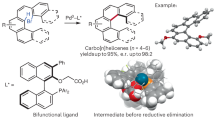
Abstract
Deracemization is an emerging strategy for generating enantioenriched compounds wherein the two enantiomers of a readily available racemic starting material are transformed into a single enantiomer, typically through the action of a light-induced catalyst1,2. Excellent proof of principle for this potentially powerful approach to asymmetric catalysis has been described3,4,5,6,7,8; nevertheless, substantial challenges have not yet been addressed, including the exploitation of carbon–heteroatom (rather than only carbon–hydrogen and carbon–carbon) bond cleavage to achieve deracemization, as well as the development of processes that provide broad classes of useful enantioenriched compounds and tetrasubstituted stereocentres. Here we describe a straightforward method that addresses these challenges, using a chiral copper catalyst, generated in situ from commercially available components, to achieve the photoinduced deracemization of tertiary (and secondary) alkyl halides through carbon–halogen bond cleavage. Mechanistic studies (including the independent synthesis of postulated intermediates, photophysical, spectroscopic and reactivity studies, and density functional theory calculations) provide support for the key steps and intermediates in our proposed catalytic cycle, as well as insight into the origin of enantioselectivity.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$32.99 / 30 days
cancel any time
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
Data availability
The data that support the findings of this study are available within the main text and its Supplementary Information, as well as from the Cambridge Crystallographic Data Centre (https://www.ccdc.cam.ac.uk/structures); crystallographic data are available free of charge under CCDC reference numbers 2285310, 2285314–2285317, 2285320, 2285322, 2285324, 2285325, 2368201, 2368202 and 2409546.
References
Huang, M., Pan, T., Jiang, X. & Luo, S. Catalytic deracemization reactions. J. Am. Chem. Soc. 145, 10917–10929 (2023).
Großkopf, J. & Bach, T. Catalytic photochemical deracemization via short-lived intermediates. Angew. Chem. Int. Edn 62, e202308241 (2023).
Hölzl-Hobmeier, A. et al. Catalytic deracemization of chiral allenes by sensitized excitation with visible light. Nature 564, 240–243 (2018).
Großkopf, J. et al. Photochemical deracemization at sp3-hybridized carbon centers via a reversible hydrogen atom transfer. J. Am. Chem. Soc. 143, 21241–21245 (2021).
Shin, N. Y., Ryss, J. M., Zhang, X., Miller, S. J. & Knowles, R. R. Light-driven deracemization enabled by excited-state electron transfer. Science 366, 364–369 (2019).
Huang, M., Zhang, L., Pan, T. & Luo, S. Deracemization through photochemical E/Z isomerization of enamines. Science 375, 869–874 (2022).
Onneken, C. et al. Light-enabled deracemization of cyclopropanes by Al-salen photocatalysis. Nature 621, 753–759 (2023).
Wen, L. et al. Multiplicative enhancement of stereoenrichment by a single catalyst for deracemization of alcohols. Science 382, 458–464 (2023).
Blackmond, D. G. “If pigs could fly” chemistry: a tutorial on the principle of microscopic reversibility. Angew. Chem. Int. Edn 48, 2648–2654 (2009).
Li, X. et al. Photochemically induced ring opening of spirocyclopropyl oxindoles: evidence for a triplet 1,3-diradical intermediate and deracemization by a chiral sensitizer. Angew. Chem. Int. Edn 59, 21640–21647 (2020).
Wang, J. et al. Enantioselective [2 + 2] photocycloreversion enables de novo deracemization synthesis of cyclobutanes. J. Am. Chem. Soc. 146, 22840–22849 (2024).
Cossy, J. (ed.) Comprehensive Chirality (Academic, 2024).
Mizuta, S., Kitamura, K., Kitagawa, A., Yamaguchi, T. & Ishikawa, T. Silver-promoted fluorination reactions of α-bromoamides. Chem. Eur. J. 27, 5930–5935 (2021).
Akagawa, H. et al. Carboxamide-directed stereospecific couplings of chiral tertiary alkyl halides with terminal alkynes. ACS Catal. 12, 9831–9838 (2022).
Ishida, S., Takeuchi, K., Taniyama, N., Sunada, Y. & Nishikata, T. Copper-catalyzed amination of congested and functionalized α-bromocarboxamides with either amines or ammonia at room temperature. Angew. Chem. Int. Edn 56, 11610–11614 (2017).
Fantinati, A., Zanirato, V., Marchetti, P. & Trapella, C. The fascinating chemistry of α-haloamides. ChemistryOpen 9, 100–170 (2020).
Nishikata, T. α-Halocarbonyls as a valuable functionalized tertiary alkyl source. ChemistryOpen 13, e202400108 (2024).
Gribble, G. W. Naturally Occurring Organohalogen Compounds—A Comprehensive Review (Springer, 2023).
Gribble, G. W. Biological activity of recently discovered halogenated marine natural products. Mar. Drugs 13, 4044–4136 (2015).
Chiodi, D. & Ishihara, Y. “Magic chloro”: profound effects of the chlorine atom in drug discovery. J. Med. Chem. 66, 5305–5331 (2023).
Gerebtzoff, G., Li-Blatter, X., Fischer, H., Frentzel, A. & Seelig, A. Halogenation of drugs enhances membrane binding and permeation. ChemBioChem 5, 676–684 (2004).
Hernandes, M. Z., Cavalcanti, S. M. T., Moreira, D. R. M., de Azevedo Junior, W. F. & Leite, A. C. L. Halogen atoms in the modern medicinal chemistry: hints for the drug design. Curr. Drug Targets 11, 303–314 (2010).
Bouzbouz, S. & Cahard, D. in Comprehensive Chirality (ed. Cossy, J.) Ch. 7.09 (Academic, 2024).
Shibatomi, K. Alternative synthetic strategies for enantioselective construction of halogenated chiral carbon centers. Synthesis 2010, 2679–2702 (2010).
Gómez-Martinez, M., Alonso, D. A., Pastor, I. M., Guillena, G. & Baeza, A. Organocatalyzed assembly of chlorinated quaternary stereogenic centers. Asian J. Org. Chem. 5, 1428–1437 (2016).
Liu, Y., Leng, H.-J., Li, Q.-Z. & Li, J.-L. Catalytic strategies for the asymmetric construction of cyclic frameworks with a halogenated tetrasubstituted stereocenter. Adv. Synth. Catal. 362, 3926–3947 (2020).
Zhang, X. & Tan, C.-H. Stereospecific and stereoconvergent nucleophilic substitution reactions at tertiary carbon centers. Chem 7, 1451–1486 (2021).
Smith, A. M. R. & Hii, K. K. Transition metal catalyzed enantioselective α-heterofunctionalization of carbonyl compounds. Chem. Rev. 111, 1637–1656 (2011).
Shibatomi, K. & Narayama, A. Catalytic enantioselective α-chlorination of carbonyl compounds. Asian J. Org. Chem. 2, 812–823 (2013).
Wang, M. et al. Asymmetric hydrogenation of ketimines with minimally different alkyl groups. Nature 631, 556–562 (2024).
D’Angeli, F. & Marchetti, P. 2-Bromoamides. Stereocontrolled substitution and application to the synthesis of compounds of biological interest. Industr. Chem. Libr. 7, 160–170 (1995).
Wu, D., Fan, W., Wu, L., Chen, P. & Liu, G. Copper-catalyzed enantioselective radical chlorination of alkenes. ACS Catal. 12, 5284–5291 (2022).
Li, Z. et al. Catalytic enantioselective nucleophilic α-chlorination of ketones with NaCl. J. Am. Chem. Soc. 146, 2779–2788 (2024).
Zhu, Y. et al. Modern approaches for asymmetric construction of carbon−fluorine quaternary stereogenic centers: synthetic challenges and pharmaceutical needs. Chem. Rev. 118, 3887–3964 (2018).
Tredwell, M. & Gouverneur, V. in Comprehensive Chirality (eds Carreira, E. M. & Yamamoto, H.) Ch. 1.5 (Academic, 2012).
Butcher, T. W. et al. Desymmetrization of difluoromethylene groups by C–F bond activation. Nature 583, 548–553 (2020).
Zhanel, G. G. et al. Solithromycin: a novel fluoroketolide for the treatment of community-acquired bacterial pneumonia. Drugs 76, 1737–1757 (2016).
Minko, Y. & Marek, I. Stereodefined acyclic trisubstituted metal enolates towards the asymmetric formation of quaternary carbon stereocentres. Chem. Commun. 50, 12597–12611 (2014).
Jia, Z. & Luo, S. in Comprehensive Chirality (ed. Cossy, J.) Ch. 7.07 (Academic, 2024).
Zhang, Y., Vanderghinste, J., Wang, J. & Das, S. Challenges and recent advancements in the synthesis of α,α-disubstituted α-amino acids. Nat. Commun. 15, 1474 (2024).
Friis, S. D., Pirnot, M. T., Dupuis, L. N. & Buchwald, S. L. A dual palladium and copper hydride catalyzed approach for alkyl–aryl cross‐coupling of aryl halides and olefins. Angew. Chem. Int. Edn 56, 7242–7246 (2017).
Xi, Y. & Hartwig, J. F. Mechanistic studies of copper-catalyzed asymmetric hydroboration of alkenes. J. Am. Chem. Soc. 139, 12758–12772 (2017).
Cho, H., Suematsu, H., Oyala, P. H., Peters, J. C. & Fu, G. C. Photoinduced, copper-catalyzed enantioconvergent alkylations of anilines by racemic tertiary electrophiles: synthesis and mechanism. J. Am. Chem. Soc. 144, 4550–4558 (2022).
Eaton, G. R., Eaton, S. S., Barr, D. P. & Weber, R. T. Quantitative EPR (Springer, 2010).
Schneebeli, S. T., Hall, M. L., Breslow, R. & Friesner, R. Quantitative DFT modeling of the enantiomeric excess for dioxirane-catalyzed epoxidations. J. Am. Chem. Soc. 131, 3965–3973 (2009).
Fang, C. et al. Mechanistically guided predictive models for ligand and initiator effects in copper-catalyzed atom transfer radical polymerization (Cu-ATRP). J. Am. Chem. Soc. 141, 7486–7497 (2019).
Chen, B., Fang, C., Liu, P. & Ready, J. M. Rhodium-catalyzed enantioselective radical addition of CX4 reagents to olefins. Angew. Chem. Int. Edn 56, 8780–8784 (2017).
Acknowledgements
This work has been supported by the National Institutes of Health (National Institute of General Medical Sciences, R35–GM145315 (G.C.F.) and R35–GM128779 (P.L.). We thank the Beckman Institute and the Dow Next-Generation Educator Fund for support and Takasago International Corporation for providing DTBM-SEGPHOS. We thank P. H. Oyala, M. K. Takase, D. Vander Velde, S. C. Virgil, J. R. Winkler (National Institutes of Health grant 1S10–OD032151), R. Anderson, H. Cho and Z.-Y. Wang for assistance and discussions. DFT calculations were carried out at the University of Pittsburgh Center for Research Computing and the Advanced Cyberinfrastructure Coordination Ecosystem: Services & Support (ACCESS) programme, supported by NSF award numbers OAC-2117681, OAC-1928147 and OAC-1928224.
Author information
Authors and Affiliations
Contributions
R.L. discovered and optimized the reaction. F.Z. and R.L. carried out the experiments illustrated in Figs. 2–4. B.K.M. performed the DFT calculations illustrated in Fig. 5. G.C.F. and P.L. directed the project. All authors contributed to the data analysis and the writing of the paper.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature thanks Ángel Rentería-Gómez and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Zhong, F., Li, R., Mai, B.K. et al. Photoinduced copper-catalysed deracemization of alkyl halides. Nature 640, 107–113 (2025). https://doi.org/10.1038/s41586-025-08784-8
Received:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41586-025-08784-8
This article is cited by
-
Unlocking enzymatic deracemization of structurally diverse allenes by designed photoenzymes
Nature Communications (2026)
-
Radical ligand transfer catalysis of photoexcited dinuclear gold complexes
Nature Catalysis (2026)
-
Redox-powered autonomous directional C–C bond rotation under enzyme control
Nature (2025)