Abstract
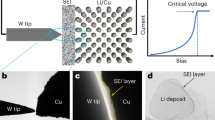
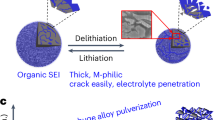
Solid-state lithium metal batteries are facing huge challenges under practical working conditions1,2. Even when the ionic conductivity of composite solid-state electrolytes is increased to 1 mS cm−1, it is still difficult to realize long-life cycling of solid-state batteries above a current density of 1 mA cm−2 and an areal capacity of 1 mAh cm−2 (ref. 3). The fundamental cause is the brittle nature of the solid–electrolyte interphase (SEI) with sluggish lithium-ion transport and the resulting lithium dendrites and severe side reactions. Here we report a ductile inorganic-rich SEI that retains its structural integrity while allowing easy ion diffusion at high current densities and areal capacities. The ductility of the SEI is ascribed to the Ag2S and AgF components, which are formed by a substitution reaction between Li2S/LiF in the SEI and AgNO3 in the dielectric composite electrolytes. Even at a high current density of 15 mA cm−2 and an areal capacity of 15 mAh cm−2, a symmetrical lithium cell with such an SEI has a long cycle life of over 4,500 hours. Furthermore, the ductile SEI also works over 7,000 hours at −30 °C, even under practical conditions of 5 mA cm−2 and 5 mAh cm−2.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$32.99 / 30 days
cancel any time
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
Data availability
All data are available in the paper or the Supplementary Information. Other raw data are available from the corresponding authors on request. Source data are provided with this paper.
References
Kalnaus, S., Dudney, N. J., Westover, A. S., Herbert, E. & Hackney, S. Solid-state batteries: the critical role of mechanics. Science 381, eabg5998 (2023).
Alexander, G. V., Shi, C., O’Neill, J. & Wachsman, E. D. Extreme lithium-metal cycling enabled by a mixed ion- and electron-conducting garnet three-dimensional architecture. Nat. Mater. 22, 1136–1143 (2023).
Yang, C. et al. Copper-coordinated cellulose ion conductors for solid-state batteries. Nature 598, 590–596 (2021).
Liu, J. et al. Pathways for practical high-energy long-cycling lithium metal batteries. Nat. Energy 4, 180–186 (2019).
Wan, H., Wang, Z., Zhang, W., He, X. & Wang, C. Interface design for all-solid-state lithium batteries. Nature 623, 739–744 (2023).
Hitz, G. T. et al. High-rate lithium cycling in a scalable trilayer Li-garnet-electrolyte architecture. Mater. Today 22, 50–57 (2019).
Wan, J. et al. Ultrathin, flexible, solid polymer composite electrolyte enabled with aligned nanoporous host for lithium batteries. Nat. Nanotechnol. 14, 705–711 (2019).
Zhang, W. et al. Single-phase local-high-concentration solid polymer electrolytes for lithium-metal batteries. Nat. Energy 9, 386–400 (2024).
Yang, K. et al. Determining the role of ion transport throughput in solid-state lithium batteries. Angew. Chem. Int. Ed. 135, e202302586 (2023).
Albertus, P., Babinec, S., Litzelman, S. & Newman, A. Status and challenges in enabling the lithium metal electrode for high-energy and low-cost rechargeable batteries. Nat. Energy 3, 16–21 (2017).
Yang, K. et al. Weak-interaction environment in a composite electrolyte enabling ultralong-cycling high-voltage solid-state lithium batteries. J. Am. Chem. Soc. 16, 11371–11381 (2024).
Wan, H. et al. Interface design for high-performance all-solid-state lithium batteries. Adv. Energy Mater. 14, 2303046 (2023).
Xu, R. et al. Artificial soft–rigid protective layer for dendrite-free lithium metal anode. Adv. Funct. Mater. 28, 1705838 (2018).
Vitos, L., Korzhavyi, P. A. & Johansson, B. Elastic property maps of austenitic stainless steels. Phys. Rev. Lett. 88, 155501 (2002).
Pugh, S. F. Relations between the elastic moduli and the plastic properties of polycrystalline pure metals. Lond. Edinb. Dubl. Philos. Mag. 45, 823–843 (1954).
Jin, S. et al. Solid–solution-based metal alloy phase for highly reversible lithium metal anode. J. Am. Chem. Soc. 142, 8818–8826 (2020).
Zhang, S. et al. Phase diagram determined lithium plating/stripping behaviors on lithiophilic substrates. ACS Energy Lett. 6, 4118–4126 (2021).
Pecharromán, C. & Moya, J. S. Experimental evidence of a giant capacitance in insulator–conductor composites at the percolation threshold. Adv. Mater. 12, 294–297 (2000).
Qi, L., Lee, B. I., Chen, S., Samuels, W. D. & Exarhos, G. J. High-dielectric-constant silver–epoxy composites as embedded dielectrics. Adv. Mater. 17, 1777–1781 (2005).
Krylova, V. & Dukštienė, N. Synthesis and characterization of Ag2S layers formed on polypropylene. J. Chem. 2013, 987879 (2013).
Wolan, J. T. & Hoflund, G. B. Surface characterization study of AgF and AgF2 powders using XPS and ISS. Appl. Surf. Sci. 125, 251–258 (1998).
Shi, X. et al. Room-temperature ductile inorganic semiconductor. Nat. Mater. 17, 421–426 (2018).
Guo, Z. et al. Combining solid solution strengthening and second phase strengthening for thinning Li metal foils. ACS Nano 17, 14136–14143 (2023).
Zhang, S. et al. The lasting impact of formation cycling on the Li-ion kinetics between SEI and the Li-metal anode and its correlation with efficiency. Sci. Adv. 10, eadj8889 (2024).
Huang, H. et al. Bonded interface enabled durable solid-state lithium metal batteries with ultra-low interfacial resistance of 0.25 Ω cm2. Adv. Funct. Mater. 34, 2407619 (2024).
Zhang, X. et al. Self-suppression of lithium dendrite in all-solid-state lithium metal batteries with poly(vinylidene difluoride)-based solid electrolytes. Adv. Mater. 31, 1806082 (2019).
Deng, T. et al. In situ formation of polymer-inorganic solid-electrolyte interphase for stable polymeric solid-state lithium-metal batteries. Chem 7, 3052–3068 (2021).
Hu, C. et al. Superionic conductors via bulk interfacial conduction. J. Am. Chem. Soc. 142, 18035–18041 (2020).
Ma, Y. et al. Scalable, ultrathin, and high-temperature-resistant solid polymer electrolytes for energy-dense lithium metal batteries. Adv. Energy Mater. 12, 2103720 (2022).
Wang, Z. et al. Lithium anode interlayer design for all-solid-state lithium-metal batteries. Nat. Energy 9, 251–262 (2024).
Han, X. et al. Negating interfacial impedance in garnet-based solid-state Li metal batteries. Nat. Mater. 16, 572–579 (2016).
Chen, B. et al. Achieving the high capacity and high stability of Li-rich oxide cathode in garnet-based solid-state battery. Angew. Chem. Int. Ed. 63, e202315856 (2023).
Huo, H. et al. A flexible electron-blocking interfacial shield for dendrite-free solid lithium metal batteries. Nat. Commun. 12, 176 (2021).
Ni, Y., Huang, C., Liu, H., Liang, Y. & Fan, L. Z. A high air-stability and Li-metal-compatible Li3+2xP1−xBixS4−1.5xO1.5x sulfide electrolyte for all-solid-state Li-metal batteries. Adv. Funct. Mater. 32, 2205998 (2022).
Zeng, D. et al. Promoting favorable interfacial properties in lithium-based batteries using chlorine-rich sulfide inorganic solid-state electrolytes. Nat. Commun. 13, 1909 (2022).
Ye, L. & Li, X. A dynamic stability design strategy for lithium metal solid state batteries. Nature 593, 218–222 (2021).
Fan, X. et al. Fluorinated solid electrolyte interphase enables highly reversible solid-state Li metal battery. Sci. Adv. 4, 2375–7548 (2018).
Wang, C. et al. A universal wet-chemistry synthesis of solid-state halide electrolytes for all-solid-state lithium-metal batteries. Sci. Adv. 7, eabh1896 (2021).
Li, S. et al. Manipulation of charge transfer in vertically aligned epitaxial ferroelectric KNbO3 nanowire array photoelectrodes. Nano Energy 35, 92–100 (2017).
Yao, Y. X. et al. Regulating interfacial chemistry in lithium-ion batteries by a weakly solvating electrolyte. Angew. Chem. Int. Ed. 60, 4090–4097 (2020).
Pecharromán, C., Esteban-Betegón, F., Bartolomé, J. F., López-Esteban, S. & Moya, J. S. New percolative BaTiO3–Ni composites with a high and frequency-independent dielectric constant (εr≈80000). Adv. Mater. 13, 1541–1544 (2001).
Ding, J. F. et al. Non-solvating and low-dielectricity cosolvent for anion-derived solid electrolyte interphases in lithium metal batteries. Angew. Chem. Int. Ed. 60, 11442–11447 (2021).
Shi, P. et al. A dielectric electrolyte composite with high lithium-ion conductivity for high-voltage solid-state lithium metal batteries. Nat. Nanotechnol. 18, 602–610 (2023).
Medlin, D. L., Yang, N., Spataru, C. D., Hale, L. M. & Mishin, Y. Unraveling the dislocation core structure at a van der Waals gap in bismuth telluride. Nat. Commun. 10, 1820 (2019).
Xie, Y., Shibata, K. & Mizoguchi, T. A brute-force code searching for cell of non-identical displacement for CSL grain boundaries and interfaces. Comput. Phys. Commun. 273, 108260 (2022).
Xie, Y. et al. InterOptimus: an AI-assisted robust workflow for screening ground-state heterogeneous interface structures in lithium batteries. J. Energy Chem. 106, 631–641 (2025).
Acknowledgements
This work was supported by the National Science Fund for Distinguished Young Scholars (number 52325206), the National Key Research and Development Program of China (number 2021YFF0500600), the National Natural Science Foundation of China (numbers U2001220, 52203298 and 92470110), the Shenzhen Outstanding Talents Training Fund (number RCJC20200714114436091), the Shenzhen Technical Plan Project (numbers JCYJ20220530143012027, JCYJ20220818101003008, JCYJ20241202124002004 and JCYJ20220818101003007), the Shenzhen Pengrui Young Faculty Program Research Plan (number SZPR2023006), the Shenzhen Stable Support Program for Higher Education Institutions (number WDZC20231126215806001), and the Shenzhen All-Solid-State Lithium Battery Electrolyte Engineering Research Centre Upgrade Project (number XMHT20240108008). We thank the Testing Technology Center of Materials and Devices, Tsinghua Shenzhen International Graduate School (https://mdtc.sz.tsinghua.edu.cn) for the atomic force microscopy, Raman and XPS measurements.
Author information
Authors and Affiliations
Contributions
Y.-B.H., J. Mi, Q.-H.Y. and F.K. conceived of the idea. Y.-B.H., T.H., W.L., Q.-H.Y. and F.K. supervised the project. Y.-B.H., J. Mi, L.C, W.L., T.H. and F.K. designed the experiments. J. Mi performed the experiments with help from L.C., X.A., J. Ma, K.Y., J.B., Y. Long, H.G., G.X., D.Z., X.C. and Y.H. W.C., L.G., R.K. and B.H. performed the cryo-TEM experiments. J.Y., S.T., T.H. and Y.X. performed the theoretical calculations. Y. Li and M.L. performed the NMR measurements. All authors discussed the results in the paper. J. Mi, Y.-B.H., T.H., W.L., L.C., Q.-H.Y. and F.K. wrote and revised the initial paper, which was approved by all authors.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature thanks George Alexander, Fugen Sun, Zhaoyin Wen and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Mi, J., Yang, J., Chen, L. et al. A ductile solid electrolyte interphase for solid-state batteries. Nature 647, 86–92 (2025). https://doi.org/10.1038/s41586-025-09675-8
Received:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41586-025-09675-8
This article is cited by
-
Functionally gradient ductile solid electrolyte interphase
Science China Materials (2025)