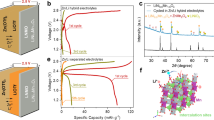
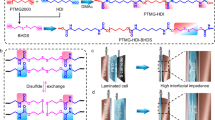
Abstract
Polymer electrolytes paired with lithium-rich manganese-based layered oxide (LRMO) cathodes and anode-free cell design are considered one of the most promising high-energy-density and high-safety systems1,2,3,4. However, the unstable anode morphological changes and the irreversible anionic reactions at the electrolyte–cathode interfaces induce oxygen escape and catalytic decomposition of polymer electrolytes, resulting in severe interfacial degradation and poor cycling stability. Here we design an in-built fluoropolyether-based polymer electrolyte composed of strongly solvating polyether and weakly solvating fluorohydrocarbon pendants, creating an anion-rich solvation structure and thus anion-derived fluorine-rich interfacial layers on the cathode and anode to resist interfacial issues. The LRMO cathode exhibits improved oxygen redox reversibility with substantially reduced oxygen-involving interfacial side reactions. This quasi-solid-state polymer electrolyte with 30 wt% trimethyl phosphate enables an LRMO cathode with a reversible high-areal-capacity cycling (>8 mAh cm−2) in pouch cells and long-term stability (>500 cycles at 25 °C) in coin cells, respectively. The pouch cells exhibit an energy density of 604 Wh kg−1 (1,027 Wh l−1) and excellent safety under a nail penetration at a fully charged condition. Our work, therefore, provides a promising direction for creating practical high-energy-density and high-safety lithium batteries.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$32.99 / 30 days
cancel any time
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
Data availability
The data that support the findings of this study are available from the corresponding author upon request. Source data are provided with this paper.
References
Luo, D. et al. A Li-rich layered oxide cathode with negligible voltage decay. Nat. Energy 8, 1078–1087 (2023).
Wang, L., Liu, T., Wu, T. & Lu, J. Strain-retardant coherent perovskite phase stabilized Ni-rich cathode. Nature 611, 61–67 (2022).
Lee, Y.-G. et al. High-energy long-cycling all-solid-state lithium metal batteries enabled by silver–carbon composite anodes. Nat. Energy 5, 299–308 (2020).
Deysher, G. et al. Design principles for enabling an anode-free sodium all-solid-state battery. Nat. Energy 9, 1161–1172 (2024).
Krauskopf, T., Richter, F. H., Zeier, W. G. & Janek, J. Physicochemical concepts of the lithium metal anode in solid-state batteries. Chem. Rev. 120, 7745–7794 (2020).
Ning, Z. et al. Dendrite initiation and propagation in lithium metal solid-state batteries. Nature 618, 287–293 (2023).
Wan, H., Wang, Z., Zhang, W., He, X. & Wang, C. Interface design for all-solid-state lithium batteries. Nature 623, 739–744 (2023).
Tarascon, J.-M. & Armand, M. Issues and challenges facing rechargeable lithium batteries. Nature 414, 359–367 (2001).
Winter, M., Barnett, B. & Xu, K. Before Li ion batteries. Chem. Rev. 118, 11433–11456 (2018).
Zhang, Q.-K. et al. Homogeneous and mechanically stable solid–electrolyte interphase enabled by trioxane-modulated electrolytes for lithium metal batteries. Nat. Energy 8, 725–735 (2023).
Enyuan, H. et al. Evolution of redox couples in Li- and Mn-rich cathode materials and mitigation of voltage fade by reducing oxygen release. Nat. Energy 3, 690–698 (2018).
Seo, D.-H. et al. The structural and chemical origin of the oxygen redox activity in layered and cation-disordered Li-excess cathode materials. Nat. Chem. 8, 692–697 (2016).
Sharpe, R. et al. Redox chemistry and the role of trapped molecular O2 in Li-rich disordered rocksalt oxyfluoride cathodes. J. Am. Chem. Soc. 142, 21799–21809 (2020).
Albertus, P. et al. Challenges for and pathways toward Li-metal-based all-solid-state batteries. ACS Energy Lett. 6, 1399–1404 (2021).
Li, Q., Yang, Y., Yu, X. & Li, H. A 700 W·h·kg−1 rechargeable pouch type lithium battery. Chinese Phys. Lett. 40, 048201 (2023).
Chen, B. et al. Achieving the high capacity and high stability of Li‐rich oxide cathode in garnet‐based solid‐state battery. Angew. Chem. Int. Ed. 63, e202315856 (2024).
Kong, W.-J. et al. From liquid to solid-state batteries: Li-rich Mn-based layered oxides as emerging cathodes with high energy density. Adv. Mater. 36, 2310738 (2024).
Sun, S. et al. Eliminating interfacial O-involving degradation in Li-rich Mn-based cathodes for all-solid-state lithium batteries. Sci. Adv. 8, eadd5189 (2022).
Zhao, Q., Liu, X., Stalin, S., Khan, K. & Archer, L. A. Solid-state polymer electrolytes with in-built fast interfacial transport for secondary lithium batteries. Nat. Energy 4, 365–373 (2019).
Liu, Y. et al. In situ polymerization of 1,3-dioxane as a highly compatible polymer electrolyte to enable the stable operation of 4.5 V Li-metal batteries. Energy Environ. Sci. 16, 6110–6119 (2023).
Zhu, J. et al. Long-cycling and high-voltage solid state lithium metal batteries enabled by fluorinated and crosslinked polyether electrolytes. Angew. Chem. Int. Ed. 63, e202400303 (2024).
Yu, J. et al. In situ fabricated quasi-solid polymer electrolyte for high-energy-density lithium metal battery capable of subzero operation. Adv. Energy Mater. 12, 2102932 (2022).
Yao, N. et al. Identifying the lithium bond and lithium ionic bond in electrolytes. Chem 11, 102254 (2025).
Guo, D. et al. Foldable solid-state batteries enabled by electrolyte mediation in covalent organic frameworks. Adv. Mater. 34, 2201410 (2022).
Liu, T. et al. Origin of structural degradation in Li-rich layered oxide cathode. Nature 606, 305–312 (2022).
Zhang, H., Liu, H., Piper, L. F. J., Whittingham, M. S. & Zhou, G. Oxygen loss in layered oxide cathodes for Li-ion batteries: mechanisms, effects, and mitigation. Chem. Rev. 122, 5641–5681 (2022).
Peng, J. et al. Phase compatible NiFe2O4 coating tunes oxygen redox in Li-rich layered oxide. ACS Nano 15, 11607–11618 (2021).
Fan, W. et al. “Peapod-like” fiber network: a universal strategy for composite solid electrolytes to inhibit lithium dendrite growth in solid-state lithium metal batteries. Nano Lett. 24, 9050–9057 (2024).
Lee, W. et al. Advanced parametrization for the production of high-energy solid-state lithium pouch cells containing polymer electrolytes. Nat. Commun. 15, 5860 (2024).
Hu, J. K. et al. High energy density solid‐state lithium metal batteries enabled by in situ polymerized integrated ultrathin solid electrolyte/cathode. Adv. Funct. Mater. 34, 2311633 (2024).
Wang, H. et al. A strongly complexed solid polymer electrolyte enables a stable solid state high-voltage lithium metal battery. Energy Environ. Sci. 15, 5149–5158 (2022).
Guo, J.-C. et al. A self-reconfigured, dual-layered artificial interphase toward high-current-density quasi-solid-state lithium metal batteries. Adv. Mater. 35, 2300350 (2023).
Xu, P. et al. Solvation regulation reinforces anion‐derived inorganic‐rich interphase for high‐performance quasi‐solid‐state Li metal batteries. Adv. Mater. 36, 2409489 (2024).
Kong, W.-J. et al. Bulk/interfacial structure design of Li-rich Mn-based cathodes for all-solid-state lithium batteries. J. Am. Chem. Soc. 146, 28190–28200 (2024).
Frisch, M. J., et al. Gaussian 16 Rev. C.01 (Gaussian, 2016).
Becke, A. D. Density-functional thermochemistry. III. The role of exact exchange. J. Chem. Phys. 98, 5648–5652 (1993).
Marenich, A. V., Cramer, C. J. & Truhlar, D. G. Universal solvation model based on solute electron density and on a continuum model of the solvent defined by the bulk dielectric constant and atomic surface tensions. J. Phys. Chem. B 113, 6378–6396 (2009).
Reed, A. E. et al. Intermolecular interactions from a natural bond orbital, donor-acceptor viewpoint. Chem. Rev. 88, 899–926 (1988).
Trasatti, S. The absolute electrode potential: an explanatory note. Pure Appl. Chem. 58, 955–966 (1986).
Acknowledgements
This work was supported by the National Key Research and Development Program (2021YFB2500300), the National Natural Science Foundation of China (22393900, 22393903, 22409114, 21825501 and T2322015), the Beijing Municipal Natural Science Foundation (L247015, L233004 and L243019), the China Postdoctoral Science Foundation (2023M731920), the Discipline Breakthrough Precursor Project of the Ministry of Education of China and the Tsinghua University Initiative Scientific Research Program.
Author information
Authors and Affiliations
Contributions
X.-Y.H. C.-Z.Z. and Q.Z. proposed the research. X.-Y.H. performed the electrochemical measurements, characterized materials, analysed the data and wrote the paper. W.-J.K. performed the Raman characterization and supervised the analysis of LRMO cathodes. Y.L. helped with the electrochemical experiments. J.-L.L. and L.S. helped with the XPS tests. N.Y. and X.C. performed the theoretical calculations. P.X. helped with the TOF-SIMS tests. Z.-Y.S. and X.-Y.H. assembled the pouch cells. C.-Z.Z., S.S., W.-Z.H., X.C., J.-Q. H., L.A.A. and Q.Z. revised the paper. All authors engaged in the discussion of the results.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature thanks Xiulin Fan, Jinsoo Kim, Faezeh Makhlooghiazad and Yingjin Wei for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information (download PDF )
This file contains Supplementary Figs. 1–53, Supplementary Notes 1–9 and Supplementary Table 1.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Huang, XY., Zhao, CZ., Kong, WJ. et al. Tailoring polymer electrolyte solvation for 600 Wh kg−1 lithium batteries. Nature 646, 343–350 (2025). https://doi.org/10.1038/s41586-025-09565-z
Received:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41586-025-09565-z
This article is cited by
-
Flame-retardant quasi-solid polymer electrolytes for building stable and dendrite-free lithium metal batteries
Science China Chemistry (2026)
-
Artificial intelligence for energy materials research: From classical machine learning to large models
ENGINEERING Energy (2026)