Abstract
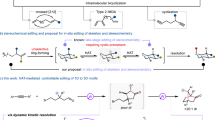
Non-aromatic heterocycles and carbocycles form the skeleton of countless bioactive and functional molecules1,2. Of note, four-membered saturated cyclic molecules such as azetidines, thietanes and cyclobutanes have garnered increasing attention in medicinal chemistry3,4,5,6,7. These molecules often have physicochemical properties relevant to drug discovery: potency, stability, metabolic stability and target specificity3. The replacement of oxygen atoms in readily available oxetanes would offer a direct route to a variety of these cyclic pharmacophores, yet such atom swapping has been rarely reported for non-aromatic molecules. Here we report a general photocatalytic strategy that selectively substitutes the oxygen atom of an oxetane with a nitrogen-based, sulfur-based or carbon-based moiety, transforming it into a diverse range of saturated cyclic building blocks in a single operation. This atom-swapping method exhibits high functional group compatibility and is applicable to late-stage functionalization, substantially simplifying the synthesis of pharmaceuticals and complex drug analogues that would otherwise require multistep routes. Mechanistic investigations unveil insights on the origin of chemoselectivity that allows the endocyclic oxygen atom to react preferentially to generate an acyclic dihalide intermediate, which then undergoes efficient ring reconstruction in the presence of a nucleophilic species.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$32.99 / 30 days
cancel any time
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
Data availability
Crystallographic data are available free of charge from the Cambridge Crystallographic Data Centre under reference nos. CCDC-2446264 (23), CCDC-2446104 (49), CCDC-2446091 (51), CCDC-2446094 (61), CCDC-2446107 (66) and CCDC-2446108 (67). All other data are available in the main text or the Supplementary information.
References
Taylor, R. D., MacCoss, M. & Lawson, A. D. G. Rings in drugs. J. Med. Chem. 57, 5845–5859 (2014).
Marson, C. M. Saturated heterocycles with applications in medicinal chemistry. Adv. Heterocycl. Chem. 121, 13–33 (2017).
Bauer, M. R. et al. Put a ring on it: application of small aliphatic rings in medicinal chemistry. RSC Med. Chem. 12, 448–471 (2021).
Parmar, D. R. et al. Azetidines of pharmacological interest. Arch. Pharm. 354, e2100062 (2021).
Francisco, K. R. & Ballatore, C. Thietanes and derivatives thereof in medicinal chemistry. Curr. Top. Med. Chem. 22, 1219–1234 (2022).
van der Kolk, M. R., Janssen, M. A. C. H., Rutjes, F. P. J. T. & Blanco-Ania, D. Cyclobutanes in small-molecule drug candidates. ChemMedChem 17, e202200020 (2022).
Rojas, J. J. & Bull, J. A. Oxetanes in drug discovery campaigns. J. Med. Chem. 66, 12697–12709 (2023).
Malarney, K. P., KC, S. & Schmidt, V. A. Recent strategies used in the synthesis of saturated four-membered heterocycles. Org. Biomol. Chem. 19, 8425–8441 (2021).
Villar, H., Frings, M. & Bolm, C. Ring closing enyne metathesis: a powerful tool for the synthesis of heterocycles. Chem. Soc. Rev. 36, 55–66 (2007).
Li, C.-L. & Liu, R.-S. Synthesis of heterocyclic and carbocyclic compounds via alkynyl, allyl, and propargyl organometallics of cyclopentadienyl iron, molybdenum, and tungsten complexes. Chem. Rev. 100, 3127–3161 (2000).
Spatola, E., Frateloreto, F., Giudice, D. D., Olivo, G. & Stefano, S. D. Cyclization reactions in confined space. Curr. Opin. Colloid Interface Sci. 64, 101680 (2023).
Tang, K., Wang, S., Gao, W., Song, Y. & Yu, B. Harnessing the cyclization strategy for new drug discovery. Acta Pharm. Sin. B 12, 4309–4326 (2022).
Lou, Y. Sulfonamido derivatives as cyclin-dependent kinase 2 inhibitors. US patent US20230303509 A1 (2023).
Jurczyk, J. et al. Single-atom logic for heterocycle editing. Nat. Synth. 1, 352–364 (2022).
Paschke, A.-S. et al. Carbon-to-nitrogen atom swap enables direct access to benzimidazoles from drug-like indoles. Nat. Chem. 17, 1750–1756 (2025).
Wu, F.-P. et al. Nitrogen-to-functionalized carbon atom transmutation of pyridine. Chem. Sci. 15, 15205–15211 (2024).
Kim, D. et al. Photocatalytic furan-to-pyrrole conversion. Science 386, 99–105 (2024).
Woo, J., Stein, C., Christian, A. H. & Levin, M. D. Carbon-to-nitrogen single-atom transmutation of azaarenes. Nature 623, 77–82 (2023).
Lyu, H., Kevlishvili, I., Yu, X., Liu, P. & Dong, G. Boron insertion into alkyl ether bonds via zinc/nickel tandem catalysis. Science 372, 175–182 (2021).
Nogi, K. & Yorimitsu, H. Aromatic metamorphosis: conversion of an aromatic skeleton into a different ring system. Chem. Commun. 53, 4055–4065 (2017).
Bhanuchandra, M., Murakami, K., Vasu, D., Yorimitsu, H. & Osuka, A. Transition-metal-free synthesis of carbazoles and indoles by an SNAr-based “aromatic metamorphosis” of thiaarenes. Angew. Chem. Int. Ed. 54, 10234–10238 (2015).
Chan-Penebre, E. et al. A selective inhibitor of PRMT 5 with in vivo and in vitro potency in MCL models. Nat. Chem. Biol. 11, 432–437 (2015).
Shen, Y. et al. Discovery of first-in-class protein arginine methyltransferase 5 (PRMT5) degraders. J. Med. Chem. 63, 9977–9989 (2020).
Gadekar, P. K. et al. Design, synthesis and biological evaluation of novel azaspiro analogs of linezolid as antibacterial and antitubercular agents. Eur. J. Med. Chem. 122, 475–487 (2016).
Ahmad, S. et al. Synthesis and antiobesity properties of 6-(4-chlorophenyl)-3-(4-((3,3-difluoro-1-hydroxycyclobutyl)methoxy)-3-methoxyphenyl)thieno[3,2-d]pyrimidin-4(3H)-one (BMS-814580): a highly efficacious melanin concentrating hormone receptor 1 (MCHR1) inhibitor. J. Med. Chem. 59, 8848–8858 (2016).
Liu, F. et al. Discovery of an in vivo chemical probe of the lysine methyltransferases G9a and GLP. J. Med. Chem. 56, 8931–8942 (2013).
Dai, C., Narayanam, J. M. R. & Stephenson, C. R. J. Visible-light-mediated conversion of alcohols to halides. Nat. Chem. 3, 140–145 (2011).
Benazza, M., Uzan, R., Beaupère, D. & Demailly, G. Direct regioselective chlorination of unprotected hexitols and pentitols by Vilsmeier and Haack’s salt. Tetrahedron Lett. 33, 4901–4904 (1992).
Penczek, S. & Kubisa, P. in Comprehensive Polymer Science and Supplements (eds Allen, G. & Bevington, J. C.) 751–786 (Pergamon, 1989).
Sander, M. Thietanes. Chem. Rev. 66, 341–353 (1966).
Liang, Y. & Demarest, K. T. GPR40 agonists in anti-diabetic drug combinations. US patent US20170290800 A1 (2017).
Davis, L. O. Recent developments in the synthesis and applications of pyrazolidines. A review. Org. Prep. Proced. Int. 45, 437–464 (2013).
Berthet, M., Cheviet, T., Dujardin, G., Parrot, I. & Martinez, J. Isoxazolidine: a privileged scaffold for organic and medicinal chemistry. Chem. Rev. 116, 15235–15283 (2016).
Felding, J., Nielsen, S. F., Larsen, J. C. H. & Babu, B. R. Novel phosphodiesterase inhibitors. International patent WO2008104175 A2 (2008).
Last, S. J., Raboisson, P. J.-M. B., Rombouts, G., Vandyck, K. & Verschueren, W. G. Sulfamoyl-arylamides and the use thereof as medicaments for the treatment of hepatitis B. International patent WO2014033176 A1 (2014).
Malashchuk, A., Chernykh, A. V., Dobrydnev, A. V. & Grygorenko, O. O. Fluorine-labelled spiro[3.3]heptane-derived building blocks: is single fluorine the best? Eur. J. Org. Chem. 2021, 4897–4910 (2021).
Olifir, O. S. et al. Multigram synthesis of advanced 6,6-difluorospiro[3.3]heptane-derived building blocks. Eur. J. Org. Chem. 2021, 6541–6550 (2021).
Miao, L. et al. Discovery of new difluorocyclobutyl derivatives as effective glucagon-like peptide-1 receptor agonists with reduced hERG inhibitory activities. J. Med. Chem. 68, 7662–7692 (2025).
Lu, G. et al. Novel aza-oxo-indoles for the treatment and prophylaxis of respiratory syncytial virus infection. International patent WO2015022263 A1 (2015).
Acknowledgements
This research was supported by the U.S. Air Force Office of Scientific Research: FA2386-25-1-4031 (M.J.K.), the Ministry of Education of Singapore Academic Research Fund Tier 1: A-8001693-00-00 (M.J.K.), National University of Singapore Foresight Grant: A-8002845-00-00, A-8002845-01-00, A-8002845-02-00 (M.J.K.), National Research Foundation, Prime Minister’s Office, Singapore under the NRF Investigatorship Programme: NRF-NRFI10-2024-0009 (M.J.K.), Novartis Early Career Award in Chemistry Unrestricted Grant: E-143-00-0072-01 (M.J.K.), the Chinese University of Hong Kong (CUHK) Vice-Chancellor Early Career Professorship Scheme Research Startup Fund: 4933634 (X.Z.) and Research Startup Matching Support: 5501779 (X.Z.). I. I. Roslan assisted with X-ray crystallographic measurements.
Author information
Authors and Affiliations
Contributions
M.J.K. and Y.-Q.Z. conceived the work. Y.-Q.Z. and S.-H.L. conducted the optimization, reaction scope and mechanistic studies. X.Z. designed and performed the DFT studies. M.J.K. directed the research and wrote the manuscript, with revisions provided by the other authors.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature thanks the anonymous reviewers for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data figures and tables
Extended Data Fig. 1 DFT-computed energetics of key reaction barriers and intermediates.
Values in parentheses indicate relative free energies in kcal mol−1 referenced to intermediate I.
Extended Data Fig. 2 Further studies in O-to-N swap with complex amines.
L-Phenylalanine methyl ester, mexiletine and oseltamivir underwent reaction with 8 to give the corresponding azetidine products. Ac, acetyl.
Extended Data Fig. 3 Previously reported synthetic routes to advanced drug intermediates.
In past protocols, extensive functional group interconversions, redox manipulations and protecting group strategies were necessary to access medicinally relevant building blocks 79 and 2. Ms, mesyl.
Supplementary information
Supplementary Information (download PDF )
This file contains Supplementary Information.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Zhang, YQ., Li, SH., Zhang, X. et al. Photocatalytic oxygen-atom transmutation of oxetanes. Nature 647, 906–912 (2025). https://doi.org/10.1038/s41586-025-09723-3
Received:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41586-025-09723-3