Abstract
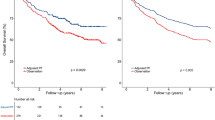
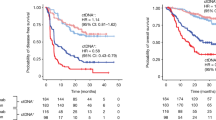
Despite promising data showing that circulating tumour DNA (ctDNA) dynamics during treatment can inform real-time tumour response and recurrence risk1, how best to translate these insights into actionable clinical decision-making remains unclear. Here we report results from the EP-STAR trial—a multi-centre, ctDNA-driven, risk-adapted, non-randomized phase II study (NCT04072107; ClinicalTrials.gov) testing whether a risk-adaptive treatment (RAT) strategy guided by on-treatment ctDNA dynamics can meaningfully improve survival, using nasopharyngeal carcinoma as a model. Eligible patients were enrolled and began treatment with standard-of-care gemcitabine–cisplatin neoadjuvant chemotherapy (GP-NAC; the P in this abbreviation stands for platinum)2, followed by RAT or standard-of-care chemoradiotherapy guided by ctDNA clearance trajectory during GP-NAC. Protocol-eligible patients who did not receive RAT, drawn from a prospectively registered ctDNA biomarker cohort (NCT03855020)3, served as a non-randomized, contemporaneous no-RAT external cohort. The primary end-point was failure-free survival (FFS) in the RAT group. After a median follow-up of 47.3 months, the 3-year FFS was 89.1% (83.2–95.0%) in the RAT group (n = 110). Patients who received RAT showed significantly improved FFS (P = 0.003, log–rank test) compared with the no-RAT external cohort (hazard ratio = 0.41 [0.23–0.75]; P = 0.004, Cox regression model). The RAT strategy was well-tolerated with no treatment-related deaths. Collectively, these data show that a ctDNA-driven RAT paradigm could be a promising strategy to improve survival, challenging the conventional fixed-course, static treatment approach.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$32.99 / 30 days
cancel any time
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
Data availability
De-identified, anonymized, participant-level data have been deposited in the Mendeley Data repository (https://doi.org/10.17632/wk275rgy98.1) and are publicly available as of the date of publication. The scRNA-seq sequencing data were obtained from published research5, and downloaded from the CNGB Sequence Archive (https://db.cngb.org/cnsa/) under the accession number CNP0001341. The public database MSigDB, which provides a resource of annotated gene sets for use, is available at https://www.gsea-msigdb.org. Source data are provided with this paper.
References
Ignatiadis, M., Sledge, G. W. & Jeffrey, S. S. Liquid biopsy enters the clinic—implementation issues and future challenges. Nat. Rev. Clin. Oncol. 18, 297–312 (2021).
Zhang, Y. et al. Gemcitabine and cisplatin induction chemotherapy in nasopharyngeal carcinoma. N. Engl. J. Med. 381, 1124–1135 (2019).
Lv, J. et al. Longitudinal on-treatment circulating tumor DNA as a biomarker for real-time dynamic risk monitoring in cancer patients: the EP-SEASON study. Cancer Cell 42, 1401–1414 (2024).
Kurtz, D. M. et al. Dynamic risk profiling using serial tumor biomarkers for personalized outcome prediction. Cell 178, 699–713 (2019).
Lv, J. et al. The tumor immune microenvironment of nasopharyngeal carcinoma after gemcitabine plus cisplatin treatment. Nat. Med. 29, 1424–1436 (2023).
Wang, X. Q. et al. Spatial predictors of immunotherapy response in triple-negative breast cancer. Nature 621, 868–876 (2023).
Cohen, S. A., Liu, M. C. & Aleshin, A. Practical recommendations for using ctDNA in clinical decision making. Nature 619, 259–268 (2023).
Nabet, B. Y. et al. Noninvasive early identification of therapeutic benefit from immune checkpoint inhibition. Cell 183, 363–376 (2020).
Pan, Y. et al. Dynamic circulating tumor DNA during chemoradiotherapy predicts clinical outcomes for locally advanced non-small cell lung cancer patients. Cancer Cell 41, 1763–1773 (2023).
Lv, J. et al. Liquid biopsy tracking during sequential chemo-radiotherapy identifies distinct prognostic phenotypes in nasopharyngeal carcinoma. Nat. Commun. 10, 3941 (2019).
Lv, J. et al. Improving on-treatment risk stratification of cancer patients with refined response classification and integration of circulating tumor DNA kinetics. BMC Med 20, 268 (2022).
Brierley, J. D. et al. (eds) TNM Classification of Malignant Tumours 8th edn (Wiley-Blackwell, 2016).
Chen, Y. P. et al. Low-dose metronomic chemotherapy improves tumor control in nasopharyngeal carcinoma. Cancer Commun. 42, 909–912 (2022).
Liu, X. et al. Induction-concurrent chemoradiotherapy with or without sintilimab in patients with locoregionally advanced nasopharyngeal carcinoma in China (CONTINUUM): a multicentre, open-label, parallel-group, randomised, controlled, phase 3 trial. Lancet 403, 2720–2731 (2024).
Liang, Y.-L. et al. Adjuvant PD-1 blockade with camrelizumab for nasopharyngeal carcinoma: the DIPPER randomized clinical trial. J. Am. Med. Assoc. 333, 1589–1598 (2025).
Dong, S. et al. Circulating tumor DNA-guided de-escalation targeted therapy for advanced non-small cell lung cancer: a nonrandomized controlled trial. JAMA Oncol. 10, 932–940 (2024).
Tie, J. et al. Circulating tumor DNA analysis guiding adjuvant therapy in stage II colon cancer. N. Engl. J. Med. 386, 2261–2272 (2022).
Tie, J. et al. Circulating tumor DNA analysis guiding adjuvant therapy in stage II colon cancer: 5-year outcomes of the randomized DYNAMIC trial. Nat. Med. 31, 1509–1518 (2025).
Nakamura, Y. et al. ctDNA-based molecular residual disease and survival in resectable colorectal cancer. Nat. Med. 30, 3272–3283 (2024).
Tie, J. et al. Circulating tumor DNA-guided adjuvant therapy in locally advanced colon cancer: the randomized phase 2/3 DYNAMIC-III trial. Nat. Med. 31, 4291–4300 (2025).
Chen, L. et al. Postoperative human papilloma virus circulating tumor DNA guided adjuvant therapy for human papilloma virus-related oropharyngeal carcinoma (PATH study). Int. J. Radiat. Oncol. Biol. Phys. 124, 676–685 (2026).
Huang, Z. et al. Plasma Epstein–Barr virus DNA temporal clearance pattern during induction-concurrent (chemo)radiation therapy for risk stratification in nasopharyngeal carcinoma. Int. J. Radiat. Oncol. Biol. Phys. 123, 129–140 (2025).
Chen, Y. P. et al. Metronomic capecitabine as adjuvant therapy in locoregionally advanced nasopharyngeal carcinoma: a multicentre, open-label, parallel-group, randomised, controlled, phase 3 trial. Lancet 398, 303–313 (2021).
Tang, L.-L. et al. CACA guidelines for holistic integrative management of nasopharyngeal carcinoma. Holist. Integr. Oncol. 2, 24, (2023).
Liu, S. L. et al. Neoadjuvant and adjuvant toripalimab for locoregionally advanced nasopharyngeal carcinoma: a randomised, single-centre, double-blind, placebo-controlled, phase 2 trial. Lancet Oncol. 25, 1563–1575 (2024).
Langer, C. J. et al. Carboplatin and pemetrexed with or without pembrolizumab for advanced, non-squamous non-small-cell lung cancer: a randomised, phase 2 cohort of the open-label KEYNOTE-021 study. Lancet Oncol. 17, 1497–1508 (2016).
Rha, S. Y. et al. Pembrolizumab plus chemotherapy versus placebo plus chemotherapy for HER2-negative advanced gastric cancer (KEYNOTE-859): a multicentre, randomised, double-blind, phase 3 trial. Lancet Oncol. 24, 1181–1195 (2023).
Le, Q. T. et al. An international collaboration to harmonize the quantitative plasma Epstein–Barr virus DNA assay for future biomarker-guided trials in nasopharyngeal carcinoma. Clin. Cancer Res. 19, 2208–2215 (2013).
Tang, S. Q. et al. Identifying optimal clinical trial candidates for locoregionally advanced nasopharyngeal carcinoma: analysis of 9468 real-world cases and validation by two phase 3 multicentre, randomised controlled trial. Radiother. Oncol. 167, 179–186 (2022).
Lin, J. C. et al. Quantification of plasma Epstein–Barr virus DNA in patients with advanced nasopharyngeal carcinoma. N. Engl. J. Med. 350, 2461–2470 (2004).
Chan, K. C. A. et al. Analysis of plasma Epstein–Barr virus DNA to screen for nasopharyngeal cancer. N. Engl. J. Med. 377, 513–522 (2017).
Lo, Y. M. et al. Quantitative analysis of cell-free Epstein–Barr virus DNA in plasma of patients with nasopharyngeal carcinoma. Cancer Res. 59, 1188–1191 (1999).
Schmidt, R. et al. Sample size calculation for the one-sample log-rank test. Stat. Med. 34, 1031–1040 (2015).
Borgan, Ø & Liestøl, K. A note on confidence intervals and bands for the survival function based on transformations. Scand. J. Stat. 17, 35–41 (1990).
Schoenfeld, D. A. Sample-size formula for the proportional-hazards regression model. Biometrics 39, 499–503 (1983).
Kurz, C. F., Krzywinski, M. & Altman, N. Propensity score matching. Nat. Methods 21, 1770–1772 (2024).
Osoba, D., Rodrigues, G., Myles, J., Zee, B. & Pater, J. Interpreting the significance of changes in health-related quality-of-life scores. J. Clin. Oncol. 16, 139–144 (1998).
Bertram, M. Y. et al. Cost-effectiveness thresholds: pros and cons. Bull. World Health Organ. 94, 925–930 (2016).
Acknowledgements
This trial was supported by grants from the Science Fund for Creative Research Groups of the National Natural Science Foundation of China (82521003 to Y.S.), the National Natural Science Foundation of China for Excellent Young Scientists Fund (82522065 to J.L.), the National Natural Science Foundation of China (82441026 to Y.S., 92259202 to Y.S. and 81803105 to J.-B.L.), the Fundamental and Interdisciplinary Disciplines Breakthrough Plan of the Ministry of Education of China (JYB2025XDXM611 to J.M.), the Guangdong Basic and Applied Basic Research Foundation (2024A1515030248 to G.-Q.Z.), the Young Talents Program of SYSUCC (YTP-SYSUCC-0103 to J.L.), the Cancer Innovative Research Program of SYSUCC (CIRP-SYSUCC-0010 to Y.S.), the Overseas Expertise Introduction Project for Discipline Innovation (111 Project) (B14035 to J.M.) and the National Medical Research Council Singapore Clinician Scientist Award (NMRC/CSA-INV/0027/2018, CSAINV20-nov-0021 to M.L.K.C.). We thank all of the participants and their families in this study. We also thank Innovent Pharmaceutical for providing free sintilimab and logistical support for all participants; J. T. S. Wee and T. S. Huey for their insightful comments on the trial design; and Y.-X. Li, X.-Z. Chen and Y. Guo for their contributions as members of the independent data monitoring committee. The trial sponsors did not have roles in data collection, interpretation or manuscript preparation.
Author information
Authors and Affiliations
Contributions
The trial was investigator-initiated and designed by Y.S. and J.L. Lead investigators from each centre (Y.S., J.-H. L. and N.Z.) contributed to patient recruitment and data acquisition. J.L., D.-X.Z., Z.-L.Y., J.-B.L., J.M. and Y.S. drafted the article and performed the data interpretation. J.L., D.-X.Z., Z.-L.Y., X.-D.X., M.L.K.C., S.H.H., J.-B.L., G.-Q.Z., J.M. and Y.S. revised the manuscript. G.-Q.Z., L.-L.T., W.-F.L., L-S.C., B.D., T.-S.G., J.-Y.Y., L.C., Y.-P.M., R.G., L.L., Y.-P.C., Y.Z., X.L., Z.-X.L., L.-X.X., P.-Y.Y., K.C., H.-Y.Z., Y.-S.J., H.-L.H. and X.-H.T. contributed to clinical management and/or QoL surveys. Z.-L.Y., Z.-C.Z., Z.-M.D. and L.-L.Z. contributed to ctDNA testing. J.-P.Y., L.-Z.L., L.T. and H.-J.L. contributed to the diagnosis and efficacy evaluation of patients. All authors reviewed, revised and approved the final manuscript.
Corresponding authors
Ethics declarations
Competing interests
M.L.K.C. reports personal fees for advisory board and education activities and funding support from BeiGene (tislelizumab) for an investigator-initiated trial; personal fees from TopAlliance Biosciences (toripalimab) for advisory board activities; personal fees from Astellas, Pfizer, MSD, AstraZeneca, Varian, Janssen, IQVIA and Telix Pharmaceuticals; nonfinancial support from AstraZeneca; nonfinancial support from Veracyte; and personal fees and grants from Bayer. M.L.K.C. consults for ImmunoSCAPE; is a co-inventor on the patent ‘High sensitivity lateral flow immunoassay for detection of analyte in sample’ (10202107837T, Singapore); and serves on the board of directors of Digital Life Line, which owns the licensing agreement of the patent. The remaining authors declare no competing interests.
Peer review
Peer review information
Nature thanks the anonymous reviewers for their contribution to the peer review of this work. Peer reviewer reports are available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data figures and tables
Extended Data Fig. 1 Blood sample collection for ctDNA assessment.
Blood samples for ctDNA assessment were collected at pretreatment (T0), after each cycle of GP-NAC (T1–T3, 21 days after the prior cycle and before the next), and weekly during the adaptive phase (T4–T15) for patients with detectable ctDNA, continuing until clearance was confirmed in two consecutive tests). At the completion of CCRT, ctDNA assessment was conducted for all enrolled patients. DDP, cisplatin; RT, radiotherapy.
Extended Data Fig. 2 Outline of the participant selection process in the no-RAT external cohort.
Patients from the EP-SEASON study who met the same eligibility criteria as the EP-STAR trial and received SOC without modifications were included as a pre-planned non-randomized external cohort for efficacy comparison.
Extended Data Fig. 3 Secondary survival outcomes of patients in the intention-to-treat population.
a–c, Kaplan–Meier curves of OS (a), DMFS (b) and LRFS (c) in the RAT group and SOC arm.
Extended Data Fig. 4 Biological correlative analysis of the risk-adaptive interventions across ctDNA-defined risk subgroups.
a, Violin plot showing the signature score for chemotherapy sensitivity across ctDNA-defined risk subgroups (n = 4 samples for low-risk subgroup, n = 6 samples for intermediate-risk subgroup, and n = 5 samples for high-risk subgroup). b, Venn plot showing the overlap of upregulated genes after GP-NAC across ctDNA-defined risk subgroups. c, Gene Ontology (GO) and Kyoto Encyclopedia of Genes and Genomes (KEGG) enrichment plot of tumour cell upregulated signalling pathways after GP-NAC in low-risk patients. d, Percentage of innate-like B cells (ILBs) after GP-NAC in low-risk patients (n = 4 pairs). e, Violin plot showing the signature score for cytotoxic T cells after GP-NAC in low-risk patients (n = 4 pairs). f, Violin plot showing the signature score for CSC after GP-NAC in intermediate-risk patients (n = 4 samples for low-risk subgroup, n = 6 samples for intermediate-risk subgroup, and n = 5 samples for high-risk subgroup). g, Violin plot showing the signature score for exhaustion T cells after GP-NAC in high-risk patients (n = 4 samples for low-risk subgroup, n = 6 samples for intermediate-risk subgroup, and n = 5 samples for high-risk subgroup). The box plot indicates the median (centre), 25th and 75th percentiles (box boundaries), and minimum and maximum (the whiskers) in a,d–g. Significance was determined by a two-sided Wilcoxon rank-sum test for a,e–g, a two-sided, hypergeometric test for c without correction for multiple comparisons and a two-sided t-test for d.
Supplementary information
Supplementary Information (download PDF )
This file contains Supplementary Methods, Supplementary Tables 1–26 and Supplementary Figs. 1−4
Supplementary Data (download ZIP )
Source data for Supplementary Figs. 1, 2 and 4
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Lv, J., Zheng, DX., Liang, JH. et al. Risk-adaptive therapy guided by dynamic ctDNA in nasopharyngeal carcinoma. Nature (2026). https://doi.org/10.1038/s41586-026-10244-w
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41586-026-10244-w