tuberculosis. IDCases 1, 92–94 (2014).
Mahajan, N., Naidu, P. & Deep Kaur, S. Insight into the diagnosis and management of subclinical genital tuberculosis in women with infertility. J. Hum. Reprod. Sci. 9, 135–144 (2016).
Das, P., Ahuja, A. & Gupta, S. D. Incidence, etiopathogenesis and pathological aspects of genitourinary tuberculosis in India: a journey revisited. Indian J. Urol. 24, 356–361 (2008).
Türkmen, I. C. et al. Female genital system tuberculosis: a retrospective clinicopathological study of 1,548 cases in Turkish women. Arch. Gynecol. Obstet. 286, 379–384 (2012).
Sharma, J. B. Current diagnosis and management of female genital tuberculosis. J. Obstet. Gynaecol. India 65, 362–371 (2015).
Akbulut, S., Arikanoglu, Z. & Basbug, M. Tubercular tubo-ovarian cystic mass mimicking acute appendicitis: a case report. J. Med. Case Rep. 5, 363 (2011).
Adsuar, N., Blanchette, H. & Kilchevsky, E. Tuberculosis peritonitis mimicking ovarian cancer in a 20-year-old woman. A case report. J. Reprod. Med. 49, 52–54 (2004).
Neonakis, I. et al. Genital tuberculosis in a tamoxifen-treated postmenopausal woman with breast cancer and bloody vaginal discharge. Ann. Clin. Microbiol. Antimicrob. 5, 20 (2006).
Shirazi, M. et al. Tuberculosis endometritis presenting as a leiomyoma. Int. J. Fertil. Steril. 8, 481–484 (2015).
Samal, S., Gupta, U. & Agarwal, P. Menstrual disorders in genital tuberculosis. J. Indian Med. Assoc. 98, 126–127,129 (2000).
Perdhana, R. et al. Patients with secondary amenorrhea due to tuberculosis endometritis towards the induced anti-tuberculosis drug category 1. Pan Afr. Med. J. 24, 121 (2016).
Nezar, M. et al. Genital tract tuberculosis among infertile women: an old problem revisited. Arch. Gynecol. Obstet. 280, 787–791 (2009).
Singh, N., Sumana, G. & Mittal, S. Genital tuberculosis: a leading cause for infertility in women seeking assisted conception in North India. Arch. Gynecol. Obstet. 278, 325–327 (2008).
Sharma, J. B. et al. Laparoscopic findings in female genital tuberculosis. Arch. Gynecol. Obstet. 278, 359–364 (2008).
Sharma, J. B. et al. High prevalence of Fitz-Hugh-Curtis Syndrome in genital tuberculosis. Int. J. Gynaecol. Obstet. 99, 62–63 (2007).
Coremans, L. & de Clerck, F. Fitz-Hugh-Curtis syndrome associated with tuberculous salpingitis and peritonitis: a case presentation and review of literature. BMC Gastroenterol. 18, 42 (2018).
Singh, S. et al. Tuberculosis of uterine cervix: a report of two cases with variable clinical presentation. Trop. Doct. 40, 125–126 (2010).
Mukerji, S. et al. Difficulties in diagnosing tuberculosis of the cervix in a post menopausal woman: case report and literature review. Australas. Med. J. 6, 367–370 (2013).
Sharma, N., Singh, A. S., Khonglah, Y. & Mishra, J. Primary tuberculosis of cervix: a coincidental finding. J. Reprod. Infertil. 17, 247–249 (2016).
Rodpenpear, N. Tuberculosis of cervix resembling cervical. Cancer J. Med. Assoc. Thai. 99, S249–S252 (2016).
Jaiprakash, P., Pai, K. & Rao, L. Diagnosis of tuberculous cervicitis by Papanicolaou-stained smear. Ann. Saudi Med. 33, 76–78 (2013).
Sabita, S., Sharmila, V., Arun Babu, T., Sinhasan, S. & Darendra, S. A rare case of cervical tuberculosis which simulated carcinoma of the cervix. J. Clin. Diagn. Res. 7, 1189–1190 (2013).
Nanjappa, V. et al. Vulval tuberculosis — an unusual presentation of disseminated tuberculosis. J. Assoc. Physicians India 60, 49–52 (2012).
Mallya, V., Yadav, Y. K. & Gupta, K. Vulval tuberculosis masquerading as vulval carcinoma. J. Postgrad. Med. 58, 307–308 (2012).
Wang, J., Fan, S., Xiao, J. & Liang, C.-Z. Renal tuberculosis tends to be low symptoms: how to improve the diagnosis and treatment of renal tuberculosis. Asian J. Androl. 18, 145–146 (2016).
Wise, G. J. & Marella, V. K. Uro-genital manifestations of tuberculosis. Urol. Clin. N. Am. 30, 111–121 (2003).
Kapoor, R., Ansari, M. S., Mandhani, A. & Gulia, A. Clinical presentation and diagnostic approach in cases of uro-genital tuberculosis. Indian J. Urol. 24, 401–405 (2008).
Zarrabi, A. D. & Heyns, C. F. Clinical features of confirmed versus suspected urogenital tuberculosis in region with extremely high prevalence of pulmonary tuberculosis. Urology 74, 41–45 (2009).
Kulchavenya, E. & Kholtobin, D. Diseases masking and delaying the diagnosis of urogenital tuberculosis. Ther. Adv. Urol. 7, 331–338 (2015).
Bacci, M. R., Namura, J. J. & Lera, A. T. Complicated urinary infection and extrapulmonary tuberculosis. BMJ Case Rep. 2012, bcr2012007553 (2012).
Kulchavenya E., Kholtobin D., Shevchenko S. Challenges in urogenital tuberculosis. World J. Urol. https://doi.org/10.1007/s00345-019-02767-x (2019).
Waikhom, R., Sarkar, D., Bennikal, M. & Pandey, R. Rapidly progressive glomerulonephritis in tuberculosis. Saudi J. Kidney Dis. Transpl. 25, 872–875 (2014).
Kanodia, K. V., Vanikar, A. V., Patel, R. D., Suthar, K. S. & Trivedi, H. L. Crescentic glomerulonephritis associated with pulmonary tuberculosis. J. Clin. Diagn. Res. 10, ED01–ED02 (2016).
Shang, M. H. et al. Membranous nephropathy associated with tuberculosis. Chin. Med. J. (Engl). 129, 622–623 (2016).
Çakar, B. & Çiledağ, A. Evaluation of coexistence of cancer and active tuberculosis; 16 case series. Respir. Med. Case Rep. 23, 33–37 (2017).
Malik, R., Pathak, N. & Sharma, S. Secondary renal amyloidosis in pulmonary tuberculosis — a classic revisited. J. Case Rep. Stud. 5, 404 (2017).
Bouziane, Z. et al. Tuberculosis of the renal artery: a rare cause of renovascular arterial hypertension. Ann. Vasc. Surg. 23, 786.e7–786.e9 (2009).
Kulchavenya, E. Best practice in the diagnosis and management of urogenital tuberculosis. Ther. Adv. Urol. 5, 143–151 (2013).
Trauzzi, S. J. et al. Management of prostatic abscess in patients with human immunodeficiency syndrome. Urology 43, 629–633 (1994).
Aziz, E. M., Abdelhak, K. & Hassan, F. M. Tuberculous prostatitis: mimicking a cancer. Pan Afr. Med. J. 25, 130 (2016).
Suankwan, U. et al. A clinicopathologic study of tuberculous epididymo-orchitis in Thailand. Southeast Asian J. Trop. Med. Publ. Health 43, 951–958 (2012).
Kulchavenya, E., Kim, C. S., Bulanova, O. & Zhukova, I. Male genital tuberculosis: epidemiology and diagnostic. World J. Urol. 30, 15–21 (2012).
Yang, D. M. et al. Comparison of tuberculous and pyogenic epididymal abscesses: clinical, gray-scale sonographic, and color doppler sonographic features. AJR Am. J. Roentgenol. 177, 1131–1135 (2001).
Pryor, J. P. & Hendry, W. F. Ejaculatory duct obstruction in subfertile males: analysis of 87 patients. Fertil. Steril. 65, 725–730 (1991).
Agarwal, A., Kumar, N. & Kishore, K. Evaluation of manual Mycobacterium growth indicator tube for isolation and susceptibility testing of Mycobacterium tuberculosis for implementation in low and medium volume laboratories. Med. J. Armed Forces India 74, 220–226 (2018).
Prakash, G. et al. Primary tuberculosis of urethra presenting as stricture urethra and watering can perineum: a rarity. Urol. Ann. 8, 493–495 (2016).
Ilmer, M., Bergauer, F., Friese, K. & Mylonas, I. Genital tuberculosis as the cause of tuboovarian abscess in an immunosuppressed patient. Infect. Dis. Obstet. Gynecol. 2009, 745060 (2009).
Romaniuk, A. et al. A rare case of tuberculous salpingitis. Interv. Med. Appl. Sci. 8, 131–134 (2016).
Nkhili, H., Abdelhak, M. & Benhmamouch, M. N. Pelvic tuberculosis simulating ovarian cancer in children about a case. Pediatrie Pratique http://www.pediatrie-pratiquecom/journal/article/chez-lenfant-propos-dun (2012).
Lantheaume, S. et al. Peritoneal tuberculosis simulating advanced ovarian carcinoma: a case report. Gynecol. Obstet. Fertil. 31, 624–626 (2003).
Djuwantono, T. et al. Female genital tuberculosis and infertility: serial cases report in Bandung, Indonesia and literature review. BMC Res. Notes 10, 683 (2017).
Sharma, J. B. et al. Genital tuberculosis: an important cause of Asherman’s syndrome in India. Arch. Gynecol. Obstet. 277, 37–41 (2008).
Gupta, B., Shree, S., Rajaram, S. & Goel, N. Genital tuberculosis: unusual presentations. Int. J. Mycobacteriol. 3, 357–359 (2016).
Koller, A. B. Granulomatous lesions of the cervix uteri in Black patients. S. Afr. Med. J. 49, 1228–1232 (1975).
Gupta, R., Dey, P., Jain, V. & Gupta, N. Cervical tuberculosis detection in Papanicolaou-stained smear: case report with review of literature. Diagn. Cytopathol. 37, 592–595 (2009).
Paudel, V., Chudal, D. & Pokhrel, D. B. Tuberculosis and HIV co-infection; the deadly duos in vulva. Indian J. Tuberc. 65, 277–279 (2018).
Gilpin, C., Korobitsyn, A., Migliori, G. B., Raviglione, M. C. & Weyer, K. The World Health Organization standards for tuberculosis care and management. Eur. Respir. J. 51, 1800098 (2018).
Lawn, S. D. et al. Advances in tuberculosis diagnostics: the Xpert MTB/RIF assay and future prospects for a point-of-care test. Lancet Infect. Dis. 13, 349–361 (2013).
Walzl, G. et al. Tuberculosis: advances and challenges in development of new diagnostics and biomarkers. Lancet Infect. Dis. 18, e199–e210 (2018).
Hongler, J. et al. Comparison of Löwenstein-Jensen and BACTEC MGIT 960 culture for Mycobacterium tuberculosis in people living with HIV. HIV Med. 19, 654–661 (2018).
Davis, J. L., Cattamanchi, A., Cuevas, L. E., Hopewell, P. C. & Steingart, K. R. Diagnostic accuracy of same-day microscopy versus standard microscopy for pulmonary tuberculosis: a systematic review and meta-analysis. Lancet Infect. Dis. 13, 147–154 (2013).
Bhalla, M. et al. Performance of light-emitting diode fluorescence microscope for diagnosis of tuberculosis. Int. J. Mycobacteriol. 2, 174–178 (2013).
Colabawalla, B. N. Reflections on urogential tuberculosis. Indian J. Urol. 6, 51–59 (1990).
Berta, M. et al. Bacteriological diagnosis of renal tuberculosis: an experience at the regional tuberculosis laboratory in Córdoba Province. Argentina. Rev. Argentina Microbiología 43, 191–194 (2011).
Hemal, A. K. et al. Polymerase chain reaction in clinically suspected genitourinary tuberculosis: comparison with intravenous urography, bladder biopsy, and urine acid fast bacilli culture. Urology 56, 570–574 (2000).
McNerney, R. & Zumla, A. Impact of the Xpert MTB/RIF diagnostic test for tuberculosis in countries with a high burden of disease. Curr. Opin. Pulm. Med. 21, 304–308 (2015).
Tortoli, E., Benedetti, M., Fontanelli, A. & Simonetti, M. T. Evaluation of automated BACTEC MGIT 960 system for testing susceptibility of Mycobacterium tuberculosis to four major antituberculous drugs: comparison with the radiometric BACTEC 460TB method and the agar plate method of proportion. J. Clin. Microbiol. 40, 607–610 (2002).
World Health Organization. Policy statement: automated real-time nucleic acid amplification technology for rapid and simultaneous detection of tuberculosis and rifampicin resistance: Xpert MTB/RIF system (WHO, Geneva, 2011).
Bates, M. & Zumla, A. The development. Evaluation and performance of molecular diagnostics for detection of Mycobacterium tuberculosis. Expert Rev. Mol. Diagn. 16, 307–322 (2016).
Green, C. et al. Rapid diagnosis of tuberculosis through the detection of mycobacterial DNA in urine by nucleic acid amplification methods. Lancet Infect. Dis. 9, 505–511 (2009).
Atherton, R. R. et al. Detection of Mycobacterium tuberculosis in urine by Xpert MTB/RIF Ultra: a useful adjunctive diagnostic tool in HIV-associated tuberculosis. Int. J. Infect. Dis. 75, 92–94 (2018).
Babafemi, E. O. et al. Complicated urinary infection and extrapulmonary tuberculosis. BMJ Case Rep. 2012, bcr2012007553 (2012).
Pang, Y. et al. GeneXpert MTB/RIF assay in the diagnosis of urinary tuberculosis from urine specimens. Sci. Rep. 7, 6181 (2017).
Gupta-Wright, A. et al. Rapid urine-based screening for tuberculosis in HIV-positive patients admitted to hospital in Africa (STAMP): a pragmatic, multicentre, parallel-group, double-blind, randomised controlled trial. Lancet 392, 292–301 (2018).
WHO. The use of next-generation sequencing technologies for the detection of mutations associated with drug resistance in Mycobacterium tuberculosis complex: technical guide. WHO https://apps.who.int/iris/bitstream/handle/10665/274443/WHO-CDS-TB-2018.19-eng.pdf (2018).
Lawn, S. D. & Gupta-Wright, A. Detection of lipoarabinomannan (LAM) in urine is indicative of disseminated TB with renal involvement in patients living with HIV and advanced immunodeficiency: evidence and implications. Trans. R. Soc. Trop. Med. Hyg. 110, 180–185 (2016).
Colijn, C. & Cohen, T. Whole-genome sequencing of Mycobacterium tuberculosis for rapid diagnostics and beyond. Lancet Respir. Med. 4, 6–8 (2016).
Doyle, R. M. et al. Direct whole-genome sequencing of sputum accurately identifies drug-resistant Mycobacterium tuberculosis faster than MGIT culture sequencing. J. Clin. Microbiol. 56, e00666-18 (2018).
Traşcă, E., Traşcă, E. T., Buzulică, R., Drăgoi, G. & Nicolescu, I. The place and the role of histological examination in diagnostic algorithm of urinary system tuberculosis. Rom. J. Morphol. Embryol. 46, 105–108 (2005).
Kulchavenya, E. & Khomyakov, V. Male genital tuberculosis in Siberians. World J. Urol. 24, 74–78 (2006).
Viswaroop, B. et al. Fine-needle aspiration cytology versus open biopsy for evaluation of chronic epididymal lesions: a prospective study. Scand. J. Urol. Nephrol. 39, 219–221 (2005).
Sah, S. P., Bhadani, P. P., Regmi, R., Tewari, A. & Raj, G. A. Fine needle aspiration cytology of tubercular epididymits and epididymo-orchitis. Acta Cytol. 50, 243–249 (2006).
Kumar, P., Owji, S. & Kherzi, A. Tuberculous orchitis diagnosed by fine needle aspiration cytology. Acta Cytol. 40, 1253–1256 (1996).
Comiter, C. V. et al. Nonpalpable intratesticular masses detected sonographically. J. Urol. 154, 1367–1369 (1995).
Cek, M. et al. EAU guidelines for the management of genitourinary tuberculosis. Eur. Urol. 48, 353–362 (2005).
Valentini, A. L., Summaria, V. & Marano, P. Diagnostic imaging of uro-genital tuberculosis. Rays 23, 126–143 (1998).
Muttarak, M., ChiangMai, W. N. & Lojanapiwat, B. Tuberculosis of the uro-genital tract: imaging features with pathological correlation. Singapore Med. J. 46, 568–574 (2005).
Jung, Y. Y., Kim, J. K. & Cho, K. S. Uro-genital tuberculosis: comprehensive cross-sectional imaging. AJR Am. J. Roentgenol. 184, 143–150 (2005).
Tirumani, S. H. et al. Imaging of tuberculosis of the abdominal viscera: beyond the intestines. J. Clin. Imag. Sci. 3, 17 (2013).
Kollins, S. A., Hartman, G. W., Carr, D. T., Segura, J. W. & Hattery, R. R. Roentgenographic findings in urinary tract tuberculosis. A 10-year review. Am. J. Roentgenol. Radium Ther. Nucl. Med. 121, 487–499 (1974).
Dyer, R. B., Chen, M. Y. & Zagoria, R. J. Abnormal calcifications in the urinary tract. Radiographics 18, 1405–1424 (1998).
Vijayaraghavan, S. B. et al. Spectrum of high-resolution sonographic features of urinary tuberculosis. J. Ultrasound Med. 23, 585–594 (2004).
Rui, X., Li, X. D., Cai, S., Chen, G. & Cai, B. Ultrasonographic diagnosis and typing of renal tuberculosis. Int. J. Urol. 15, 135–139 (2008).
Hamrick-Turner, J., Abbitt, P. L. & Ros, P. R. Tuberculosis of the lower uro-genital tract: findings on sonography and MR. AJR Am. J. Roentgenol. 158, 919 (1992).
Engin, G., Acunas, B., Acunas, G. & Tunaci, M. Imaging of extrapulmonary tuberculosis. Radiographics 20, 471–488 (2000).
Chung, J. J., Kim, M.-J., Lee, T., Yoo, H. S. & Lee, J. T. Sonographic findings in tuberculous epididymitis and epididymo-orchitis. J. Clin. Ultrasound 25, 390–394 (1997).
Awasthi, S., Saxena, M., Ahmad, F., Kumar, A. & Dutta, S. Abdominal tuberculosis: a diagnostic dilemma. J. Clin. Diagn. Res. 9, EC01–EC03 (2015).
Navarro-Vilasaró, M. et al. Genitourinary mycobacteriosis: retrospective study of 45 cases in a general hospital. Enferm. Infecc. Microbiol. Clin. 9, 540–545 (2008).
Sallami, S. et al. Imaging findings of urinary tuberculosis on computerized tomography versus excretory urography: through 46 confirmed cases. Tunis. Med. 12, 743–747 (2014).
Kenney, P. J. Imaging of chronic renal infections. Am. J. Roentgenol. 3, 485–494 (1990).
Gaudiano, C. et al. Multidetector CT urography in urogenital tuberculosis: use of reformatted images for the assessment of the radiological findings. A pictorial essay. Abdom. Radiol. 9, 2314–2324 (2017).
Goel, A. & Dalela, D. Options in the management of tuberculous ureteric stricture. Indian J. Urol. 24, 376–381 (2008).
Aswathaman, K. & Devasia, A. Thimble bladder. ANZ J. Surg. 78, 1049 (2008).
Wang, L.-J., Wong, Y.-C., Chen, C.-J. & Lim, K.-E. CT features of genitourinary tuberculosis. J. Comput. Assist. Tomogr. 21, 254–258 (1997).
Lu, P., Li, C. & Zhou, X. Significance of the CT scan in renal tuberculosis. Zhonghua Jie He He Hu Xi Za Zhi 24, 407–409 (2001).
Wang, L. J. et al. Imaging findings of urinary tuberculosis on excretory urography and computerized tomography. J. Urol. 169, 524–528 (2003).
Sinan, T., Sheikh, M., Ramadan, S., Sahwney, S. & Behbehani, A. CT features in abdominal tuberculosis: 20 years’ experience. BMC Med. Imaging 2, 3 (2002).
Kukrej, N., Cook, G. J. & Pattison, J. M. Positron-emission tomography used to diagnose tuberculosis in a renal transplant patient. Am. J. Transplant. 2, 105–107 (2002).
Mahajan, M. S., Bedmutha, A. & Singh, N. 18F-fludeoxyglucose positron emission tomography computed tomography-guided diagnosis of prostatic and leptomeningeal tuberculosis. Indian J. Urol. 33, 325–327 (2017).
Coleman, M. T. et al. Early changes by (18) fluorodeoxyglucose positron emission tomography co-registered with computed tomography predict outcome after Mycobacterium tuberculosis infection in cynomolgus macaques. Infect. Immun. 82, 2400–2404 (2014).
Martin, C. et al. Prospective serial FDG PET/CT during treatment of extrapulmonary tuberculosis in HIV-infected patients: an exploratory study. Clin. Nucl. Med. 43, 635–640 (2018).
Gambhir, S. et al. Imaging in extrapulmonary tuberculosis. Int. J. Infect. Dis. 56, 237–247 (2017).
Subramanyam, P. & Palaniswamy, S. S. Dual time point (18)F-FDG PET/CT imaging identifies bilateral renal tuberculosis in an immunocompromised patient with an unknown primary malignancy. Infect. Chemother. 47, 117–119 (2015).
da Rocha, E. L. et al. Abdominal tuberculosis: a radiological review with emphasis on computed tomography and magnetic resonance imaging findings. Radiol. Bras. 48, 181–191 (2015).
Rais-Bahrami, S. et al. Clinical and multiparametric MRI signatures of granulomatous prostatitis. Abdom. Radiol. 42, 1956–1962 (2017).
Cheng, Y., Huang, L., Zhang, X., Ji, Q. & Shen, W. Multiparametric magnetic resonance imaging characteristics of prostate tuberculosis. Korean J. Radiol. 16, 846–852 (2015).
Liao, L. G. et al. Etiology of 305 cases of refractory hematospermia and therapeutic options by emerging endoscopic technology. Sci. Rep. 9, 5018 (2019).
Ahmadi, F., Zafarani, F. & Shahrzad, G. Hysterosalpingographic appearances of female genital tract tuberculosis. I. Fallopian tube. Int. J. Fertil. Steril. 7, 245–252 (2014).
Ahmadi, F., Zafarani, F. & Shahrzad, G. S. Hysterosalpingographic appearances of female genital tract tuberculosis. II. Uterus. Int. J. Fertil. Steril. 8, 13–20 (2014).
Farrokh, D., Layegh, P., Afzalaghaee, M., Mohammadi, M. & Fallah Rastegar, Y. Hysterosalpingographic findings in women with genital tuberculosis. Iran. J. Reprod. Med. 13, 297–304 (2015).
Maynard-Smith, L., Fernando, B., Hopkins, S., Harber, M. & Lipman, M. Managing latent tuberculosis in UK renal transplant units: how does practice compare with published guidance? Clin. Med. 14, 26–29 (2014).
Shu, C. C. et al. Predictors and prevalence of latent tuberculosis infection in patients receiving long-term hemodialysis and peritoneal dialysis. PLOS ONE 7, e42592 (2012).
Hadaya, K. et al. Contribution of interferon-γ release assays (IGRAs) to the diagnosis of latent tuberculosis infection after renal transplantation. Transplantation 95, 1485–1490 (2013).
WHO. Latent TB infection: updated and consolidated guidelines for programmatic management. WHO https://www.who.int/tb/publications/2018/latent-tuberculosis-infection/en/ (2018).
British Thoracic Society. Guidelines for the prevention and management of Mycobacterium tuberculosis infection and disease in adult patients with chronic kidney disease BTS Guideline Group on behalf of The British Thoracic Society Standards of Care Committee and Joint Tuberculosis Committee. Thorax 65, 559–570 (2010).
WHO. WHO consolidated guidelines on drug-resistant tuberculosis treatment. WHO https://www.who.int/tb/publications/2019/consolidated-guidelines-drug-resistant-TB-treatment/en/ (2019).
Tiberi, S. et al. Tuberculosis: progress and advances in development of new drugs, treatment regimens, and host-directed therapies. Lancet Infect. Dis. 18, e183–e198 (2018).
World Health Organization. Improving the diagnosis and treatment of smear-negative pulmonary and extrapulmonary tuberculosis among adults and adolescents. WHO https://www.who.int/tb/publications/2006/tbhiv_recommendations.pdf (2006).
Sharma, J. B. et al. Six months versus nine months anti-tuberculous therapy for female genital tuberculosis: a randomized controlled trial. Eur. J. Obstet. Gynecol. Reprod. Biol. 203, 264–273 (2016).
Wagaskar, V. G. et al. Urinary tuberculosis with renal failure: challenges in management. J. Clin. Diagn. Res. 10, PC01–PC03 (2016).
Chang, C. H. et al. Acute kidney injury due to anti-tuberculosis drugs: a five-year experience in an aging population. BMC Infect. Dis. 14, 23 (2014).
Fischer, M. & Flamm, J. The value of surgical therapy in the treatment of urogenital tuberculosis. Urologe A 29, 261–264 (1990).
Carl, P. & Stark, L. Indications for surgical management of genitourinary tuberculosis. World J. Surg. 21, 505–510 (1997).
Bansal, P. & Bansal, N. The surgical management of urogenital tuberculosis our experience and long-term follow-up. Urol. Ann. 7, 49–52 (2015).
Li, X. et al. A clinical comparative analysis of retroperitoneal laparoscopic tuberculous nephrectomy and open tuberculous nephrectomy. J. Laparoendosc. Adv. Surg. Tech. A 29, 909–913 (2019).
Gupta, R. et al. Laparoscopic ablative and reconstructive surgeries in uro-genital tuberculosis. JSLS 18, e2014.00203 (2014).
O’Flynn, D. Surgical treatment of genito-urinary tuberculosis: a report on 762 cases. Br. J. Urol. 42, 667–671 (1970).
Mochalova, T. P. & Starikov, I. Y. Reconstructive surgery for treatment of urogenital tuberculosis: 30 years of observation. World J. Surg. 21, 511–515 (1997).
Zwergel, U., Wullich, B., Rohde, V. & Zwergel, T. Surgical management of urinary tuberculosis: a review of 341 patients. J. Urol. 161, 9 (1999).
Krishnamoorthy, S. & Gopalakrishnan, G. Surgical management of renal tuberculosis. Indian J. Urol. 24, 369–375 (2008).
Rizzo, M. et al. Twenty-years experience on genitourinary tuberculosis. Arch. Ital. Urol. Androl. 76, 83–87 (2004).
Mcaleer, S. J., Johnson, C. W. & Johnson, W. D. in Campbell-Walsh Urology 9th edn (ed. Wein, A. J.) 436–470 (Saunders Elsevier, 2007).
Gow, J. G. Renal calcification in genitourinary tuberculosis. Br. J. Surg. 52, 283–288 (1965).
Kim, H. H., Lee, K. S., Park, K. & Ahn, H. Laparoscopic nephrectomy for nonfunctioning tuberculous kidney. J. Endourol. 14, 433–437 (2000).
Rassweiler, J. et al. Laparoscopic nephrecthe: the experience of the laparoscopy working group of the German Urologic Association. J. Urol. 160, 18–21 (1998).
Gupta, S. et al. Acute renal failure in bilateral urinary tract tuberculosis. Pediatr. Surg. Int. 13, 200–201 (1998).
Vasdev, N., Moon, A. & Thorpe, A. C. Metabolic complications of urinary intestinal diversion. Indian J. Urol. 29, 310–315 (2013).
Gupta, R., Singh, P. & Kumar, R. Should men with idiopathic obstructive azoospermia be screened for genitourinary tuberculosis? J. Hum. Reprod. Sci. 8, 43–47 (2015).
Nickel, J. C. Chronic epididymitis: a practical approach to understanding and managing a difficult urologic enigma. Rev. Urol. 5, 209–215 (2003).
Cho, Y. S., Joo, K. J., Kwon, C. H. & Park, H. J. Tuberculosis of testis and prostate that mimicked testicular cancer in young male soccer player. J. Exerc. Rehabil. 9, 389–393 (2013).
Weiner, J. 3rd et al. Metabolite changes in blood predict the onset of tuberculosis. Nat. Commun. 9, 5208 (2018).
Duffy, F. J. et al. Immunometabolic signatures predict risk of progression to active tuberculosis and disease outcome. Front. Immunol. 10, 52 (2019).
Fitzgerald, B. L. et al. Elucidation of a human urine metabolite as a seryl-leucine glycopeptide and as a biomarker of effective anti-tuberculosis therapy. ACS Infect. Dis. 5, 353–364 (2019).
MacLean, E. et al. A systematic review of biomarkers to detect active tuberculosis. Nat. Microbiol. 4, 748–758 (2019).
Zumla, A. et al. Host-directed therapies for infectious diseases: current status, recent progress, and future prospects. Lancet Infect. Dis. 16, e47–e63 (2016).
Liu, C. et al. Quantification of circulating Mycobacterium tuberculosis antigen peptides allows rapid diagnosis of active disease and treatment monitoring. Proc. Natl. Acad. Sci. USA 114, 3969–3974 (2017).
Rao, M. et al. Improving treatment outcomes for MDR-TB — novel host-directed therapies and personalised medicine of the future. Int. J. Infect. Dis. 80S, S62–S67 (2019).
Skrahin, A. et al. Autologous mesenchymal stromal cell infusion as adjunct treatment in patients with multidrug and extensively drug-resistant tuberculosis: an open-label phase 1 safety trial. Lancet Respir. Med. 2, 108–122 (2014).
Yudintceva, N. M. et al. Application of the allogenic mesenchymal stem cells in the therapy of the bladder tuberculosis. J. Tissue Eng. Regen. Med. 12, e1580–e1593 (2018).
Kadhiravan, T. & Sharma, S. K. Medical management of genitourinary tuberculosis. Indian J. Urol. 24, 362–368 (2008).
WHO. Guidelines on the management of latent tuberculosis infection. WHO https://www.who.int/tb/publications/latent-tuberculosis-infection/en/ (2019).
White, R. G., Hanekom, W. A., Vekemans, J. & Harris, R. C. The way forward for tuberculosis vaccines. Lancet Respir. Med. 7, 204–206 (2019).
Zumla, A. & Petersen, E. The historic and unprecedented United Nations General Assembly High Level Meeting on Tuberculosis (UNGA-HLM-TB) —‘United to End TB: An Urgent Global Response to a Global Epidemic’. Int. J. Infect. Dis. 75, 118–120 (2018).
Acknowledgements
A.Z. receives a UK National Institutes of Health Research (NIHR) senior investigator award. A.Z. acknowledges support from the PANDORA-ID-NET grant from the EDCTP Reg/Grant RIA2016E-1609), CANTAM2, TESA2 and EACCR2 EDCTP Networks of Excellence grants, all funded by the European and Developing Countries Clinical Trials Partnership (EDCTP2) programme, which is supported under Horizon 2020, the European Union’s Framework Programme for Research and Innovation. A.Z. and A.M. acknowledge support from the NIHR Biomedical Research Centre at UCLH. Figures 3a, 3b, 3c, 3d, 3i, 7c, 7d and 7e were kindly provided by Professor Sebastian Lucas, St Thomas’s Hospital, London.
Author information
Authors and Affiliations
Contributions
All authors researched data for the article, wrote the manuscript and reviewed and edited the manuscript before submission. B.M., S.K. and A.Z. made substantial contributions to discussions of content.
Corresponding author
Ethics declarations
Competing interests
A.Z. serves on the WHO and other global tuberculosis (TB) expert advisory groups and committees and is editor of two textbooks on TB, which were used as references. The other authors declare no competing interests.
Additional information
Peer review information
Nature Reviews Urology thanks E. Kulchavenya and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Review criteria
We reviewed publications in English on the MEDLINE, EMBASE and GOOGLE SCHOLAR for the period up to 1 September 2018 using the search terms “tuberculosis” or “TB” in combination with the terms “urological”, “genital”, “uro-genital”, “epididymo-orchitis”, “kidney”, or “renal” or “ureter” or “bladder” or “testes” or “urethra” or “penis” or “infertility”; websites of global and national public health agencies such as WHO, US-Centres for Disease Control, UK-Public Health England, European Centre for Disease Prevention and Control (ECDC); The 2018 WHO Global TB Report; specialist textbooks on tuberculosis; clinical guidelines developed by specialist societies; and relevant substantive reviews to inform readers of more references.
Rights and permissions
About this article
Cite this article
Muneer, A., Macrae, B., Krishnamoorthy, S. et al. Urogenital tuberculosis — epidemiology, pathogenesis and clinical features. Nat Rev Urol 16, 573–598 (2019). https://doi.org/10.1038/s41585-019-0228-9
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41585-019-0228-9
This article is cited by
-
Ileal ureteral replacement for tuberculous ureteral strictures: 11 cases of experience
International Urology and Nephrology (2026)
-
A rare presentation of urogenital tuberculosis leading to obstructive uropathy and renal failure: a case report
BMC Urology (2025)
-
Clinical analysis of the onset characteristics and inducing factors of atypical renal tuberculosis
BMC Infectious Diseases (2025)
-
Invasive ventilator exhaust in critical care: aerosol transmission risks and management strategies-a narrative review
Critical Care (2025)
-
Molecular diagnosis of urogenital tuberculosis among patients attending tuberculosis chest clinic in kogi state specialist hospital, Lokoja, Kogi State, Nigeria
Mycobacteria (2025)