Abstract
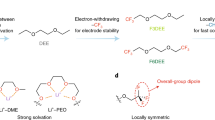
Electrolyte solvents for electrochemical devices have been dominated by oxygen (O)-based and nitrogen (N)-based ligands over the past decades1,2,3,4,5, for which the dipole–ion (Li+, Na+ and so on) interaction usually lays the foundations of ion dissociation and transport but frustrates the charge transfer process at the electrolyte–electrode interface6,7,8,9. Here, by synthesizing alkanes with monofluorinated structures, we show that fluorine (F)-based ligands with designed steric hindrance and Lewis basicity enable salt dissolution of more than 2 mol l−1. Among them, 1,3-difluoro-propane (DFP)-based Li-ion electrolyte is endowed with all merits for energy-dense and low-temperature batteries, including low viscosity (0.95 cp), high oxidation stability (>4.9 V) and ionic conductivity of 0.29 mS cm−1 at −70 °C. By incorporating F atoms in the first solvation shell, the weak F–Li+ coordination facilitates the Li plating/stripping process with Coulombic efficiency (CE) up to 99.7% and exchange current density one magnitude larger than O–Li+ coordination at −50 °C. The electrolytes further enable the operation of lithium-metal pouch cells under an electrolyte amount of less than 0.5 g Ah−1, achieving energy densities greater than 700 Wh kg−1 at room temperature and about 400 Wh kg−1 at −50 °C. The hydrofluorocarbon (HFC) electrolytes in this work provide a feasible approach to building electrochemical systems beyond traditional coordination chemistry.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$32.99 / 30 days
cancel any time
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
Data availability
The data that support the findings of this study are available within this article and its Supplementary information. Further data are available from the corresponding authors on request. Source data are provided with this paper.
References
Lu, D. et al. Ligand-channel-enabled ultrafast Li-ion conduction. Nature 627, 101–107 (2024).
Fan, X. & Wang, C. High-voltage liquid electrolytes for Li batteries: progress and perspectives. Chem. Soc. Rev. 50, 10486–10566 (2021).
Jie, Y. et al. Towards long-life 500 Wh kg−1 lithium metal pouch cells via compact ion-pair aggregate electrolytes. Nat. Energy 9, 987–998 (2024).
Shen, L. et al. Creating lithium-ion electrolytes with biomimetic ionic channels in metal–organic frameworks. Adv. Mater. 30, 1707476 (2018).
Song, X. et al. Practical lithium-sulfur batteries: beyond the conventional electrolyte concentration. ACS Energy Lett. 9, 5576–5586 (2024).
Holoubek, J. et al. Tailoring electrolyte solvation for Li metal batteries cycled at ultra-low temperature. Nat. Energy 6, 303–313 (2021).
Piao, N. et al. Designing temperature-insensitive solvated electrolytes for low-temperature lithium metal batteries. J. Am. Chem. Soc. 146, 18281–18291 (2024).
Shi, J. et al. An amphiphilic molecule-regulated core-shell-solvation electrolyte for Li-metal batteries at ultra-low temperature. Angew. Chem. Int. Ed. 135, e202218151 (2023).
Chen, Y. et al. Steric effect tuned solvation enabling stable cycling of high-voltage lithium metal battery. J. Am. Chem. Soc. 143, 18703–18713 (2021).
Zhang, J. et al. Lithium metal anodes with nonaqueous electrolytes. Chem. Rev. 120, 13312–13348 (2020).
Xu, K. Nonaqueous liquid electrolytes for lithium-based rechargeable batteries. Chem. Rev. 104, 4303–4418 (2004).
Yu, Z. et al. Rational solvent molecule tuning for high-performance lithium metal battery electrolytes. Nat. Energy 7, 94–106 (2022).
Amanchukwu, C. V. et al. A new class of ionically conducting fluorinated ether electrolytes with high electrochemical stability. J. Am. Chem. Soc. 142, 7393–7403 (2020).
Ma, P. et al. Effect of building block connectivity and ion solvation on electrochemical stability and ionic conductivity in novel fluoroether electrolytes. ACS Cent. Sci. 7, 1232–1244 (2021).
Zhang, S. et al. Oscillatory solvation chemistry for a 500 Wh kg−1 Li metal pouch cell. Nat. Energy 9, 1285–1296 (2024).
Yin, Y. et al. Fire-extinguishing, recyclable liquefied gas electrolytes for temperature-resilient lithium-metal batteries. Nat. Energy 7, 548–559 (2022).
Wu, Z. et al. Deciphering and modulating energies of solvation structure of solvation structure enables aggressive high-voltage chemistry of Li metal batteries. Chem 9, 656–664 (2023).
Rustomji, C. S. et al. Liquefied gas electrolytes for electrochemical energy storage devices. Science 356, eaal4263 (2017).
Staley, R. H. et al. Intrinsic acid-base properties of molecules. Binding energies of lithium(1+) ion to .pi.- and n-donor bases. J. Am. Chem. Soc. 97, 5920–5921 (1975).
Li, Z. et al. Non-polar ether-based electrolyte solutions for stable high-voltage non-aqueous lithium metal batteries. Nat. Commun. 14, 868 (2023).
Yang, S. et al. Regulating the electrochemical reduction kinetics by the steric hindrance effect for a robust Zn metal anode. Energy Environ. Sci. 17, 1095–1106 (2024).
Crabb, E. et al. Electrolyte dependence of Li+ transport mechanisms in small molecule solvents from classical molecular dynamics. J. Phys. Chem. B 128, 3427–3441 (2024).
Son, C. Y. et al. Ion transport in small-molecule and polymer electrolytes. J. Chem. Phys. 153, 100903 (2020).
Efaw, C. M. et al. Localized high-concentration electrolytes get more localized through micelle-like structures. Nat. Mater. 22, 1531–1539 (2023).
Cao, X. et al. Optimization of fluorinated orthoformate based electrolytes for practical high-voltage lithium metal batteries. Energy Storage Mater. 34, 76–84 (2021).
Park, E. et al. Exploiting the steric effect and low dielectric constant of 1,2-dimethoxypropane for 4.3 V lithium metal batteries. ACS Energy Lett. 8, 179–188 (2023).
Zhang, G. et al. A monofluoride ether-based electrolyte solution for fast-charging and low temperature non-aqueous lithium metal batteries. Nat. Commun. 14, 1081 (2023).
Xu, J. et al. Revealing the anion–solvent interaction for ultralow temperature lithium metal batteries. Adv. Mater. 36, 2306462 (2024).
Li, T. et al. Stable anion-derived solid electrolyte interphase in lithium metal batteries. Angew. Chem. Int. Ed. 133, 22865–22869 (2021).
Wu, Q. et al. Effect of the electric double layer (EDL) in multicomponent electrolyte reduction and solid electrolyte interphase (SEI) formation in lithium batteries. J. Am. Chem. Soc. 145, 2473–2484 (2023).
Kim, S. C. et al. Potentiometric measurement to probe solvation energy and its correlation to lithium battery cyclability. J. Am. Chem. Soc. 143, 10301–10308 (2021).
Tang, T. Long-lifespan 522 Wh kg−1 lithium metal pouch cell enabled by compound additives engineering. Angew. Chem. Int. Ed. 64, e202417471 (2025).
Qiao, R. et al. Non-fluorinated electrolytes with micelle-like solvation for ultra-high-energy density lithium metal batteries. Chem 11, 102306 (2025).
Ji, H. et al. Liquid–liquid interfacial tension stabilized Li-metal batteries. Nature 643, 1255–1262 (2025).
Troup, R. I. et al. Skipped fluorination motifs, synthesis of building blocks and comparison of lipophilicity trends with vicinal and isolated fluorinated motifs. J. Org. Chem. 86, 1882–1900 (2021).
Frisch, M. et al. Gaussian 16 Rev. C.01. Gaussian Inc. (2016).
Weigend, F. et al. Balanced basis sets of split valence, triple zeta valence and quadruple zeta valence quality for H to Rn: design and assessment of accuracy. Phys. Chem. Chem. Phys. 7, 3297–3305 (2005).
Kresse, G. et al. Efficient iterative schemes for ab initio total-energy calculations using a plane-wave basis set. Phys. Rev. B 54, 11169–11186 (1996).
Perdew, J. P. et al. Generalized gradient approximation made simple. Phys. Rev. Lett. 77, 3865–3868 (1996).
Berendsen, H. J. C. et al. GROMACS: a message-passing parallel molecular dynamics implementation. Comput. Phys. Commun. 91, 43–56 (1995).
Wang, J. et al. Development and testing of a general amber force field. J. Comput. Chem. 25, 1157–1174 (2004).
Singh, U. C. et al. An approach to computing electrostatic charges for molecules. J. Comput. Chem. 5, 129–145 (1984).
Ravikumar, B. et al. Molecular dynamics investigation of electric field altered behavior of lithium ion battery electrolytes. J. Mol. Liq. 300, 112252 (2020).
Kühne, T. D. et al. CP2K: an electronic structure and molecular dynamics software package - Quickstep: efficient and accurate electronic structure calculations. J. Chem. Phys. 152, 194103 (2020).
Acknowledgements
This work was supported by the National Natural Science Foundation of China (nos. 22393900, 22372083, 52201259, 22121005, 92472122), the National Key R&D Program of China (nos. 2021YFB2500300, 2024YFB3814500), Fundamental and Interdisciplinary Disciplines Breakthrough Plan of the Ministry of Education of China (no. JYB2025XDXM410), the Natural Science Foundation of Tianjin (nos. 22JCZDJC00380, 24ZXZSSS00390), Young Elite Scientist Sponsorship Program by CAST and the Natural Science Foundation of Chongqing (no. CSTB2023NSCQ-LZX0084).
Author information
Authors and Affiliations
Contributions
L.W., J.C. and Q.Z. conceived the idea. L.W. prepared and characterized the solvents and electrolytes, as well as the electrochemical performances. Jinyu Zhang, W.X. and Y.N. performed the theoretical calculations. Yong Li performed the pouch-cell evaluations. Y. Lu conducted the ultrasonic scanning test. Z.F., S.R., Jie Zhang and Yawen Li conducted the Raman, differential scanning calorimetry and SAXS tests. L.W., J.C. and Q.Z. co-wrote the paper. All authors discussed the results and commented on the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature thanks Dong Zhou and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Wu, L., Zhang, J., Li, Y. et al. Hydrofluorocarbon electrolytes for energy-dense and low-temperature batteries. Nature 651, 383–389 (2026). https://doi.org/10.1038/s41586-026-10210-6
Received:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41586-026-10210-6