Abstract
Caribbean reefs have experienced major human-driven changes to their coral and fish communities1,2,3,4, yet how these changes have affected trophic dynamics remains poorly understood owing to challenges in reconstructing the trophic structure of pre-human-impact reefs. Advances in fossil-bound protein nitrogen isotope (15N/14N) analysis now enable the reconstruction of ancient trophic dynamics5,6, as the 15N to 14N ratio reflects an animal’s trophic position7. Here we apply this method to modern and prehistoric (7,000-year-old) fish otoliths (ear stones) and corals from Caribbean Panama and the Dominican Republic, focusing on fishes occupying low to middle trophic levels. We find that although the trophic level typically declined in high-trophic-level fishes over time, it increased or remained unchanged in low-trophic-level fishes, indicating that modern food chains are 60–70% shorter than on the prehistoric reefs in both Panama and the Dominican Republic. Furthermore, across all trophic groups, we observed a marked reduction in dietary variation, with a 20–70% lower trophic range on the modern reefs compared to the prehistoric reefs. This pattern is best explained by less dietary specialization in modern reefs, consistent with less ecological complexity than in prehistoric reefs. These differences document and quantify the trophic simplification that has occurred on modern Caribbean reefs, a change that may increase their vulnerability to ecosystem collapse.
Similar content being viewed by others
Main
Coral reefs are among the most vital marine ecosystems in low-latitude regions. These biodiverse environments provide essential habitats for at least 25% of marine species, including many reef fishes8. Moreover, the health of coral reefs is closely linked to the health, storm protection and nutrition of approximately one billion people—about 13% of the global population—who rely on reefs owing to proximity (that is, living within 100 km of coral reefs)9. Yet global coral cover across reef ecosystems has declined owing to climate change, eutrophication, overfishing and disease1,2. For instance, average stony coral cover in the Caribbean has decreased by 50% since the 1970s3, leading to a shift from coral- to algae-dominated ecosystems.
One crucial aspect of coral reef biodiversity and resilience is trophic diversity—the range and specialization of feeding roles within the community. Reef fishes perform critical and varied ecological functions, including ectoparasite cleaning, algae farming, symbiotic relationships with invertebrates and poaching10,11. Even in fishes considered generalists, high-resolution studies show intraspecific prey specialization12,13,14. This wide range of trophic interactions in these ecosystems underpins their extraordinary biodiversity and productivity15,16 and thus supports the services that they provide17.
Despite well-documented losses in coral cover and fish biomass, it remains unclear whether the diets of fish communities have been altered by ecological degradation of coral reefs. Many of the impacts leading to coral reef ecosystem change, including overfishing, eutrophication and shifts in coral community composition, began before modern recordkeeping3,4. Thus, reconstructing pre-human trophic structures is critical for understanding whether and how human activities have altered energy flow in these and other marine ecosystems compared to their historical baselines.
Stable isotopes are commonly used in modern ecological studies as tracers of trophic structure and energy flow7,18. Recent methodological advancements5,6,19 have enabled isotopic analyses of organic nitrogen in proteins bound within the fossil skeletons of diverse organisms, including fish otoliths, coral, foraminifera and teeth. These innovations make it feasible to reconstruct past marine food web structure and trophic diversity using an ecosystem-based nitrogen-isotope approach, particularly when multiple taxa co-exist at a given fossil reef site. Otoliths—ear stones involved in vestibular function in bony fishes—have taxon-specific shapes and are composed of calcium carbonate (>99.6% by mass) and organic constituents (<0.4%)20 (Supplementary Methods). In fossil and subfossil specimens, the mineral lattice protects the organic constituents from factors leading to poor preservation. The high-sensitivity otolith-bound nitrogen isotope (δ15Noto, where δ15N = [(15N/14Nsample)/(15N/14Nair) – 1] × 1,000) approach enables individual- and community-level δ15N analyses from fossil specimens but has only begun to be applied to study ecosystems5,21. The δ15N patterns in otoliths—as in muscle tissue—convey information about the proportional contribution of food items with distinct isotopic values (Methods).
Here we measure the δ15N of fossil-bound organic matter in ~7,000-year-old Caribbean coral reef deposits (Fig. 1), one in Panama (Bocas del Toro) and the other in the Dominican Republic (Enriquillo Basin), to reconstruct coral reef trophic structure before widespread human impacts for comparison with the trophic structure of nearby modern reefs. Fish otoliths and corals are preserved within the coral reef sediments at these sites22, alongside other biogenic hard parts (for example, spines, denticles, teeth, spicules, shells) from diverse organisms such as molluscs, urchins, sharks and sponges4. We focus on coral (Porites furcata; family Poritidae) and four fish families (Fig. 2a), gobies (Gobiidae), cardinalfishes (Apogonidae), silversides (Atherinidae) and grunts (Haemulidae), all of which are important prey fish. As prey fish otoliths are primarily deposited onto reef sediments through predator excretion23, otolith abundances reflect the relative contributions of different fishes to energy flow in the overlying food web averaged over the timescale of sedimentary deposition. These families were therefore chosen for their high abundances in sedimentary deposits as well as their distinct ecological roles. Moreover, apart from grunts, these families are not routinely targeted by fisheries, reducing fishing-driven biases in the dataset24.
a, Caribbean regional map and sampling locations within Bocas del Toro, Panama (southwest Caribbean, left) and the Dominican Republic (eastern Caribbean, right) (refer to Extended Data Fig. 1 and Supplementary Table 3 for detailed locality information for modern (red) and fossil (blue) sites). b, Sediment sampling of the coral reef framework in modern and fossil coral reefs. c, Assorted specimens of coral reef matrix-sourced fossil fish otoliths viewed under a dissecting microscope and inset showing example scanning electron microscopy (SEM) images of the cleaned otoliths and coral skeletal material. From top to bottom in the inset we show representative otoliths from each family (Fig. 2a) in the current study: grunts (Haemulidae), cardinalfishes (Apogonidae), silversides (Atherinidae) and gobies (Gobiidae), with the bottom-most image showing fragments of branching finger coral (Poritidae). Scale bars, 1 mm. Fossil and modern reef images in b and images of assorted specimens and otolith SEM in c reproduced from ref. 22; PLoS, under a CC0 1.0 Creative Commons license.
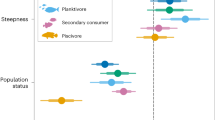
a, Descriptions of the taxa analysed in both time periods to generate an ecosystem nitrogen isotope distribution for coral reef trophic reconstruction. Top row: larger reef-associated fishes; middle rows, smaller reef-associated fishes; bottom row, primary consumers. b, Measurements of otolith- and coral-bound nitrogen isotopes (δ15N) for each fish family, where each symbol denotes an individual coral fragment or fish otolith. Each symbol represents the mean ± 1 s.d. of replicate measurements from individual otoliths or coral fragments. The n value below each category reflects the number of biologically independent individuals (coral fragments or fish) measured for each family, time period and region. c, Family-level patterns in δ15N (trophic level) for fossil and modern time periods (where n is the same as for b). Statistical tests for whether mean δ15N has changed since the mid-Holocene are shown below each fish silhouette (Wilcoxon, two-sided; *P < 0.05, **P < 0.01, ***P < 0.001). δ15N declined for Dominican Republic cardinalfishes (P = 2.5 × 10−4, W = 100, r = 0.873) and grunts (P = 0.048, W = 20, r = 0.82) (Extended Data Table 2). Data from Bocas del Toro, Panama, are shown on the left; data from the southeast Dominican Republic are shown on the right for b and c. d, Regional and family-level patterns in isotopic niche width (diet-driven δ15N diversity), calculated as the 1 s.d. of the individual-level measurements in b. The percent decline compared with the mid-Holocene niche width is shown above each modern bar. Statistical tests for whether the variances have changed since the mid-Holocene are shown below each fish silhouette (F-test, two-sided; *P < 0.05, **P < 0.01, ***P < 0.001). Panama gobies (P = 0.043, F = 3.0), silversides (P = 0.0042, F = 8.53) and grunts (P = 0.0004, F = 10.22) declined significantly (Extended Data Table 4). Grey arrows (from b, pointing towards c and d) indicate that the metrics shown in c (trophic position) and d (dietary diversity) are calculated from all data shown in b. Insets in each of c and d illustrate how each metric was calculated. Fish silhouettes in a–d reproduced from ref. 51, GitHub, under a GPL-2 license.
We use δ15N to quantify three complementary metrics: mean trophic level (MTL; the mean of δ15Noto measurements for a family or the whole community), isotopic niche width (as reflected by the standard deviation of δ15Noto measurements for a given family or the whole community25) and food chain length (FCL; the range in mean family δ15Noto) (Supplementary Fig. 1). Below, population refers to individual otoliths within a family, whereas community refers to the assemblage of the studied families. Together, these metrics trace fish behaviour, dietary diversity and energy-flow pathways across individuals (each otolith), families (means and variance across individuals within each family) and the partial communities (mean and variance across the families). As diets are averaged within individual fish, high levels of specialization—each fish relying on a distinct subset of prey—can yield relatively large within-family δ15N variance compared with prey δ15N variance14,25. For example, a large isotopic niche width within a given family, in this context, indicates dietary specialization at the level of individual fish. If fish engage in more generalist foraging, or if resource diversity (that is, the ‘menu’) has become more homogenous, then we would observe greater trophic similarity among individuals and a narrow within-family isotopic niche width12,25. On modern reefs, we predicted that coral loss, predator depletion and habitat fragmentation would reduce resource diversity and promote generalist foraging, leading to greater trophic similarity among individuals. If fish diets also become more similar among families, then FCL is expected to decrease. Ecological theory also predicts that decreased habitat connectivity should lead to decreased FCL26,27.
We analysed 136 fish otoliths and co-occurring corals from both fossil (mid-Holocene, 7,000 years ago; ka) and modern Caribbean reefs to reconstruct prehistoric and contemporary reef fish trophic structures (Supplementary Table 1). Cleaning and analysis techniques ensured that only fish- and coral-native organic matter was analysed for δ15N (Methods). The δ15N of coral-bound organics (δ15Ncb) co-varies with regional patterns in the isotopic baseline (δ15Nbase), that is, the nitrogen isotope composition of nutrients consumed by primary producers at the base of the food chain (Methods). We found that δ15Nbase did not change significantly over time in either region (Fig. 2c; Wilcoxon, Panama, P = 0.45, t = 109, effect size r = 0.3; Dominican Republic, P = 0.62, t = 14, r = 0.33; Extended Data Table 1), enabling us to examine trophic-level patterns without adjusting for δ15Nbase shifts.
Otolith δ15N tracks trophic roles
The resulting δ15N data of fossil fishes (Fig. 2b) generally conformed to expected trophic levels (refer to the ‘δ15Noto comparison with known dietary patterns’ section in the Supplementary Information), with grunts and cardinalfishes occupying higher trophic levels compared with gobies and silversides. Gobies showed the lowest relative trophic level, consistent with known patterns based on prey items from stomach-content studies28,29 and tissue-stable-isotope studies (for example, ref. 30) of modern fishes in the Caribbean. Past isotope studies have found that these species were 3–4‰ lower than co-occurring grunts30, similar to our results. Silversides (small pelagic schooling fishes that primarily consume copepods and fish larvae) had higher δ15N than gobies, but lower δ15N than cardinalfish and grunts. In previous studies, modern silverside tissue δ15N and trophic level were typically higher than that of co-occurring herbivores31,32, probably due to the higher δ15N of pelagic versus benthic organisms in coral reef settings, including in Bocas del Toro, Panama33. Thus, the δ15N elevation of silversides relative to gobies in our study is consistent with pelagic foraging by silversides and primarily benthic foraging by gobies29. Grunts, the most abundant representative of higher-trophic-level fishes in our fossil sediment record, are omnivorous or carnivorous predators that move among adjacent habitats, where available, ranging up to 5 km to feed on diverse crustaceans, molluscs and some fish34,35. Modern tissue studies on this group of fishes show that their δ15N ranges from 8–10‰ in the Caribbean, similar to our otolith results33,34,35. Cardinalfishes showed a trophic level that was similar to that of grunts. There are few modern tissue isotope studies on this family36, but their classification as generalist carnivores is consistent with the relatively high otolith δ15N observed here. This alignment with known dietary behaviours supports the validity of our fossil trophic-level estimates.
Trophic-level decline in higher-trophic-level fishes
δ15Noto indicates that the different fish families have experienced different degrees of dietary change since the mid-Holocene (Fig. 2c). In Panama, we observed no significant changes in family trophic levels (Fig. 2c, Extended Data Tables 1 and 2). In the Dominican Republic, modern grunts and cardinalfishes occupied lower trophic levels (that is, had lower average δ15Noto) than their fossil counterparts (Fig. 2c). Specifically, during the modern time period, mean δ15N was 2.4‰ lower in grunts (Wilcoxon, P = 0.048, W = 20, r = 0.82) and 3.7‰ lower in cardinalfishes (P = 2.5 × 10−4, W = 100, r = 0.873; Extended Data Table 2). As the trophic level changes in Panama were relatively subtle and required more samples to increase the statistical power, we used a resampling approach to generate bootstrapped δ15N data (Methods). With the bootstrapped resampling (1,000 iterations), Panama’s data echo the changes observed in the Dominican Republic, with higher mid-Holocene δ15N in grunts and cardinalfishes in Panama, but with a much lower amplitude (grunts: 0.3‰ decline, Wilcoxon, P = 0.033, W = 26, r = 0.572; cardinalfishes: 1.0‰ decline, P = 2.2 × 10−16, W = 8,281, r = 0.722; Extended Data Table 3). Gobies and silversides were similar to or higher than their fossil counterparts in both regions. Specifically, modern goby δ15Noto was higher than fossil by 1.4‰ (Wilcoxon, P = 2.2 × 10−16, W = 0, r = 0.828) in Panama and lower by 0.8‰ in the Dominican Republic (P = 2.2 × 10−16, W = 21,732, r = 0.840); silversides were lower by 0.3‰ in Panama (P = 6.52 × 10−6, W = 1,169, r = 0.342) and were not measured in the Dominican Republic owing to a lack of specimens (Extended Data Table 3).
Community shifts in mean trophic level
We also examined community metrics (Fig. 3), including the MTL. Declining MTL in commercial fisheries—termed fishing down the food chain—is well-documented globally, yet most modern studies assume fixed trophic levels for each species, potentially overlooking dynamic changes in trophic level, trophic niche width and FCL. By contrast, nitrogen isotopes capture ‘realized’ trophic diversity, akin to an Eltonian niche, thus reflecting dynamic patterns in prey availability and consumer behaviour7,25,37.
a, Measured MTL, calculated as the mean of mean family δ15N (± 1 s.e.) in each region and time period (where n represents families per average) (Supplementary Fig. 1). Measured MTL did not change for Panama (left, Wilcoxon, two-sided, P = 1, W = 8, r = 0) or the Dominican Republic (right, Wilcoxon, P = 0.4, W = 7, r = 0.445) (Extended Data Table 2). Grey symbols show the taxon trophic level (± 1 s.e.) (the symbols are the same as for Fig. 2b). NS, not significant. b, Isotopic niche width, calculated as the mean of all family s.d. values within each region and time period. To identify changes, we first normalized the isotope niche widths from Fig. 2d. We found that the community mean niche widths declined in both Panama (Paired t-test, two-sided, t = 4.23, d.f. = 3, P = 0.0242) and the Dominican Republic (Paired t-test, t = 17.073, d.f. = 2, P = 0.00341; data were screened for normality using the Shapiro–Wilk test for Panama, P = 0.42, W = 0.90; and for the Dominican Republic, P = 0.12, W = 0.80). For modern data, the mean niche ± 1 s.e. values are plotted; for the fossil data, no s.e. values are shown because data were scaled to mid-Holocene data from each region, removing variation. Grey symbols show the isotopic niche width for each family (same data as Fig. 2d). c, FCL, calculated as the range of mean family δ15N (see Supplementary Fig. 1). As the FCL is a calculated range, we did not conduct statistical tests. d, Family abundance-weighted δ15N distributions were modelled by applying bootstrapped δ15N to each otolith in the sedimentary record (refer to the ‘Ecosystem nitrogen isotope distribution’ section in the Methods). A smoothing function (4× binwidth kernel) was applied to the bootstrapped data to illustrate the distributional changes and therefore focus on the large-scale δ15N distribution. We calculated community metrics: the family abundance-weighted MTL and the family abundance-weighted FCL (95% interquartile range; Extended Data Table 5 and Supplementary Fig. 1).
Here we estimated MTL in fossil and modern reef fish communities using two approaches: (1) the ‘measured MTL’ (Fig. 3a) of the partial fish community, calculated by averaging the mean δ15N values of each of the measured fish families, and (2) the family ‘abundance-weighted MTL’ (Fig. 3d) that incorporates the numerical abundances of each family (Extended Data Table 5). The latter approach uses the previously described abundances22 of our target fish groups and applies a resampled (bootstrapped) δ15N measurement to each individual fish (otolith) recovered from the fossil and modern reefs, generating a modelled family-weighted (or scaled) distribution of fish community δ15N (refer to the ‘Ecosystem nitrogen isotope distribution’ section in the Methods). The abundance-weighted MTL is then calculated as the mean of the resulting distribution (Supplementary Fig. 1). As the relative abundances of different component families as well as the measured δ15Noto can shift the δ15Noto distribution, the ecosystem nitrogen isotope approach can be considered a dynamic metric for tracking MTL. We included only the three fish families found in both time periods and regions, thus excluding silversides, which were found in high abundances only in Panama.
We found that measured MTL (Fig. 3a) remained stable in Panama (Wilcoxon, P = 1.0, W = 8, r = 0) and in the Dominican Republic (Wilcoxon, P = 0.4, W = 7, r = 0.445) (Fig. 3a, Extended Data Table 2 and Supplementary Table 2). The family abundance-weighted MTL (Fig. 3d), in contrast, increased in Panama from 5.8 to 7.2‰ (Wilcoxon, P = 2.2 × 10−16, W = 56,693, r = 0.669) and decreased in the Dominican Republic from 6.1 to 5.2‰ (Wilcoxon, P = 8.4 × 10−5, W = 33,018, r = 0.176; Extended Data Table 5). Notably, the significant MTL increase in Panama emerged only in the abundance-weighted metric, driven by elevated goby δ15N, which outweighed the δ15N declines in cardinalfishes and grunts owing to the high abundance of gobies during both time periods (84–91% of the partial community in Panama; Supplementary Tables 4 and 5). The elevated δ15N of modern gobies is consistent with increased reliance on pelagic prey (refer to the ‘Ecological explanations for reduced δ15N variability’ section in the Supplementary Information). In the Dominican Republic, MTL declined owing to the δ15N declines in each family, although the numerical dominance of gobies (51–95% of the partial community in the Dominican Republic) set the magnitude (0.81‰) of the decline. Goby δ15N declined only modestly (0.42‰) compared to the larger magnitude declines (2.4‰ in grunts, 3.7‰ in cardinalfishes) in the less abundant taxa, thus yielding the observed 0.81‰ family abundance-weighted MTL decline. We conclude that the comparison of the measured MTL, which is stable through time, to the family-weighted MTL, which showed divergent trends by region, adds context to the ecosystem nitrogen isotope approach for determining changes in reef community trophic structure.
Declines in dietary breadth within taxa
Isotopic niche width (Fig. 2d), calculated from δ15N variation within each family (1 s.d.; ref. 25) and at the community level (Fig. 3c), was consistently greater in fossil fishes than in modern ones. These niche widths during the mid-Holocene (19 to 69%; Extended Data Table 4) were statistically significant for gobies, silversides and grunts in Panama (F-test to compare variances, P < 0.05 to P < 0.001 depending on species; Extended Data Table 4). No significant niche width changes were observed in the Dominican Republic, probably owing to limited statistical power. Nevertheless, bootstrapped results revealed modern declines in niche widths for Dominican Republic gobies (F-test to compare variances, 55% decline, P < 2.2 × 10−16, F = 4.91, d.f. = 125) and cardinalfishes (100% decline, P < 2.2 × 10−16, F = Inf, d.f. = 26) and significant shifts in the δ15N distributions for all Dominican Republic families (Kolmogorov–Smirnov, gobies, P < 2.2 × 10−16, D = 0.93; cardinalfishes, P < 2.2 × 10−16, D = 1; and grunts, P = 0.0031, D = 1; Extended Data Table 3).
Within a population, isotopic niche width reflects dietary variation among individual fish. The observed declines therefore suggest a reduction in individual-level dietary specialization on modern reefs (Fig. 3b and Extended Data Table 4). This is consistent with optimal foraging theory38, which predicts that specialization improves energy efficiency by reducing handling time for each prey item. On degraded modern reefs, limited access to preferred prey probably forces fish to consume a broader array of suboptimal prey, resulting in individuals with more generalized diets39. Unintuitively, this leads to narrower population-level isotopic niches, as dietary overlap among individuals increases and δ15N converges12,14,25,40 (Fig. 4b). We emphasize that we observed no niche widening in any family, contrary to predictions from the distributed stress model41. This suggests that reduced specialization, not diversification, is the dominant trend.
a, Trophic simplification of modern Caribbean coral reefs. Community patterns in MTL and FCL for the mid-Holocene (left) and modern (right) coral reefs. In the modern reef, FCL declines, whereas community MTL is relatively stable compared with in the fossil reef. b, Decreased family-level dietary diversity in modern populations. Individual-fish-level foraging patterns affect family-level isotopic niche width (that is, diet-driven δ15N diversity). Consuming prey items in common with other fish individuals increases their isotopic similarity to one another, and this isotopic similarity also reflects a homogenization of functional roles and energy flow pathways on coral reefs today. By contrast, fish on mid-Holocene reefs, owing to higher specialization and less overlap, supplied a greater diversity of unique pathways by which energy could enter coral reef food webs. As a greater diversity of energy channels is known to increase food web stability, the loss of heterogeneity on modern reefs may reduce ecosystem stability. Fish silhouettes in a and b reproduced from ref. 51, GitHub, under a GPL-2 license.
Alternatively, the narrowing of isotopic niches may reflect a loss in δ15N diversity among available prey, rather than a behavioural shift in foraging; however, this scenario is less likely, given evidence that many reef fishes increase the number of prey items consumed when preferred prey decline (for example, ref. 39). It is also improbable that remaining prey would coincidentally span a δ15N range similar to prehistoric means across multiple fish families. In any case, the δ15N of a prey item generally reflects its trophic position. Thus, regardless of the mechanism, the outcome is the same: increased trophic similarity among individual fish and reduced isotopic niche width at the population level. This emergent pattern highlights a trend towards homogenized trophic roles on modern Caribbean reefs. The decline in family-level niche widths might cause decreased stability as changes in the availability of a given prey item would affect many individual fish in the consumer population40,42, as discussed below. Overall, the observed changes reveal previously unknown aspects of how human-caused alteration of coral reef ecosystems has affected energy flow on modern reefs, potentially leading to new ecosystem-based indicators of ecosystem health.
Reduced food chain lengths
In both Panama and the Dominican Republic, FCL—the range in mean δ15Noto across taxa—on the modern reefs was about 40% shorter than during the mid-Holocene (Fig. 3c and Extended Data Table 2). To further assess community-level trophic structure, we calculated FCL from the ecosystem nitrogen isotope distribution (bootstrapped δ15N data), which uses a resampling approach to weight δ15N to the proportional abundances of each family (as described above and in the Methods). The analysis (Fig. 3d) revealed a marked decline in community-level δ15N variance in both regions (Kolmogorov–Smirnov test, D = 0.91, P = 2.2 × 10−16 for Panama; D = 0.47, P = 2.2 × 10−16 for Dominican Republic; Extended Data Table 5). The modern δ15N distributions were compressed (Fig. 4), with inter-quartile ranges (95%) reduced by 53% (Panama) and 60% (Dominican Republic) (Extended Data Table 5). The family-weighted distributions show decreased variance (by 2.68–4.05-fold, F-test; Panama, P = 2.2 × 10−16, F = 2.68, d.f. = 558; Dominican Republic, P = 2.2 × 10−16, F = 4.04, d.f. = 158) and had lower kurtosis and skewness (Extended Data Table 5), further recording the contraction of trophic diversity observed in the FCL results.
The reduction in FCL reflects a loss of trophic diversity at both the upper and lower extremes of the food web, suggesting a simplification of energy flow pathways among the reef fishes (Fig. 4). This observation might be explained by changes in structural habitat complexity and predation pressure (refer to the ‘Ecological explanations for reduced δ15N variability’ section in the Supplementary Information). For example, historically abundant predatory fishes4 may have created a ‘landscape of fear’ that constrained prey movement, leading to localized foraging behaviour and supporting fine-scale trophic partitioning43. Likewise, greater structural complexity in mid-Holocene coral reefs before human-driven shifts in coral community composition2,3 probably increased both refugia (from predators) and prey diversity2,44 by supporting distinct microhabitats with different δ15N (refs. 36,37). Access to mangrove foraging grounds, which contribute to macroscale habitat complexity and contain relatively high δ15N prey34,35, may have further supported longer food chains during the mid-Holocene. In food web theory, declines in habitat connectivity (that is, habitat fragmentation) should generate shorter FCLs26,27. As an empirical example of how habitat connectivity affects FCL, coastal habitat fragmentation in the Bahamas results in decreased FCL (and population niche widths) of predators12. Fragmentation reduces the diversity of prey, especially higher-trophic-level prey items, decreasing the specialization opportunities for the predators while lowering their trophic level and the ecosystem FCL12, echoing the results from freshwater ecosystems showing that reduced connectivity decreases the FCL in some contexts26,27,45. The greater trophic changes in the Dominican Republic are consistent with the greater intensity of human impacts in the Dominican Republic compared with Panama, including greater losses of mangroves and higher degrees of overfishing8 (refer to the ‘Panama versus Dominican Republic coastal health comparisons’ section in the Supplementary Information). Although the relative contribution of each driver (habitat diversity, predator presence, prey availability) is not yet clear, modern coral reef ecosystems clearly show reduced heterogeneity in energy channels.
Importantly, trophic changes were observed across all fish taxa analysed in both regions, including gobies—the lowest trophic level fish species examined. These small, short-lived fishes have outsized roles in energy cycling on coral reefs as both consumers28,29 and prey29, comprising up to 80% of fish consumed on coral reefs46. Thus, the reconstructed changes in energy flow (prey availability and consumption) would also have impacted the sources, rates and quality of energy supplied to large fishes.
Implications for ecosystem resilience
Using a new ‘ecosystem nitrogen isotope’ approach that analyses co-existing otolith and coral fossils, this study provides the first direct reconstruction of coral reef fish trophic structure before substantial human impact, and it reveals ecosystem-wide trophic compression across family, community and regional scales. We find that trophic diversity, indicating the breadth of resource use within and across coral reef taxa, has declined markedly. This reduction seems to reflect a loss of dietary specialization and reduced prey availability at the upper and lower ends of food webs.
Our findings show that recent changes on these reefs have shortened reef food chains while also reducing the dietary specialization of reef-associated fishes, similar to comparisons of fished and unfished coral reef food webs in remote oceanic atolls in the Pacific47. Modern comparison studies typically find that trophic responses to coral reef degradation are highly context- and species-specific18,32,48,49 in contrast to our results showing universally reduced niche widths across each family and decreased FCL in both regions.
Our findings have implications for ecosystem stability. The observed family-level shifts in trophic niche width and trophic level suggest that reef fish have already adapted their behaviour to changing prey availability, foraging opportunities and habitat connectivity, demonstrating a degree of resilience (refer to the ‘Niche width and ecosystem stability’ section in the Supplementary Information). However, as anthropogenic impacts become progressively more severe, the intrinsic buffering capacity of the food web network is now limited. The reductions in both FCL and niche diversity indicate a contraction in the number of distinct energy pathways that support the ecosystem. Ecological theory predicts that such a decline in diversity can erode stability by weakening the network’s ability to buffer shocks1,27,50. If all individual fish in a population rely on an overlapping, common pool of resources, as opposed to highly individualized feeding preferences, the population will no longer be buffered against changes in food availability. By contrast, under the mid-Holocene condition of greater intra-population specialization, a given change in food availability would have affected only a subset of individuals. Our reconstruction suggests that modern coral reef ecosystems operate with fewer trophic pathways and reduced functional redundancy, reducing their capacity to withstand ongoing and future stressors and increasing the risk of ecosystem collapse.
Methods
Taxa
Our main goal was to assess long-term changes in the trophic structure of coral reef-associated fishes compared to prehistoric trophic structure before widespread human impacts. As different stressors can affect different components of marine food webs, we analysed otoliths from key taxa representing the most abundant species from different ecological niches. Gobies (Gobiidae) are abundant, small, cryptobenthic fishes that forage in reef crevices. Cardinalfishes (Apogonidae) are cryptobenthic taxa that exhibit nocturnal off-reef foraging29. Silversides (Atherinidae) are small, pelagic planktivores that feed in schools above reefs52 and grunts (Haemulidae) are relatively larger omnivores that forage in both reef and adjacent seagrass and mangrove habitats35. For taxonomic identification based on otoliths, we used a Caribbean otolith reference collection comprised of locally obtained modern specimens22. Owing to the diagnostic limitations of identifying very small otoliths from early life history stages, only a portion (30–100%, depending on the family; Supplementary Table 4) could be identified to the genus or species, whereas the remaining specimens were assigned to family (8–60%).
Study regions
Otoliths were extracted from two coral reef sedimentary deposits that are the only known mid-Holocene otolith-bearing reef sediments in the Caribbean, located in Bocas del Toro, Panama (9° N × 82° W; Fig. 1 and Supplementary Table 3), and the Enriquillo Basin, Dominican Republic (18° N × 71° W). These sites contain well-preserved fossil reef assemblages spanning the mid-Holocene (around 7000 years before present; bp) and modern (around 100 bp) time periods22,53. Multiple sub-localities were sampled in each region (Extended Data Fig. 1). Bulk sediments, around 9 kg per sample, were collected from reef frameworks using different methods depending on the time period as described in ref. 22. At the mid-Holocene sites, sediments were excavated from 3-m-deep trenches, whereas in modern reefs, SCUBA divers collected sediment from 10–15-cm-deep strata adjacent to living corals. Samples were sieved into size fractions (2 mm, 500 µm, 250 µm and 106 µm) and then otoliths were manually extracted under a dissecting microscope.
Dating
Radiometric dating of coral skeletal material using U-Th and calibrated radiocarbon techniques was previously published for Panama4,22,53 and the Dominican Republic54,55,56. For both regions and time periods, fossil samples spanned around 100 years. U-Th dates showed a larger date range than ages obtained from radiocarbon dating, with ages falling between 6345 and 7164 bp (819 y range) for U-Th, and between 6533 to 6638 bp for 14C (105 y range4). Similarly, dates for the modern samples ranged from 1152 to 1984 bp (832 year range3) for U-Th, with the majority of 14C dates between 1926 to 2014 CE for modern (that is, approximately the past 100 years22,53,57).
Otolith specimen selection
We selected otolith and coral specimens across multiple reef sub-localities to capture spatial and temporal variability (Supplementary Table 3). In total, 91 fish otolith specimens were analysed from Panama (42 mid-Holocene, 49 modern) and 42 from the Dominican Republic (21 mid-Holocene, 21 modern). To select otoliths from the collection, a list of sample numbers (specifying a unique combination of locality + depth + bulk bag replicate) were selected on the basis of obtaining samples from a wide distribution of sub-localities in each region. Otoliths were then randomly chosen from each sample number until n = 10–30 otoliths for each taxon from each country and time period were selected (Supplementary Fig. 2 shows the spatial distribution of specimens across different sub-localities). Localities close to banana plantations (for example, Punto Donato) were avoided, as were sites with known time-averaging problems (Airport Point) or distinct exposures (Cayo Adriana, which is oceanographically connected); however, for Atherinidae and Haemulidae, samples were needed from each of these localities for sufficient sample size.
Furthermore, five modern grunts (family Haemulidae) were measured from the reference collection. Other reef fishes (n = 35) were also measured but not included owing to a lack of conspecifics for comparison, including fishes in the Engraulidae (21), Lutjanidae (8), Serranidae (4) and Gerreidae (2) families (Supplementary Fig. 3). These data were not included in the main text owing to a lack of comparisons across time periods or regions.
Coral specimen selection
The ecosystem nitrogen isotope approach allowed for direct comparisons of fish-trophic-level shifts using otolith-bound δ15N from multiple taxa while controlling for possible isotopic baseline δ15N variability using coral-bound δ15N. δ15Ncb patterns are known to reflect variations in δ15Nbase, the nitrogen isotope composition of nutrients consumed by primary producers at the base of the food chain58,59. Although corals are mixotrophic, and heterotrophic feeding can increase the δ15N due to decreased reliance on symbionts60, the primary signal is the δ15N of inorganic nutrients58,61,62. We used coral fragments (as per ref. 63), allowing for similar spatiotemporal sampling of otoliths and corals across sites. We analysed fragments (n = 3–5 per locality and time period) of branching Porites furcata (Panama, n = 26; Dominican Republic, n = 9). Where possible, coeval corals and otoliths from the same sub-localities and depth horizons bags were analysed (Supplementary Fig. 2 and Fig. 3).
Pre-processing of coral and otolith specimens
Three regions of each coral fragment were exposed with a rotary disk drill (Dremel 4000) and visually inspected for encrusting organisms. Powders were removed by drilling with a rotary tool using a 1.4 mm diamond drill bit (Shofu). The powder drilled from each fragment was homogenized and subsampled into two subsamples that were analysed separately for coral-bound δ15N using previously established methods19,58. Subsamples were then averaged to generate a mean value for each coral fragment.
Otoliths were photographed (Leica KL1500 LCD), weighed and prepared for δ15N analysis. Specimens were pulverized and homogenized before chemical cleaning, following established protocols5. Otoliths with initial masses of over 5.5 mg were subsampled into masses between 3.5 and 4.5 mg. For otoliths with initial masses of less than 5.5 mg, the otolith was pulverized and the powder from the entire otolith was analysed. Initial nitrogen isotope analyses involved pooled otoliths (Extended Data Fig. 2) to ensure sufficient nitrogen content for measurements out of an abundance of caution to ensure sufficient nitrogen for analysis. All subsequent isotopic analyses were conducted on individual otoliths, including specimens as small as 0.06 mg (60 µg) (Supplementary Table 1).
Previous studies show that the δ15N patterns in otoliths—for example, muscle tissue—convey information about the proportional contribution of food items with distinct isotopic values64. This method has been validated through calibration studies showing consistent trophic information compared with the commonly measured tissues20,64. δ15Noto studies have reconstructed trophic information on centennial and million-year timescales5.
δ15N measurements
For coral and otolith powders, cleaning of the biomineral powder entailed sequential treatments with sodium polyphosphate (clay removal), dithionite-citric acid (metal oxide removal) and sodium hypochlorite (non-bound organic removal), as described in ref. 19 and ref. 5. Once cleaned, powdered samples were dissolved in ACS-grade 4 N HCl, and organic nitrogen was oxidized with basic persulfate oxidizing reagent. Nitrate concentrations were determined using chemiluminescence65. To convert nitrate to N2O, sample aliquots amounting to 10 nmol nitrogen were injected into vials of concentrated denitrifying bacteria and digested at room temperature for 3–6 h (refs. 66,67). The 15N to 14N ratio of the sample analyte, N2O, was analysed on a MAT253 GC-IRMS interfaced with a purpose-built N2O purification and extraction system at Princeton University (Isodat v.3.0)66,67. Long-term analytical precision (1 s.d.) was 0.08‰ for N2O standards and 0.3‰ for in-house otolith standards.
Food web metrics
To assess shifts in trophic structure, we calculated several metrics (Supplementary Fig. 1). For each family, we calculated the taxon trophic level and the isotopic niche width (1 s.d.; for example, ref. 68). For the fish community, we report (1) the MTL, which is calculated as the mean of each family’s trophic level, and (2) the FCL, the number of trophic steps in the food web, calculated as the isotopic difference between the lowest- and highest-trophic-level family means from each time period and region. Our calculations, based on family-level trophic-level as opposed to the trophic-level of individual fish, are a conservative measure of the trophic distance between the highest- and lowest-trophic-level taxa due to the low sample sizes of the fish measured. Isotopic niche width, calculated as the variation (1 s.d.) in δ15N within each family, is an indicator of breadth—that is, the dietary dissimilarity—of the community. Baseline-corrected δ15N were standardized by the respective regional (Panama or Dominican Republic) and temporal δ15Ncb (Extended Data Table 2). As FCL is a relative, not absolute metric, baseline δ15N does not affect its value.
When examining our data by species- and genus-level, where otolith-based identification allowed for higher taxonomic resolution, we found that δ15N followed the same patterns as the family-level δ15N patterns (Extended Data Fig. 3). DNA metabarcoding of stomach contents also supports this observation, showing that family-level patterns in coral reef fish diet are consistently better predictors of fish diet than trophic group or guild69,70. Consequently, for the purposes of interpreting the family-level isotope data, family-level patterns can be considered indicators of species- or population-level trophic patterns.
Potential artifacts—such as changes in fish community composition, differences in sample time averaging or preservation-related effects on δ15N—could influence family- or community-level patterns (refer to the ‘Community composition’, ‘Time averaging’ and ‘Preservation’ sections in the Supplementary Information). Shifts in the size distribution of the sampled fishes could also influence trophic structure, as size and trophic level are tightly linked in most fish (refer to the ‘Fish size’ section in the Supplementary Information); however, our collected samples reveal consistency in each variable, suggesting stable species distributions, temporal sampling (80–100 years; refer to the ‘Dating’ section above), fish size (Extended Data Fig. 4a and Supplementary Fig. 4) and preservation (Extended Data Fig. 4b and Supplementary Fig. 5) across periods. Thus, consistent with previous studies showing fossil versus modern declines in aspects of coral reef ecosystem health in the region (Supplementary Fig. 6), the trophic changes are more likely explained by the multiple stressors acting upon modern coral reefs.
Ecosystem nitrogen isotope distribution
To assess the impacts of our observed δ15N changes at the scale of the fish community, we modelled taxon-weighted distributions of δ15N for each region and time period. Community composition data was used to weight the δ15N to generate a model of community-weighted δ15N distributions. Matrices containing the proportional contributions of each of the three taxonomic groups found in both regions for both time periods—gobies, grunts and cardinalfishes—and their corresponding mean and median δ15N were used to model δ15N distribution at the community level (Supplementary Table 5).
Pseudo-replicated δ15N values (that is, for samples where multiple otoliths were combined to make one measurement and each otolith treated separately for the purposes of the main text) were removed before calculating the means and medians. To minimize the influence of outliers, localities with n ≤ 2 were excluded. The mean δ15N was calculated from the raw data in those cases.
To obtain proportional contributions, each locality was treated as a sample owing to the low otolith counts in some sub-locality bulk sample bags. Then, the proportions were averaged across all localities within each period and region.
Bootstrapped sampling distributions were generated to capture more of the variation across δ15N measurements. For each family-region-age group, the original δ15N values were sampled (sample size n = 100) with replacement, and a mean was calculated from each bootstrapped sample (with n = 1,000 iterations). This generated a distribution of means for each group, from which values were randomly pulled and then assigned to each otolith found in the bulk samples. The new distributions were then plotted from these values. Following these analyses, we provide a figure summarizing the patterns observed in each region (Supplementary Fig. 7).
Statistics
We used R (R Core Team, 2024) to perform statistical tests and plot our data. Fish shapes for grunts, gobies and cardinalfishes were obtained from the R package fishualize51 (v.0.2.3). All data analysis was conducted using R software (v.4.1.2), via the tidyverse (v.1.3.1), boot (v.1.3-28), rlist (v.0.4.6.2), onewaytests (v.2.7), rstatix (v.0.7.0), forcats (v.0.5.1) and tidyr (v.1.3.1) packages. We used the following packages for other statistical and mapping operations in R: ggmap (v.3.0.0), ggrepel (v.0.9.3), ggsn (v.0.5.3), cowplot (v.1.1) and grid (v.4.1.1).
Reporting summary
Further information on research design is available in the Nature Portfolio Reporting Summary linked to this article.
Data availability
All data used in this study are available from FigShare at https://doi.org/10.6084/m9.figshare.28811663.
Code availability
The code used for bootstrapping (Fig. 3d and Extended Data Fig. 5) and other R scripts used to create figures are available at https://doi.org/10.6084/m9.figshare.28811663.
References
Hughes, T. P. et al. Climate change, human impacts, and the resilience of coral reefs. Science 301, 929–933 (2003).
Alvarez-Filip, L., Dulvy, N. K., Gill, J. A., Côté, I. M. & Watkinson, A. R. Flattening of Caribbean coral reefs: region-wide declines in architectural complexity. Proc. R. Soc. B 276, 3019–3025 (2009).
Cramer, K. L. et al. Widespread loss of Caribbean Acroporid corals was underway before coral bleaching and disease outbreaks. Sci. Adv. 6, eaax9395 (2020).
Dillon, E. M. et al. Fossil dermal denticles reveal the preexploitation baseline of a Caribbean coral reef shark community. Proc. Natl Acad. Sci. USA 118, e2017735118 (2021).
Lueders-Dumont, J. A., Wang, X. T., Jensen, O. P., Sigman, D. M. & Ward, B. B. Nitrogen isotopic analysis of carbonate-bound organic matter in modern and fossil fish otoliths. Geochim. Cosmochim. Acta 224, 200–222 (2018).
Kast, E. R. et al. Cenozoic megatooth sharks occupied extremely high trophic positions. Sci. Adv. 8, 7–18 (2022).
Skinner, C., Cobain, M. R. D., Zhu, Y., Wyatt, A. S. J. & Polunin, N. V. C. Progress and direction in the use of stable isotopes to understand complex coral reef ecosystems: a review. Oceanogr. Mar. Biol. An Annu. Rev. 60, 373–432 (2022).
Burke, L., Reytar, K., Spalding, M. & Perry, A. Reefs at risk revisited. World Resour. Inst. 74, 1–130 (2011).
Sing Wong, A., Vrontos, S. & Taylor, M. L. An assessment of people living by coral reefs over space and time. Glob. Chang. Biol. 28, 7139–7153 (2022).
Aronson, R. B. Foraging behavior of the west Atlantic trumpetfish, Aulostomus maculatus: use of large, herbivorous reef fishes as camouflage. Bull. Mar. Sci. 33, 166–171 (1983).
Lukoschek, V. & McCormick, M. I. A review of multi-species foraging associations in fishes and their ecological significance. Proc. Ninth Int. Coral Reef Symp. I, 467–474 (2000).
Layman, C. A., Quattrochi, J. P., Peyer, C. M. & Allgeier, J. E. Niche width collapse in a resilient top predator following ecosystem fragmentation. Ecol. Lett. 10, 937–944 (2007).
Frédérich, B., Lehanse, O., Vandewalle, P. & Lepoint, G. Trophic niche width, shift, and specialization of Dascyllus aruanus in Toliara Lagoon, Madagascar. Copeia 2010, 218–226 (2010).
Layman, C. A. & Allgeier, J. E. Characterizing trophic ecology of generalist consumers: A case study of the invasive lionfish in the Bahamas. Mar. Ecol. Prog. Ser. 448, 131–141 (2012).
Odum, H. T. & Odum, E. P. Trophic structure and productivity of a windward coral reef community on Eniwetok Atoll. Ecol. Monogr. 25, 291–320 (1995).
Polovina, J. J. Model of a coral reef ecosystem: I. The ECOPATH model and its application to French Frigate Shoals. Coral Reefs 3, 1–11 (1984).
Woodhead, A. J., Hicks, C. C., Norström, A. V., Williams, G. J. & Graham, N. A. J. Coral reef ecosystem services in the Anthropocene. Funct. Ecol. 33, 1023–1034 (2019).
Letourneur, Y., Briand, M. J. & Graham, N. A. J. Coral reef degradation alters the isotopic niche of reef fishes. Mar. Biol. 164, 224 (2017).
Wang, X. T. et al. Isotopic composition of carbonate-bound organic nitrogen in deep-sea scleractinian corals: a new window into past biogeochemical change. Earth Planet. Sci. Lett. 400, 243–250 (2014).
Lueders-Dumont, J. A. et al. Comparison of the isotopic composition of fish otolith-bound organic N with host tissue. Can. J. Fish. Aquat. Sci. 77, 264–275 (2020).
Rao, Z. C. et al. A nitrogen isotopic shift in fish otolith–bound organic matter during the Late Cretaceous. Proc. Natl Acad. Sci. 121, e2322863121 (2024).
Lin, C.-H. et al. Reconstructing reef fish communities using fish otoliths in coral reef sediments. PLoS ONE 14, e0218413 (2019).
Leonhard, I. & Agiadi, K. Addressing challenges in marine conservation with fish otoliths and their death assemblages. Geol. Soc. London 529, 243–262 (2023).
Salas, S., Chuenpagdee, R., Charles, A. T. & Seijo, J. C. Coastal Fisheries of Latin America and the Caribbean (Food and Agriculture Organization of the United Nations, 2011).
Bearhop, S., Adams, C. E., Waldron, S., Fuller, R. A. & Macleod, H. Determining trophic niche width: a novel approach using stable isotope analysis. J. Anim. Ecol. 73, 1007–1012 (2004).
Post, D. M. The long and short of food-chain length. Trends Ecol. Evol. 17, 269–277 (2002).
Ward, C. L. & McCann, K. S. A mechanistic theory for aquatic food chain length. Nat. Commun. 8, 2028 (2017).
Depczynski, M. & Bellwood, D. R. The role of cryptobenthic reef fishes in coral reef trophodynamics. Mar. Ecol. Prog. Ser. 256, 183–191 (2003).
Brandl, S. J., Goatley, C. H. R., Bellwood, D. R. & Tornabene, L. The hidden half: ecology and evolution of cryptobenthic fishes on coral reefs. Biol. Rev. 93, 1846–1873 (2018).
Zhu, Y., Newman, S. P., Reid, W. D. K. & Polunin, N. V. C. Fish stable isotope community structure of a Bahamian coral reef. Mar. Biol. 166, 160 (2019).
Tilley, A., López-Angarita, J. & Turner, J. R. Diet reconstruction and resource partitioning of a Caribbean marine mesopredator using stable isotope Bayesian modelling. PLoS ONE 8, e79560 (2013).
Hempson, T. N. et al. Coral reef mesopredators switch prey, shortening food chains, in response to habitat degradation. Ecol. Evol. 7, 2626–2635 (2017).
Stuthmann, L. E. & Castellanos-Galindo, G. A. Trophic position and isotopic niche of mangrove fish assemblages at both sides of the Isthmus of Panama. Bull. Mar. Sci. 96, 449–467 (2020).
Vaslet, A., Phillips, D. L., France, C. A. M., Feller, I. C. & Baldwin, C. C. Trophic behaviour of juvenile reef fishes inhabiting interlinked mangrove-seagrass habitats in offshore mangrove islets. J. Fish Biol. 87, 256–273 (2015).
Cocheret de la Morinière, E. et al. Ontogenetic dietary changes of coral reef fishes in the mangrove-seagrass-reef continuum: stable isotopes and gut-content analysis. Mar. Ecol. Prog. Ser. 246, 279–289 (2003).
Frédérich, B. et al. Comparative feeding ecology of cardinalfishes (Apogonidae) at Toliara reef, Madagascar. Zool. Stud. 56, e10 (2017).
Rader, J. A. et al. Isotopic niches support the resource breadth hypothesis. J. Anim. Ecol. 86, 405–413 (2017).
Schoener, T. W. Theory of feeding strategies. Annu. Rev. Ecol. Syst. 2, 369–404 (1971).
Clever, F. et al. The gut microbiome variability of a butterflyfish increases on severely degraded Caribbean reefs. Commun. Biol. 5, 770 (2022).
Stewart, S. D., Kelly, D., Biessy, L., Laroche, O. & Wood, S. A. Individual diet specialization drives population trophic niche responses to environmental change in a predator fish population. Food Webs 27, e00193 (2021).
Steube, T. R., Altenritter, M. E. & Walther, B. D. Distributive stress: individually variable responses to hypoxia expand trophic niches in fish. Ecology 102, e03356 (2021).
Stouffer, D. B. & Bascompte, J. Compartmentalization increases food-web persistence. Proc. Natl Acad. Sci. USA 108, 3648–3652 (2011).
Hixon, M. A. & Beets, J. P. Predation, prey refuges, and the structure of coral-reef fish assemblages. Ecol. Monogr. 63, 77–101 (1993).
Beukers, J. S. & Jones, G. P. Habitat complexity modifies the impact of piscivores on a coral reef fish population. Oecologia 114, 50–59 (1998).
Tunney, T. D., McCann, K. S., Lester, N. P. & Shuter, B. J. Food web expansion and contraction in response to changing environmental conditions. Nat. Commun. 3, 1105 (2012).
Brandl, S. J. et al. Demographic dynamics of the smallest marine vertebrates fuel coral reef ecosystem functioning. Science 364, 1189–1192 (2019).
Young, H. S., McCauley, F. O., Micheli, F., Dunbar, R. B. & McCauley, D. J. Shortened food chain length in a fished versus unfished coral reef. Ecol. Appl. 34, e3002 (2024).
Morillo-Velarde, P. S. et al. Habitat degradation alters trophic pathways but not food chain length on shallow Caribbean coral reefs. Sci. Rep. 8, 4109 (2018).
Briones-Fourzán, P. et al. Coral reef degradation differentially alters feeding ecology of co-occurring congeneric spiny lobsters. Front. Mar. Sci. 5, 516 (2019).
Schindler, D. E. et al. Population diversity and the portfolio effect in an exploited species. Nature 465, 609–612 (2010).
Schiettekatte, N. M. D., Brandl, S. J. & Casey, J. M. fishualize: Color Palettes Based on Fish Species (GitHub, 2019); https://nschiett.github.io/fishualize/index.html.
Randall, J. Food Habits of Fishes of the West Indies (NOAA, 1967).
O’Dea, A. et al. Defining variation in pre-human ecosystems can guide conservation: an example from a Caribbean coral reef. Sci. Rep. 10, 2922 (2020).
Mann, P., Taylor, F. W., Burke, K. & Kulstad, R. Subaerially exposed Holocene coral reef, Enriquillo Valley, Dominican Republic. Bull. Geol. Soc. Am. 95, 1084–1092 (1984).
Greer, L. & Swart, P. K. Decadal cyclicity of regional mid-Holocene precipitation: evidence from Dominican coral proxies. Paleoceanography 21, PA2020 (2006).
Cuevas, D. N., Sherman, C. E., Ramírez, W. & Hubbard, D. K. Coral growth rates from the Holocene Cañada Honda fossil reef, southwestern Dominican Republic: comparisons with modern counterparts in high sedimentation settings. Caribb. J. Sci. 45, 94–109 (2009).
Fredston-Hermann, A. L., O’Dea, A., Rodriguez, F., Thompson, W. G. & Todd, J. A. Marked ecological shifts in seagrass and reef molluscan communities since the mid-Holocene in the southwestern Caribbean. Bull. Mar. Sci. 89, 983–1002 (2013).
Wang, X. T. et al. Isotopic composition of skeleton-bound organic nitrogen in reef-building symbiotic corals: a new method and proxy evaluation at Bermuda. Geochim. Cosmochim. Acta 148, 179–190 (2015).
Wang, X. T. et al. Influence of open ocean nitrogen supply on the skeletal δ15N of modern shallow-water scleractinian corals. Earth Planet. Sci. Lett. 441, 125–132 (2016).
Donnelly, H. A. et al. Groundtruthing nitrogen isotopes as a symbiosis proxy using the facultatively symbiotic coral Oculina arbuscula. Front. Mar. Sci. 11, 1433382 (2024).
Luu, V. H. et al. Nitrogen isotope ratios across the Bermuda coral reef: implications for coral nitrogen sources and the coral-bound nitrogen isotope proxy. Front. Mar. Sci. 12, 1554418 (2025).
Sims, Z. C., Cohen, A. L., Luu, V. H., Wang, X. T. & Sigman, D. M. Uptake of groundwater nitrogen by a near-shore coral reef community on Bermuda. Coral Reefs 39, 215–228 (2020).
Wang, X. T. et al. Deep-sea coral evidence for lower Southern Ocean surface nitrate concentrations during the last ice age. Proc. Natl Acad. Sci. USA 114, 3352–3357 (2017).
Lueders-Dumont, J. A. et al. Controls on the nitrogen isotopic composition of fish otolith organic matter: lessons from a controlled diet switch experiment. Geochim. Cosmochim. Acta 316, 69–86 (2022).
Braman, R. S. & Hendrix, S. A. Nanogram nitrite and nitrate determination in environmental and biological materials by vanadium (III) reduction with chemiluminescence detection. Anal. Chem. 61, 2715–2718 (1989).
Sigman, D. M. et al. A bacterial method for the nitrogen isotopic analysis of nitrate in seawater and freshwater. Anal. Chem. 73, 4145–4153 (2001).
Weigand, M. A., Foriel, J., Barnett, B., Oleynik, S. & Sigman, D. M. Updates to instrumentation and protocols for isotopic analysis of nitrate by the denitrifier method. Rapid Commun. Mass Spectrom. 30, 1365–1383 (2016).
Boecklen, W. J., Yarnes, C. T., Cook, B. A. & James, A. C. On the use of stable isotopes in trophic ecology. Annu. Rev. Ecol. Evol. Syst. 42, 411–440 (2011).
Casey, J. M. et al. Reconstructing hyperdiverse food webs: gut content metabarcoding as a tool to disentangle trophic interactions on coral reefs. Methods Ecol. Evol. 10, 1157–1170 (2019).
Parravicini, V. et al. Delineating reef fish trophic guilds with global gut content data synthesis and phylogeny. PLoS Biol. 18, e3000702 (2020).
Kahle, D. & Wickham, H. ggmap: spatial visualization with ggplot2. R J. 5, 144–161 (2013).
Acknowledgements
This work was funded by the Smithsonian MarineGEO programme (J.A.L.-D.); the Smithsonian Tropical Research Institute (J.A.L.-D. and A.O.); the Smithsonian Institution Scholarly Studies (A.O.); the US National Science Foundation through grants EAR-2347773 (A.O., B.D.G., E.M.D. and S.F.) and EAR-1325683 (A.O.); the Secretaría Nacional de Ciencia Tecnología e Innovación, Panamá (A.O., E.M.D., B.D.G. and J.A.L.-D.); the Scott Fund of the Department of Geosciences, Princeton University (J.A.L.-D.); the David and Lucile Packard Foundation (S.F.); the Army Engineer Research and Development Center through grants W912HZ2020061-RA2 and W912HZ2520020 (J.A.L.-D., X.T.W.); the Simons Foundation through Grant No. 00016323 (X.T.W.); and Boston College start-up funds (X.T.W.). We are grateful to the Anders, Bytnar and Selin families for donations that supported Panama field and laboratory work. We acknowledge the logistical and technical support from the following people: A. Bilgray, P. Gomez, M.R. Russo, E. Groves, N. Zelizer, S. Castillo, F. Rodriguez, M. Lepore, O. Aguilera, S. Mattson, M. Hynes, S. dos Santos, M. Alvarez, P. Gondola, U. Gonzalez, J. Edlinger, D. Doughty, E. Grossman, G. Jacome, G. Abreu, V. Galvan, R. de Leon, Y. del Valle, G. Quijano, K. Griswold, K. Ripley-Dunlap, I. Ingemi, C. Vergara-Chan, A. Verdurmen, M. Pinzon, M. Herrera, R. Robertson, D. Cedeño, K. McComas, Y. Samara, C. Courtier, M. Ureña, H. Hernández, C. De Leon, J. Mate, R. Torres and the team at Sweet Bocas, Panama. We thank S.F. Gale for discussion and insights regarding Fig. 4. Collection permits for bulk sediment sampling were issued by the Ministerio de Ambiente, República de Panamá (permit SE/AO-4-18) and the Ministerio de Medio Ambiente y Recursos Naturales, República Dominicana (permit VAPB-02374). Goby, cardinalfish and grunt silhouettes are from the R package fishualize (v.0.2.3); branching coral outlines in Fig. 1b are from D. Kleine, Marine Botany UQ (ian.umces.edu/media-library) under the Attribution-ShareAlike 4.0 International (CC BY-SA 4.0) license (https://creativecommons.org/licenses/by-sa/4.0/#). This is contribution 158 from the Smithsonian’s MarineGEO and Tennenbaum Marine Observatories Network.
Author information
Authors and Affiliations
Contributions
This study was designed by J.A.L.-D. and A.O. with input from B.D.G., X.T.W., E.M.D. and D.M.S. Field and sediment sampling was done by B.D.G. and A.O. Taxonomic identification was done by B.D.G. and C.-H.L. J.A.L.-D. sampled the material and conducted the isotope analysis with help from S.O. J.A.L.-D. and E.M.D. performed the statistical analysis with input from A.O., X.T.W., S.F. and D.M.S. J.A.L.-D., A.O. and X.T.W wrote the manuscript with input from all authors. All authors were involved in discussion of the data at different stages and contributed to the final manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature thanks David Baker, Peter Roopnarine and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data figures and tables
Extended Data Fig. 1 Detailed maps of localities from which otoliths and corals were measured.
a, overview map of the Caribbean Region, b, Dominican Republic map showing both fossil and modern localities, c, Panama map showing both fossil and modern localities, d, zoomed map on Panama fossil localities, e, Dominican Republic fossil localities, and f, Dominican Republic modern localities. b–f, Google Earth imagery copyright 2022. All base maps were created using the R package ggmap71 (v.3.0.0).
Extended Data Fig. 3 δ15Noto (mean ± 1 s.d.) plotted by the most highly taxonomically resolved identification (species, genus, or family).
Data shown for a, gobies (Gobiidae; n = 45 biologically independent samples/ animals), b, cardinalfishes (Apogonidae; n = 40 biologically independent samples/ animals), and c, grunts (Haemulidae; n = 32 biologically independent samples/ animals) for fossil (F) and modern (M) time periods. δ15Noto patterns, which in the main text (Fig. 2) are aggregated by family, are retained at each level of taxonomic identity. Thus, family-specific patterns reported in the main text are representative of more highly resolved taxonomic identity, where available, of fishes in both regions and time periods. Taxonomic identity of all otoliths in the study was challenged by the fact that otoliths of young fishes have not developed some of the diagnostic morphological features, limiting the taxonomic identification of all otoliths to genus or species level. For this reason, the δ15Noto data in the main text are examined at their coarsest, family-level taxonomic identity. Fish silhouettes reproduced from ref. 51, GitHub, under a GPL-2 license.
Extended Data Fig. 4 δ15N versus fish size and versus the N content for each otolith.
a, δ15N versus reconstructed fish size. Each symbol corresponds to an individual fish otolith for which fish standard length (mm) was reconstructed. Where error bars are shown, they denote the 1 s.d. of two (n = 2) whole-process replicates from the same otolith specimen. A total of n = 131 biologically independent otolith specimens were measured for fish length for Panama (44 fossil, 44 modern) and for the Dominican Republic (21 fossil, 22 modern). Fossil and modern fish size (standard length, in mm) did not change in Panama (Wilcoxon, two-sided, P = 0.796, W = 936, r = 0.0280) or the Dominican Republic (P = 1, W = 231, r = 0). For Panama, fossil fish size was 24.2 ± 2.1 mm (1 s.e.) and modern was 26.5 ± 2.1 mm (1 s.e.); the Dominican Republic fossil fish size was 32 ± 4.9 mm (1 s.e.) and modern Dominican Republic was 22.6 ± 4.8 mm (1 s.e.). Symbols shapes as for Fig. 2. See also Supplementary Fig. S4 for intra-family patterns in δ15N versus fish size. b, Comparing N content for otoliths shows no difference between time periods for Panama (Wilcoxon, two-sided, P = 0.13, W = 1275, r = 0.157) but does show a difference for the Dominican Republic (P = 0.03, W = 319, r = 0.327) (Extended Data Table 1) (n = 136 biologically independent samples for which fish δ15N and N content were reconstructed for Panama (44 fossil, 49 modern) and the Dominican Republic (21 fossil, 22 modern). For coral N content data, see Extended Data Table 1 and Supplementary Fig. S5.
Extended Data Fig. 5 Distribution and statistical significance of bootstrapped resampling for each family.
For Panama, shifts in mean δ15N were significant for grunts (Wilcoxon, two-sided, P = 0.033, W = 26, r = 0.572), cardinalfishes (P = 3.3 × 10−26, W = 8281, r = 0.722), silversides (P = 8.7 × 10−6, W = 1169, r = 0.342), and gobies (P = 2.1 × 10−216, W = 0, r = 0.828) and shifts in the variance were significant for cardinalfishes (Kolmogorov-Smirnov, two-sided, P = 2.2 × 10−16, D = 1), silversides (P = 2.9 × 10−6, D = 1), and gobies (P = 2.2 × 10−16, D = 1). In the Dominican Republic, shifts in mean δ15N were significant for grunts (P = 0.003, W = 42, r = 0.834), cardinalfishes (P = 4.6 × 10−42, W = 4347, r = 0.993), and gobies (P = 1.9 × 10−47, W = 21732, r = 0.840) and shifts in the variance were significant for grunts (P = 0.0031, D = 1), cardinalfishes (P = 2.2 × 10−16, D = 1), and gobies (P = 2.2 × 10−16, D = 0.93) (Extended Data Table 3). Fish silhouettes reproduced from ref. 51, GitHub, under a GPL-2 license.
Supplementary information
Supplementary Information (download PDF )
Supplementary Sections 1–6 (including the Supplementary Methods, δ15Noto comparisons with known dietary patterns, alternative explanations for reduced δ15N variability, ecological explanations for reduced δ15N variability, Panama versus Dominican Republican coastal health comparisons, niche width and ecosystem stability, and Supplementary references), Figs 1–7 and Tables 1–5.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Lueders-Dumont, J.A., O’Dea, A., Dillon, E.M. et al. Fossil isotope evidence for trophic simplification on modern Caribbean reefs. Nature 651, 967–973 (2026). https://doi.org/10.1038/s41586-025-10077-z
Received:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41586-025-10077-z