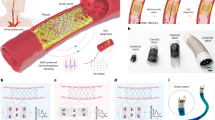
Abstract
Conventional approaches for vascular graft stenosis diagnostics, including X-ray angiography, magnetic resonance imaging and Doppler ultrasound, although highly accurate, are cumbersome, used intermittently and often do not detect stenosis early enough, leading to diagnosis only after substantial narrowing. Here we report a magnetoelastic vascular graft (MVG) for post-implantation stenosis diagnosis that is hemodynamics-driven, biocompatible and waterproof. It enables wireless, real-time and continuous diagnosis of stenosis by converting arterial hemodynamics into high-fidelity electrical signals. The MVGs were scalably manufactured with customizable diameters and tested in vivo in the femoral arteries of rats and swine through microsurgical anastomosis. The anastomosed MVGs restored blood flow and identified the location and severity of induced stenosis through artificial intelligence-assisted analysis. Furthermore, a 4-month in vivo study in rats verified the stability and biocompatibility of the MVGs in the host, with no evident signs of an adverse immune response. The MVG is expected to advance existing vascular graft solutions and improve vascular disease management.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$32.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
Data availability
All data supporting the findings of this study are available within the article and its Supplementary Information. Raw imaging data are available in a publicly accessible repository (https://doi.org/10.6084/m9.figshare.28326497)53.
Code availability
As the code was adopted and modified on the basis of a previous publication, the source of the code is given in the Supplementary Information.
References
Timmis, A. et al. European Society of Cardiology: cardiovascular disease statistics 2021. Eur. Heart J. 43, 716–799 (2022).
Tsao, C. W. et al. Heart disease and stroke statistics—2023 update: a report from the American Heart Association. Circulation 147, e93–e621 (2023).
Beerkens, F. J. et al. Contemporary coronary artery bypass graft surgery and subsequent percutaneous revascularization. Nat. Rev. Cardiol. 19, 195–208 (2022).
Gutowski, P. et al. Arterial reconstruction with human bioengineered acellular blood vessels in patients with peripheral arterial disease. J. Vasc. Surg. 72, 1247–1258 (2020).
Vachharajani, T. J., Taliercio, J. J. & Anvari, E. New devices and technologies for hemodialysis vascular access: a review. Am. J. Kidney Dis. 78, 116–124 (2021).
Niklason, L. E. & Lawson, J. H. Bioengineered human blood vessels. Science 370, eaaw8682 (2020).
Kirkton, R. D. et al. Bioengineered human acellular vessels recellularize and evolve into living blood vessels after human implantation. Sci. Transl. Med. 11, eaau6934 (2019).
Wang, D. et al. Microfluidic bioprinting of tough hydrogel-based vascular conduits for functional blood vessels. Sci. Adv. 8, eabq6900 (2022).
Seifu, D. G., Purnama, A., Mequanint, K. & Mantovani, D. Small-diameter vascular tissue engineering. Nat. Rev. Cardiol. 10, 410–421 (2013).
Drews, J. D. et al. Spontaneous reversal of stenosis in tissue-engineered vascular grafts. Sci. Transl. Med. 12, eaax6919 (2020).
Moore, M. J., Tan, R. P., Yang, N., Rnjak-Kovacina, J. & Wise, S. G. Bioengineering artificial blood vessels from natural materials. Trends Biotechnol. 40, 693–707 (2022).
Nezarati, R. M., Eifert, M. B., Dempsey, D. K. & Cosgriff-Hernandez, E. Electrospun vascular grafts with improved compliance matching to native vessels. J. Biomed. Mater. Res. B Appl. Biomater. 103, 313–323 (2015).
Quin, J. A. et al. Coronary artery bypass grafting transit time flow measurement: graft patency and clinical outcomes. Ann. Thorac. Surg. 112, 701–707 (2021).
Kim, K.-B. et al. Twenty-year experience with off-pump coronary artery bypass grafting and early postoperative angiography. Ann. Thorac. Surg. 109, 1112–1119 (2020).
Wolf, F. et al. MR and PET–CT monitoring of tissue-engineered vascular grafts in the ovine carotid artery. Biomaterials 216, 119228 (2019).
Fananapazir, G. et al. Screening for transplant renal artery stenosis: ultrasound-based stenosis probability stratification. Am. J. Roentgenol. 209, 1064–1073 (2017).
Frija, G. et al. How to improve access to medical imaging in low- and middle-income countries? eClinicalMedicine 38, 101034 (2021).
Weiss, D. J. et al. Global maps of travel time to healthcare facilities. Nat. Med. 26, 1835–1838 (2020).
Elbadawi, A. et al. Outcomes of reoperative coronary artery bypass graft surgery in the United States. J. Am. Heart Assoc. 9, e016282 (2020).
Zhou, Y. et al. Giant magnetoelastic effect in soft systems for bioelectronics. Nat. Mater. 20, 1670–1676 (2021).
Zhao, X. et al. A reconfigurable and conformal liquid sensor for ambulatory cardiac monitoring. Nat. Commun. 15, 8492 (2024).
Zhao, X. et al. Permanent fluidic magnets for liquid bioelectronics. Nat. Mater. 23, 703–710 (2024).
Kim, Y. & Zhao, X. Magnetic soft materials and robots. Chem. Rev. 122, 5317–5364 (2022).
Liu, J. et al. Bioresorbable shape-adaptive structures for ultrasonic monitoring of deep-tissue homeostasis. Science 383, 1096–1103 (2024).
Walden, R., L’Italien, G. J., Megerman, J. & Abbott, W. M. Matched elastic properties and successful arterial grafting. Arch. Surg. 115, 1166–1169 (1980).
Ku, H.-C., Lee, S.-Y., Wu, Y.-K. A., Yang, K.-C. & Su, M.-J. A model of cardiac remodeling through constriction of the abdominal aorta in rats. J. Vis. Exp. 2, e54818 (2016).
Rocha, R. V. et al. Multiple arterial grafting is associated with better outcomes for coronary artery bypass grafting patients. Circulation 138, 2081–2090 (2018).
Ma, Y. et al. Relation between blood pressure and pulse wave velocity for human arteries. Proc. Natl Acad. Sci. USA 115, 11144–11149 (2018).
Seo, Y. K. et al. Correlation between scaffold in vivo biocompatibility and in vitro cell compatibility using mesenchymal and mononuclear cell cultures. Cell Biol. Toxicol. 25, 513–522 (2009).
Gelman, S., Warner, D. S. & Warner, M. A. Venous function and central venous pressure: a physiologic story. Anesthesiology 108, 735–748 (2008).
Oglat, A. A. et al. A review of medical doppler ultrasonography of blood flow in general and especially in common carotid artery. J. Med. Ultrasound 26, 3–13 (2018).
Shorten, C. & Khoshgoftaar, T. M. A survey on image data augmentation for deep learning. J. Big Data 6, 60 (2019).
Szegedy, C. et al. Going deeper with convolutions. In Proc. 2015 IEEE Conference On Computer Vision and Pattern Recognition 1–9 (Curran Associates, 2015).
MATLAB. Classify time series using wavelet analysis and deep learning. MATLAB Help Center https://www.mathworks.com/help/wavelet/ug/classify-time-series-using-wavelet-analysis-and-deep-learning.html (2024).
Delwatta, S. L. et al. Reference values for selected hematological, biochemical and physiological parameters of Sprague–Dawley rats at the animal house, Faculty of Medicine, University of Colombo, Sri Lanka. Animal Model Exp. Med. 1, 250–254 (2018).
Charles River Laboratories. CD IGS rat model information sheet https://www.criver.com/resources/cd-igs-rat-model-information-sheet (Charles River, 2007).
de Kort, M. et al. Historical control data for hematology parameters obtained from toxicity studies performed on different Wistar rat strains: acceptable value ranges, definition of severity degrees, and vehicle effects. Toxicol. Res. Appl. 4, 2397847320931484 (2020).
Seo, S. et al. Artificial neural network for Slice Encoding for Metal Artifact Correction (SEMAC) MRI. Magn. Reson. Med. 84, 263–276 (2020).
Lee, E. M. et al. Improving MR image quality in patients with metallic implants. Radiographics 41, E126–E137 (2021).
Kwon, K., Kim, D., Kim, B. & Park, H. Unsupervised learning of a deep neural network for metal artifact correction using dual-polarity readout gradients. Magn. Reson. Med. 83, 124–138 (2020).
Alexander, J. H. & Smith, P. K. Coronary-artery bypass grafting. N. Engl. J. Med. 374, 1954–1964 (2016).
Enriquez-Marulanda, A. et al. The evolution and future directions of bypass surgery. J. Neurosurg. 142, 40–51 (2025).
Ates, H. C. et al. End-to-end design of wearable sensors. Nat. Rev. Mater. 7, 887–907 (2022).
Moin, A. et al. A wearable biosensing system with in-sensor adaptive machine learning for hand gesture recognition. Nat. Electron. 4, 54–63 (2021).
Ballard, Z., Brown, C., Madni, A. M. & Ozcan, A. Machine learning and computation-enabled intelligent sensor design. Nat. Mach. Intell. 3, 556–565 (2021).
Varghese, C., Harrison, E. M., O’Grady, G. & Topol, E. J. Artificial intelligence in surgery. Nat. Med. 30, 1257–1268 (2024).
Kwon, K. et al. A battery-less wireless implant for the continuous monitoring of vascular pressure, flow rate and temperature. Nat. Biomed. Eng. 7, 1215–1228 (2023).
Boutry, C. M. et al. Biodegradable and flexible arterial-pulse sensor for the wireless monitoring of blood flow. Nat. Biomed. Eng. 3, 47–57 (2019).
Park, K. et al. Resealable antithrombotic artificial vascular graft integrated with a self-healing blood flow sensor. ACS Nano 17, 7296–7310 (2023).
Wang, C. et al. Monitoring of the central blood pressure waveform via a conformal ultrasonic device. Nat. Biomed. Eng. 2, 687–695 (2018).
Lin, M. et al. A fully integrated wearable ultrasound system to monitor deep tissues in moving subjects. Nat. Biotechnol. 42, 448–457 (2024).
Sykes, M. & Sachs, D. H. Progress in xenotransplantation: overcoming immune barriers. Nat. Rev. Nephrol. 18, 745–761 (2022).
Chen, G. et al. Raw imaging data. figshare https://doi.org/10.6084/m6089.figshare.28326497 (2025).
Acknowledgements
J.C. acknowledges the Vernroy Makoto Watanabe Excellence in Research Award at the UCLA Samueli School of Engineering, the American Heart Association Innovative Project Award (award ID, 23IPA1054908), the American Heart Association Transformational Project Award (award ID, 23TPA1141360), the American Heart Association’s Second Century Early Faculty Independence Award (award ID, 23SCEFIA1157587), the Office of Naval Research Young Investigator Award (award ID, N00014-24-1-2065), National Institutes of Health (NIH) grant (award ID, R01 HL175135 and R01 CA287326) and National Science Foundation Grant (award no. 2425858). S.L. acknowledges the support from NIH (grant no. NS126918) and from the Broad Stem Cell Research Center, the Jonsson Comprehensive Cancer Center and California NanoSystems Institute at UCLA. G.C. acknowledges the Amazon Doctoral Student Fellowship from Amazon AWS and the UCLA Science Hub for Humanity and Artificial Intelligence. G.C. also acknowledges the Predoctoral Fellowship from the American Heart Association and The VIVA Foundation (award ID, 24PRE1193744). Y.-R.L. acknowledges the UCLA MIMG M. John Pickett Post-Doctoral Fellow Award, a CIRM-BSCRC Postdoctoral Fellowship and a UCLA Sydney Finegold Postdoctoral Award. We also acknowledge the MRI guidance provided by S. Xu and the Crump Preclinical Imaging Technology Center with the support from NIH Cancer Center Support Grant (grant no. 2 P30 CA016042-44) and NIH S10 Shared Instrumentation for Animal Research Grant (grant no. 1 S10 OD026917-01A1).
Author information
Authors and Affiliations
Contributions
J.C. and S.L. conceived the idea and guided the entire project. G.C., J.C., T.C., Z.L. and S.L. designed the experiments, analyzed the data, drew the figures and composed the manuscript. G.C., T.C., X.Z., W.K., S.P. and G.P.C. contributed to the animal study. T.C., Z.L. and S.L. performed the biocompatibility tests. G.C., X.Z., J.C., Y.Z. and X.G. contributed to the device design, fabrication and characterization. Y.-R.L. and G.C. performed the immune profiling experiment. J.C., S.L., G.P.C. and S.P. contributed to the funding acquisition. S.P., K.S., G.C., G.P.C., J.C. and S.L. revised the manuscript. All authors have read the paper, agreed to its content and approved the final submission.
Corresponding authors
Ethics declarations
Competing interests
A patent application related to this work has been filed by UCLA, listing J.C., S.L. and G.C. as the inventors. The other authors declare no competing interests.
Peer review
Peer review information
Nature Biotechnology thanks Tohid Didar, Narutoshi Hibino, Bozhi Tian and Sheng Xu for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data
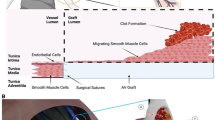
Extended Data Fig. 1 Arterial anastomosis on the swine femoral artery.
a, b, Schematic illustration of femoral artery anastomosis in a swine model, illustrating (a) surgical exposure of the femoral artery and (b) the end-to-end arterial anastomosis procedure. Partially created with BioRender. c, Photograph of the surgeon’s hands performing the end-to-end arterial anastomosis on the swine femoral artery to anastomose the MVG. Scale bar, 6 mm. d–f, Clear surgical view of the step-by-step procedures of anastomosing the MVG to the swine femoral artery. (d) Precise dissection of muscle and connective tissues was performed to expose the targeted segment of the femoral artery for replacement. Scale bar, 4 mm. (e) MVG was anastomosed to the proximal end of the femoral artery, ensuring precise vascular graft alignment, secure suturing, and thorough flushing and inspection. Scale bar, 4 mm. (f) Similarly, the MVG was anastomosed to the distal end of the femoral artery, completing vascular graft integration and ensuring seamless arterial flow to maintain physiological circulatory function. Scale bar, 4 mm. g, Clear surgical view of the successfully anastomosed MVG, which resulted in the immediate and seamless resumption of blood circulation through the graft. Scale bar, 2 mm.
Extended Data Fig. 2 Diagnostic angiogram and postoperative hemodynamic sensing.
a–d, Digital subtraction angiography before the arterial anastomoses. (a) Contrast dye was administered through the glide catheter to improve the visualization of arterial structures. (b) The blood flowed through the femoral artery, (c) revealing the target implantation site, (d) and reaching the distal vasculature. Scale bars, 8 mm. e–h, Digital subtraction angiography after the arterial anastomoses. (e) Contrast dye was administered through the glide catheter to improve the visualization of arterial structures. (f) These images outline the patency of the MVG, verifying the vessel’s openness, structural integrity, (g) and anterograde blood flow following the MVG anastomoses. (h) The final image represents the late arterial phase and demonstrates the contrast agent’s progression distally into the vasculature, indicative of good flow and proper functioning of the MVG. Furthermore, the lack of evidence suggesting unwanted platelet aggregation and thrombus formation indicates appropriate biocompatibility. Scale bars, 8 mm. i–l, The MVG-generated hemodynamic signals were recorded after arterial anastomosis under different barrier conditions, including (i) no barrier, (j) one layer, (k) two layers, and (l) three layers of barriers. Representative of three (a-h) experiments.
Extended Data Fig. 3 Long-term studies on rats.
a, b, The rats with MVGs anastomosed to the femoral artery behaved and moved normally with no appreciable skin necrosis or swelling for up to 120 days post-implantation. Scale bars, 2 cm. c, The ultrasound probe is positioned above the femoral incision area to evaluate the patency of the anastomosed MVG. Partially created with BioRender. d, e, (d) The original Doppler ultrasound image alongside (e) the vascular color flow image captured the state of the anastomosed MVG two weeks post-surgery. f, g, (f) The original Doppler ultrasound image alongside (g) the vascular color flow image captured the state of the anastomosed MVG four weeks post-surgery. The use of color gradients in these images illustrates the blood flow, demonstrating the MVG’s patency during the early stages of recovery. These images provide critical insights into the graft’s immediate function and its integration within the host’s vascular system, showcasing the preservation of blood flow and the absence of apparent major obstructions. h, The photograph depicts the femoral incision area post-implantation, showcasing wound healing with no obvious signs of wound infection or inflammation. Scale bar, 1 cm. i, An infrared thermal image of the femoral incision area reveals an even temperature distribution. This uniform thermal profile is indicative of a healthy healing process, with no obvious signs of excessive heat that could denote infection or inflammation. Scale bar, 2 cm.
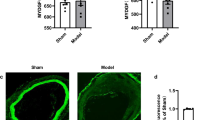
Extended Data Fig. 4 Immunofluorescence analysis of rat tissue samples after 120 days of implantation.
a, b, (a) Nuclei (DAPI) and (b) macrophage (CD68) markers in surrounding tissue samples from the MVG group. Scale bars, 25 µm. c, d, (c) Nuclei (DAPI) and (d) T cell (CD3) markers in surrounding tissue samples from the MVG group. Scale bars, 25 µm. e, f, (e) Nuclei (DAPI) and (f) macrophage (CD68) markers in surrounding tissue samples from the control group. Scale bars, 25 µm. g, h, (g) Nuclei (DAPI) and (h) T cell (CD3) markers in surrounding tissue samples from the control group. Scale bars, 25 µm. The dashed boxes (b, d, f, h) highlight the corresponding markers. Representative of three (a-h) experiments. i–l, Endothelial cell markers in the vessel sample near the MVG. Scale bars, 25 µm. These four immunofluorescence images represent different regions of the vascular endothelium within vessel samples. m–p, Endothelial cell markers in the natural vessel sample from the control group. Scale bars, 25 µm. These four immunofluorescence images represent different regions of the vascular endothelium within vessel samples.
Extended Data Fig. 5 Histology analysis of rat organ samples after 120 days of implantation.
a–e, (a) Histology analysis of rat heart samples after 120 days of implantation, including H&E-stained sections of the (b) MVG group and (c) the control group, and MT-stained sections of the (d) MVG group and (e) the control group. Scale bars, 40 µm. f–j, (f) Histology analysis of rat brain samples after 120 days of implantation, including H&E-stained sections of the (g) MVG group and (h) the control group, and MT-stained sections of the (i) MVG group and (j) the control group. Scale bars, 40 µm. k–o, (k) Histology analysis of rat kidney samples after 120 days of implantation, including H&E-stained sections of the (l) MVG group and (m) the control group, and MT-stained sections of the (n) MVG group and (o) the control group. Scale bars, 40 µm. p–t, (p) Histology analysis of rat liver samples after 120 days of implantation, including H&E-stained sections of the (q) MVG group and (r) the control group, and MT-stained sections of the (s) MVG group and (t) the control group. Scale bars, 40 µm. Representative of three (b-e, g-j, l-o, q-t) experiments. Partially created with BioRender.com.
Extended Data Fig. 6 Quantification of T cell phenotypes.
a–c, Immune cells from (a) blood, (b) spleen, and (c) surrounding tissue samples were analyzed for T cell phenotype quantification. Partially created with BioRender. d–f, Percentages of activated CD4+ T cell (left) and naive CD4+ T cell (right) relative to CD4+ T cells in (d) blood, (e) spleen, and (f) surrounding tissue samples. g–i, Percentages of helper T cell (left) and regulatory T cell (right) relative to the CD4+ T cells in (g) blood, (h) spleen, and (i) surrounding tissue samples. j–l, Percentages of activated CD8+ T cell (left) and naive CD8+ T cell (right) percentages relative to CD8+ T cells in (j) blood, (k) spleen, and (l) surrounding tissue samples. Data (d-l) are presented as mean ± s.d.; n = 3 from three rats. Significance was determined by a two-tailed t-test. N.S., not significant.
Extended Data Fig. 7 Quantification of macrophage polarization.
a, b, Immune cells from (a) spleen and (b) surrounding tissue samples were analyzed for macrophage polarization. Partially created with BioRender. c, d, Mean fluorescence intensity (MFI) of M1 macrophage marker CD80 in (c) spleen and (d) surrounding tissue samples. e, f, MFI of M2 macrophage marker CD163 in (e) spleen and (f) surrounding tissue samples. Data (c-f) are presented as mean ± s.d.; n = 3 from three rats. Significance was determined by a two-tailed t-test. N.S., not significant.
Extended Data Fig. 8 Hematology analysis of rat blood samples after 120 days of implantation.
a-b, Detailed experimental procedure for hematology analysis, (a) encompassing the collection of blood samples from the animals, (b) serum isolation, and comprehensive analytical assessments. Partially created with BioRender. c, Albumin concentration of the experiment animal and the control animal. d, Alanine transaminase concentration of the experiment animal and the control animal. e, Aspartate aminotransferase concentration of the experiment animal and the control animal. f, Total bilirubin concentration of the experiment animal and the control animal. g, Total protein concentration of the experiment animal and the control animal. h, Blood urea nitrogen (BUN) concentration of the experiment animal and the control animal. i, Globulin concentration of the experiment animal and the control animal. j, BUN/creatinine ratio of the experiment animal and the control animal. k, Mean corpuscular hemoglobin concentration of the experiment animal and the control animal. l, Mean corpuscular volume of the experiment animal and the control animal. m, Mean corpuscular hemoglobin of the experiment animal and the control animal. n, Red blood cells of the experiment animal and the control animal. Data (c-n) are presented as mean ± s.d.; n = 3 from three blood samples.
Supplementary information
Supplementary Information (download PDF )
Supplementary Figs. 1–84, Supplementary Tables 1–5, Supplementary Notes 1–31 and Supplementary Video Captions 1–4.
Supplementary Video 1 (download MP4 )
Doppler ultrasound demonstrating patency of the MVG anastomosed to the rat femoral artery.
Supplementary Video 2 (download MP4 )
A clear surgical view of the MVG anastomosed to the swine femoral artery.
Supplementary Video 3 (download MP4 )
Digital subtraction angiography demonstrating blood flow through the MVG anastomosed to the swine femoral artery.
Supplementary Video 4 (download MP4 )
Diagnostic angiogram demonstrating blood flow through the anastomosed MVG into the distal swine vasculature.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Chen, G., Chung, T., Liu, Z. et al. Hemodynamics-driven magnetoelastic vascular grafts for stenosis diagnosis. Nat Biotechnol 44, 406–417 (2026). https://doi.org/10.1038/s41587-025-02619-7
Received:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41587-025-02619-7
This article is cited by
-
AI-enabled vascular grafts monitor blood flow
Nature Biotechnology (2026)