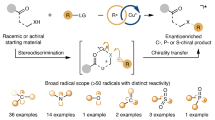
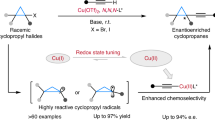
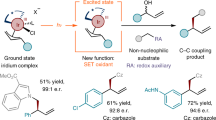
Abstract
Free radicals were first discovered more than 120 years ago by Gomberg1 and the first radical cross-couplings demonstrated by Kochi in the 1970s (ref. 2). In contrast to widely used polar cross-coupling chemistry to forge C(sp2)–C(sp2) bonds (such as Suzuki, Negishi and Kumada), radical cross-coupling is advantageous when applied to the coupling of saturated systems because of the mild conditions used and enhanced chemoselectivity associated with single-electron chemistry. The ability to use ubiquitous carbon-based fragments (such as carboxylic acids, alcohols, amines and olefins) in cross-coupling has greatly simplified access to various complex molecules3,4,5,6,7,8,9. Apart from these advantages, enantiospecific coupling reactions involving free radicals are unknown and generally believed to be challenging because of their near-instantaneous racemization (picosecond timescale)10. As a result, controlling the stereochemical outcome of radical cross-coupling can be achieved only on a case-by-case basis using bespoke chiral ligands11 or in a diastereoselective fashion guided by nearby stereocentres12. Here we show how readily accessible enantioenriched sulfonylhydrazides and low loadings of an inexpensive achiral Ni catalyst can be used to solve this challenge, thereby enabling enantiospecific, stereoretentive radical cross-coupling between enantioenriched alkyl fragments and (hetero)aryl halides without exogenous redox chemistry or chiral ligands. Calculations support the intermediacy of a unique Ni-bound diazene-containing transition state with C–C bond formation driven by loss of N2.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$32.99 / 30 days
cancel any time
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
Data availability
All the data are available within the main text or the Supplementary Information. Experimental and characterization data for all new compounds prepared during this study are provided in the Supplementary Information. The X-ray crystallographic coordinate for Ni(4-Cl-bpy)(NO3)2·2H2O and compound 13-Ts has been deposited at the Cambridge Crystallographic Data Centre (CCDC) with accession codes 2425076 and 2411035, respectively. Copies of the data can be obtained free of charge at https://www.ccdc.cam.ac.uk/structures/.
References
Gomberg, M. An instance of trivalent carbon: triphenylmethyl. J. Am. Chem. Soc. 22, 757–771 (1900).
Morrell, D. G. & Kochi, J. K. Mechanistic studies of nickel catalysis in the cross coupling of aryl halides with alkylmetals. Role of arylalkylnickel(II) species as intermediates. J. Am. Chem. Soc. 97, 7262–7270 (1975).
Smith, J. M., Harwood, S. J. & Baran, P. S. Radical retrosynthesis. Acc. Chem. Res. 51, 1807–1817 (2018).
Yan, M., Lo, J. C., Edwards, J. T. & Baran, P. S. Radicals: reactive intermediates with translational potential. J. Am. Chem. Soc. 138, 12692–12714 (2016).
Palkowitz, M. D., Emmanuel, M. A. & Oderinde, M. S. A paradigm shift in catalysis: electro- and photomediated nickel-catalyzed cross-coupling reactions. Acc. Chem. Res. 56, 2851–2865 (2023).
Laudadio, G., Palkowitz, M. D., El-Hayek Ewing, T. & Baran, P. S. Decarboxylative cross-coupling: a radical tool in medicinal chemistry. ACS Med. Chem. Lett. 13, 1413–1420 (2022).
Harwood, S. J. et al. Modular terpene synthesis enabled by mild electrochemical couplings. Science 375, 745–752 (2022).
Zhang, B. et al. Complex molecule synthesis by electrocatalytic decarboxylative cross-coupling. Nature 623, 745–751 (2023).
Zhang, B. et al. Ni-electrocatalytic Csp3–Csp3 doubly decarboxylative coupling. Nature 606, 313–318 (2022).
Johnston, L. J. & Ingold, K. U. Kinetics of cyclopropyl radical reactions. 2. Studies on the inversion of cyclopropyl and 1-methylcyclopropyl radicals and on the kinetics of some addition and abstraction reactions of 1-methylcyclopropyl and 1-methoxycyclopropyl radicals. J. Am. Chem. Soc. 108, 2343–2348 (1986).
Fu, G. C. Transition-metal catalysis of nucleophilic substitution reactions: a radical alternative to SN1 and SN2 processes. ACS Cent. Sci. 3, 692–700 (2017).
Yang, Y.-H. & Sibi, M. P. in Encyclopedia of Radicals in Chemistry, Biology and Materials (eds Chatgilialoglu, C. & Studer, A.) 655–692 (Wiley, 2012).
Bar, G. & Parsons, A. F. Stereoselective radical reactions. Chem. Soc. Rev. 32, 251–263 (2003).
Buckmelter, A. J., Kim, A. I. & Rychnovsky, S. D. Conformational memory in enantioselective radical reductions and a new radical clock reaction. J. Am. Chem. Soc. 122, 9386–9390 (2000).
Resendiz, M. J. E. et al. Radical reactions with double memory of chirality (2MOC) for the enantiospecific synthesis of adjacent stereogenic quaternary centers in solution: cleavage and bonding faster than radical rotation. J. Am. Chem. Soc. 131, 8425–8433 (2009).
Gloor, C. S., Dénès, F. & Renaud, P. Memory of chirality in reactions involving monoradicals. Free Radic. Res. 50, 102–111 (2016).
Curran, D. P., Porter, N. A. & Giese, B. Stereochemistry of Radical Reactions: Concepts, Guidelines, and Synthetic Applications (Wiley, 2008).
Biswas, S. & Weix, D. J. Mechanism and selectivity in nickel-catalyzed cross-electrophile coupling of aryl halides with alkyl halides. J. Am. Chem. Soc. 135, 16192–16197 (2013).
Toriyama, F. et al. Redox-active esters in Fe-catalyzed C–C coupling. J. Am. Chem. Soc. 138, 11132–11135 (2016).
Chen, T.-G. et al. Building C(sp3)-rich complexity by combining cycloaddition and C–C cross-coupling reactions. Nature 560, 350–354 (2018).
Mondal, S. et al. Enantioselective radical reactions using chiral catalysts. Chem. Rev. 122, 5842–5976 (2022).
Yus, M., Nájera, C., Foubelo, F. & Sansano, J. M. Metal-catalyzed enantioconvergent transformations. Chem. Rev. 123, 11817–11893 (2023).
Kennedy, S. H., Dherange, B. D., Berger, K. J. & Levin, M. D. Skeletal editing through direct nitrogen deletion of secondary amines. Nature 593, 223–227 (2021).
Chi, Y., Tang, W. & Zhang, X. in Modern Rhodium-Catalyzed Organic Reactions (ed. Evans, A.) 1–31 (Wiley, 2005).
Minnaard, A. J., Feringa, B. L., Lefort, L. & de Vries, J. G. Asymmetric hydrogenation using monodentate phosphoramidite ligands. Acc. Chem. Res. 40, 1267–1277 (2007).
Roseblade, S. J. & Pfaltz, A. Iridium-catalyzed asymmetric hydrogenation of olefins. Acc. Chem. Res. 40, 1402–1411 (2007).
Noyori, R., Kitamura, M. & Ohkuma, T. Toward efficient asymmetric hydrogenation: architectural and functional engineering of chiral molecular catalysts. Proc. Natl Acad. Sci. USA 101, 5356–5362 (2004).
Knowles, W. S. & Noyori, R. Pioneering perspectives on asymmetric hydrogenation. Acc. Chem. Res. 40, 1238–1239 (2007).
Gong, Y., Hu, J., Qiu, C. & Gong, H. Insights into recent nickel-catalyzed reductive and redox C–C coupling of electrophiles, C(sp3)–H bonds and alkenes. Acc. Chem. Res. 57, 1149–1162 (2024).
Chan, A. Y. et al. Metallaphotoredox: the merger of photoredox and transition metal catalysis. Chem. Rev. 122, 1485–1542 (2022).
Payamifar, S., Behrouzi, L. & Poursattar Marjani, A. The electrochemical coupling reactions of organic halides compound in a valuable and practical manner for C–C and C–heteroatom formation: an overview. Arab. J. Chem. 17, 105822 (2024).
Sun, J. et al. Sulfonyl hydrazides as a general redox-neutral platform for radical cross-coupling. Science 387, 1377–1383 (2025).
Schultz, P. G. & Dervan, P. B. Photochemistry of 1,1-diazenes. Direct and sensitized photolyses of N-(2,2,5,5-tetramethylpyrrolidyl)nitrene, dl-N-(2,5-diethyl-2,5-dimethylpyrrolidyl)nitrene, and N-(2,2,6,6-tetramethylpiperidyl)nitrene. J. Am. Chem. Soc. 104, 6660–6668 (1982).
Movassaghi, M., Ahmad, O. K. & Lathrop, S. P. Directed heterodimerization: stereocontrolled assembly via solvent-caged unsymmetrical diazene fragmentation. J. Am. Chem. Soc. 133, 13002–13005 (2011).
Horning, B. et al. Compounds selective for JAK1 and methods of use. WO 2022/251280 A1 (2022).
Nagaraj, H. K. M. et al. Piperidine ureas as cathepsin cysteine protease inhibitors. WO 2016/027284 A3 (2016).
Buesking, A. et al. CDK Inhibitors and their use as pharmaceuticals. WO 2022/061273 A1 (2022).
Liu, P., Miller, C. A., Yu, M. & Zhang, Z. Heteroaryl carboxamide compounds as inhibitors of RIPK2. US patent 2018/0072717 A1 (2018).
Brooijmans, N. et al. Inhibitors of activin receptor-like kinase. WO 2017/181117 A1 (2017).
Whitten, J. P., Schwaebe, M. & Regan, C. Chiral pyrrolidine derivatives, and methods for preparing compounds thereof. US 2006/0025468 A1 (2006).
Palkowitz, M. D. et al. Overcoming limitations in decarboxylative arylation via Ag–Ni electrocatalysis. J. Am. Chem. Soc. 144, 17709–17720 (2022).
Neese, F. Software update: the ORCA program system—version 5.0. WIRES Comput. Mol. Sci. 12, e1606 (2022).
Myers, A. G., Movassaghi, M. & Zheng, B. Mechanistic studies of the free-radical fragmentation of monoalkyl diazenes. Tetrahedron Lett. 38, 6569–6572 (1997).
Cagan, D. A., Bím, D., Kazmierczak, N. P. & Hadt, R. G. Mechanisms of photoredox catalysis featuring nickel–bipyridine complexes. ACS Catal. 14, 9055–9076 (2024).
Lin, Q., Spielvogel, E. H. & Diao, T. Carbon-centered radical capture at nickel(II) complexes: spectroscopic evidence, rates, and selectivity. Chem 9, 1295–1308 (2023).
Yuan, M., Song, Z., Badir, S. O., Molander, G. A. & Gutierrez, O. On the nature of C(sp3)–C(sp2) bond formation in nickel-catalyzed tertiary radical cross-couplings: a case study of Ni/photoredox catalytic cross-coupling of alkyl radicals and aryl halides. J. Am. Chem. Soc. 142, 7225–7234 (2020).
Brookhart, M., Green, M. L. H. & Parkin, G. Agostic interactions in transition metal compounds. Proc. Natl Acad. Sci. USA 104, 6908–6914 (2007).
Newcomb, M. in Encyclopedia of Radicals in Chemistry, Biology and Materials (eds Chatgilialoglu, C. & Studer, A.) 1–18 (Wiley, 2012).
Shevick, S. L., Obradors, C. & Shenvi, R. A. Mechanistic interrogation of Co/Ni-dual catalyzed hydroarylation. J. Am. Chem. Soc. 140, 12056–12068 (2018).
Tasker, S. Z., Standley, E. A. & Jamison, T. F. Recent advances in homogeneous nickel catalysis. Nature 509, 299–309 (2014).
Acknowledgements
This project is financially supported by NIH (GM-118176, P.S.B.) and the Gates Foundation (INV-056603, P.S.B. and Y.K.). L.M. thanks the Swedish Research Council (Vetenskapsrådet, VR-2023-00499) for a postdoctoral fellowship. F.C.A. acknowledges the ARC Centre of Excellence for Innovation in Peptide and Protein Science for Capacity Building (grant CBG117). We also thank D.-H. Huang and L. Pasternack (Scripps Research) for NMR spectroscopic assistance; M. Gembicky (UCSD) for X-ray analysis; and B. Orzolek, B. Sanchez and Q. N. Wong (Scripps Automated Synthesis Facility). We thank B. Jiang, M. Costantini and Á. Péter for their discussions. The computations presented here were conducted in the Garibaldi High Performance Computing (HPC) cluster, a facility supported by Scripps Research, La Jolla.
Author information
Authors and Affiliations
Contributions
J.S., J.H., L.M., Y.K. and P.S.B. conceptualized the study. J.S., J.H., L.M., D.A.C., J.T., Y.W., F.C.A., J.E.S. and J.S.L. conducted the experimental investigation. J.S., J.H., L.M., D.A.C., J.T., Y.W., J.E.S., J.S.L., Y.K. and P.S.B. performed the data analysis. J.S., J.H., L.M., D.A.C., J.T., Y.K. and P.S.B. wrote the paper. Y.K. and P.S.B. helped with the fund acquisition. P.S.B. was in charge of the project administration.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature thanks the anonymous reviewers for their contribution to the peer review of this work. Peer reviewer reports are available
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information (download PDF )
This file contains Supplementary Text and NMR Spectra data; see contents pages for details.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Sun, J., He, J., Massaro, L. et al. Stereoretentive radical cross-coupling. Nature 642, 85–91 (2025). https://doi.org/10.1038/s41586-025-09011-0
Received:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41586-025-09011-0