Abstract
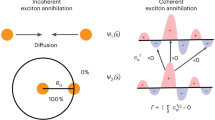
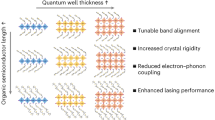
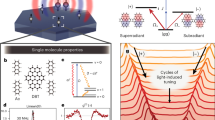
Intermolecular distance largely determines the optoelectronic properties of organic matter. Conventional organic luminescent molecules are commonly used either as aggregates or as single molecules that are diluted in a foreigner matrix. They have garnered great research interest in recent decades for a variety of applications, including light-emitting diodes1,2, lasers3,4,5 and quantum technologies6,7, among others8,9,10. However, there is still a knowledge gap on how these molecules behave between the aggregation and dilution states. Here we report an unprecedented phase of molecular aggregate that forms in a two-dimensional hybrid perovskite superlattice with a near-equilibrium distance, which we refer to as a single-molecule-like aggregate (SMA). By implementing two-dimensional superlattices, the organic emitters are held in proximity, but, surprisingly, remain electronically isolated, thereby resulting in a near-unity photoluminescence quantum yield, akin to that of single molecules. Moreover, the emitters within the perovskite superlattices demonstrate strong alignment and dense packing resembling aggregates, allowing for the observation of robust directional emission, substantially enhanced radiative recombination and efficient lasing. Molecular dynamics simulations together with single-crystal structure analysis emphasize the critical role of the internal rotational and vibrational degrees of freedom of the molecules in the two-dimensional lattice for creating the exclusive SMA phase. This two-dimensional superlattice unifies the paradoxical properties of single molecules and aggregates, thus offering exciting possibilities for advanced spectroscopic and photonic applications.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$32.99 / 30 days
cancel any time
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
Data availability
Crystallographic data for the structure of FBTT-contained and FBTP-contained 2D SLs reported in this article have been deposited at the Cambridge Crystallographic Data Centre under deposition numbers CCDC 2289715 and 2283333, respectively. Copies of the data can be obtained free of charge at https://www.ccdc.cam.ac.uk/structures/. All other data supporting the findings of this study are available in the article and its Supplementary Information. Source data are provided with this paper.
References
Tang, C. W. & VanSlyke, S. A. Organic electroluminescent diodes. Appl. Phys. Lett. 51, 913–915 (1987).
Uoyama, H., Goushi, K., Shizu, K., Nomura, H. & Adachi, C. Highly efficient organic light-emitting diodes from delayed fluorescence. Nature 492, 234–238 (2012).
Tessler, N., Denton, G. J. & Friend, R. H. Lasing from conjugated-polymer microcavities. Nature 382, 695–697 (1996).
Kozlov, V. G., Bulović, V., Burrows, P. E. & Forrest, S. R. Laser action in organic semiconductor waveguide and double-heterostructure devices. Nature 389, 362–364 (1997).
Samuel, I. D. W. & Turnbull, G. A. Organic semiconductor lasers. Chem. Rev. 107, 1272–1295 (2007).
Toninelli, C. et al. Single organic molecules for photonic quantum technologies. Nat. Mater. 20, 1615–1628 (2021).
Hail, C. U. et al. Nanoprinting organic molecules at the quantum level. Nat. Commun. 10, 1880 (2019).
Carter, K. P., Young, A. M. & Palmer, A. E. Fluorescent sensors for measuring metal ions in living systems. Chem. Rev. 114, 4564–4601 (2014).
Yang, Y., Zhao, Q., Feng, W. & Li, F. Luminescent chemodosimeters for bioimaging. Chem. Rev. 113, 192–270 (2013).
Cosco, E. D. et al. Flavylium polymethine fluorophores for near- and shortwave infrared imaging. Angew. Chem. Int. Ed. 56, 13126–13129 (2017).
Hong, Y., Lam, J. W. Y. & Tang, B. Z. Aggregation-induced emission: phenomenon, mechanism and applications. Chem. Commun. 29, 4332–4353 (2009).
Lane, P. A. et al. Origin of electrophosphorescence from a doped polymer light emitting diode. Phys. Rev. B 63, 235206 (2001).
Wang, H. et al. Doped organic crystals with high efficiency, color-tunable emission toward laser application. Cryst. Growth Des. 9, 4945–4950 (2009).
Mischok, A., Hillebrandt, S., Kwon, S. & Gather, M. C. Highly efficient polaritonic light-emitting diodes with angle-independent narrowband emission. Nat. Photon. 17, 393–400 (2023).
Vietze, U. et al. Zeolite-dye microlasers. Phys. Rev. Lett. 81, 4628–4631 (1998).
Yu, J. et al. Confinement of pyridinium hemicyanine dye within an anionic metal-organic framework for two-photon-pumped lasing. Nat. Commun. 4, 2719 (2013).
Fang, Q. et al. Designed synthesis of large-pore crystalline polyimide covalent organic frameworks. Nat. Commun. 5, 4503 (2014).
Huang, Y. et al. Reducing aggregation caused quenching effect through co-assembly of PAH chromophores and molecular barriers. Nat. Commun. 10, 169 (2019).
Hua, B. et al. Supramolecular solid-state microlaser constructed from pillar[5]arene-based host–guest complex microcrystals. J. Am. Chem. Soc. 140, 15651–15654 (2018).
Kim, D.-H. et al. High-efficiency electroluminescence and amplified spontaneous emission from a thermally activated delayed fluorescent near-infrared emitter. Nat. Photon. 12, 98–104 (2018).
Mitzi, D. B., Feild, C. A., Harrison, W. T. A. & Guloy, A. M. Conducting tin halides with a layered organic-based perovskite structure. Nature 369, 467–469 (1994).
Gao, Y. et al. Molecular engineering of organic–inorganic hybrid perovskites quantum wells. Nat. Chem. 11, 1151–1157 (2019).
Smith, M. D., Connor, B. A. & Karunadasa, H. I. Tuning the luminescence of layered halide perovskites. Chem. Rev. 119, 3104–3139 (2019).
Leng, K., Fu, W., Liu, Y., Chhowalla, M. & Loh, K. P. From bulk to molecularly thin hybrid perovskites. Nat. Rev. Mater. 5, 482–500 (2020).
Li, X., Hoffman, J. M. & Kanatzidis, M. G. The 2D halide perovskite rulebook: how the spacer influences everything from the structure to optoelectronic device efficiency. Chem. Rev. 121, 2230–2291 (2021).
Gong, X. et al. Electron–phonon interaction in efficient perovskite blue emitters. Nat. Mater. 17, 550–556 (2018).
Passarelli, J. V. et al. Enhanced out-of-plane conductivity and photovoltaic performance in n = 1 layered perovskites through organic cation design. J. Am. Chem. Soc. 140, 7313–7323 (2018).
Yan, L., Gloor, C. J., Moran, A. M. & You, W. Non-covalent interactions involving π effect between organic cations in low-dimensional organic/inorganic hybrid perovskites. Appl. Phys. Lett. 122, 240501 (2023).
Wang, N. et al. Perovskite light-emitting diodes based on solution-processed self-organized multiple quantum wells. Nat. Photon. 10, 699–704 (2016).
Wang, K. et al. Suppressing phase disproportionation in quasi-2D perovskite light-emitting diodes. Nat. Commun. 14, 397 (2023).
Tsai, H. et al. High-efficiency two-dimensional Ruddlesden–Popper perovskite solar cells. Nature 536, 312–316 (2016).
Feng, J. et al. Single-crystalline layered metal-halide perovskite nanowires for ultrasensitive photodetectors. Nat. Electron. 1, 404–410 (2018).
Qin, C. et al. Stable room-temperature continuous-wave lasing in quasi-2D perovskite films. Nature 585, 53–57 (2020).
Era, M., Maeda, K. & Tsutsui, T. Enhanced phosphorescence from naphthalene-chromophore incorporated into lead bromide-based layered perovskite having organic–inorganic superlattice structure. Chem. Phys. Lett. 296, 417–420 (1998).
Chondroudis, K. & Mitzi, D. B. Electroluminescence from an organic–inorganic perovskite incorporating a quaterthiophene dye within lead halide perovskite layers. Chem. Mater. 11, 3028–3030 (1999).
Braun, M., Tuffentsammer, W., Wachtel, H. & Wolf, H. C. Pyrene as emitting chromophore in organic–inorganic lead halide-based layered perovskites with different halides. Chem. Phys. Lett. 307, 373–378 (1999).
Ema, K., Inomata, M., Kato, Y., Kunugita, H. & Era, M. Nearly perfect triplet-triplet energy transfer from Wannier excitons to naphthalene in organic-inorganic hybrid quantum-well materials. Phys. Rev. Lett. 100, 257401 (2008).
Karl, M. et al. Flexible and ultra-lightweight polymer membrane lasers. Nat. Commun. 9, 1525 (2018).
Silver, S., Yin, J., Li, H., Brédas, J.-L. & Kahn, A. Characterization of the valence and conduction band levels of n = 1 2D perovskites: a combined experimental and theoretical investigation. Adv. Energy Mater. 8, 1703468 (2018).
Gryn’ova, G., Lin, K.-H. & Corminboeuf, C. Read between the molecules: computational insights into organic semiconductors. J. Am. Chem. Soc. 140, 16370–16386 (2018).
Mitzi, D. B., Chondroudis, K. & Kagan, C. R. Design, structure, and optical properties of organic–inorganic perovskites containing an oligothiophene chromophore. Inorg. Chem. 38, 6246–6256 (1999).
Chung, C., Lee, M. & Choe, E. K. Characterization of cotton fabric scouring by FT-IR ATR spectroscopy. Carbohydr. Polym. 58, 417–420 (2004).
Hong, Y., Lam, J. W. Y. & Tang, B. Z. Aggregation-induced emission. Chem. Soc. Rev. 40, 5361–5388 (2011).
Gómez-Castaño, M. et al. Energy transfer and interference by collective electromagnetic coupling. Nano Lett. 19, 5790–5795 (2019).
Rainò, G. et al. Superfluorescence from lead halide perovskite quantum dot superlattices. Nature 563, 671–675 (2018).
Cherniukh, I. et al. Perovskite-type superlattices from lead halide perovskite nanocubes. Nature 593, 535–542 (2021).
Spano, F. C., Kuklinski, J. R. & Mukamel, S. Temperature-dependent superradiant decay of excitons in small aggregates. Phys. Rev. Lett. 65, 211–214 (1990).
Blach, D. D. et al. Superradiance and exciton delocalization in perovskite quantum dot superlattices. Nano Lett. 22, 7811–7818 (2022).
Findik, G. et al. High-temperature superfluorescence in methyl ammonium lead iodide. Nat. Photon. 15, 676–680 (2021).
Dursun, I. et al. Temperature-dependent optical and structural properties of chiral two-dimensional hybrid lead-iodide perovskites. J. Phys. Chem. C 127, 15423–15434 (2023).
Chen, H. et al. Structural and spectral dynamics of single-crystalline Ruddlesden-Popper phase halide perovskite blue light-emitting diodes. Sci. Adv. 6, eaay4045 (2020).
Aubrey, M. L. et al. Directed assembly of layered perovskite heterostructures as single crystals. Nature 597, 355–359 (2021).
Qian, Q. et al. Chiral molecular intercalation superlattices. Nature 606, 902–908 (2022).
Yuan, Z. et al. One-dimensional organic lead halide perovskites with efficient bluish white-light emission. Nat. Commun. 8, 14051 (2017).
Zhou, C. et al. Blue emitting single crystalline assembly of metal halide clusters. J. Am. Chem. Soc. 140, 13181–13184 (2018).
Hestand, N. J. & Spano, F. C. Molecular aggregate photophysics beyond the Kasha model: novel design principles for organic materials. Acc. Chem. Res. 50, 341–350 (2017).
Kaufmann, C., Bialas, D., Stolte, M. & Würthner, F. Discrete π-stacks of perylene bisimide dyes within folda-dimers: insight into long- and short-range exciton coupling. J. Am. Chem. Soc. 140, 9986–9995 (2018).
Shi, E. et al. Two-dimensional halide perovskite lateral epitaxial heterostructures. Nature 580, 614–620 (2020).
Ilavsky, J. Nika: software for two-dimensional data reduction. J. Appl. Crystallogr. 45, 324–328 (2012).
Gaussian 16, Revision C.01 (Gaussian, Inc., 2016).
Stephens, P. J., Devlin, F. J., Chabalowski, C. F. & Frisch, M. J. Ab initio calculation of vibrational absorption and circular dichroism spectra using density functional force fields. J. Phys. Chem. 98, 11623–11627 (1994).
Weigend, F. & Ahlrichs, R. Balanced basis sets of split valence, triple zeta valence and quadruple zeta valence quality for H to Rn: design and assessment of accuracy. Phys. Chem. Chem. Phys. 7, 3297–3305 (2005).
Lu, T. & Chen, F. Multiwfn: a multifunctional wavefunction analyzer. J. Comput. Chem. 33, 580–592 (2012).
Brunner, K., Tortschanoff, A., Warmuth, C., Bässler, H. & Kauffmann, H. F. Site torsional motion and dispersive excitation hopping transfer in π-conjugated polymers. J. Phys. Chem. B 104, 3781–3790 (2000).
Meskers, S. C. J., Hübner, J., Oestreich, M. & Bässler, H. Dispersive relaxation dynamics of photoexcitations in a polyfluorene film involving energy transfer: experiment and Monte Carlo simulations. J. Phys. Chem. B 105, 9139–9149 (2001).
Acknowledgements
This work is supported by the National Science Foundation (grant nos. 2131608-ECCS and 2143568-DMR). A.D. acknowledges the support for spectroscopy measurements from the US Department of Energy, Office of Science, Office of Basic Energy Sciences under award no. DE-SC0022082. Z.-Y.L. and B.M.S. acknowledge the support for MD simulations from the US Department of Energy, Office of Energy Efficiency and Renewable Energy (EERE) under the Solar Energy Technologies Office award DE-EE0009519. The views expressed herein do not necessarily represent the views of the US Department of Energy or the United States Government. Work at Yale was supported by the Air Force Office of Scientific Research under grant no. FA9550-22-1-0209. C.B.F., A.B. and V.M.S. acknowledge the support of the Office of Naval Research (ONR) under award no. N00014-21-1-2026 and the Air Force Office of Scientific Research under grant no. FA9550-22-1-0372. We thank J. Simon for establishing the angle-dependent PL setup, Q. Hu for the help with mass-spectrometry measurements, L. Huang and M. Gather for discussions, and H. Liu for spatial-coherence measurements.
Author information
Authors and Affiliations
Contributions
L.D. conceived the idea. L.D. and B.M.S. supervised the project. K.W. carried out the materials synthesis, structural characterizations, film fabrication and data analysis. Z.-Y.L. and B.M.S. performed MD simulations and data analysis. A.D. conducted the low-temperature PL and time-resolved PL measurements and data analysis. C.A.K. and P.G. carried out streak-camera measurements. W.S. helped with DFT calculations on band alignment. H.Y. carried out FLIM measurements and data analysis. Z.H. and Y.S.Z. helped with DBR device fabrication and lasing characterizations. A.H.C. and C.Z. performed GIWAXS measurements. Y.T. conducted FTIR spectroscopy measurements. A.B. and V.M.S. supervised the optical-characterization-related activities and C.B.F. conducted angle-resolved PL measurements. D.V. contributed to ligand design and synthesis. K.W. and L.D. wrote the manuscript. All authors discussed the results and revised the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature thanks the anonymous reviewers for their contribution to the peer review of this work. Peer reviewer reports are available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data figures and tables
Extended Data Fig. 1 DFT and TDDFT simulation on the energy level of molecular emitters.
a, Molecular structures of four different organic emitters. b, The calculated HOMO–LUMO and S1 level of organic molecules relative to the valance band maximum (VBM)–conduction band minimum (CBM) of lead-bromide matrix. The VBM–CBM value of [PbBr4]2− was adapted from the literature39. c, Scheme of the band alignment in organic–inorganic perovskite 2D SL structures including type I, type II and reversed type I.
Extended Data Fig. 2 The extracted azimuthal distribution of GIWAXS intensity for 2D perovskites at a typical q near 0.5 A−1 (dashed rings in Fig. 2b,f).
a, FBTT 2D SLs. b, FBTP 2D SLs. 0° and 180° correspond to the qxy plane (vertical orientation) and 90° corresponds to the qz direction (horizontal orientation). For the FBTT sample, it shows a peak scattering intensity of about 2,200 at 90°, which is very similar to that of FBTP (about 2,100). At 0° or 180°, FBTT shows substantial scattering with an intensity of about 410, whereas FBTP has a negligible scattering. Here the orientation factor was defined as the scattering intensity ratio between 0° (180°) and 90°, which yields a value of 0.19 and roughly 0 for FBTT and FBTP, respectively. A greater orientation factor value suggests a preferred orientation along the qxy direction, which corresponds to a vertically oriented [PbX4]2− inorganic sublattice relative to the substrate. Therefore, in our scenario, the FBTT sample is dominated by horizontal orientation with a small fraction of vertical orientation, whereas the FBTP sample exhibits an almost perfectly horizontal orientation.
Extended Data Fig. 3 FBTP emitters confined in 2D SLs with tunable inorganic sublattices.
a, PL spectra. b, UV–vis spectra. c, XRD of FBTP-contained 2D SLs with different halide compositions.
Extended Data Fig. 4 The atomic force microscopy surface morphology study on both molecular aggregates and 2D perovskites thin-film samples.
a, FBTT. b, FBTP. c, PBTP. d, BBTP. The dimensions of all images are 4 μm × 4 μm.
Extended Data Fig. 5 2D perovskite nanocrystals and their corresponding PL spectra.
a, (PEA)2PbBr4 nanocrystal. b, (FBTT)2PbBr4 nanocrystal. c, (FBTP)2PbBr4 nanocrystal. The scale bars are 5 μm. The PEA sample shows a square shape with violet emission from the [PbBr4]2− inorganic sublattice, which is consistent with the results from the literature58, demonstrating the successful growth of single-crystalline (PEA)2PbBr4 2D perovskite. By replacing the PEA with FBTT or FBTP, we also obtained rectangular nanocrystals using a similar crystal-growth procedure, implying the formation of single-crystalline FBTT-based and FBTP-based 2D perovskite nanocrystals. Moreover, FBTT-based and FBTP-based nanocrystals exhibit strong orange and green emission, respectively, which is probably from the organic molecules. By taking a closer look at their PL spectra, we found that the PL peak of the (FBTT)2PbBr4 nanocrystal is closer to that of FBTT aggregates, whereas the PL peak of the (FBTP)2PbBr4 nanocrystal closely resembles that of FBTP monomer. These findings align well with our observations from thin-film samples, thus ruling out the influence of crystallinity on different emission properties.
Extended Data Fig. 6 Schematic illustration of MD simulations on organic molecules rotation in the perovskite lattice.
a, FBTT-contained 2D SLs. b, FBTP-contained 2D SLs. The dashed boxes highlight the molecules under rotation investigation.
Extended Data Fig. 7 The packing styles of organic molecules within 2D SLs.
a, View down the long molecular axis of FBTT molecules in the SLs, which clearly demonstrates a traditional herringbone packing style. b, View down the long molecular axis of partial FBTP molecules in the SLs, revealing a new molecular packing style. H atoms and inorganic sublattices are omitted for clarity.
Extended Data Fig. 8 Structural characterizations and UV–vis spectra of PBTP-based and BBTP-based thin films.
XRD (a) and GIWAXS (b,c) patterns for perovskite thin films incorporated with PBTP (b) and BBTP (c) molecules. XRD and out-of-plane (qz) GIWAXS patterns help us verify that both PBTP and BBTP have been successfully incorporated into the lead-bromide matrix, forming a layered structure. The in-plane spacing is determined to be 5.98 and 5.76 Å from the qxy direction at 1.05 and 1.09 Å−1 for PBTP and BBTP (similar to the 3D perovskite lattice), respectively, which indicates that the 2D structures of these organic emitters incorporated 2D SLs. d, UV–vis spectra of PBTP-doped PMMA (monomers), neat PBTP aggregates and PBTP 2D SLs films. e, UV–vis spectra of BBTP-doped PMMA (monomers), neat BBTP aggregates and BBTP 2D SLs films. The sharp UV–vis peak around 400 nm and the nearby shoulder peak from 2D SLs films can be indexed to the excitonic peak of perovskites and organic emitters, respectively, further supporting the formation of 2D perovskite SLs.
Extended Data Fig. 9 Streak-camera results at different temperatures.
a,b, FBTP monomers at 250 K (a) and 150 K (b). c,d, FBTP 2D SLs at 250 K (c) and 150 K (d). After carefully examining our FBTP SL sample and its spectroscopic features, we found several reasons to rule out the dispersive energy transfer explanation64,65. (1) Our organic emitters in the perovskite lattice are the identical molecules, typically exhibiting homogeneous energy distribution. This is different from the conjugated polymer cases, which show dispersive energy transfer from high-energy sites to low-energy sites owing to the inhomogeneously broadened density of states with different effective conjugation length. (2) Forster-type energy transfer usually requires effective spectral overlap between the absorption and emission spectra. The spectral overlap is minimal in our 2D SL system (Fig. 1g and Supplementary Fig. 5c), which may result in the dispersive energy transport being less effective. Furthermore, the excitation wavelengths used for time-resolved PL and streak-camera measurements are 447 and 440 nm, respectively, which are near the tail end of the absorption band. This suggests that the dispersive energy transfer is unlikely to play a notable role in the observed bathochromic shift over time. (3) If the dispersive energy transport were occurring in FBTP 2D SLs, the PL spectra would be expected to continuously redshift and reach a stationary energy in a nanosecond timescale. By contrast, our streak-camera results (Fig. 5e and Extended Data Fig. 9c,d) show redshifting only within about 100 ps. More importantly, the final emission state reached in FBTP SLs is exactly the same as that of the FBTP monomer (Fig. 5d and Extended Data Fig. 9a,b). This behaviour implies that the emerging rapid decay at the short-wavelength side originates from the high-energy emission state (that is, locally excited state), rather than from the high-energy fraction of the emitters (broadened density of states).
Extended Data Fig. 10 Lasing characterization on FBTP monomer and FBTP aggregates.
a, Experimentally measured reflectance spectra for DBRs, in which the pump laser was directed into the device from the top DBR to excite the sample, followed by the collection of emission from the same side. The wavelength range of the top DBR with high reflectance (>98%) fully covers the optical gain region of FBTP monomer, 2D SLs and aggregates. b, Simulated transmittance spectra of a 2D SLs-based DBR device, which matches well with the lasing spectra from this device. Inset, electric-field distribution of the 536-nm optical standing wave inside the device. c, PL images of a 2D SLs-based DBR device with pump fluence below, near and above the threshold. Scale bar, 100 μm. d, PL spectra evolution of FBTP monomer under different pump fluences. e, Corresponding PL intensity against increasing pump fluences in log–log scale, showing a clear ‘kink’ at a threshold energy density of 2.71 μJ cm−2. The superlinear intensity dependence is fitted to a power law xp with pmon = 1.70 above the threshold. f, PL spectra evolution of FBTP aggregates under different pump fluences. g, Corresponding PL intensity against increasing pump fluences in log–log scale, which reveals linear (pagg = 1.01) and sublinear (pagg = 0.78) growth across a broad pump fluence range, suggesting the absence of lasing from FBTP aggregates.
Supplementary information
Supplementary Information (download PDF )
This file contains the materials synthesis routes, Supplementary Figs. 1–25, Supplementary Tables 1–3, 1H, 13C-NMR and high-resolution mass spectrometry spectra and Supplementary References.
Supplementary data (download ZIP )
This zipped file contains single-crystal data in cif files for FBTT-based and FBTP-based 2D SLs.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Wang, K., Lin, ZY., De, A. et al. Two-dimensional-lattice-confined single-molecule-like aggregates. Nature 633, 567–574 (2024). https://doi.org/10.1038/s41586-024-07925-9
Received:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41586-024-07925-9
This article is cited by
-
Efficient solution-processed light-emitting diodes based on organic-inorganic hybrid antimony halides
Nature Communications (2026)
-
Ultrahigh-radiance TTA-based OLED with 13 kA cm−2 current injection
Light: Science & Applications (2026)
-
Organic parallel grouping crystals without grain boundary
Nature Communications (2025)