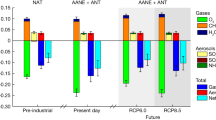
Abstract
Observations have demonstrated the ubiquity of short-lived halogens (SLHs)—defined as organic and inorganic chlorine, bromine and iodine compounds with an overall atmospheric lifetime of less than 6 months—in the global atmosphere. They are primarily emitted naturally from the ocean, cryosphere, volcanoes, salt lakes and the biosphere. However, unregulated anthropogenic sources are increasingly contributing to their atmospheric loading. Some of their natural emissions have increased over time due to anthropogenic pollution, for example, the increased oceanic emissions of iodine compounds due to the deposition of ozone on the sea surface. SLHs affect chemical processes, such as ozone and methane chemistry, and therefore influence air quality and climate. Nevertheless, some of their sources and chemistry are not included in air-quality and climate models used in international assessment reports. Here we describe in detail the various impacts of SLHs on air quality and climate, and make a case for the inclusion of more comprehensive SLH chemistry in future atmospheric, air-quality and climate assessments. In doing so, we also identify gaps in our knowledge of SLH emissions, chemistry, and environmental and climate impacts.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$32.99 / 30 days
cancel any time
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
Code availability
No specific code was used for this study.
References
Molina, M. J. & Rowland, F. S. Stratospheric sink for chlorofluoromethanes—chlorine atomic-catalysed destruction of ozone. Nature 249, 810–812 (1974).
Wofsy, C., Mcelroy, M. B. & Yung, Y. L. The chemistry of atmospheric bromine. Geophys. Res. Lett. 2, 215–218 (1975).
McElroy, M. B., Salawitch, R. J., Wofsy, S. C. & Logan, J. A. Reductions of Antarctic ozone due to synergistic interactions of chlorine and bromine. Nature 321, 759–762 (1986). Antarctic stratospheric ozone loss was attributed to synergistic chemical reactions between chlorine and bromine compounds.
Scientific Assessment of Ozone Depletion: 2022 (WMO, 2022); library.wmo.int/idurl/4/58360.
Yung, Y. L., Mcelroy, M. B. & Wofsy, S. C. Atmospheric halocarbons: a discussion with emphasis on chloroform. Geophys. Res. Lett. 2, 397–399 (1975).
Dorf, M. et al. Bromine in the tropical troposphere and stratosphere as derived from balloon-borne BrO observations. Atmos. Chem. Phys. 8, 7265–7271 (2008).
Salawitch, R. J. et al. A new interpretation of total column BrO during Arctic spring. Geophys. Res. Lett. 37, L21805 (2010).
Koenig, T. K. et al. Quantitative detection of iodine in the stratosphere. Proc. Natl Acad. Sci. USA https://doi.org/10.1073/pnas.1916828117 (2020). First direct observations of reactive iodine compounds in the stratosphere.
Textor, C., Graf, H. F., Herzog, M. & Oberhuber, J. M. Injection of gases into the stratosphere by explosive volcanic eruptions. J. Geophys. Res. Atmos. 108, 4606 (2003).
Sturges, W. T., Oram, D. E., Carpenter, L. J., Penkett, S. A. & Engel, A. Bromoform as a source of stratospheric bromine. Geophys. Res. Lett. 27, 2081–2084 (2000). The study showed that bromine-containing SLHs can contribute to stratospheric ozone depletion.
Saiz-Lopez, A. et al. Injection of iodine to the stratosphere. Geophys. Res. Lett. 42, 6852–6859 (2015).
Fernandez, R. P., Salawitch, R. J., Kinnison, D. E., Lamarque, J.-F. & Saiz-Lopez, A. Bromine partitioning in the tropical tropopause layer: implications for stratospheric injection. Atmos. Chem. Phys. 14, 13391–13410 (2014).
Salawitch, R. J. Sensitivity of ozone to bromine in the lower stratosphere. Geophys. Res. Lett. 32, L05811 (2005).
Kovalenko, L. J. et al. Validation of aura microwave limb sounder BrO observations in the stratosphere. J. Geophys. Res. Atmos. 112, D24S41 (2007).
Fu, X. et al. Anthropogenic short-lived halogens increase human exposure to mercury contamination due to enhanced mercury oxidation over continents. Proc. Natl Acad. Sci. USA 121, e2315058121 (2024).
Sinnhuber, B.-M. Global observations of stratospheric bromine monoxide from SCIAMACHY. Geophys. Res. Lett. 32, L20810 (2005).
Solomon, S., Garcia, R. R. & Ravishankara, A. R. On the role of iodine in ozone depletion. J. Geophys. Res. 99, 20491 (1994). This work hypothesised the importance of iodine-containing compounds in the depletion of stratospheric ozone.
Butz, A. et al. Constraints on inorganic gaseous iodine in the tropical upper troposphere and stratosphere inferred from balloon-borne solar occultation observations. Atmos. Chem. Phys. 9, 7229–7242 (2009).
Pundt, I., Pommereau, J. P. J.-P. P., Phillips, C. & Lateltin, E. Upper limit of iodine oxide in the lower stratosphere. J. Atmos. Chem. 30, 173–185 (1998).
Sinnhuber, B. M., Sheode, N., Sinnhuber, M., Chipperfield, M. P. & Feng, W. The contribution of anthropogenic bromine emissions to past stratospheric ozone trends: a modelling study. Atmos. Chem. Phys. 9, 2863–2871 (2009).
Cuevas, C. A. et al. The influence of iodine on the Antarctic stratospheric ozone hole. Proc. Natl Acad. Sci. USA 119, e2110864119 (2022).
Fernandez, R. P., Kinnison, D. E., Lamarque, J. F., Tilmes, S. & Saiz-Lopez, A. Impact of biogenic very short-lived bromine on the Antarctic ozone hole during the 21st century. Atmos. Chem. Phys. 17, 1673–1688 (2017).
Oman, L. D. et al. The effect of representing bromine from VSLS on the simulation and evolution of Antarctic ozone. Geophys. Res. Lett. 43, 9869–9876 (2016).
Villamayor, J. et al. Very short-lived halogens amplify ozone depletion trends in the tropical lower stratosphere. Nat. Clim. Change 13, 554–560 (2023).
Bureau, H. et al. Modern and past volcanic degassing of iodine. Geochim. Cosmochim. Acta 173, 114–125 (2016).
Østerstrøm, F. F., Klobas, J. E., Kennedy, R. P., Cadoux, A. & Wilmouth, D. M. Sensitivity of stratospheric ozone to the latitude, season, and halogen content of a contemporary explosive volcanic eruption. Sci. Rep. 13, 6457 (2023).
Tilmes, S. et al. Impact of very short-lived halogens on stratospheric ozone abundance and UV radiation in a geo-engineered atmosphere. Atmos. Chem. Phys. 12, 10945–10955 (2012).
Barrie, L. A., Bottenheim, J. W., Schnell, R. C., Crutzen, P. J. & Rasmussen, R. A. Ozone destruction and photochemical reactions at polar sunrise in the lower Arctic atmosphere. Nature 334, 138–141 (1988). First ODEs observed in the lower troposphere, bromine-containing SLHs were identified as the main driver of this destruction.
Finlayson-Pitts, B. J., Livingston, F. E. & Berko, H. N. Ozone destruction and bromine photochemistry at ground-level in the arctic spring. Nature 343, 622–625 (1990).
Fan, S. M. & Jacob, D. J. Surface ozone depletion in arctic spring sustained by bromine reactions on aerosols. Nature 359, 522–524 (1992).
McConnell, J. C. et al. Photochemical bromine production implicated in arctic boundary layer ozone depletion. Nature 355, 150–152 (1992).
Kreher, K., Johnston, P. V., Wood, S. W., Nardi, B. & Platt, U. Ground-based measurements of tropospheric and stratospheric BrO at Arrival Heights, Antarctica. Geophys. Res. Lett. 24, 3021–3024 (1997).
Wagner, T. & Platt, U. Satellite mapping of enhanced BrO concentrations in the troposphere. Nature 395, 486–490 (1998). Satellite-based observations of bromine-containing SLHs reported in this study have enabled an improved understanding of their geographical distribution.
Saiz-Lopez, A. & von Glasow, R. Reactive halogen chemistry in the troposphere. Chem. Soc. Rev. https://doi.org/10.1039/c2cs35208g (2012).
von Glasow, R. et al. Impact of reactive bromine chemistry in the troposphere. Atmos. Chem. Phys. 4, 2481–2497 (2004).
Yang, M. et al. Tropospheric bromine chemistry and its impacts on ozone: a model study. J. Geophys. Res. Atmos. 110, D23311 (2005).
Wang, S. et al. Direct detection of atmospheric atomic bromine leading to mercury and ozone depletion. Proc. Natl Acad. Sci. USA 116, 14479–14484 (2019).
Pratt, K. A. et al. Photochemical production of molecular bromine in Arctic surface snowpacks. Nat. Geosci. 6, 351–356 (2013).
Simpson, W. R., Brown, S. S., Saiz-Lopez, A., Thornton, J. A. & Von Glasow, R. Tropospheric halogen chemistry: sources, cycling, and impacts. Chem. Rev. https://doi.org/10.1021/cr5006638 (2015).
Richter, A., Wittrock, F., Eisinger, M. & Burrows, J. P. GOME observations of tropospheric BrO in northern hemispheric spring and summer 1997. Geophys. Res. Lett. 25, 2683–2686 (1998).
Chance, K. Analysis of BrO measurements from the global ozone monitoring experiment. Geophys. Res. Lett. 25, 3335–3338 (1998).
Saiz-Lopez, A. et al. Boundary layer halogens in coastal Antarctica. Science 317, 348–351 (2007). This study first showed that iodine-containing SLHs can contribute to ozone destruction in the polar regions and enhance the effect of bromine compounds.
Benavent, N. et al. Substantial contribution of iodine to Arctic ozone destruction. Nat. Geosci. 15, 770–773 (2022).
Mahajan, A. S. et al. Evidence of reactive iodine chemistry in the Arctic boundary layer. J. Geophys. Res. 115, D20 (2010).
Raso, A. R. W. et al. Active molecular iodine photochemistry in the Arctic. Proc. Natl Acad. Sci. USA 114, 10053–10058 (2017).
Schönhardt, A. et al. Observations of iodine monoxide columns from satellite. Atmos. Chem. Phys. 8, 637–653 (2008).
Saiz-Lopez, A., Chance, K. V., Liu, X., Kurosu, T. P. & Sander, S. P. First observations of iodine oxide from space. Geophys. Res. Lett. 34, L12812 (2007).
Mahajan, A. S. et al. Differences in iodine chemistry over the Antarctic continent. Polar Sci. https://doi.org/10.1016/j.polar.2023.101014 (2023).
Saiz-Lopez, A. & Blaszczak-Boxe, C. S. The polar iodine paradox. Atmos. Environ. 145, 72–73 (2016).
Baccarini, A. et al. Frequent new particle formation over the high Arctic pack ice by enhanced iodine emissions. Nat. Commun. 11, 4924 (2020).
Sipilä, M. et al. Molecular-scale evidence of aerosol particle formation via sequential addition of HIO3. Nature 537, 532–534 (2016).
Liao, J. et al. High levels of molecular chlorine in the Arctic atmosphere. Nat. Geosci. 7, 91–94 (2014).
Abbatt, J. P. D. et al. Halogen activation via interactions with environmental ice and snow in the polar lower troposphere and other regions. Atmos. Chem. Phys. 12, 6237–6271 (2012).
Buys, Z. et al. High temporal resolution Br2, BrCl and BrO observations in coastal Antarctica. Atmos. Chem. Phys. 13, 1329–1343 (2013).
Foster, K. L. et al. The Role of Br2 and BrCl in surface ozone destruction at Polar sunrise. Science 291, 471–474 (2001).
Roberts, J. M. et al. Observations of cyanogen bromide (BrCN) in the global troposphere and their relation to polar surface O3 destruction. Atmos. Chem. Phys. 24, 3421–3443 (2024).
Fernandez, R. P. et al. Arctic halogens reduce ozone in the northern mid-latitudes. Proc. Natl Acad. Sci. USA 121, e2401975121 (2024).
Chameides, W. L. & Davis, D. D. Iodine: its possible role in tropospheric photochemistry. J. Geophys. Res. 85, 7383–7398 (1980). This was the first study to show that iodine-containing SLHs can be emitted directly from the ocean surface, suggesting a global source.
Garland, J. A. & Curtis, H. Emission of iodine from the sea surface in the presence of ozone. J. Geophys. Res. 86, 3183–3186 (1981).
Read, K. A. et al. Extensive halogen-mediated ozone destruction over the tropical Atlantic Ocean. Nature 453, 1232–1235 (2008). Observations of iodine- and bromine-containing SLHs showed that SLH are the most important sink of ozone in the remote marine boundary layer after photolysis.
Mahajan, A. S. et al. Measurement and modelling of tropospheric reactive halogen species over the tropical Atlantic Ocean. Atmos. Chem. Phys. 10, 4611–4624 (2010).
Mahajan, A. S. et al. Latitudinal distribution of reactive iodine in the Eastern Pacific and its link to open ocean sources. Atmos. Chem. Phys. 12, 11609–11617 (2012).
Prados-Roman, C. et al. Iodine oxide in the global marine boundary layer. Atmos. Chem. Phys. 15, 583–593 (2015).
Saiz-Lopez, A. et al. Estimating the climate significance of halogen-driven ozone loss in the tropical marine troposphere. Atmos. Chem. Phys. 12, 3939–3949 (2012).
Dix, B. et al. Detection of iodine monoxide in the tropical free troposphere. Proc. Natl Acad. Sci. USA 110, 2035–2040 (2013).
Großmann, K. et al. Iodine monoxide in the Western Pacific marine boundary layer. Atmos. Chem. Phys. 13, 3363–3378 (2013).
Leser, H., Honninger, G. & Platt, U. MAX-DOAS measurements of BrO and NO2 in the marine boundary layer. Geophys. Res. Lett. 30, 1537 (2003).
Mahajan, A. S. et al. Understanding iodine chemistry over the northern and equatorial Indian Ocean. J. Geophys. Res. Atmos. https://doi.org/10.1029/2018JD029063 (2019).
van Herpen, M. M. J. W. et al. Photocatalytic chlorine atom production on mineral dust–sea spray aerosols over the North Atlantic. Proc. Natl Acad. Sci. USA 120, e2303974120 (2023). This study showed that heterogeneous reactions on dust and sea spray are a major source of chlorine-containing SLHs.
Salawitch, R. J. Biogenic bromine. Nature 439, 275–277 (2006). This study highlighted that biogenic sources of bromine also contribute to tropospheric ozone reduction and may therefore be linked to climate change.
Badia, A. et al. Importance of reactive halogens in the tropical marine atmosphere: A regional modelling study using WRF-Chem. Atmos. Chem. Phys. 19, 3161–3189 (2019).
Mahajan, A. S. et al. Modelling the impacts of iodine chemistry on the Northern Indian Ocean marine boundary layer. Atmos. Chem. Phys. https://doi.org/10.5194/acp-2020-1219 (2021).
Li, Q. et al. Impact of halogen chemistry on summertime air quality in coastal and continental Europe: application of the CMAQ model and implications for regulation. Atmos. Chem. Phys. 19, 15321–15337 (2019).
Sherwen, T. et al. Global impacts of tropospheric halogens (Cl, Br, I) on oxidants and composition in GEOS-Chem. Atmos. Chem. Phys. 16, 12239–12271 (2016).
Ordóñez, C. et al. Bromine and iodine chemistry in a global chemistry-climate model: description and evaluation of very short-lived oceanic sources. Atmos. Chem. Phys. 12, 1423–1447 (2012).
Sherwen, T. et al. Iodine’s impact on tropospheric oxidants: a global model study in GEOS-Chem. Atmos. Chem. Phys. 16, 1161–1186 (2016).
Hossaini, R. et al. A global model of tropospheric chlorine chemistry: organic versus inorganic sources and impact on methane oxidation. J. Geophys. Res. 121, 14271–14297 (2016).
Iglesias-Suarez, F. et al. Natural halogens buffer tropospheric ozone in a changing climate. Nat. Clim. Change 10, 147–154 (2020).
Saiz-Lopez, A. et al. Iodine chemistry in the troposphere and its effect on ozone. Atmos. Chem. Phys. 14, 13119–13143 (2014).
Fernandez, R. P. et al. Intercomparison between surrogate, explicit, and full treatments of vsl bromine chemistry within the CAM-Chem chemistry-climate model. Geophys. Res. Lett. 48, e2020GL091125 (2021).
Badia, A. et al. The role of natural halogens in global tropospheric ozone chemistry and budget under different 21st century climate scenarios. J. Geophys. Res. Atmos. 126, e2021JD034859 (2021).
Bossolasco, A. et al. Key role of short-lived halogens on global atmospheric oxidation during historical periods. Environ. Sci. Atmos. https://doi.org/10.1039/D4EA00141A (2025).
Li, Q. et al. Reactive halogens increase the global methane lifetime and radiative forcing in the 21st century. Nat. Commun. 13, 2768 (2022). This study showed that SLHs affect the lifetime of methane and therefore have an indirect effect on radiative forcing.
Saiz-Lopez, A. et al. Natural short-lived halogens exert an indirect cooling effect on climate. Nature 618, 967–973 (2023). This study showed that SLHs affect climate and that this effect is changing over time due to changes in emissions.
Finlayson-Pitts, B. J., Ezell, M. J. & Pitts, J. N. Formation of chemically active chlorine compounds by reactions of atmospheric NaCl particles with gaseous N2O5 and ClONO2. Nature 337, 241–244 (1989). This study showed that heterogeneous reactions in polluted environments can lead to the emission of SLHs from sea salt particles, suggesting a feedback between air quality and SLHs.
Schroeder, W. H. & Drone, P. Formation of nitrosyl chloride from salt particles in air. Environ. Sci. Technol. 8, 756–758 (1974).
Spicer, C. W. et al. Unexpectedly high concentrations of molecular chlorine in coastal air. Nature 394, 353–356 (1998). This study showed the presence of high levels of chlorine-containing SLHs in polluted environments.
Osthoff, H. D. et al. High levels of nitryl chloride in the polluted subtropical marine boundary layer. Nat. Geosci. 1, 324–328 (2008).
Alicke, B., Hebestreit, K., Stutz, J. & Platt, U. Iodine oxide in the marine boundary layer. Nature 397, 572–573 (1999).
O’Dowd, C. D. et al. Marine aerosol formation from biogenic iodine emissions. Nature 417, 632–636 (2002). This study showed that iodine-containing SLHs can lead to new particle formation, acting as a source of aerosols.
Saiz-Lopez, A. & Plane, J. M. C. Novel iodine chemistry in the marine boundary layer. Geophys. Res. Lett. 31, L04112 (2004).
McFiggans, G. B. Marine aerosols and iodine emissions. Nature 433, E13 (2005).
Thornton, J. A. et al. A large atomic chlorine source inferred from mid-continental reactive nitrogen chemistry. Nature 464, 271–274 (2010). This study showed that chlorine chemistry is highly active even in polluted continental environments far from marine sources.
Li, Q. et al. Halogens enhance haze pollution in China. Environ. Sci. Technol. 55, 13625–13637 (2021).
Wang, T. et al. Observations of nitryl chloride and modeling its source and effect on ozone in the planetary boundary layer of southern China. J. Geophys. Res. Atmos. 121, 2476–2489 (2016).
Brown, S. S. & Stutz, J. Nighttime radical observations and chemistry. Chem. Soc. Rev. 41, 6405–6447 (2012).
Saiz-Lopez, A. et al. Nighttime atmospheric chemistry of iodine. Atmos. Chem. Phys. 16, 15593–15604 (2016).
Hebestreit, K. et al. DOAS measurements of tropospheric bromine oxide in mid-latitudes. Science 283, 55–57 (1999).
Stutz, J., Ackermann, R., Fast, J. D. & Barrie, L. A. Atmospheric reactive chlorine and bromine at the Great Salt Lake, Utah. Geophys. Res. Lett. 29, 1380 (2002).
Zingler, J. & Platt, U. Iodine oxide in the Dead Sea Valley: evidence for inorganic sources of boundary layer IO. J. Geophys. Res. Atmos. 110, D07307 (2005).
Bobrowski, N., Hönninger, G. H., Galle, B. & Platt, U. Detection of bromine monoxide in a volcanic plume. Nature 423, 273–276 (2003).
Claxton, T. et al. A synthesis inversion to constrain global emissions of two very short lived chlorocarbons: dichloromethane, and perchloroethylene. J. Geophys. Res. Atmos. 125, e2019JD031818 (2020).
An, M. et al. Rapid increase in dichloromethane emissions from China inferred through atmospheric observations. Nat. Commun. 12, 7279 (2021). New emissions of chlorine-containing SLH were identified in this study, showing that anthropogenic activities are a direct source of SLH.
Fang, X. et al. Rapid increase in ozone-depleting chloroform emissions from China. Nat. Geosci. 12, 89–93 (2019).
Hossaini, R. et al. Growth in stratospheric chlorine from short-lived chemicals. Geophys. Res. Lett. 42, 4573–4580 (2015).
Hossaini, R. et al. The contribution of natural and anthropogenic very short-lived species to stratospheric bromine. Atmos. Chem. Phys. 12, 371–380 (2012).
Peng, X. et al. An unexpected large continental source of reactive bromine and chlorine with significant impact on wintertime air quality. Natl Sci. Rev. 8, nwaa304 (2021).
Tsai, C. et al. Nocturnal loss of NOx during the 2010 CalNex-LA study in the Los Angeles Basin. J. Geophys. Res. Atmos. 119, 13004–13025 (2014).
Sarwar, G., Simon, H., Xing, J. & Mathur, R. Importance of tropospheric ClNO2 chemistry across the Northern Hemisphere. Geophys. Res. Lett. 41, 4050–4058 (2014).
Bannan, T. J. et al. The first UK measurements of nitryl chloride using a chemical ionizationmass spectrometer in central London in the summer of 2012, and an investigation of the role of Cl atom oxidation. J. Geophys. Res. Atmos. 120, 5638–5657 (2015).
Baker, A. K. et al. Evidence for strong, widespread chlorine radical chemistry associated with pollution outflow from continental Asia. Sci. Rep. 6, 36821 (2016).
Riedel, T. P. et al. Chlorine activation within urban or power plant plumes: vertically resolved ClNO2 and Cl2 measurements from a tall tower in a polluted continental setting. J. Geophys. Res. https://doi.org/10.1002/jgrd.50637 (2013).
Gunthe, S. S. et al. Enhanced aerosol particle growth sustained by high continental chlorine emission in India. Nat. Geosci. https://doi.org/10.1038/s41561-020-00677-x (2021).
Li, Q. et al. Chemical interactions between ship-originated air pollutants and ocean-emitted halogens. J. Geophys. Res. Atmos. 126, e2020JD034175 (2021).
Womack, C. C. et al. Midlatitude ozone depletion and air quality impacts from industrial halogen emissions in the Great Salt Lake basin. Environ. Sci. Technol. 57, 1870–1881 (2023).
McNamara, S. M. et al. Observation of N2O5 deposition and ClNO2 production on the saline snowpack. ACS Earth Sp. Chem. 5, 1020–1031 (2021).
Young, C. J. et al. Chlorine as a primary radical: evaluation of methods to understand its role in initiation of oxidative cycles. Atmos. Chem. Phys. 14, 3427–3440 (2014).
Decker, Z. C. J. et al. Airborne observations constrain heterogeneous nitrogen and halogen chemistry on tropospheric and stratospheric biomass burning aerosol. Geophys. Res. Lett. 51, e2023GL107273 (2024).
Solomon, S. et al. Chlorine activation and enhanced ozone depletion induced by wildfire aerosol. Nature 615, 259–264 (2023). This study showed that the injection of organic aerosols from wildfires can lead to the activation of reactive chlorine species in the stratosphere.
Lobert, J. M., Keene, W. C., Logan, J. A. & Yevich, R. Global chlorine emissions from biomass burning: reactive chlorine emissions inventory. J. Geophys. Res. Atmos. 104, 8373–8389 (1999).
Peng, X. et al. Photodissociation of particulate nitrate as a source of daytime tropospheric Cl2. Nat. Commun. 13, 939 (2022).
Xia, M. et al. Pollution-derived Br2 boosts oxidation power of the coastal atmosphere. Environ. Sci. Technol. 56, 12055–12065 (2022).
Shah, V. et al. Improved mechanistic model of the atmospheric redox chemistry of mercury. Environ. Sci. Technol. https://doi.org/10.1021/acs.est.1c03160 (2021).
Schroeder, W. H. et al. Arctic springtime depletion of mercury. Nature 394, 331–332 (1998).
Saiz-Lopez, A. et al. The chemistry of mercury in the stratosphere. Geophys. Res. Lett. 49, e2022GL097953 (2022).
IPCC. Climate Change 2021: The Physical Science Basis. Contribution of Working Group I to the Sixth Assessment Report of the Intergovernmental Panel on Climate Change (eds Masson-Delmotte, V. et al.) (Cambridge Univ. Press, 2021).
Hossaini, R. et al. Efficiency of short-lived halogens at influencing climate through depletion of stratospheric ozone. Nat. Geosci. 8, 186–190 (2015).
Kok, J. F. et al. Mineral dust aerosol impacts on global climate and climate change. Nat. Rev. Earth Environ. 4, 71–86 (2023).
Thornhill, G. D., Smith, L. A. & Shine, K. P. Radiative forcing from halogen reservoir and halocarbon breakdown products. J. Geophys. Res. Atmos. 129, e2024JD040912 (2024).
Cuevas, C. A. et al. Rapid increase in atmospheric iodine levels in the North Atlantic since the mid-20th century. Nat. Commun. 9, 1452 (2018). First ice core iodine observations showed that atmospheric iodine levels have significantly increased since pre-industrial times.
Legrand, M. et al. Alpine ice evidence of a three-fold increase in atmospheric iodine deposition since 1950 in Europe due to increasing oceanic emissions. Proc. Natl Acad. Sci. USA 115, 12136–12141 (2018).
Zhao, X., Hou, X. & Zhou, W. Atmospheric iodine (127I and 129I) record in spruce tree rings in the northeast Qinghai-Tibet Plateau. Environ. Sci. Technol. 53, 8706–8714 (2019).
Carpenter, L. J. et al. Atmospheric iodine levels influenced by sea surface emissions of inorganic iodine. Nat. Geosci. 6, 108–111 (2013). This laboratory study provided a parameterisation for the emission of iodine-containing SLHs from the ocean surface, enabling modelling the global atmospheric impacts of these compounds.
MacDonald, S. M. et al. A laboratory characterisation of inorganic iodine emissions from the sea surface: dependence on oceanic variables and parameterisation for global modelling. Atmos. Chem. Phys. 14, 5841–5852 (2014).
Prados-Roman, C. et al. A negative feedback between anthropogenic ozone pollution and enhanced ocean emissions of iodine. Atmos. Chem. Phys. 15, 2215–2224 (2015).
Corella, J. P. et al. Climate changes modulated the history of Arctic iodine during the Last Glacial Cycle. Nat. Commun. 13, 6–14 (2022).
Vallelonga, P. et al. Sea-ice reconstructions from bromine and iodine in ice cores. Quat. Sci. Rev. 269, 107133 (2021).
Zhai, S. et al. Anthropogenic influence on tropospheric reactive bromine since the pre-industrial: implications for arctic ice-core bromine trends. Geophys. Res. Lett. 51, e2023GL107733 (2024).
Zhai, S. et al. Anthropogenic impacts on tropospheric reactive chlorine since the preindustrial. Geophys. Res. Lett. 48, e2021GL093808 (2021).
Segato, D. et al. Arctic mercury flux increased through the Last Glacial Termination with a warming climate. Nat. Geosci. 16, 439–445 (2023).
Pratt, K. A. Tropospheric halogen photochemistry in the rapidly changing Arctic. Trends Chem. 1, 545–548 (2019).
Mahajan, A. S. et al. High bromine oxide concentrations in the semi-polluted boundary layer. Atmos. Environ. 43, 3811–3818 (2009).
Salierno, G. On the chemical pathways influencing the effective global warming potential of commercial hydrofluoroolefin gases. ChemSusChem 17, e202400280, (2024).
Adcock, K. E. et al. Aircraft-based observations of ozone-depleting substances in the upper troposphere and lower stratosphere in and above the Asian summer monsoon. J. Geophys. Res. Atmos. 126, e2020JD033137 (2021).
Hossaini, R. et al. Recent trends in stratospheric chlorine from very short-lived substances. J. Geophys. Res. Atmos. 124, 2318–2335 (2019).
Wang, C. et al. Chloramines as an important photochemical source of chlorine atoms in the urban atmosphere. Proc. Natl Acad. Sci. USA 120, 2017 (2023).
Angelucci, A. A. et al. Elevated levels of chloramines and chlorine detected near an indoor sports complex. Environ. Sci. Process. Impacts 25, 304–313 (2023).
Feng, W., Plane, J. M. C., Chipperfield, M. P., Saiz-Lopez, A. & Booth, J. P. Potential stratospheric ozone depletion due to iodine injection from small satellites. Geophys. Res. Lett. 50, e2022GL102300 (2023).
McDuffie, E. E. et al. ClNO2 yields from aircraft measurements during the 2015 WINTER campaign and critical evaluation of the current parameterization. J. Geophys. Res. Atmos. 123, 12,994–13,015 (2018).
Jorga, S. D. et al. Kinetics of hypochlorous acid reactions with organic and chloride-containing tropospheric aerosol. Environ. Sci. Process. Impacts 25, 1645–1656 (2023).
Royer, H. M. et al. The role of hydrates, competing chemical constituents, and surface composition on ClNO2 formation. Environ. Sci. Technol. 55, 2869–2877 (2021).
Li, Q. et al. Global environmental implications of atmospheric methane removal through chlorine-mediated chemistry-climate interactions. Nat. Commun. 14, 4045 (2023).
Carpenter, L. J. et al. Marine iodine emissions in a changing world. Proc. R. Soc. London. A. https://doi.org/10.1098/rspa.2020.0824 (2021).
Brown, L. et al. Iodine speciation in snow during the MOSAiC Expedition and its implications for Arctic iodine emissions. Faraday Discuss. https://doi.org/10.1039/D4FD00178H (2024).
von Glasow, R., Bobrowski, N. & Kern, C. The effects of volcanic eruptions on atmospheric chemistry. Chem. Geol. 263, 131–142 (2009).
Saunders, R. W. et al. Studies of the formation and growth of aerosol from molecular iodine precursor. Z. Phys. Chem. 224, 1095–1117 (2010).
Finkenzeller, H. et al. The gas-phase formation mechanism of iodic acid as an atmospheric aerosol source. Nat. Chem. 15, 129–135 (2023).
He, X. et al. Role of iodine oxoacids in atmospheric aerosol nucleation. Science 371, 589–595 (2021).
Gómez Martín, J. C. et al. A gas-to-particle conversion mechanism helps to explain atmospheric particle formation through clustering of iodine oxides. Nat. Commun. 11, 4521 (2020).
Pickard, H. M. et al. Ice core record of persistent short-chain fluorinated alkyl acids: evidence of the impact from global environmental regulations. Geophys. Res. Lett. 47, e2020GL087535 (2020).
Tham, Y. J. et al. Widespread detection of chlorine oxyacids in the Arctic atmosphere. Nat. Commun. 14, 1769 (2023).
Höpfner, M., Orphal, J., Von Clarmann, T., Stiller, G. & Fischer, H. Stratospheric BrONO2 observed by MIPAS. Atmos. Chem. Phys. 9, 1735–1746 (2009).
Tham, Y. J. et al. Direct field evidence of autocatalytic iodine release from atmospheric aerosol. Proc. Natl Acad. Sci. USA 118, e2009951118 (2021).
Schneider, S. R., Lakey, P. S. J., Shiraiwa, M. & Abbatt, J. P. D. Iodine emission from the reactive uptake of ozone to simulated seawater. Environ. Sci. Process. Impacts 25, 254–263 (2023).
Nordmeyer, T. et al. Unique products of the reaction of isoprene with atomic chlorine: potential markers of chlorine atom chemistry. Geophys. Res. Lett. 24, 1615–1618 (1997).
Griffiths, P. T. et al. Tropospheric ozone in CMIP6 simulations. Atmos. Chem. Phys. 21, 4187–4218 (2021).
Naik, V. et al. Preindustrial to present-day changes in tropospheric hydroxyl radical and methane lifetime from the Atmospheric Chemistry and Climate Model Intercomparison Project (ACCMIP). Atmos. Chem. Phys. 13, 5277–5298 (2013).
Bonan, G. B. & Doney, S. C. Climate, ecosystems, and planetary futures: the challenge to predict life in Earth system models. Science 359, eaam8328 (2018).
Karagodin-Doyennel, A. et al. Iodine chemistry in the chemistry-climate model SOCOL-AERv2-I. Geosci. Model Dev. 14, 6623–6645 (2021).
Caram, C. et al. Sensitivity of tropospheric ozone to halogen chemistry in the chemistry-climate model LMDZ-INCA vNMHC. Geosci. Model Dev. 16, 4041–4062 (2023).
Voulgarakis, A. et al. Analysis of present day and future OH and methane lifetime in the ACCMIP simulations. Atmos. Chem. Phys. 13, 2563–2587 (2013).
Iglesias-Suarez, F. et al. Key drivers of ozone change and its radiative forcing over the 21st century. Atmos. Chem. Phys. 18, 6121–6139 (2018).
Horowitz, H. M. et al. Effects of sea salt aerosol emissions for marine cloud brightening on atmospheric chemistry: implications for radiative forcing. Geophys. Res. Lett. 47, e2019GL085838 (2020).
Acknowledgements
This work has been funded by the European Research Council Executive Agency under Horizon 2020 Research and Innovation programme project ERC-2016-COG 726349 CLIMAHAL and grant PID2023-152856OB-I00 funded by MICIU/AEI/10.13039/501100011033 and by ERDF/EU. The Indian Institute of Tropical Meteorology is funded by the Ministry of Earth Sciences, Government of India. B.J.F.-P. acknowledges support from the US National Science Foundation (2030175, 2303948, 2327825). L.J.C. thanks the support from the European Research Council (ERC) under the European Union’s Horizon 2020 Program (grant agreement no. 833290). M.P.C. thanks NERC for funding through projects NE/V011863/X and NE/X003450/1. R.H. was supported by NERC (grant NE/V011863/1). T.W. thanks the support from the Hong Kong Research Grants Council (T24-504/17-N and 15207421). K.A.P. acknowledges support from the US National Science Foundation (OPP-2000493). S.S. appreciates support from the NSF grant 2128617. R.P.F. thanks the financial support from MinCyT (REMATE IF-2023-85161983-APN) and CONICET. R.J.S. acknowledges the support of the NASA Atmospheric Composition Modelling and Analysis Program (grant 80NSSC19K098). We thank the staff at NorArte Visual Science for the design of Fig. 1. A.S.-L. would like to acknowledge the support of Silverlining and Spark Climate Solutions.
Author information
Authors and Affiliations
Contributions
A.S.-L. and A.S.M. initiated this study. A.S.-L., A.S.M., C.A.C., R.P.F., J.P.B., L.J.C., M.P.C., B.J.F.-P., J.M.C.P., A.R.R., R.J.S. and S.S. performed the data compilation and analysis. A.S.M., C.A.C. and R.P.F. produced the figures. All of the authors discussed and commented on the findings. A.S.-L. and A.S.M. wrote the manuscript with contributions from J.A., N.B., S.S.B., J.P.B., L.J.C., M.P.C., C.A.C., R.P.F., R.H., D.E.K., J.-F.L., B.J.F.-P., J.M.C.P., U.P., K.A.P., A.R.R, R.J.S., E.S.S., W.R.S., S.S., J.A.T. and T.W.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature thanks the anonymous reviewers for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Saiz-Lopez, A., Mahajan, A.S., Abbatt, J. et al. The influence of short-lived halogens on atmospheric chemistry and climate. Nature 648, 289–299 (2025). https://doi.org/10.1038/s41586-025-09753-x
Received:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41586-025-09753-x