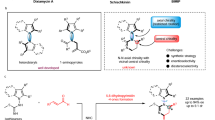
Abstract
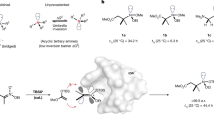
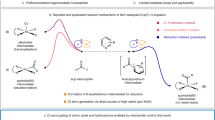
Chirality is central to life, and controlling the formation of one of a pair of mirror-image molecules (enantiomers) is a central tenet of synthetic chemistry. Although controlling stereogenic carbon1,2,3, silicon4,5, phosphorus6,7 and sulfur8,9 centres is commonplace, nitrogen centres in amines are not typically stable. Limited achievements in the enantioselective construction of nitrogen chirality have primarily been established in quaternary ammonium salts10,11,12 and bridged bicyclic amines13,14,15,16,17, which have a restricted pyramidal configuration. The asymmetric synthesis of non-bridged pyramidal nitrogen-chirogenic compounds suffers from a super-stoichiometric chiral source and exhibits poor stereoselectivity18,19,20,21,22,23,24. Here we present a catalytic enantioselective strategy for construction of acyclic nitrogen stereocentres via a chiral Brønsted acid-catalysed chlorination reaction. We designed a stereospecific intramolecular reaction to overcome the structural and configurational instabilities of nitrogen-chlorinated hydroxylamines. The resulting 2-alkoxy-1,2-oxazolidines showed good enantiopurities, and density functional theory calculations confirmed successful enantiocontrol of nitrogen chirality during the chlorination process. Furthermore, this strategy has been applied successfully to synthesize the enantioselective N-chloroaziridines with a configurationally stable nitrogen stereogenic centre. Control experiments provide evidence for an SN2 pathway for the intramolecular nucleophilic substitution event.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$32.99 / 30 days
cancel any time
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
Data availability
Crystallographic data for the structures reported in this article have been deposited at the Cambridge Crystallographic Data Centre, under deposition numbers CCDC 2330815 (20), 2405212 (32), 2405215 (ent-33′), 2330813 (34), 2330814 (45) and 2405209 (49). Copies of the data can be obtained free of charge via https://www.ccdc.cam.ac.uk/structures/. The data supporting the findings of this work are provided in Supplementary Information, including experimental procedures and characterization of new compounds.
References
Quasdorf, K. W. & Overman, L. E. Catalytic enantioselective synthesis of quaternary carbon stereocentres. Nature 516, 181–191 (2014).
Zeng, X.-P., Cao, Z.-Y., Wang, Y.-H., Zhou, F. & Zhou, J. Catalytic enantioselective desymmetrization reactions to all-carbon quaternary stereocenters. Chem. Rev. 116, 7330–7396 (2016).
Feng, J., Holmes, M. & Krische, M. J. Acyclic quaternary carbon stereocenters via enantioselective transition metal catalysis. Chem. Rev. 117, 12564–12580 (2017).
Wu, Y. & Wang, P. Silicon-stereogenic monohydrosilane: synthesis and applications. Angew. Chem. Int. Ed. 61, e202205382 (2022).
Xu, L.-W., Li, L., Lai, G.-Q. & Jiang, J.-X. The recent synthesis and application of silicon-stereogenic silanes: a renewed and significant challenge in asymmetric synthesis. Chem. Soc. Rev. 40, 1777–1790 (2011).
Grabulosa, A., Granell, J. & Muller, G. Preparation of optically pure P-stereogenic trivalent phosphorus compounds. Coord. Chem. Rev. 251, 25–90 (2007).
Dutartre, M., Bayardon, J. & Jugé, S. Applications and stereoselective syntheses of P-chirogenic phosphorus compounds. Chem. Soc. Rev. 45, 5771–5794 (2016).
Fernández, I. & Khiar, N. Recent developments in the synthesis and utilization of chiral sulfoxides. Chem. Rev. 103, 3651–3706 (2003).
Han, J., Soloshonok, V. A., Klika, K. D., Drabowicz, J. & Wzorek, A. Chiral sulfoxides: advances in asymmetric synthesis and problems with the accurate determination of the stereochemical outcome. Chem. Soc. Rev. 47, 1307–1350 (2018).
Walsh, M. P., Phelps, J. M., Lennon, M. E., Yufit, D. S. & Kitching, M. O. Enantioselective synthesis of ammonium cations. Nature 597, 70–76 (2021).
Luo, Z. et al. Ionic hydrogen bond-assisted catalytic construction of nitrogen stereogenic center via formal desymmetrization of remote diols. Angew. Chem. Int. Ed. 63, e202404979 (2024).
Bhadra, S. & Yamamoto, H. Catalytic asymmetric synthesis of N-chiral amine oxides. Angew. Chem. Int. Ed. 55, 13043–13046 (2016).
Chattopadhyay, A. K. & Hanessian, S. Recent progress in the chemistry of daphniphyllum alkaloids. Chem. Rev. 117, 4104–4146 (2017).
Yuan, R. et al. The first direct synthesis of chiral Tröger’s bases catalyzed by chiral glucose-containing pyridinium ionic liquids. Chem. Eng. J. 316, 1026–1034 (2017).
Huang, S. et al. Organocatalytic enantioselective construction of chiral azepine skeleton bearing multiple-stereogenic elements. Angew. Chem. Int. Ed. 60, 21486–21493 (2021).
Ma, C., Sun, Y., Yang, J., Guo, H. & Zhang, J. Catalytic asymmetric synthesis of Tröger’s base analogues with nitrogen stereocenter. ACS Cent. Sci. 9, 64–71 (2023).
Yu, T. et al. Immobilizing stereogenic nitrogen center in doubly fused triarylamines through palladium-catalyzed asymmetric C−H activation/seven-membered-ring formation. ACS Catal. 13, 9688–9694 (2023).
Annunziata, R., Fornasier, R. & Montanari, F. Compounds with molecular asymmetry due solely to a tercovalent non-bridgehead nitrogen atom: optically active N-chloro-2,2-diphenylaziridine. J. Chem. Soc. Chem. Commun. 1972, 1133–1134 (1972).
Forni, A., Moretti, I., Prosyanik, A. V. & Torre, G. Optically active trifluoromethylcarbinols as chiral solvating agents for asymmetric transformations at a ring-nitrogen atom. J. Chem. Soc. Chem. Commun. 1981, 588–590 (1981).
Bucciarelli, M., Forni, A., Moretti, I. & Torre, G. Optically active trifluoromethylcarbinols as chiral solvating agents for asymmetric transformations at a ring-nitrogen atom. Synthesis of optically active N-chloroaziridines and stereochemical aspects of chiral solvent-aziridine solute complexes. J. Org. Chem. 48, 2640–2644 (1983).
Shustov, G. V. et al. Asymmetric nitrogen. 72. Geminal systems. 46. N-chlorooxaziridines: optical activation, absolute configuration, and chiroptical properties. J. Am. Chem. Soc. 111, 4210–4215 (1989).
Montanari, F., Moretti, I. & Torre, G. Asymmetric introduction at trivalent nitrogen. Optically active 2-methyl-3,3-diphenyloxaziridine, a compound with molecular asymmetry due solely to the nitrogen atom. Chem. Commun. 1968, 1694–1695 (1968).
Boyd, D. R. Optically active oxaziridines. Tetrahedron Lett. 9, 4561–4564 (1968).
Kostyanovsky, R. G., Rudchenko, V. F., Shtamburg, V. G., Chervin, I. I. & Nasibov, S. S. Asymmetrical nonbridgehead nitrogen—XXVI. Synthesis, configurational stability, and resolution of N,N-dialkoxyamines into antipodes. Tetrahedron 37, 4245–4254 (1981).
Smith, O. et al. Control of stereogenic oxygen in a helically chiral oxonium ion. Nature 615, 430–435 (2023).
Porto, C. M., de Barros, G. A., Santana, L. C., Moralles, A. C. & Morgon, N. H. Ammonia quantum tunneling in cold rare-gas He and Ar clusters and factorial design approach for methodology evaluation. J. Mol. Model. 28, 293 (2022).
Adams, R. & Cairns, T. L. Attempts to prepare optically active ethyleneimine derivatives containing an asymmetric nitrogen atom. J. Am. Chem. Soc. 61, 2464–2467 (1939).
Dunlop, H. G. & Tucker, S. H. Attempts to prepare optically active tervalent nitrogen compounds. Part I. Syntheses of 1:9-phenylenecarbazole and derivatives. J. Chem. Soc. 1939, 1945–1956 (1939).
Brois, S. J. Aziridines. XII. Isolation of a stable nitrogen pyramid. J. Am. Chem. Soc. 90, 508–509 (1968).
Rauk, A., Allen, L. C. & Mislow, K. Pyramidal inversion. Angew. Chem. Int. Ed. 9, 400–414 (1970).
Zaitseva, S. & Köhler, V. Pyramidal stereogenic nitrogen centers (SNCs). Synthesis 57, 1237–1254 (2025).
Shtamburg, V. G. et al. Reactions of N-chloro-N-alkoxy-tert-alkylamines with isobutylene and methanol. Russ. Chem. Bull. 40, 951–954 (1991).
Rudchenko, V. F. & Kostyanovskii, R. G. Geminal oxygen–nitrogen–halogen systems. N-halohydroxylamine derivatives. Russ. Chem. Rev. 67, 179–192 (1998).
Wendlandt, A. E., Vangal, P. & Jacobsen, E. N. Quaternary stereocentres via an enantioconvergent catalytic SN1 reaction. Nature 556, 447–451 (2018).
Singh, V. K. et al. Taming secondary benzylic cations in catalytic asymmetric SN1 reactions. Science 382, 325–329 (2023).
Zhang, X. et al. An enantioconvergent halogenophilic nucleophilic substitution (SN2X) reaction. Science 363, 400–404 (2019).
Lovinger, G. J., Sak, M. H. & Jacobsen, E. N. Catalysis of an SN2 pathway by geometric preorganization. Nature 632, 1052–1059 (2024).
Denmark, S. E., Kuester, W. E. & Burk, M. T. Catalytic, asymmetric halofunctionalization of alkenes—a critical perspective. Angew. Chem. Int. Ed. 51, 10938–10953 (2012).
Akiyama, T., Itoh, J., Yokota, K. & Fuchibe, K. Enantioselective Mannich-type reaction catalyzed by a chiral Brønsted acid. Angew. Chem. Int. Ed. 43, 1566–1568 (2004).
Uraguchi, D. & Terada, M. Chiral Brønsted acid-catalyzed direct Mannich reactions via electrophilic activation. J. Am. Chem. Soc. 126, 5356–5357 (2004).
Akiyama, T. Stronger Brønsted acids. Chem. Rev. 107, 5744–5758 (2007).
Parmar, D., Sugiono, E., Raja, S. & Rueping, M. Complete field guide to asymmetric BINOL-phosphate derived Brønsted acid and metal catalysis: history and classification by mode of activation; Brønsted acidity, hydrogen bonding, ion pairing and metal phosphates. Chem. Rev. 114, 9047–9153 (2014).
Reisman, S. E., Doyle, A. G. & Jacobsen, E. N. Enantioselective thiourea-catalyzed additions to oxocarbenium ions. J. Am. Chem. Soc. 130, 7198–7199 (2008).
Banik, S. M., Levina, A., Hyde, A. M. & Jacobsen, E. N. Lewis acid enhancement by hydrogen-bond donors for asymmetric catalysis. Science 358, 761–764 (2017).
Zhou, H. et al. Organocatalytic stereoselective cyanosilylation of small ketones. Nature 605, 84–89 (2022).
Wang, M. et al. Asymmetric hydrogenation of ketimines with minimally different alkyl groups. Nature 631, 556–562 (2024).
Frisch, M. J. et al. Gaussian 16, Revision A.03 (Gaussian, 2016).
Becke, A. D. Density‐functional thermochemistry. III. The role of exact exchange. J. Chem. Phys. 98, 5648–5652 (1993).
Lee, C., Yang, W. & Parr, R. G. Development of the Colle–Salvetti correlation-energy formula into a functional of the electron density. Phys. Rev. B 37, 785–789 (1988).
Grimme, S., Antony, J., Ehrlich, S. & Krieg, H. A consistent and accurate ab initio parametrization of density functional dispersion correction (DFT-D) for the 94 elements H–Pu. J. Chem. Phys. 132, 154104 (2010).
Grimme, S., Ehrlich, S. & Goerigk, L. Effect of the damping function in dispersion corrected density functional theory. J. Comput. Chem. 32, 1456–1465 (2011).
Chai, J.-D. & Head-Gordon, M. Long-range corrected hybrid density functionals with damped atom–atom dispersion corrections. Phys. Chem. Chem. Phys. 10, 6615–6620 (2008).
Marenich, A. V., Cramer, C. J. & Truhlar, D. G. Universal solvation model based on solute electron density and on a continuum model of the solvent defined by the bulk dielectric constant and atomic surface tensions. J. Phys. Chem. B 113, 6378–6396 (2009).
Bickelhaupt, F. M. & Houk, K. N. Analyzing reaction rates with the distortion/interaction‐activation strain model. Angew. Chem. Int. Ed. 56, 10070–10086 (2017).
Duan, M. et al. Chiral phosphoric acid catalyzed conversion of epoxides into thiiranes: mechanism, stereochemical model, and new catalyst design. Angew. Chem. Int. Ed. 61, e202113204 (2022).
Acknowledgements
We are grateful for financial support from the National Natural Science Foundation of China (22425011 to B.T., 22231004 to B.T. and 22271135 to S.-H.X.), the National Key R&D Program of China (2022YFA1503700 to B.T.), the Guangdong Innovative Program (2019BT02Y335 to B.T.), the Guangdong Basic and Applied Basic Research Foundation (2024B1515020055 to S.-H.X.), the New Cornerstone Science Foundation through the Xplorer Prize (to B.T.), the Shenzhen Science and Technology Program (KQTD20210811090112004 to B.T.), High level of special funds (G03050K003 to B.T.) and the US National Science Foundation (CHE-2153972 to K.N.H.). We appreciate the assistance of SUSTech Core Research Facilities.
Author information
Authors and Affiliations
Contributions
B.T. conceived of and directed the project. S.W. and P.C. designed and performed the experiments. P.-Y.J. and S.-H.X. helped with the collection of some compounds and data analysis. K.N.H. directed the DFT calculations and mechanism analysis. M.D. performed the DFT calculations. M.D. and Q.Z. conducted the mechanism analysis. B.T., S.W. and S.-H.X. wrote the paper. All authors discussed the results and commented on the paper. S.W., P.C. and M.D. contributed equally to this work.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature thanks Jolene Reid and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data figures and tables
Extended Data Fig. 1 Research status of catalytic enantioselective construction of stereocentres and strategies to stabilize the configuration of nitrogen-chirogenic molecules.
a, Research status of catalytic asymmetric construction of various stereogenic centres. b, Conventional means to enhance the pyramidal inversion barrier of N-stereogenic centres.
Supplementary information
Supplementary Information (download PDF )
The Supplementary Information file contains Supplementary Methods, characterization details, optimized Cartesian coordinates, Supplementary Figs. 1–18, Tables 1–5 and Refs. 1–4.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Wu, S., Chen, P., Duan, M. et al. Controlling pyramidal nitrogen chirality by asymmetric organocatalysis. Nature 647, 897–905 (2025). https://doi.org/10.1038/s41586-025-09607-6
Received:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41586-025-09607-6
This article is cited by
-
Taming pyramidal nitrogen chirality
Science China Chemistry (2025)