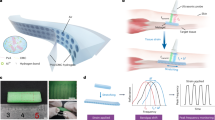
Abstract
Monitoring internal physiological signals is essential for effective medical care1, yet most current technologies rely on external measurements or imaging systems that cannot capture enough deep-tissue dynamics2,3,4,5,6. Implantable devices offer a solution, but conventional designs often require batteries or magnets7,8,9,10,11, which carry risks during removal, and existing biodegradable sensors based on passive inductor–capacitor circuits are limited by short readout distances and unstable communication issues12,13,14,15,16,17,18,19. Here we describe a soft, biodegradable, wireless sensing device that can monitor pressure, temperature and strain over long distances (up to 16 cm), maintaining accuracy across varying positions and angles. This is achieved through a ‘pole-moving sweeping’ readout system combined with a folded structure that integrates mechanical flexibility with electromagnetic function. In vivo tests in the abdominal cavity of horses reliably captured deep-tissue pressure and temperature, and ex vivo measurements demonstrated accurate strain monitoring without strict positional control. The long-distance and wide-angle readout of soft biodegradable implants holds translational promise for accessing deep-tissue physiological signals.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$32.99 / 30 days
cancel any time
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
Data availability
The data that support the findings of this study are available in the paper, Supplementary Information and source data. Source data are provided with this paper.
Code availability
Custom codes for collection of the phase–frequency response data are available at GitHub (https://github.com/lanyuqun/Long-distance-wireless-sensing-platform).
References
Lin, M. Y., Hu, H. J., Zhou, S. & Xu, S. Soft wearable devices for deep-tissue sensing. Nat. Rev. Mater. 7, 850–869 (2022).
Suetens, P. Fundamentals of Medical Imaging 3rd edn (Cambridge Univ. Press, 2017).
Hu, H. J. et al. A wearable cardiac ultrasound imager. Nature 613, 667–675 (2023).
Lin, M. Y. et al. A fully integrated wearable ultrasound system to monitor deep tissues in moving subjects. Nat. Biotechnol. 42, 448–457 (2024).
Du, W. Y. et al. Conformable ultrasound breast patch for deep tissue scanning and imaging. Sci. Adv. 9, eadh5325 (2023).
Zhou, S. et al. Transcranial volumetric imaging using a conformal ultrasound patch. Nature 629, 810–818 (2024).
Kwon, K. et al. A battery-less wireless implant for the continuous monitoring of vascular pressure, flow rate and temperature. Nat. Biomed. Eng. 7, 1215–1228 (2023).
Ouyang, W. et al. A wireless and battery-less implant for multimodal closed-loop neuromodulation in small animals. Nat. Biomed. Eng. 7, 1252–1269 (2023).
Song, J. W. et al. Bioresorbable, wireless, and battery-free system for electrotherapy and impedance sensing at wound sites. Sci. Adv. 9, eade4687 (2023).
Gleich, B., Schmale, I., Nielsen, T. & Rahmer, J. Miniature magneto-mechanical resonators for wireless tracking and sensing. Science 380, 966–971 (2023).
Wan, J. et al. Millimeter-scale magnetic implants paired with a fully integrated wearable device for wireless biophysical and biochemical sensing. Sci. Adv. 10, eadm9314 (2024).
Lu, D. et al. Bioresorbable Wireless Sensors as Temporary Implants for In Vivo Measurements of Pressure. Adv. Funct. Mater. 30, 2003754 (2020).
Boutry, C. M. et al. Biodegradable and flexible arterial-pulse sensor for the wireless monitoring of blood flow. Nat. Biomed. Eng. 3, 47–57 (2019).
Lu, D. et al. Bioresorbable, wireless, passive sensors as temporary implants for monitoring regional body temperature. Adv. Healthcare Mater. 9, 2000942 (2020).
Li, S. et al. Bioresorbable, wireless, passive sensors for continuous pH measurements and early detection of gastric leakage. Sci. Adv. 10, eadj0268 (2024).
Collins, C. C. Miniature passive pressure transensor for implanting in the eye. IEEE Trans. Biomed. Eng. BME-14, 74–83 (1967).
Huang, X. et al. Materials and designs for wireless epidermal sensors of hydration and strain. Adv. Funct. Mater. 24, 3846–3854 (2014).
Lee, J. et al. Flexible, sticky, and biodegradable wireless device for drug delivery to brain tumors. Nat. Commun. 10, 5205 (2019).
Herbert, R., Lim, H. R., Rigo, B. & Yeo, W. H. Fully implantable wireless batteryless vascular electronics with printed soft sensors for multiplex sensing of hemodynamics. Sci. Adv. 8, eabm1175 (2022).
Madhvapathy, S. R. et al. Implantable bioelectronic systems for early detection of kidney transplant rejection. Science 381, 1105–1112 (2023).
Kim, Y. et al. Chip-less wireless electronic skins by remote epitaxial freestanding compound semiconductors. Science 377, 859–869 (2022).
Assawaworrarit, S., Yu, X. F. & Fan, S. H. Robust wireless power transfer using a nonlinear parity-time-symmetric circuit. Nature 546, 387–390 (2017).
Chen, P. Y. et al. Generalized parity-time symmetry condition for enhanced sensor telemetry. Nat. Electron. 1, 297–304 (2018).
Dong, Z. Y., Li, Z. P., Yang, F. Y., Qiu, C. W. & Ho, J. S. Sensitive readout of implantable microsensors using a wireless system locked to an exceptional point. Nat. Electron. 2, 335–342 (2019).
Bogatin, E. Signal and Power Integrity - Simplified 2nd edn (Prentice Hall, 2010).
Kurs, A. et al. Wireless power transfer via strongly coupled magnetic resonances. Science 317, 83–86 (2007).
Haus, H. A. Waves and Fields in Optoelectronics (Prentice Hall, 1984).
Karalis, A., Joannopoulos, J. D. & Soljacic, M. Efficient wireless non-radiative mid-range energy transfer. Ann. Phys. 323, 34–48 (2008).
Timoshenko, S. Strength of Materials (CBS, 1986).
Kim, D.-H. et al. Epidermal electronics. Science 333, 838–843 (2011).
Gyawali, D., Tran, R. T., Guleserian, K. J., Tang, L. & Yang, J. Citric-acid-derived photo-cross-linked biodegradable elastomers. J. Biomater. Sci. Polym. Ed. 21, 1761–1782 (2010).
Hwang, S. W. et al. High-performance biodegradable/transient electronics on biodegradable polymers. Adv. Mater. 26, 3905–3911 (2014).
Wang, Y. D., Ameer, G. A., Sheppard, B. J. & Langer, R. A tough biodegradable elastomer. Nat. Biotechnol. 20, 602–606 (2002).
Choi, Y. S. et al. Biodegradable polyanhydrides as encapsulation layers for transient electronics. Adv. Funct. Mater. 30, 2000941 (2020).
Boutry, C. M. et al. A stretchable and biodegradable strain and pressure sensor for orthopaedic application. Nat. Electron. 1, 314–321 (2018).
Yuk, H. et al. Dry double-sided tape for adhesion of wet tissues and devices. Nature 575, 169–174 (2019).
Boutry, C. M. et al. A sensitive and biodegradable pressure sensor array for cardiovascular monitoring. Adv. Mater. 27, 6954–6961 (2015).
Xiong, Y. X. et al. A flexible, ultra-highly sensitive and stable capacitive pressure sensor with convex microarrays for motion and health monitoring. Nano Energy 70, 104436 (2020).
Yang, Q. S. et al. High-speed, scanned laser structuring of multi-layered eco/bioresorbable materials for advanced electronic systems. Nat. Commun. 13, 6518 (2022).
Acknowledgements
We thank S. Xie, J. Yang, X. Wei, Q. Zhou, R. Ding, Q. Li, K. Chen, J. Du, D. Jiang and M. Wang for providing technical support. We also thank the Advanced Research and Testing Laboratory, Center of Nanofabrication, Tsinghua University and Keysight Technologies for providing technical support for the impedance analyser. We acknowledge the support from the National Natural Science Foundation of China (12172359, 11921002, 12032014, 61421002 and T2488101). Y.S. acknowledges the support from the Key Research Program of Frontier Sciences of the Chinese Academy of Sciences (ZDBS-LY-JSC014) and the CAS Interdisciplinary Innovation Team (JCTD-2020-03). X.Y. acknowledges the support from the Research Grants Council of the Hong Kong Special Administrative Region (11213721, 11215722, 11211523 and RFS2324-1S03) and the City University of Hong Kong (9229055, 9610444, 9678274 and 9680322).
Author information
Authors and Affiliations
Contributions
S.L., Y.L. and Y.S. conceptualized the study. Y.L., S.L., Y.S. and X.Y. developed the methodology. Y.L. wrote the software. Y.L. and S.L. validated the results. Y.L. and S.L. conducted formal analysis. Y.L., S.L., H. Guo, Q.L., T.W., L.Z., Y. Zhao, Y. Zhu, Jing Li, Z.Z., Q.W., R.S., X.W., X.X., Yuhong Wu, Z.W., B.L., Jiaqi Li, H.L. and H. Gao performed the investigation. Y.S., Q.G., Yuchen Wu, S.L. and H. Gao provided the resources. Y.L. and S.L. curated the data. Y.L. and S.L. wrote the original draft. S.L., Y.S., X.Y. and X.-Q.F. reviewed and edited the manuscript. Y.L., J.F. and S.L. created the visualizations. Y.S., S.L. and X.Y. supervised the project. Y.S., X.-Q.F. and X.Y. acquired the funding.
Corresponding authors
Ethics declarations
Competing interests
S.L. and X.-Q.F. are inventors on a patent application (CN202311773783.2) that describes an early version of the readout system. Y.S., Y.L. and S.L. are inventors on two other patent applications (CN202511447742.3 and CN202511447887.3) that are more closely related to the readout system and sensor design reported in this study. All patents are pending. The other authors declare no competing interests.
Peer review
Peer review information
Nature thanks Ada Shuk Yan Poon and the other, anonymous, reviewer(s) for their contribution to the peer review of this work. Peer reviewer reports are available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data figures and tables
Extended Data Fig. 1 Effect of distance and angle on the coupling coefficient of two coupled coils.
a, Schematic diagram of the studied distance l and angle α. b, Finite element results of coupling coefficients with distance and angle. The effect of distance l is analyzed for a fixed angle α = 0° and the effect of angle α is analyzed for a fixed distance l = 50 mm.
Extended Data Fig. 2 Theoretical phase diagram analysis and examples of our readout system.
a, Phase diagram of the derivatives of the phase-frequency curves for different coupling coefficients k and frequency f. b, Examples of the phase-frequency curves with different coupling coefficients k.
Extended Data Fig. 3
Different methods of passive wireless sensing based on LC circuits.
Extended Data Fig. 4 Comparison of DP, PT-symmetric, and EP-locked systems.
The pole-zero plots and the phase-frequency curves of a,d, DP system (\({\gamma }_{{\rm{r}}}={\gamma }_{{\rm{s}}}=0.01\), ωr = 1), b,e, PT-symmetric system (\({\gamma }_{{\rm{r}}}\to -{g}_{1}=-\,{\gamma }_{{\rm{s}}}-0.01\), ωr = 1) and c,f, EP-locked system (ωr1 = ωr2 = 1, \({g}_{{\rm{r}}1}={\gamma }_{{\rm{r}}2}=\mu \) = 0.95) under the same parameters κ = 0.003, ωs = 1. g-k, S11 of different systems, with the parameters of each system corresponding to Fig. 2e,f and above d-f. l,m, The phase-frequency curves of our system and EP-locked system for κ = 0.0001 and κ = 0 (without sensors) at resonance. Other parameters: ωr1 = ωr2 = 1, \({g}_{{\rm{r}}1}={\gamma }_{{\rm{r}}2}=\mu =0.95\). n, The zoomed-in view of m.
Extended Data Fig. 5 Coupling of the copper-coil reader and copper-coil “sensor”.
a, Photographs of the reader coil and sensor coil. b, Illustration of the geometric parameters. c, COMSOL simulation results of the coupling coefficient k as a function of distance. d, Relationship between the program instructions (code) and output voltage of digital-to-analogue converter. e, Relationship between the output voltage of digital-to-analogue converter and the resonant frequency of reader. f, Measured results at different distances with an angle of 0 degrees. g, Measured results at different angles with a distance of 10 cm.
Extended Data Fig. 6
Folding process of capacitor’s electrodes.
Extended Data Fig. 7 Geometric details and experimental results on distance and angle insensitivity of the pressure sensor.
a, Planar dimensional diagram of the pressure sensor. b, Explosion diagram of the pressure sensor. c, The measured frequency change of the pressure sensor caused by the distance variation from 5 cm to 16 cm. d, The corresponding phase-frequency curve. e, The allowable ranges of instrument accuracy and readout distance for our readout system and the standard readout system under a 1% total error requirement. f, Comparison of signals between our readout system and the standard readout system at a distance of 7 cm and an angle of 0 degrees.
Extended Data Fig. 8 Geometric details of the temperature sensor.
a, Planar dimensional diagram of the temperature sensor. b, Explosion diagram of the temperature sensor.
Supplementary information
Supplementary Information (download PDF )
This file contains Supplementary Notes 1–15, Supplementary Figs. 1–36 and Supplementary References.
Supplementary Video 1 (download MP4 )
Peristalsis of horse’s intestine, procedure of suturing the sensor and signal measurement (MP4).
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Lan, Y., Li, S., Guo, H. et al. Soft biodegradable implants for long-distance and wide-angle sensing. Nature 649, 366–374 (2026). https://doi.org/10.1038/s41586-025-09874-3
Received:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41586-025-09874-3