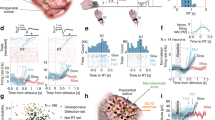
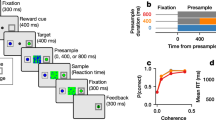
Abstract
We use our experiences to form and update beliefs about the hidden states of the world1,2,3. When possible, we also gather evidence by observing others. However, how the brain integrates experiential and observational evidence is not understood. We studied the dynamics of evidence integration in a two-player game with volatile hidden states. Both humans and monkeys successfully updated their beliefs while playing the game and observing their partner, although less effectively when observing. Electrophysiological recordings in animals revealed that the anterior cingulate cortex integrates independent sources of experiential and observational evidence into a coherent neural representation of dynamic belief about the environment’s state. The geometry of population activity revealed the computational architecture of this integration and provided a neural account of the behavioural asymmetry between experiential and observational evidence accumulation. This work lays the groundwork for understanding the neural mechanisms underlying evidence accumulation in social contexts in the primate brain.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$32.99 / 30 days
cancel any time
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
Data availability
The data are available via DANDI at https://dandiarchive.org/dandiset/001435. Source data are provided with this paper.
Code availability
The code for analysis in this paper is available via GitHub at https://github.com/jazlab/RC_SR_NV_SBY_MJ_2025.
References
Hayden, B. Y., Pearson, J. M. & Platt, M. L. Neuronal basis of sequential foraging decisions in a patchy environment. Nat. Neurosci. 14, 933–939 (2011).
Shenhav, A., Botvinick, M. M. & Cohen, J. D. The expected value of control: an integrative theory of anterior cingulate cortex function. Neuron 79, 217–240 (2013).
Sarafyazd, M. & Jazayeri, M. Hierarchical reasoning by neural circuits in the frontal cortex. Science 364, eaav8911 (2019).
Carter, C. et al. Anterior cingulate cortex, error detection, and the online monitoring of performance. Science 280, 747–749 (1998).
Ito, S., Stuphorn, V., Brown, J. W. & Schall, J. D. Performance monitoring by the anterior cingulate cortex during saccade countermanding. Science 302, 120–122 (2003).
Seo, H. & Lee, D. Temporal filtering of reward signals in the dorsal anterior cingulate cortex during a mixed-strategy game. J. Neurosci. 27, 8366–8377 (2007).
Monosov, I. E. Anterior cingulate is a source of valence-specific information about value and uncertainty. Nat. Commun. 8, 1–12 (2017).
Kennerley, S. W., Behrens, T. E. J. & Wallis, J. D. Double dissociation of value computations in orbitofrontal and anterior cingulate neurons. Nat. Neurosci. 14, 1581–1589 (2011).
Kennerley, S. W. & Wallis, J. D. Evaluating choices by single neurons in the frontal lobe: outcome value encoded across multiple decision variables. Eur. J. Neurosci. 29, 2061–2073 (2009).
Shima, K. & Tanji, J. Role for cingulate motor area cells in voluntary movement selection based on reward. Science 282, 1335–1338 (1998).
Amiez, C., Joseph, J.-P. & Procyk, E. Anterior cingulate error-related activity is modulated by predicted reward. Eur. J. Neurosci. 21, 3447–3452 (2005).
Hadland, K. A., Rushworth, M. F. S., Gaffan, D. & Passingham, R. E. The anterior cingulate and reward-guided selection of actions. J. Neurophysiol. 89, 1161–1164 (2003).
Williams, Z. M., Bush, G., Rauch, S. L., Cosgrove, G. R. & Eskandar, E. N. Human anterior cingulate neurons and the integration of monetary reward with motor responses. Nat. Neurosci. 7, 1370–1375 (2004).
Rushworth, M. F. S. & Behrens, T. E. J. Choice, uncertainty and value in prefrontal and cingulate cortex. Nat. Neurosci. 11, 389–397 (2008).
Akam, T. et al. The anterior cingulate cortex predicts future states to mediate model-based action selection. Neuron 109, 149–163 (2021).
Vertechi, P. et al. Inference-based decisions in a hidden state foraging task: differential contributions of prefrontal cortical areas. Neuron 106, 166–176 (2020).
Shidara, M. & Richmond, B. J. Anterior cingulate: single neuronal signals related to degree of reward expectancy. Science 296, 1709–1711 (2002).
Narayanan, N. S., Cavanagh, J. F., Frank, M. J. & Laubach, M. Common medial frontal mechanisms of adaptive control in humans and rodents. Nat. Neurosci. 16, 1888–1895 (2013).
Quilodran, R., Rothé, M. & Procyk, E. Behavioral shifts and action valuation in the anterior cingulate cortex. Neuron 57, 314–325 (2008).
Tervo, D. G. R. et al. The anterior cingulate cortex directs exploration of alternative strategies. Neuron 109, 1876–1887 (2021).
Tervo, D. G. R. et al. Behavioral variability through stochastic choice and its gating by anterior cingulate cortex. Cell 159, 21–32 (2014).
Bernacchia, A., Seo, H., Lee, D. & Wang, X.-J. A reservoir of time constants for memory traces in cortical neurons. Nat. Neurosci. 14, 366–372 (2011).
Kerns, J. G. et al. Anterior cingulate conflict monitoring and adjustments in control. Science 303, 1023–1026 (2004).
Gu, B.-M. et al. Neural correlates of cognitive inflexibility during task-switching in obsessive-compulsive disorder. Brain 131, 155–164 (2008).
Kennerley, S. W., Walton, M. E., Behrens, T. E. J., Buckley, M. J. & Rushworth, M. F. S. Optimal decision making and the anterior cingulate cortex. Nat. Neurosci. 9, 940–947 (2006).
Chen, W., Liang, J., Wu, Q. & Han, Y. Anterior cingulate cortex provides the neural substrates for feedback-driven iteration of decision and value representation. Nat. Commun. 15, 6020 (2024).
Joiner, J., Piva, M., Turrin, C. & Chang, S. W. C. Social learning through prediction error in the brain. NPJ Sci. Learn. 2, 8 (2017).
Grabenhorst, F., Báez-Mendoza, R., Genest, W., Deco, G. & Schultz, W. Primate amygdala neurons simulate decision processes of social partners. Cell 177, 986–998 (2019).
Báez-Mendoza, R., Harris, C. J. & Schultz, W. Activity of striatal neurons reflects social action and own reward. Proc. Natl Acad. Sci. USA 110, 16634–16639 (2013).
Burke, C. J., Tobler, P. N., Baddeley, M. & Schultz, W. Neural mechanisms of observational learning. Proc. Natl Acad. Sci. USA 107, 14431–14436 (2010).
Cooper, J. C., Dunne, S., Furey, T. & O’Doherty, J. P. Human dorsal striatum encodes prediction errors during observational learning of instrumental actions. J. Cogn. Neurosci. 24, 106–118 (2012).
Azzi, J. C. B., Sirigu, A. & Duhamel, J.-R. Modulation of value representation by social context in the primate orbitofrontal cortex. Proc. Natl Acad. Sci. USA 109, 2126–2131 (2012).
Jeon, D. et al. Observational fear learning involves affective pain system and Cav1.2 Ca2+ channels in ACC. Nat. Neurosci. 13, 482–488 (2010).
Gariépy, J.-F. et al. Social learning in humans and other animals. Front. Neurosci. 8, 58 (2014).
de Araujo, M. F. P. et al. Neuronal activity of the anterior cingulate cortex during an observation-based decision making task in monkeys. Behav. Brain Res. 230, 48–61 (2012).
Hill, M. R., Boorman, E. D. & Fried, I. Observational learning computations in neurons of the human anterior cingulate cortex. Nat. Commun. 7, 12722 (2016).
Allsop, S. A. et al. Corticoamygdala transfer of socially derived information gates observational learning. Cell 173, 1329–1342 (2018).
Huang, Z. et al. Ventromedial prefrontal neurons represent self-states shaped by vicarious fear in male mice. Nat. Commun. 14, 3458 (2023).
Yoshida, K., Saito, N., Iriki, A. & Isoda, M. Social error monitoring in macaque frontal cortex. Nat. Neurosci. 15, 1307–1312 (2012).
Chang, S. W. C., Gariépy, J.-F. & Platt, M. L. Neuronal reference frames for social decisions in primate frontal cortex. Nat. Neurosci. 16, 243–250 (2013).
Basile, B. M., Schafroth, J. L., Karaskiewicz, C. L., Chang, S. W. C. & Murray, E. A. The anterior cingulate cortex is necessary for forming prosocial preferences from vicarious reinforcement in monkeys. PLoS Biol. 18, e3000677 (2020).
Hayden, B. Y., Pearson, J. M. & Platt, M. L. Fictive reward signals in the anterior cingulate cortex. Science 324, 948–950 (2009).
Bellebaum, C., Jokisch, D., Gizewski, E. R., Forsting, M. & Daum, I. The neural coding of expected and unexpected monetary performance outcomes: dissociations between active and observational learning. Behav. Brain Res 227, 241–251 (2012).
Morin, O., Jacquet, P. O., Vaesen, K. & Acerbi, A. Social information use and social information waste. Philos. Trans. R. Soc. Lond. B Biol. Sci. 376, 20200052 (2021).
Nicolle, A., Symmonds, M. & Dolan, R. J. Optimistic biases in observational learning of value. Cognition 119, 394–402 (2011).
Rigotti, M. et al. The importance of mixed selectivity in complex cognitive tasks. Nature 497, 585–590 (2013).
Mante, V., Sussillo, D., Shenoy, K. V. & Newsome, W. T. Context-dependent computation by recurrent dynamics in prefrontal cortex. Nature 503, 78–84 (2013).
Ebitz, R. B. & Hayden, B. Y. The population doctrine in cognitive neuroscience. Neuron 109, 3055–3068 (2021).
Elsayed, G. F., Lara, A. H., Kaufman, M. T., Churchland, M. M. & Cunningham, J. P. Reorganization between preparatory and movement population responses in motor cortex. Nat. Commun. 7, 13239 (2016).
Johnston, W. J., Fine, J. M., Yoo, S. B. M., Ebitz, R. B. & Hayden, B. Y. Semi-orthogonal subspaces for value mediate a binding and generalization trade-off. Nat. Neurosci. 27, 2218–2230 (2024).
Flesch, T., Juechems, K., Dumbalska, T., Saxe, A. & Summerfield, C. Orthogonal representations for robust context-dependent task performance in brains and neural networks. Neuron 110, 4212–4219 (2022).
Tang, C., Herikstad, R., Parthasarathy, A., Libedinsky, C. & Yen, S.-C. Minimally dependent activity subspaces for working memory and motor preparation in the lateral prefrontal cortex. eLife 9, e58154 (2020).
Bernardi, S. et al. The geometry of abstraction in the hippocampus and prefrontal cortex. Cell 183, 954–967 (2020).
Remington, E. D., Narain, D., Hosseini, E. A. & Jazayeri, M. Flexible sensorimotor computations through rapid reconfiguration of cortical dynamics. Neuron 98, 1005–1019 (2018).
Lindsey, J. W. & Issa, E. B. Factorized visual representations in the primate visual system and deep neural networks. eLife 13, RP91685 (2024).
Ito, T. et al. Compositional generalization through abstract representations in human and artificial neural networks. In Proc. 36th International Conference on Neural Information Processing Systems (eds Koyejo, S. et al.) 32225–32239 (ACM, 2022).
Johnston, W. J. & Fusi, S. Abstract representations emerge naturally in neural networks trained to perform multiple tasks. Nat. Commun. 14, 1040 (2023).
Daie, K., Fontolan, L., Druckmann, S. & Svoboda, K. Feedforward amplification in recurrent networks underlies paradoxical neural coding. Preprint at bioRxiv https://doi.org/10.1101/2023.08.04.552026 (2023).
Ganguli, S. et al. One-dimensional dynamics of attention and decision making in LIP. Neuron 58, 15–25 (2008).
Brown, J. W. & Alexander, W. H. Foraging value, risk avoidance, and multiple control signals: how the anterior cingulate cortex controls value-based decision-making. J. Cogn. Neurosci. 29, 1656–1673 (2017).
Vassena, E., Holroyd, C. B. & Alexander, W. H. Computational models of anterior cingulate cortex: at the crossroads between prediction and effort. Front. Neurosci. 11, 316 (2017).
Mardinly, A. R. et al. Precise multimodal optical control of neural ensemble activity. Nat. Neurosci. 21, 881–893 (2018).
Clark, A. M. et al. An optrode array for spatiotemporally-precise large-scale optogenetic stimulation of deep cortical layers in non-human primates. Commun. Biol. 7, 329 (2024).
Russell, L. E. et al. All-optical interrogation of neural circuits in behaving mice. Nat. Protoc. 17, 1579–1620 (2022).
Watters, N., Tenenbaum, J. & Jazayeri, M. Modular object-oriented games: a task framework for reinforcement learning, psychology, and neuroscience. Preprint at https://arxiv.org/abs/2102.12616 (2021).
Acknowledgements
R.C. was supported by the Simons Center for the Social Brain at MIT and Hock E. Tan and K. Lisa Yang Center for Autism Research. S.R. was supported by a Mathworks Graduate Fellowship and K. Lisa Yang ICoN Center Fellowship. S.B.M.Y. was supported by the Simons Center for the Social Brain at MIT. M.J. was supported by the Simons Foundation and the McGovern Institute. We thank J. Gabel, N. Watters and A. Piccato for their respective help with electrophysiology, modelling and open-sourcing.
Author information
Authors and Affiliations
Contributions
S.R., S.B.M.Y. and M.J. designed the task. S.B.M.Y. trained the animals. R.C. and N.V. performed animal experiments. S.R. performed human experiments. R.C., S.R. and N.V. analysed the data. R.C., S.R. and M.J. wrote the manuscript. M.J. supervised the project.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature thanks Kayvon Daie and the other, anonymous, reviewer(s) for their contribution to the peer review of this work. Peer reviewer reports are available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information (download PDF )
Supplementary Figs. 1–15 and Table 1.
Supplementary Data (download ZIP )
Source data for Supplementary Figs. 1–8 and 10–15.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Chen, R., Radkani, S., Valluru, N. et al. Evidence accumulation from experience and observation in the cingulate cortex. Nature 650, 681–689 (2026). https://doi.org/10.1038/s41586-025-09885-0
Received:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41586-025-09885-0