Abstract
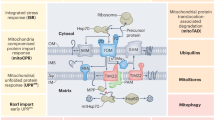
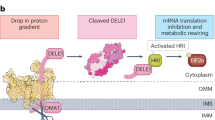
Mitochondrial protein import is required for maintaining organellar function1. Perturbations in this process are associated with various physiological and disease conditions2. Several stress responses, including the mitochondrial compromised protein import response (mitoCPR), combat damage caused by mitochondrial protein import defects2. However, how this defect is sensed remains largely unknown. Here we reveal that the conserved mitochondrial Hsp70 co-chaperone, Mge1, acts as a stress messenger in budding yeast. During mitochondrial stress, unimported Mge1 entered the nucleus and triggered the transcription of mitoCPR target genes. This was mediated by the interaction of Mge1 with the transcription factor Pdr3 on DNA regulatory elements. The mitochondrial targeting sequence of Mge1 was both sufficient and essential for mitoCPR induction, demonstrating that in addition to their roles in mitochondrial protein import, targeting sequences can also function as signalling molecules. As protein import defects are a common consequence of various types of mitochondrial damage3,4, these findings suggest a novel function for the targeting sequence of Mge1 as an indicator of mitochondrial health.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$32.99 / 30 days
cancel any time
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
Data availability
All data supporting the findings of this study are available within the paper and its Supplementary Information. Supplementary Fig. 1 contains verification of yeast strains described in the Methods section (‘Yeast strain construction’). The full version of all gels and blots is provided in Supplementary Fig. 2. Source data for the overexpression screen have been provided in Supplementary Table 1. Mass spectrometry (associated with Fig. 3d and Extended Data Fig. 6f) have been deposited to the ProteomeXchange Consortium via the PRIDE86 with the dataset identifier PXD066688 (publicly available at https://www.ebi.ac.uk/pride/archive/projects/PXD066688). Source data for the mass spectrometry have also been provided in Supplementary Table 2. RNA-seq data (associated with Fig. 4b and Extended Data Fig. 6c) can be accessed via the Gene Expression Omnibus with the accession number GSE303345. Source data for these RNA-seq analysis have also been provided in Supplementary Tables 3–5. Source data for all graphs have been provided. There are no restrictions on data availability. Source data are provided with this paper.
References
Endo, T. & Wiedemann, N. Molecular machineries and pathways of mitochondrial protein transport. Nat. Rev. Mol. Cell Biol. https://doi.org/10.1038/s41580-025-00865-w (2025).
Pfanner, N., den Brave, F. & Becker, T. Mitochondrial protein import stress. Nat. Cell Biol. 27, 188–201 (2025).
Balzarini, M., Kim, J. & Weidberg, H. Quality control of un-imported mitochondrial proteins at a glance. J. Cell Sci. 138, jcs263757 (2025).
Lee-Glover, L. P. & Shutt, T. E. Mitochondrial quality control pathways sense mitochondrial protein import. Trends Endocrinol. Metab. 35, 308–320 (2024).
Suomalainen, A. & Nunnari, J. Mitochondria at the crossroads of health and disease. Cell 187, 2601–2627 (2024).
Nashed, S. et al. Functional mapping of N-terminal residues in the yeast proteome uncovers novel determinants for mitochondrial protein import. PLoS Genet. 19, e1010848 (2023).
Vögtle, F.-N. et al. Global analysis of the mitochondrial N-proteome identifies a processing peptidase critical for protein stability. Cell 139, 428–439 (2009).
Craig, E. A. Hsp70 at the membrane: driving protein translocation. BMC Biol. 16, 11 (2018).
Boos, F., Labbadia, J. & Herrmann, J. M. How the mitoprotein-induced stress response safeguards the cytosol: a unified view. Trends Cell Biol. 30, 241–254 (2020).
Nowicka, U. et al. Cytosolic aggregation of mitochondrial proteins disrupts cellular homeostasis by stimulating the aggregation of other proteins. eLife 10, e65484 (2021).
Boos, F. et al. Mitochondrial protein-induced stress triggers a global adaptive transcriptional programme. Nat. Cell Biol. 21, 442–451 (2019).
Fessler, E. et al. A pathway coordinated by DELE1 relays mitochondrial stress to the cytosol. Nature 579, 433–437 (2020).
Fessler, E., Krumwiede, L. & Jae, L. T. DELE1 tracks perturbed protein import and processing in human mitochondria. Nat. Commun. 13, 1853 (2022).
Guo, X. et al. Mitochondrial stress is relayed to the cytosol by an OMA1-DELE1-HRI pathway. Nature 579, 427–432 (2020).
Haakonsen, D. L. et al. Stress response silencing by an E3 ligase mutated in neurodegeneration. Nature 626, 874–880 (2024).
Krämer, L. et al. MitoStores: chaperone-controlled protein granules store mitochondrial precursors in the cytosol. EMBO J. 42, e112309 (2023).
Münch, C. & Harper, J. W. Mitochondrial unfolded protein response controls matrix pre-RNA processing and translation. Nature 534, 710–713 (2016).
Shakya, V. P. et al. A nuclear-based quality control pathway for non-imported mitochondrial proteins. eLife 10, e61230 (2021).
Sutandy, F. X. R., Gößner, I., Tascher, G. & Münch, C. A cytosolic surveillance mechanism activates the mitochondrial UPR. Nature 618, 849–854 (2023).
Wang, X. & Chen, X. J. A cytosolic network suppressing mitochondria-mediated proteostatic stress and cell death. Nature 524, 481–484 (2015).
Weidberg, H. & Amon, A. MitoCPR — a surveillance pathway that protects mitochondria in response to protein import stress. Science 360, eaan4146 (2018).
Wrobel, L. et al. Mistargeted mitochondrial proteins activate a proteostatic response in the cytosol. Nature 524, 485–488 (2015).
Coyne, L. P. et al. Mitochondrial protein import clogging as a mechanism of disease. eLife 12, e84330 (2023).
Hsu, M.-C. et al. Mitochondrial YME1L1 governs unoccupied protein translocase channels. Nat. Cell Biol. 27, 309–321 (2025).
Kim, J. et al. ATAD1 prevents clogging of TOM and damage caused by un-imported mitochondrial proteins. Cell Rep. 43, 114473 (2024).
Krakowczyk, M. et al. OMA1 protease eliminates arrested protein import intermediates upon mitochondrial depolarization. J. Cell Biol. 223, e202306051 (2024).
Mårtensson, C. U. et al. Mitochondrial protein translocation-associated degradation. Nature 569, 679–683 (2019).
Buechel, E. R. & Pinkett, H. W. Transcription factors and ABC transporters: from pleiotropic drug resistance to cellular signaling in yeast. FEBS Lett. 594, 3943–3964 (2020).
Delahodde, A., Pandjaitan, R., Corral-Debrinski, M. & Jacq, C. Pse1/Kap121-dependent nuclear localization of the major yeast multidrug resistance (MDR) transcription factor Pdr1. Mol. Microbiol. 39, 304–313 (2001).
Bursać, D. & Lithgow, T. Jid1 is a J-protein functioning in the mitochondrial matrix, unable to directly participate in endoplasmic reticulum associated protein degradation. FEBS Lett. 583, 2954–2958 (2009).
Lytovchenko, O. et al. The INA complex facilitates assembly of the peripheral stalk of the mitochondrial F1Fo-ATP synthase. EMBO J. 33, 1624–1638 (2014).
Westermann, B., Prip-Buus, C., Neupert, W. & Schwarz, E. The role of the GrpE homologue, Mge1p, in mediating protein import and protein folding in mitochondria. EMBO J. 14, 3452–3460 (1995).
Nguyen Ba, A. N., Pogoutse, A., Provart, N. & Moses, A. M. NLStradamus: a simple hidden Markov model for nuclear localization signal prediction. BMC Bioinformatics 10, 202 (2009).
Kimura, M. & Imamoto, N. Biological significance of the importin-β family-dependent nucleocytoplasmic transport pathways. Traffic 15, 727–748 (2014).
Veatch, J. R., McMurray, M. A., Nelson, Z. W. & Gottschling, D. E. Mitochondrial dysfunction leads to nuclear genome instability via an iron-sulfur cluster defect. Cell 137, 1247–1258 (2009).
Devaux, F., Carvajal, E., Moye-Rowley, S. & Jacq, C. Genome-wide studies on the nuclear PDR3-controlled response to mitochondrial dysfunction in yeast. FEBS Lett. 515, 25–28 (2002).
Rödl, S. et al. A protein-specific priority code in presequences determines the efficiency of mitochondrial protein import. PLoS Biol. 23, e3003298 (2025).
Gallas, M. R., Dienhart, M. K., Stuart, R. A. & Long, R. M. Characterization of Mmp37p, a Saccharomyces cerevisiae mitochondrial matrix protein with a role in mitochondrial protein import. Mol. Biol. Cell 17, 4051–4062 (2006).
Tamura, Y. et al. Identification of Tam41 maintaining integrity of the TIM23 protein translocator complex in mitochondria. J. Cell Biol. 174, 631–637 (2006).
Tamura, Y. et al. Tam41 is a CDP-diacylglycerol synthase required for cardiolipin biosynthesis in mitochondria. Cell Metab. 17, 709–718 (2013).
Kutik, S. et al. The translocator maintenance protein Tam41 is required for mitochondrial cardiolipin biosynthesis. J. Cell Biol. 183, 1213–1221 (2008).
Abramson, J. et al. Accurate structure prediction of biomolecular interactions with AlphaFold 3. Nature 630, 493–500 (2024).
Banerjee, A., Rahman, H., Prasad, R. & Golin, J. How fungal multidrug transporters mediate hyper resistance through DNA amplification and mutation. Mol. Microbiol. 118, 3–15 (2022).
Ducett, J. K. et al. Unfolding of the C-terminal domain of the J-protein Zuo1 releases autoinhibition and activates Pdr1-dependent transcription. J. Mol. Biol. 425, 19–31 (2013).
DeRisi, J. et al. Genome microarray analysis of transcriptional activation in multidrug resistance yeast mutants. FEBS Lett. 470, 156–160 (2000).
Thakur, J. K. et al. A nuclear receptor-like pathway regulating multidrug resistance in fungi. Nature 452, 604–609 (2008).
Wu, G.-Z. et al. Control of retrograde signalling by protein import and cytosolic folding stress. Nat. Plants 5, 525–538 (2019).
Narendra, D. P. et al. PINK1 is selectively stabilized on impaired mitochondria to activate Parkin. PLoS Biol. 8, e1000298 (2010).
Nargund, A. M., Pellegrino, M. W., Fiorese, C. J., Baker, B. M. & Haynes, C. M. Mitochondrial import efficiency of ATFS-1 regulates mitochondrial UPR activation. Science 337, 587–590 (2012).
Rolland, S. G. et al. Compromised mitochondrial protein import acts as a signal for UPRmt. Cell Rep. 28, 1659–1669.e5 (2019).
Shpilka, T. et al. UPRmt scales mitochondrial network expansion with protein synthesis via mitochondrial import in Caenorhabditis elegans. Nat. Commun. 12, 479 (2021).
Konovalova, S. et al. Redox regulation of GRPEL2 nucleotide exchange factor for mitochondrial HSP70 chaperone. Redox Biol. 19, 37–45 (2018).
Marada, A. et al. Mge1, a nucleotide exchange factor of Hsp70, acts as an oxidative sensor to regulate mitochondrial Hsp70 function. Mol. Biol. Cell 24, 692–703 (2013).
DiGiovanni, L. F. et al. ROS transfer at peroxisome-mitochondria contact regulates mitochondrial redox. Science 389, 157–162 (2025).
McMinimy, R. et al. Reactive oxygen species control protein degradation at the mitochondrial import gate. Mol. Cell 84, 4612–4628.e13 (2024).
Savojardo, C., Bruciaferri, N., Tartari, G., Martelli, P. L. & Casadio, R. DeepMito: accurate prediction of protein sub-mitochondrial localization using convolutional neural networks. Bioinformatics 36, 56–64 (2020).
Srivastava, S. et al. Regulation of mitochondrial protein import by the nucleotide exchange factors GrpEL1 and GrpEL2 in human cells. J. Biol. Chem. 292, 18075–18090 (2017).
Calvo, S. E. et al. Comparative analysis of mitochondrial N-termini from mouse, human, and yeast. Mol. Cell. Proteomics 16, 512–523 (2017).
Bayne, A. N., Dong, J., Amiri, S., Farhan, S. M. K. & Trempe, J.-F. MTSviewer: a database to visualize mitochondrial targeting sequences, cleavage sites, and mutations on protein structures. PLoS ONE 18, e0284541 (2023).
Myers, A. M., Pape, L. K. & Tzagoloff, A. Mitochondrial protein synthesis is required for maintenance of intact mitochondrial genomes in Saccharomyces cerevisiae. EMBO J. 4, 2087–2092 (1985).
Schrott, S. & Osman, C. Two mitochondrial HMG-box proteins, Cim1 and Abf2, antagonistically regulate mtDNA copy number in Saccharomyces cerevisiae. Nucleic Acids Res. 51, 11813–11835 (2023).
Smoyer, C. J. et al. Analysis of membrane proteins localizing to the inner nuclear envelope in living cells. J. Cell Biol. 215, 575–590 (2016).
Fukasawa, Y. et al. MitoFates: improved prediction of mitochondrial targeting sequences and their cleavage sites. Mol. Cell. Proteomics 14, 1113–1126 (2015).
Pfanner, N., Müller, H. K., Harmey, M. A. & Neupert, W. Mitochondrial protein import: involvement of the mature part of a cleavable precursor protein in the binding to receptor sites. EMBO J. 6, 3449–3454 (1987).
Wiley, J. C., Wailes, L. A., Idzerda, R. L. & McKnight, G. S. Role of regulatory subunits and protein kinase inhibitor (PKI) in determining nuclear localization and activity of the catalytic subunit of protein kinase A. J. Biol. Chem. 274, 6381–6387 (1999).
Bragoszewski, P., Turek, M. & Chacinska, A. Control of mitochondrial biogenesis and function by the ubiquitin-proteasome system. Open Biol. 7, 170007 (2017).
Valenti, R. et al. A proteome-wide yeast degron collection for the dynamic study of protein function. J. Cell Biol. 224, e202409050 (2025).
Morawska, M. & Ulrich, H. D. An expanded tool kit for the auxin-inducible degron system in budding yeast. Yeast 30, 341–351 (2013).
Nishimura, K., Fukagawa, T., Takisawa, H., Kakimoto, T. & Kanemaki, M. An auxin-based degron system for the rapid depletion of proteins in nonplant cells. Nat. Methods 6, 917–922 (2009).
Gallego, O. et al. Detection and characterization of protein interactions in vivo by a simple live-cell imaging method. PLoS ONE 8, e62195 (2013).
Haruki, H., Nishikawa, J. & Laemmli, U. K. The anchor-away technique: rapid, conditional establishment of yeast mutant phenotypes. Mol. Cell 31, 925–932 (2008).
Gelperin, D. M. et al. Biochemical and genetic analysis of the yeast proteome with a movable ORF collection. Genes Dev. 19, 2816–2826 (2005).
Young, B. P. & Loewen, C. J. Balony: a software package for analysis of data generated by synthetic genetic array experiments. BMC Bioinformatics 14, 354 (2013).
Storey, J. D. & Tibshirani, R. Statistical methods for identifying differentially expressed genes in DNA microarrays. Methods Mol. Biol. 224, 149–157 (2003).
Meisinger, C., Pfanner, N. & Truscott, K. N. Isolation of yeast mitochondria. Methods Mol. Biol. 313, 33–39 (2006).
Glowczewski, L., Waterborg, J. H. & Berman, J. G. Yeast chromatin assembly complex 1 protein excludes nonacetylatable forms of histone H4 from chromatin and the nucleus. Mol. Cell. Biol. 24, 10180 (2004).
Hughes, C. S. et al. Single-pot, solid-phase-enhanced sample preparation for proteomics experiments. Nat. Protoc. 14, 68–85 (2019).
Rappsilber, J., Mann, M. & Ishihama, Y. Protocol for micro-purification, enrichment, pre-fractionation and storage of peptides for proteomics using StageTips. Nat. Protoc. 2, 1896–1906 (2007).
Demichev, V., Messner, C. B., Vernardis, S. I., Lilley, K. S. & Ralser, M. DIA-NN: neural networks and interference correction enable deep proteome coverage in high throughput. Nat. Methods 17, 41–44 (2020).
Teo, G. et al. SAINTq: scoring protein-protein interactions in affinity purification-mass spectrometry experiments with fragment or peptide intensity data. Proteomics 16, 2238–2245 (2016).
Livak, K. J. & Schmittgen, T. D. Analysis of relative gene expression data using real-time quantitative PCR and the 2−ΔΔCT method. Methods 25, 402–408 (2001).
Sprouffske, K. & Wagner, A. Growthcurver: an R package for obtaining interpretable metrics from microbial growth curves. BMC Bioinformatics 17, 172 (2016).
Katoh, K. & Standley, D. M. MAFFT multiple sequence alignment software version 7: improvements in performance and usability. Mol. Biol. Evol. 30, 772–780 (2013).
Waterhouse, A. M., Procter, J. B., Martin, D. M. A., Clamp, M. & Barton, G. J. Jalview Version 2 — a multiple sequence alignment editor and analysis workbench. Bioinformatics 25, 1189–1191 (2009).
Creighton, T. E. Proteins: Structures and Molecular Properties (W. H. Freeman, 1993).
Perez-Riverol, Y. et al. The PRIDE database at 20 years: 2025 update. Nucleic Acids Res. 53, D543–D553 (2025).
Acknowledgements
We thank H. Rampelt and N. Pfanner for the Mge1 antiserum; X. Jie Chen for the aac2A128P, A137D plasmid; C. Loewen and E. Conibear for comments, discussions and for sharing reagents; the University of British Columbia (UBC) LSI Imaging Core Facility for guidance regarding imaging data collection and analysis (RRID:SCR_023783); the UBC Biofactorial Core Facility (Bio!) for plate reader services; and the UBC LSI qPCR Core (UBC GREx Biological Resilience Initiative), the LSI Bioinformatics Core, the School of Biomedical Engineering Sequencing Core and the members of the UBC Proteomic Core Facility for their help with sample analysis. This work was supported by the Natural Sciences and Engineering Research Council of Canada (grant RGPIN-2020-05204 to H.W.), Michael Smith Health Research BC (SCH-2021-1524 to H.W.), the Canadian Foundation for Innovation (John R. Evans Leaders Fund Project 40467), the UBC Cell and Developmental Biology (CELL) Fellowships (to Z.Y., M.B. and M.G.), the Canadian Institutes of Health Research (Canada Graduate Scholarships-Master’s to M.G.), 2020 CFI Innovation Funds (Project 39914 to T.M.) and by the Francis Crick Institute, which receives its core funding from Cancer Research UK (CC2043), the UK Medical Research Council (CC2043) and the Wellcome Trust (CC2043). Schematics were created using BioRender (https://biorender.com).
Author information
Authors and Affiliations
Contributions
Z.Y., M.V. and H.W. conceived the study and designed the experiments. Z.Y., M.B., M.V., M.G. and T.S.P. performed and interpreted most of the experiments and analysed the data. E.H., N.N.F. and T.M. acquired and analysed the mass spectrometry data. C.K.Y. performed the AlphaFold3 analysis. W.S.A. and F.J.v.W. performed and analysed the ChIP analysis. M.H. and K.W. performed the evolutionary analysis. Z.Y., M.B., M.G. and H.W. drafted the manuscript with input and edits from all authors.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature thanks Cole Haynes, Johannes Herrmann and the other, anonymous, reviewer(s) for their contribution to the peer review of this work. Peer reviewer reports are available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data figures and tables
Extended Data Fig. 1 Overexpression of Mge1 leads to upregulation of mitoCPR target genes.
(a) Live-cell fluorescence imaging of cells expressing PTEF2-GFP-PDR3 and the nuclear marker HTB1-mCherry under control (empty vector) and impaired mitochondrial protein import (PSD1 overexpression) conditions. Zoom-in images of representative cells are shown on the right. Scale bars, 5 μm. OE, overexpression. (b) CIS1 and PSD1 mRNA level in cells used in (a) and main Fig. 1a. n = 3 biological replicates; Two-tailed Student’s t-test. OE, overexpression. (c) PSD1 mRNA level in cells used for ChIP analysis (Fig. 1b). One-way ANOVA followed by Tukey’s test. (d) CIS1 mRNA levels in control cells (empty vector, n = 3 biological repeats) and cells overexpressing the indicated mitochondrial genes (n = 3 biological repeats). Overexpression was induced by the addition of galactose for 4 h. One-way ANOVA followed by Dunnett’s test; **** P ≤ 0.0001. OE, overexpression. (e) mRNA levels of the indicated overexpressed genes (n = 3 biological repeats) compared to empty vector control (n = 2 biological repeats) following 4-h galactose induction. Two-tailed Student’s t-test. EV, empty vector, OE, overexpression (f) Same as (d). The indicated overexpressed mitochondrial genes were tagged with 2XV5 at the N-terminus. (g) Same analysis as (e). (h) Immunoblot of overexpressed Mge1-HA-ProtA and 2V5-Mge1-HA-ProtA following 4-h galactose induction in wild-type and PDR3 deleted cells. OE, overexpression. (i) mRNA levels of YGR035C, PDR5, PDR15, GRE2 and BDH2 in cells overexpressing MGE1 and 2V5-MGE1 for 4 h. n = 3 biological replicates; One-way ANOVA followed by Dunnett’s test. OE, overexpression. (b-g, i) Data represent mean +/− SD; ns, not significant.
Extended Data Fig. 2 Overexpressed Mge1 localizes to both mitochondria and the nucleus.
(a) Live-cell fluorescence imaging of cells expressing the mitochondrial marker Tom70-GFP and either Mge1-mCherry (top) or Mss116-mCherry (bottom). Overexpression of MGE1-mCherry and MSS116-mCherry from the GAL1-10 promoter was induced by the addition of galactose for 4 h. Zoom-in images of representative cells are shown on the right. Scale bars, 5 μm. Mean Pearson’s correlation coefficients is shown on the right (n = 3 independent experiments; in total of 60 cells per strain; Two-tailed Student’s t-test). (b) Large fields of cells analyzed in Fig. 1e, overexpressing Mge1-mCherry (top) or Mss116-mCherry (bottom) as well as the nuclear envelope marker Nup159-GFP. Representative zoom-in images and corresponding fluorescence intensity profiles along the white line are shown on the right. Scale bars, 5 μm. (c) Large fields of cells analyzed in Fig. 1f, expressing the nuclear protein GFP1–10-mCherry-Pus1 alone or with either Mge1-GFP11 or Mss116-GFP11. Zoom-in images of representative cells are shown on the right. Scale bars, 5 μm. Overexpression of MGE1-GFP11 and MSS116-GFP11 was confirmed by measuring their mRNA levels relative to control cells (bottom panels). n = 3 biological replicates; two-tailed Student’s t-test. (d) CIS1 mRNA levels in cells overexpressing MGE1, ILV2, HEM15 or MRP51 from the GAL1-10 promoter for 4 h (left panel; one-way ANOVA followed by Dunnett’s test). Overexpression was confirmed by qPCR analysis (right panels; two-tailed Student’s t-test). n = 3 biological replicates. EV, empty vector; OE, overexpression. (a, c, d) Data represent mean +/− SD; ns, not significant.
Extended Data Fig. 3 Mge1 precursor accumulates in the nucleus and not inside mitochondria when protein import is impaired.
(a) In vitro protein import assay confirming protein import defects in cells overexpressing PSD1. Mitochondria were isolated from control cells (empty vector) or cells overexpressing PSD1 (4 h). Mitochondria were incubated with the radiolabeled MTS-containing matrix proteins 35S-Zim17 and 35S-Mge1 for the indicated durations. Membrane potential (Δψ) was dissipated by preincubation with antimycin A, oligomycin, and valinomycin. Import rate of the intermembrane space protein 35S-Mix23, which does not depend on the TIM23 translocase, was used as a control. Quantifications from three biological replicates are shown on the right; protein levels imported into control mitochondria at the final time point was set to 100%. p, precursor; m, mature. (b) DiSC3(5) fluorescence measurement of membrane potential in mitochondria isolated from control cells (empty vector) or cells overexpressing PSD1 (4 h). Mitochondria and 2 μM of valinomycin were added as indicated. Fluorescent intensity was normalized to the maximal signal for each trace (n = 4 biological replicates; Data are shown as mean). Quantification of mitochondrial membrane potential is shown on the right (Data are shown as mean +/− SD; Two-tailed Student’s t-test). ns, not significant. (c) In vitro protein import assay of radiolabeled 35S-Mge1 and 35S-Mge1-Flag into mitochondria isolated from wild-type cells. Membrane potential (Δψ) was dissipated by preincubation with antimycin A, oligomycin, and valinomycin. Quantification of two biological replicates is shown on the right; level of imported 35S-Mge1 at the final time point was set to 100%. Data are shown as mean. p, precursor; m, mature. (d) Confirmation of PSD1 overexpression by qPCR analysis for the samples used in Fig. 2a. n = 3 biological replicates; two-tailed Student’s t-test. OE, overexpression. (e) Mitochondria were isolated by differential centrifugation from empty vector (EV) control cells or cells overexpressing PSD1 for 4 h. Immunoblot analysis was used for detecting the mature and precursor forms of Mge1-Flag in the mitochondrial and cytosolic fractions. Mitochondria were treated with 25 or 50 μg/ml proteinase K (Prot. K). Tom70-Myc served as an outer membrane control protein, Cox4 as a matrix control protein, and Pgk1 as a cytosolic control protein. OE, overexpression. (f) Immunoblot analysis of untagged Mge1 in cellular fractions from cells overexpressing PSD1 for 4 h. Htb1-mCherry and Pgk1serve as nuclear and cytosolic markers, respectively. Input and spheroplasts (Sphe.) represent total cell lysates, before and after cell wall digestion. Supernatant (Sup.) =post-nuclear supernatant; Nuc. Frac, nuclear fraction; p, precursor form; m, mature form. (g) Confirmation of PSD1 overexpression by qPCR analysis for the samples used in Fig. 2d. n = 3 biological replicates; One-way ANOVA followed by Dunnett’s test. (a, d, g) Data represent mean +/− SD.
Extended Data Fig. 4 Protein import rate, activation of the mitoCPR, and accumulation of Mge1 precursor in rho− cells.
(a) Confirmation of PSD1 overexpression by qPCR analysis for the samples used in Fig. 2f. n = 3 biological replicates; One-way ANOVA followed by Dunnett’s test. (b) Cells expressing PTEF2-V5-PDR3 and control no-V5 cells (PTEF2-GFP-PDR3) were incubated for 4 h under control or protein import stress conditions (induced by PSD1 overexpression). V5-Pdr3 was immunoprecipitated using V5-Trap beads. Untagged Mge1 was detected using Mge1 antiserum. The asterisks indicate nonspecific bands. OE, overexpression. p, precursor form. m, mature form. The corresponding PSD1 mRNA levels are shown on the right panel. n = 3 biological replicates; one-way ANOVA followed by Dunnett’s test. (c) Cells expressing MGE1-FLAG and cells expressing MGE1-FLAG and PTEF2-GFP-PDR1 were incubated for 4 h under control or protein import stress conditions. GFP-Pdr1 was immunoprecipitated using anti-GFP antibodies coupled to IgG beads from cells expressing PTEF2-GFP-PDR1. The asterisk indicates a nonspecific band and ▲ indicates the GFP antibody’s light chain. OE, overexpression; p, precursor form; m, mature form. The corresponding PSD1 mRNA levels are shown on the right panel. n = 3 biological replicates; one-way ANOVA followed by Dunnett’s test. (d) Confirmation of PSD1 overexpression by qPCR analysis for the samples used in Fig. 2g. n = 3 biological replicates; one-way ANOVA followed by Tukey’s test. (e) Serial dilutions of wild-type and rho− cells. Cells were grown to logarithmic phase and spotted on YP (1% yeast extract, 2% bactopeptone) plates supplemented by 2% glucose or 3% glycerol. (f) CIS1 mRNA levels in wild-type and rho− cells in the presence of absence of PDR3. n = 3 biological replicates; one-way ANOVA followed by Dunnett’s test. (g) In vitro protein import assay using mitochondria isolated from wild-type and rho− cells. Mitochondria were incubated with radiolabeled 35S-Mdh1 and 35S-Mge1 for the indicated durations. Membrane potential (Δψ) was dissipated by preincubation with antimycin A, oligomycin, and valinomycin. Quantifications from three biological replicates are shown on the right; protein levels imported into wild-type mitochondria at the final time point were set to 100%. p, precursor; m, mature. (h) Immunoblot analysis of Mge1-FLAG and untagged Mge1 in wild-type and rho− cells. Wild-type cells that do not contain a FLAG tag were used as a control for Mge1-FLAG. p, precursor form. m, mature forms. (i) Half-life analysis of the Mge1-FLAG precursor in rho− and PDR3-deleted rho− cells. Cells were treated with cycloheximide (CHX, 0.5 mg/ml) and samples were collected at the indicated time points. Quantification of 4 biological replicates is shown on the right. p, precursor form. m, mature form. (j) Same as (i). Half-life analysis of the untagged Mge1 precursor in rho− and PDR3 deleted rho− cells (n = 3 biological replicates). p, precursor form. m, mature form. Asterisks indicate nonspecific bands. (k) Same as in (i), rho− cells were treated with DMSO (vehicle control) or MG132 (40 µM) for 1 h prior to the addition of cycloheximide (n = 3 biological replicates). (a-d, f, g, i-k) Data represent mean +/− SD. (a-d) **** P ≤ 0.0001.
Extended Data Fig. 5 Activation of the mitoCPR requires the presence of Mge1 precursor in the nucleus.
(a) Tetrad dissection of wild-type and MGE1/mge1∆ diploid strains showing that haploid yeast deleted of MGE1 are inviable. (b) Confirmation of PSD1 overexpression by qPCR analysis for the samples used in Fig. 3a,b. n = 4 biological replicates; one-way ANOVA followed by Tukey’s test. (c) Cells expressing Mge1-Myc-AID or Ubx2-Myc-AID were treated with DMSO or auxin (1 mM indole-3-acetic acid) following 4 h induction of PSD1 overexpression by galactose. Samples were collected at the indicated time points. Levels of the Mge1 precursor as well as Ubx2 were quantified from 3 biological replicates. (d) CIS1 and PSD1 mRNA levels in cell expressing MGE1-FRB with or without the plasma membrane (PM) anchor Pil1-FKBP under control or import stress conditions (PSD1 overexpression for 4 h). Anchoring of the Mge1 precursor was induced by adding 50 nM rapamycin for the duration of PSD1 overexpression. n = 2 biological replicates; data represent mean; one-way ANOVA followed by Dunnett’s test; ns, not significant. (e) Immunoblot analysis of Mge1-NES-mCherry in cellular fractions from cells overexpressing PSD1 for 4 h. Htb1-mCherry and Pgk1serve as nuclear and cytosolic markers, respectively. Input and spheroplasts (Sphe.) represent total cell lysates, before and after cell wall digestion. Supernatant (Sup.)=post-nuclear supernatant. The asterisk indicates a band likely to be a degradation product. p, precursor form. m, mature form. (f) Confirmation of PSD1 overexpression by qPCR analysis for the samples used in Fig. 3c. n = 4 biological replicates; one-way ANOVA followed by Tukey’s test. (g) PSD1 overexpression was induced by galactose and terminated following 4 h by glucose addition. PSD1 mRNA levels were monitored over 12 h post-glucose addition (same samples as in Fig. 3e). n = 3 biological replicates; one-way ANOVA followed by Dunnett’s test; ns, not significant. (b, c, f, g) Data represent mean +/− SD.
Extended Data Fig. 6 Mge1’s presequence is required for inducing the mitoCPR.
(a) Immunoblot of overexpressed mCherry-tagged Mge1 truncation mutants and the Mss116’s MTS (4 h following galactose induction). OE, overexpression. (b) Live-cell fluorescence images of cells expressing the nuclear envelope marker Nup159-GFP and ΔMTS-Mge1-mCherry or NLS-ΔMTS-Mge1-mCherry. Expression of MGE1 variants from the GAL1-10 promoter was induced by the addition of galactose for 4 h. Scale bars, 5 μm. (c) Pdr3-dependent upregulated genes were identified by differential gene expression analysis of: 1. wild-type versus pdr3Δ cells, both overexpressing MTSMge1-mCherry, and 2. wild-type versus pdr3Δ cells, both overexpressing PSD1. The scatter plot displays genes with an adjusted p value ≤ 0.05 (Wald test in DESeq2) in at least one of analyses. PDR3 was removed from both datasets prior to the correlation analysis. (d) Growth curves of wild-type, MTSSu9-MGE1, and MTSIlv2-MGE1 cells were plotted on a semilogarithmic scale. Corresponding growth rate (r) was calculated from the growth curves of three biological replicates, each with three technical replicates; two-tailed Student’s t-test. ns, not significant. (e) Immunoblot of cells expressing Mge1-FLAG, MTSSu9-Mge1-FLAG, or MTSIlv2-Mge1-FLAG under control and PSD1 overexpression conditions. p1, Mge1-FLAG precursor; p2, MTSSu9-Mge1-FLAG precursor; p3, MTSIlv2-Mge1-FLAG precursor. m1/2/3, mature form Mge1-FLAG/MTSSu9-Mge1-FLAG/MTSIlv2-Mge1-FLAG (f) V5-Pdr3 was immunoprecipitated using V5-trap beads from the following: 1. cells under basal conditions (n = 2 biological repeats), 2. cells under impaired mitochondrial protein import conditions (PSD1 overexpression; n = 2 biological repeats), or 3. MTSIlv2-MGE1 cells under impaired mitochondrial protein import conditions (n = 3 biological repeats). Mge1 intensities were measured by mass spectrometry analysis. Data are shown as mean; one-way ANOVA followed by Dunnett’s test. (g) Left- Confirmation of PSD1 overexpression by qPCR analysis for the samples used in Fig. 4e. n = 3 biological replicates; one-way ANOVA followed by Tukey’s test. Right- Immunoblot of Mge1-FLAG and MTSIlv2-Mge1-FLAG under control or import stress (PSD1 overexpression for 4 h) conditions. Addbacks of Mge1-mCherry and Mge1-NES-FLAG into the MTSIlv2-MGE1 background are also presented. p1, Mge1-FLAG precursor; p2, MTSIlv2-Mge1-FLAG precursor; p3, Mge1-NES-FLAG precursor; p4, Mge1-mCherry precursor; m1/2, mature Mge1-FLAG/MTSIlv2-Mge1-FLAG; m3, mature Mge1-NES-FLAG; m4, mature Mge1-mCherry. OE, overexpression. (h) Left- Confirmation of PSD1 overexpression by qPCR analysis for the samples used in Fig. 4f. n = 3 biological replicates; one-way ANOVA followed by Tukey’s test. Right- Immunoblot of Mge1-FLAG and MTSSu9-Mge1-FLAG under control or import stress (PSD1 overexpression for 4 h) conditions. Addback of Mge1-mCherry into the MTSSu9-MGE1 background is also presented. p1, Mge1-FLAG precursor; p2, MTSSu9-Mge1-FLAG precursor; p3, Mge1-mCherry precursor; m1/2, mature Mge1-FLAG/MTSSu9-Mge1-FLAG; m3, mature Mge1-mCherry. OE, overexpression. (i) CIS1 and PSD1 mRNA levels in cell expressing MTSIlv2-MGE1-FLAG and MGE1-FRB with or without the plasma membrane (PM) anchor Pil1-FKBP. Cells were grown under control or import stress (PSD1 overexpression for 4 h) conditions. Anchoring of the Mge1 precursor was induced by adding 50 nM rapamycin for the duration of PSD1 overexpression. n = 3 biological replicates; one-way ANOVA followed by Tukey’s test. (j) Immunoblot of Mge1-FLAG and MTSIlv2-Mge1-FLAG in wild-type cells, rho− cells, tam41∆ cells (at both 30 °C and 37 °C), and cells expressing aac2A128P, A137D for 4 h. p1, Mge1-FLAG precursor; p2, MTSIlv2-Mge1-FLAG precursor; m1/2, mature form Mge1-FLAG/MTSIlv2-Mge1-FLAG. (d, g-i) Data represent mean +/− SD; ns, not significant; **** P ≤ 0.0001.
Extended Data Fig. 7 The N-terminus of Mge1 contains critical features for the activation of the mitoCPR.
(a) Amino acid alignment and sequence conservation of Mge1 across 10 Saccharomyces species. (b) Immunoblot of overexpressed full-length and truncated Mge1-mCherry variants (4 h galactose induction). OE, overexpression. (c) In vitro protein import assay using mitochondria isolated from control cells (empty vector) and cells overexpressing MTSMge1(1-20)-mCherry for 4 h. Mitochondria were incubated with radiolabeled 35S-Zim17 for the indicated durations. Membrane potential (Δψ) was dissipated by preincubation with antimycin A, oligomycin, and valinomycin. Quantification from three biological replicates is shown in the bottom panel; protein levels imported into control mitochondria at the final time point were set to 100%. p, precursor; m, mature. OE, overexpression. (d) Growth curves of cells overexpressing MTS116-mCherry or MTSMge1(1-20)-mCherry, in the presence or absence of PDR3, were plotted on a semilogarithmic scale. Corresponding growth rate (r) was calculated from the growth curves of three biological replicates, each with three technical replicates; two-tailed Student’s t-test. OE, overexpression. (e) Immunoblot of overexpressed MTS116-mCherry and MTSMge1(1-20)-mCherry 4 h following galactose addition. OE, overexpression. (f) Upper panel- Immunoblot of the endogenously expressed addbacks (full-length Mge1-mCherry, MTSMge1-mCherry, and MTSMge1(1-20)-mCherry) into the MTSSU9-MGE1-FLAG background. Lower panel- Confirmation of PSD1 overexpression by qPCR analysis for the samples used in the upper panel and in Fig. 5c. n = 3 biological replicates; one-way ANOVA followed by Tukey’s test. OE, overexpression. (g) The electrostatic surface potential plot shows that the presequence of Mge1(residues 1–60, in green) fits into a negatively charged patch in Pdr3 (residues 86–856). (h-i) Heatmap of the amino acid enrichment across the first twenty amino acid of the MTSs from all yeast mitochondrial proteins (h) or previously reported nuclear-localized mitochondrial proteins (i). The enrichment score represents the frequency of a residue at a given position across the set weighted by the genome-wide amino acid frequencies. A table listing the N-terminal twenty amino acids of the mitochondrial presequences used in (i) is shown on the right. (j) Immunoblot of FLAG-tagged wild-type and mutants Mge1, expressed from the native MGE1 promoter, in the MTSSU9-MGE1-FLAG background. The asterisk indicates a likely degradation product. p1, MTSSu9-Mge1-FLAG precursor; p2, Mge1-FLAG variants precursor; m1/2, mature form MTSSu9-Mge1-FLAG/Mge1-FLAG variants. OE, overexpression. (k) Confirmation of PSD1 overexpression by qPCR analysis for the samples used in Fig. 5f. n = 3 biological replicates; one-way ANOVA followed by Tukey’s test. OE, overexpression. (c, d, f, k) Data represent mean +/− SD; **** P ≤ 0.0001.
Supplementary information
Supplementary Figure 1 (download PDF )
Verification of yeast strains
Supplementary Figure 2 (download PDF )
Raw blots and gels
Supplementary Tables 1–6 (download ZIP )
Supplementary Table 1: Results of the overexpression screen for mitoCPR activators. Supplementary Table 2: Pdr3 co-IP mass spectrometry data Supplementary Table 3: Differential gene expression analysis for MTSMge1 overexpression/MTSMss116 overexpression. Supplementary Table 4: Differential gene expression analysis for MTSMge1 overexpression/ pdr3∆ MTSMge1 overexpression. Supplementary Table 5: Differential gene expression analysis for PSD1 overexpression/ pdr3∆ PSD1 overexpression. Supplementary Table 6: Yeast strains and oligonucleotides used in this study
Supplementary Data (source data supplementary figure 1) (download XLSX )
Source data for supplementary figure 1
Source data
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Yuan, Z., Balzarini, M., Volpe, M. et al. A direct role for a mitochondrial targeting sequence in signalling stress. Nature 649, 1302–1311 (2026). https://doi.org/10.1038/s41586-025-09834-x
Received:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41586-025-09834-x