Abstract
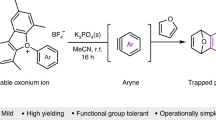
Densely substituted aromatic rings are ubiquitous in pharmaceuticals and agrochemicals1. For making aromatic molecules, aryne intermediates have synthetic potential that rivals most functional groups2. They readily react with nucleophiles, participate in pericyclic reactions and activate inert σ-bonds. Despite their potential, arynes are used at present by a specialized community for mainly niche applications. The lack of widespread adoption of arynes is due to the undesirable means to generate them. Here we report the design of an aryne precursor to overcome this prohibitive barrier. Readily available carboxylic acids are derivatized in a single step to a make a precursor that is then activated by blue light or by heat. Dozens of previously unknown aminated arynes, including pyridynes, are generated in this work, opening the door to drug discovery using aryne intermediates. We foresee that future development of this precursor platform will allow even more decorated arynes to be accessed, further expanding the reach of aryne chemistry.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$32.99 / 30 days
cancel any time
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
Data availability
All data are available in the text or Supplementary Information. Crystal structures can be found in the Cambridge Crystallographic Data Centre under accession codes 2410237–2410238. Source data are provided with this paper.
References
Nilova, A., Campeau, L.-C., Sherer, E. C. & Stuart, D. R. Analysis of benzenoid substitution patterns in small molecule active pharmaceutical ingredients. J. Med. Chem. 63, 13389–13396 (2020).
Shi, J., Li, L. & Li, Y. o-Silylaryl triflates: a journey of Kobayashi aryne precursors. Chem. Rev. 121, 3892–4044 (2021).
Roberts, J. D., Simmons, H. E., Carlsmith, L. A. & Vaughan, C. W. Rearrangement in the reaction of chlorobenzene-1-C14 with potassium amide1. J. Am. Chem. Soc. 75, 3290–3291 (1953).
Kim, N., Choi, M., Suh, S.-E. & Chenoweth, D. M. Aryne chemistry: generation methods and reactions incorporating multiple arynes. Chem. Rev. 124, 11435–11522 (2024).
Medina, J. M., Mackey, J. L., Garg, N. K. & Houk, K. N. The role of aryne distortions, steric effects, and charges in regioselectivities of aryne reactions. J. Am. Chem. Soc. 136, 15798–15805 (2014).
Sundalam, S. K., Nilova, A., Seidl, T. L. & Stuart, D. R. A selective C−H deprotonation strategy to access functionalized arynes by using hypervalent iodine. Angew. Chem. Int. Ed. 55, 8431–8434 (2016).
Nilova, A., Metze, B. & Stuart, D. R. Aryl(TMP)iodonium tosylate reagents as a strategic entry point to diverse aryl intermediates: selective access to arynes. Org. Lett. 23, 4813–4817 (2021).
Roberts, R. A., Metze, B. E., Nilova, A. & Stuart, D. R. Synthesis of arynes via formal dehydrogenation of arenes. J. Am. Chem. Soc. 145, 3306–3311 (2023).
Smith, O. et al. Harnessing triaryloxonium ions for aryne generation. Nat. Synth. 3, 58–66 (2024).
Sullivan, J. M. Explosion during preparation of benzenediazonium-2-carboxylate hydrochloride. J. Chem. Educ. 48, 419 (1971).
Gann, A. W. et al. A photoinduced, benzyne click reaction. Org. Lett. 16, 2003–2005 (2014).
Maki, Y., Furuta, T., Kuzuya, M. & Suzuki, M. Photochemistry of o-nitrobenzaldehyde N-acetyl-N-alkylhydrazones resulting in the formation of benzyne. J. Chem. Soc. Chem. Commun. https://doi.org/10.1039/C39750000616 (1975).
Goti, G., Manal, K., Sivaguru, J. & Dell’Amico, L. The impact of UV light on synthetic photochemistry and photocatalysis. Nat. Chem. 16, 684–692 (2024).
Yoshida, S., Nakamura, Y., Uchida, K., Hazama, Y. & Hosoya, T. Aryne relay chemistry en route to aminoarenes: synthesis of 3-aminoaryne precursors via regioselective silylamination of 3-(triflyloxy)arynes. Org. Lett. 18, 6212–6215 (2016).
Kennedy, S. H., Dherange, B. D., Berger, K. J. & Levin, M. D. Skeletal editing through direct nitrogen deletion of secondary amines. Nature 593, 223–227 (2021).
Fazekas, T. J. et al. Diversification of aliphatic C–H bonds in small molecules and polyolefins through radical chain transfer. Science 375, 545–550 (2022).
Qin, T. et al. A general alkyl–alkyl cross-coupling enabled by redox-active esters and alkylzinc reagents. Science 352, 801–805 (2016).
Johnston, C. P., Smith, R. T., Allmendinger, S. & MacMillan, D. W. C. Metallaphotoredox-catalysed sp3–sp3 cross-coupling of carboxylic acids with alkyl halides. Nature 536, 322–325 (2016).
Zhang, L., DeMuynck, B. M., Paneque, A. N., Rutherford, J. E. & Nagib, D. A. Carbene reactivity from alkyl and aryl aldehydes. Science 377, 649–654 (2022).
Nguyen, K. N. M. et al. Harnessing carbene polarity: unified catalytic access to donor, neutral, and acceptor carbenes. Science 389, 183–189 (2025).
Beringer, F. M. & Huang, S. J. Effects of substituents on the rearrangement of 2-aryl iodoniobenzoates and their cleavage to benzyne1,2. J. Org. Chem. 29, 1637–1638 (1964).
Beringer, F. M. & Huang, S. J. Rearrangement and cleavage of 2-aryliodoniobenzoates. Trapping agents for benzyne1-3. J. Org. Chem. 29, 445–448 (1964).
Le Goff, E. Aprotic generation of benzyne from diphenyliodonium-2-carboxylate. J. Am. Chem. Soc. 84, 3786–3786 (1962).
Brown, D. G. & Boström, J. Analysis of past and present synthetic methodologies on medicinal chemistry: where have all the new reactions gone?. J. Med. Chem. 59, 4443–4458 (2016).
Baur, A., Bustin, K. A., Aguilera, E., Petersen, J. L. & Hoover, J. M. Copper and silver benzoate and aryl complexes and their implications for oxidative decarboxylative coupling reactions. Org. Chem. Front. 4, 519–524 (2017).
Crovak, R. A. & Hoover, J. M. A predictive model for the decarboxylation of silver benzoate complexes relevant to decarboxylative coupling reactions. J. Am. Chem. Soc. 140, 2434–2437 (2018).
Hansch, C., Leo, A. & Taft, R. W. A survey of Hammett substituent constants and resonance and field parameters. Chem. Rev. 91, 165–195 (1991).
Linde, E., Bulfield, D., Kervefors, G., Purkait, N. & Olofsson, B. Diarylation of N- and O-nucleophiles through a metal-free cascade reaction. Chem 8, 850–865 (2022).
Seidl, T. L., Sundalam, S. K., McCullough, B. & Stuart, D. R. Unsymmetrical aryl(2,4,6-trimethoxyphenyl)iodonium salts: one-pot synthesis, scope, stability, and synthetic studies. J. Org. Chem. 81, 1998–2009 (2016).
Mironova, I. A., Noskov, D. M., Yoshimura, A., Yusubov, M. S. & Zhdankin, V. V. Aryl-, akynyl-, and alkenylbenziodoxoles: synthesis and synthetic applications. Molecules 28, 2136 (2023).
Yusubov, M. S., Yusubova, R. Y., Nemykin, V. N. & Zhdankin, V. V. Preparation and X-ray structural study of 1-arylbenziodoxolones. J. Org. Chem. 78, 3767–3773 (2013).
Swain, C. G. & Lupton, E. C. Field and resonance components of substituent effects. J. Am. Chem. Soc. 90, 4328–4337 (1968).
Maiti, M., Sinha, S., Deb, C., De, A. & Ganguly, T. Photophysics of 4-methoxy-benzo[b]thiophene in different environments. Its role in non-radiative transitions both as an electron and as an energy donor. J. Lumin. 82, 259–276 (1999).
Mei, T.-S., Giri, R., Maugel, N. & Yu, J.-Q. PdII-catalyzed monoselective ortho halogenation of C–H bonds assisted by counter cations: a complementary method to directed ortho lithiation. Angew. Chem. Int. Ed. 47, 5215–5219 (2008).
Stompor-Gorący, M., Bajek-Bil, A., Potocka, N. & Zawlik, I. Therapeutic perspectives of aminoflavonoids—a review. Int. J. Mol. Sci. 26, 2014 (2025).
Díaz, M. T., Cobas, A., Guitián, E. & Castedo, L. Polar control of the regioselectivity of hetaryne cycloadditions. synthesis of ellipticine. Synlett 1998, 157–158 (1998).
Díaz, M., Cobas, A., Guitián, E. & Castedo, L. Synthesis of ellipticine by hetaryne cycloadditions—control of regioselectivity. Eur. J. Org. Chem. 2001, 4543–4549 (2001).
Goetz, A. E. & Garg, N. K. Regioselective reactions of 3,4-pyridynes enabled by the aryne distortion model. Nat. Chem. 5, 54–60 (2013).
Marshall, C. M., Federice, J. G., Bell, C. N., Cox, P. B. & Njardarson, J. T. An update on the nitrogen heterocycle compositions and properties of U.S. FDA-approved pharmaceuticals (2013–2023). J. Med. Chem. 67, 11622–11655 (2024).
Bureš, F. Fundamental aspects of property tuning in push–pull molecules. RSC Adv. 4, 58826–58851 (2014).
Romero, N. A. & Nicewicz, D. A. Organic photoredox catalysis. Chem. Rev. 116, 10075–10166 (2016).
Twilton, J. et al. The merger of transition metal and photocatalysis. Nat. Rev. Chem. 1, 0052 (2017).
Chan, A. Y. et al. Metallaphotoredox: the merger of photoredox and transition metal catalysis. Chem. Rev. 122, 1485–1542 (2022).
Buglioni, L., Raymenants, F., Slattery, A., Zondag, S. D. A. & Noël, T. Technological innovations in photochemistry for organic synthesis: flow chemistry, high-throughput experimentation, scale-up, and photoelectrochemistry. Chem. Rev. 122, 2752–2906 (2022).
Nakajima, M. et al. A direct S0→T transition in the photoreaction of heavy-atom-containing molecules. Angew. Chem. Int. Ed. 59, 6847–6852 (2020).
Strieth-Kalthoff, F. & Glorius, F. Triplet energy transfer photocatalysis: unlocking the next level. Chem 6, 1888–1903 (2020).
Acknowledgements
We thank the Hoye group for use of infrared instrumentation and the Gladfelter group for use of UV–vis instrumentation. We thank A. Younis and S. S. Pullarat for help with crystallography. We thank M. Hillmyer, J. Back, M. Hintz and J. R. Lamb for help with thermogravimetric analysis and differential scanning calorimetry. We all thank the University of Minnesota for startup funding. C.C.R. acknowledges the Amgen Young Investigator Award, the BMS Unrestricted Research Grant, the Sloan Fellowship, the Dreyfus Teacher Scholar Award, the McKnight Land-Grant and 3M-Alumni Professorships. S.S.K. and C.M.S. both thank University of Minnesota for Wayland E. Noland Excellence Fellowships and Doctoral Dissertation Fellowships.
Author information
Authors and Affiliations
Contributions
Conceptualization: experimental: C.M.S., S.S.K., C.-L.Y. and C.C.R; computational: J.-N.B. and D.G. Data curation: C.M.S., S.S.K., C.-L.Y. and D.G. Formal analysis: C.M.S., S.S.K., C.-L.Y. and D.G. Funding acquisition: J.-N.B. and C.C.R. Investigation: C.M.S., S.S.K., C.L.Y. and D.G. Methodology: all authors. Project administration: J.-N.B. and C.C.R. Resources: C.M.S., S.S.K. and C.-L.Y. Supervision: J.-N.B. and C.C.R. Validation: C.M.S., S.S.K. and C.-L.Y. Writing—original draft and writing—review and editing: all authors.
Corresponding authors
Ethics declarations
Competing interests
The University of Minnesota has filed a provisional patent on this work under application no. 63/886,266 with C.M.S., S.S.K., C.-L.Y. and C.C.R. as inventors for potential commercialization of reagents.
Peer review
Peer review information
Nature thanks Christopher Jones and the other, anonymous, reviewer(s) for their contribution to the peer review of this work. Peer reviewer reports are available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information (download PDF )
All experimental procedures, characterization of new compounds, mechanistic studies and density-functional theory calculations.
Source data
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Seong, C.M., Kargbo, S.S., Yu, CL. et al. Myriad aryne derivatives from carboxylic acids. Nature 649, 91–97 (2026). https://doi.org/10.1038/s41586-025-09830-1
Received:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41586-025-09830-1