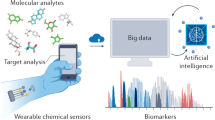
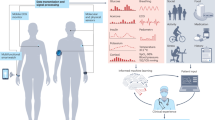
Abstract
Biomolecular profiling offers a powerful lens into human physiology, yet current diagnostics often rely on invasive sampling and delayed, centralized analysis. Advances in mass spectrometry (MS), particularly untargeted metabolomics and proteomics, have expanded molecular access to noninvasive biofluids such as sweat, saliva, tears and interstitial fluid, revealing dynamic biomarkers linked to both chronic and acute conditions. In parallel, wearable biosensors enable real-time, on-body chemical sensing, but remain limited to a narrow panel of predefined analytes. This Review highlights how MS-based molecular discovery and wearable sensing serve as complementary approaches—MS enabling high-dimensional untargeted profiling and wearables delivering longitudinal real-time data—and also discusses how their bidirectional integration and co-evolution open new possibilities for personalized noninvasive health monitoring. We discuss advances in sampling strategies, sensing modalities and system integration, and outline criteria for identifying biomarkers amenable to sensor translation. By uniting untargeted discovery with real-world deployment, this convergence shifts personalized noninvasive healthcare from episodic diagnostics to continuous, context-aware monitoring.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$32.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Frank, R. & Hargreaves, R. Clinical biomarkers in drug discovery and development. Nat. Rev. Drug Discov. 2, 566–580 (2003).
Guo, T., Steen, J. A. & Mann, M. Mass-spectrometry-based proteomics: from single cells to clinical applications. Nature 638, 901–911 (2025).
Danzi, F. et al. To metabolomics and beyond: a technological portfolio to investigate cancer metabolism. Signal Transduct. Target. Ther. 8, 137 (2023).
Gao, W. et al. Fully integrated wearable sensor arrays for multiplexed in situ perspiration analysis. Nature 529, 509–514 (2016). This article introduces a fully integrated wearable sensor array with on-board signal processing and wireless transmission that simultaneously measures multiple sweat analytes, enabling real-time, continuous physiological monitoring.
Brasier, N. et al. Applied body-fluid analysis by wearable devices. Nature 636, 57–68 (2024).
Heng, W. et al. A smart mask for exhaled breath condensate harvesting and analysis. Science 385, 954–961 (2024). This article introduces a wearable mask integrating tandem cooling, automated microfluidics and selective electrochemical sensors for continuous exhaled breath condensate harvesting, sampling and monitoring.
Chen, S. et al. Wearable flexible microfluidic sensing technologies. Nat. Rev. Bioeng. 2, 950–971 (2023).
Brunmair, J. et al. Finger sweat analysis enables short interval metabolic biomonitoring in humans. Nat. Commun. 12, 5993 (2021).
Delgado-Povedano, M. D. M., Calderón-Santiago, M., Priego-Capote, F., Jurado-Gámez, B. & Luque De Castro, M. D. Recent advances in human sweat metabolomics for lung cancer screening. Metabolomics 12, 166 (2016).
Kuwabara, H. et al. Salivary metabolomics with machine learning for colorectal cancer detection. Cancer Sci. 113, 3234–3243 (2022).
Ben, S. et al. Tear fluid-based metabolomics profiling in chronic dacryocystitis patients. J. Proteome Res. 24, 224–233 (2025).
Jiang, X., Wilkirson, E. C., Bailey, A. O., Russell, W. K. & Lillehoj, P. B. Microneedle-based sampling of dermal interstitial fluid using a vacuum-assisted skin patch. Cell Rep. Phys. Sci. 5, 101975 (2024).
Ibrahim, W. et al. Visualization of exhaled breath metabolites revreals distinct diagnostic signatures for acute cardiorespiratory breathlessness. Sci. Transl. Med. 14, eabl5849 (2022).
Mueller, D. C., Piller, M., Niessner, R., Scherer, M. & Scherer, G. Untargeted metabolomic profiling in saliva of smokers and nonsmokers by a validated GC–TOF–MS method. J. Proteome Res. 13, 1602–1613 (2014).
Ossoliński, K. et al. Targeted and untargeted urinary metabolic profiling of bladder cancer. J. Pharm. Biomed. Anal. 233, 115473 (2023).
Wu, J. et al. Glaucoma characterization by machine learning of tear metabolic fingerprinting. Small Methods 6, e2200264 (2022).
Xu, W. et al. Diagnosis of Parkinson’s disease via the metabolic fingerprint in saliva by deep learning. Small Methods 7, e2300285 (2023). This article presents a nanoparticle-enhanced MS platform capable of acquiring saliva metabolic fingerprints from nanoliter samples, enabling Parkinson’s disease diagnosis through deep learning analysis of data collected from 312 participants.
Tu, J. et al. A wireless patch for the monitoring of C-reactive protein in sweat. Nat. Biomed. Eng. 7, 1293–1306 (2023).
Xu, C. et al. A physicochemical-sensing electronic skin for stress response monitoring. Nat. Electron. 7, 168–179 (2024). This article introduces a machine-learning reinforced multimodal wearable sensor capable of continuous monitoring of vital signs alongside sweat biomarkers, enabling both stressor classification and quantitative state anxiety assessment.
Naik, A. R. et al. Printed microfluidic sweat sensing platform for cortisol and glucose detection. Lab Chip 22, 156–169 (2022).
Kunnel, B. P. & Demuru, S. An epidermal wearable microfluidic patch for simultaneous sampling, storage, and analysis of biofluids with counterion monitoring. Lab Chip 22, 1793–1804 (2022).
Lim, H.-R. et al. Smart bioelectronic pacifier for real-time continuous monitoring of salivary electrolytes. Biosens. Bioelectron. 210, 114329 (2022).
Gao, Y. et al. A flexible multiplexed immunosensor for point-of-care in situ wound monitoring. Sci. Adv. 7, eabg9614 (2021).
Koh, A. et al. A soft, wearable microfluidic device for the capture, storage, and colorimetric sensing of sweat. Sci. Transl. Med. 8, 366ra165 (2016).
Kim, J. et al. A skin-interfaced, miniaturized microfluidic analysis and delivery system for colorimetric measurements of nutrients in sweat and supply of vitamins through the skin. Adv. Sci. (Weinh.) 9, e2103331 (2022).
Jiang, X. & Lillehoj, P. B. Microneedle-based skin patch for blood-free rapid diagnostic testing. Microsyst. Nanoeng. 6, 96 (2020).
Yetisen, A. K. et al. Scleral lens sensor for ocular electrolyte analysis. Adv. Mater. 32, e1906762 (2020).
Li, X. et al. Lactate metabolism in human health and disease. Signal Transduct. Target. Ther. 7, 305 (2022).
Heikenfeld, J. et al. Accessing analytes in biofluids for peripheral biochemical monitoring. Nat. Biotechnol. 37, 407–419 (2019).
McElvaney, O. J., Curley, G. F., Rose-John, S. & McElvaney, N. G. Interleukin-6: obstacles to targeting a complex cytokine in critical illness. Lancet Respir. Med. 9, 643–654 (2021).
Martínez-Ruiz, C. et al. Genomic–transcriptomic evolution in lung cancer and metastasis. Nature 616, 543–552 (2023).
Alseekh, S. et al. Mass spectrometry-based metabolomics: a guide for annotation, quantification and best reporting practices. Nat. Methods 18, 747–756 (2021).
Mintun, M. A., Vlassenko, A. G., Rundle, M. M. & Raichle, M. E. Increased lactate/pyruvate ratio augments blood flow in physiologically activated human brain. Proc. Natl Acad. Sci. USA 101, 659–664 (2004).
Abujrais, S., Vallianatou, T. & Bergquist, J. Untargeted metabolomics and quantitative analysis of tryptophan metabolites in myalgic encephalomyelitis patients and healthy volunteers: a comparative study using high-resolution mass spectrometry. ACS Chem. Neurosci. 15, 3525–3534 (2024).
Wang, F. et al. Plasma metabolomic profiles associated with mortality and longevity in a prospective analysis of 13,512 individuals. Nat. Commun. 14, 5744 (2023).
Calderón-Santiago, M. et al. Human sweat metabolomics for lung cancer screening. Anal. Bioanal. Chem. 407, 5381–5392 (2015).
Nowak, C. et al. Glucose challenge metabolomics implicates medium-chain acylcarnitines in insulin resistance. Sci. Rep. 8, 8691 (2018).
Sonner, Z. et al. The microfluidics of the eccrine sweat gland, including biomarker partitioning, transport, and biosensing implications. Biomicrofluidics 9, 031301 (2015).
Turner, Z. & Drabovich, A. P. Opportunities and challenges of multiomics for discovery and monitoring of human pathogens. Environ. Health (Wash.) 3, 1139–1153 (2025).
Schiebenhoefer, H. et al. A complete and flexible workflow for metaproteomics data analysis based on MetaProteomeAnalyzer and Prophane. Nat. Protoc. 15, 3212–3239 (2020).
Van Den Bossche, T. et al. The microbiologist’s guide to metaproteomics. iMeta 4, e70031 (2025).
Chakraborty, N. Metabolites: a converging node of host and microbe to explain meta-organism. Front. Microbiol. 15, 1337368 (2024).
Kim, M., Yun, T. G., Noh, J., Kang, M. & Pyun, J. Photothermal structural dynamics of Au Nanofurnace for in situ enhancement in desorption and ionization. Small 17, e2103745 (2021).
Bandodkar, A. J. et al. Battery-free, skin-interfaced microfluidic/electronic systems for simultaneous electrochemical, colorimetric, and volumetric analysis of sweat. Sci. Adv. 5, eaav3294 (2019).
Harshman, S. W. et al. Rate normalization for sweat metabolomics biomarker discovery. Talanta 223, 121797 (2021).
Nyein, H. Y. Y. et al. A wearable patch for continuous analysis of thermoregulatory sweat at rest. Nat. Commun. 12, 1823 (2021).
Shin, S. et al. A bioinspired microfluidic wearable sensor for multiday sweat sampling, transport, and metabolic analysis. Sci. Adv. 11, eadw9024 (2025).
Rakusanova, S., Fiehn, O. & Cajka, T. Toward building mass spectrometry-based metabolomics and lipidomics atlases for biological and clinical research. Trends Analyt. Chem. 158, 116825 (2023).
Kim, T. et al. A hierarchical approach to removal of unwanted variation for large-scale metabolomics data. Nat. Commun. 12, 4992 (2021).
Reeder, J. T. et al. Waterproof, electronics-enabled, epidermal microfluidic devices for sweat collection, biomarker analysis, and thermography in aquatic settings. Sci. Adv. 5, eaau6356 (2019).
Haslam, D. E. et al. Stability and reproducibility of proteomic profiles in epidemiological studies: comparing the Olink and SOMAscan platforms. Proteomics 22, e2100170 (2022).
Xu, Y. et al. Comparison between dietary assessment methods and biomarkers in estimating dietary (poly)phenol intake. Food Funct. 14, 1369–1386 (2023).
Bi, H. et al. Electrostatic spray ionization-mass spectrometry for direct and fast wine characterization. ACS Omega 3, 17881–17887 (2018).
Udeshi, N. D. et al. Rapid and deep-scale ubiquitylation profiling for biology and translational research. Nat. Commun. 11, 359 (2020).
Roberg-Larsen, H., Wilson, S. R. & Lundanes, E. Recent advances in on-line upfront devices for sensitive bioanalytical nano LC methods. Trends Analyt. Chem. 136, 116190 (2021).
Bravo-Veyrat, S. & Hopfgartner, G. Mass spectrometry based high-throughput bioanalysis of low molecular weight compounds: are we ready to support personalized medicine?. Anal. Bioanal. Chem. 414, 181–192 (2022).
Kim, M. et al. Laser-induced surface reconstruction of nanoporous Au-modified TiO2 nanowires for in situ performance enhancement in desorption and ionization mass spectrometry. Adv. Funct. Mater. 31, 2102475 (2021). This article demonstrates that nanoporous Au‐modified TiO2 nanowires substantially enhance MS performance by coupling photothermal-driven analyte desorption with strain-induced suppression of charge-carrier recombination.
Meister, I. et al. High-precision automated workflow for urinary untargeted metabolomic epidemiology. Anal. Chem. 93, 5248–5258 (2021).
Bian, Y. et al. Robust microflow LC–MS/MS for proteome analysis: 38 000 runs and counting. Anal. Chem. 93, 3686–3690 (2021).
Messner, C. B. et al. Mass spectrometry-based high-throughput proteomics and its role in biomedical studies and systems biology. Proteomics 23, e2200013 (2023).
Guzman, U. H. et al. Ultra-fast label-free quantification and comprehensive proteome coverage with narrow-window data-independent acquisition. Nat. Biotechnol. 42, 1855–1866 (2024).
Dayon, L. et al. Relative quantification of proteins in human cerebrospinal fluids by MS/MS using 6-plex isobaric tags. Anal. Chem. 80, 2921–2931 (2008).
Ravindra, K. C. et al. Tandem mass tag-based quantitative proteomic profiling identifies candidate serum biomarkers of drug-induced liver injury in humans. Nat. Commun. 14, 1215 (2023).
Park, J.-M. et al. MALDI-TOF mass spectrometry based on parylene-matrix chip for the analysis of lysophosphatidylcholine in sepsis patient sera. Anal. Chem. 91, 14719–14727 (2019).
Lancaster, N. M. et al. Fast and deep phosphoproteome analysis with the Orbitrap Astral mass spectrometer. Nat. Commun. 15, 7016 (2024).
Demichev, V. et al. dia-PASEF data analysis using FragPipe and DIA-NN for deep proteomics of low sample amounts. Nat. Commun. 13, 3944 (2022).
Meier, F. et al. Online parallel accumulation–serial fragmentation (PASEF) with a novel trapped ion mobility mass spectrometer. Mol. Cell. Proteomics 17, 2534–2545 (2018).
Ding, S. et al. A fingertip-wearable microgrid system for autonomous energy management and metabolic monitoring. Nat. Electron. 7, 788–799 (2024).
Friedel, M. et al. Opportunities and challenges in the diagnostic utility of dermal interstitial fluid. Nat. Biomed. Eng. 7, 1541–1555 (2023).
Arakawa, T. et al. A wearable cellulose acetate-coated mouthguard biosensor for in vivo salivary glucose measurement. Anal. Chem. 92, 12201–12207 (2020).
Xu, J., Tao, X., Liu, X. & Yang, L. Wearable eye patch biosensor for noninvasive and simultaneous detection of multiple biomarkers in human tears. Anal. Chem. 94, 8659–8667 (2022).
Shirzaei Sani, E. et al. A stretchable wireless wearable bioelectronic system for multiplexed monitoring and combination treatment of infected chronic wounds. Sci. Adv. 9, eadf7388 (2023).
Wang, C. et al. Wound management materials and technologies from bench to bedside and beyond. Nat. Rev. Mater. 9, 550–566 (2024).
Wang, C. et al. A microfluidic wearable device for wound exudate management and analysis in human chronic wounds. Sci. Transl. Med. 17, eadt0882 (2025). This article introduces a smart bandage that integrates nanoengineered sensors and microfluidic modules for continuous sampling and analysis of wound exudate, enabling machine-learning–assisted assessment of wound status and healing in patients.
Mimee, M. et al. An ingestible bacterial-electronic system to monitor gastrointestinal health. Science 360, 915–918 (2018).
Min, J. et al. Continuous biochemical profiling of the gastrointestinal tract using an integrated smart capsule. Nat. Electron. 8, 844–855 (2025).
Ershad, F. et al. Ultra-conformal drawn-on-skin electronics for multifunctional motion artifact-free sensing and point-of-care treatment. Nat. Commun. 11, 3823 (2020).
Kim, S. et al. Soft, skin-interfaced microfluidic systems with integrated immunoassays, fluorometric sensors, and impedance measurement capabilities. Proc. Natl Acad. Sci. USA 117, 27906–27915 (2020).
Xu, K., Lu, Y. & Takei, K. Multifunctional skin-inspired flexible sensor systems for wearable electronics. Adv. Mater. Technol. 4, 1800628 (2019).
Kim, S. J. et al. Wearable UV sensor based on carbon nanotube-coated cotton thread. ACS Appl. Mater. Interfaces 10, 40198–40202 (2018).
Zhang, Y. et al. Cotton fabrics decorated with conductive graphene nanosheet inks for flexible wearable heaters and strain sensors. ACS Appl. Nano Mater. 4, 9709–9720 (2021).
Song, Y. et al. 3D-printed epifluidic electronic skin for machine learning–powered multimodal health surveillance. Sci. Adv. 9, eadi6492 (2023).
Lee, H. et al. Facile fabrication of a fully biodegradable and stretchable serpentine-shaped wire supercapacitor. Chem. Eng. J. 366, 62–71 (2019).
Liu, Y., Huang, J., Ding, G. & Yang, Z. High-performance and wearable strain sensors based on graphene microfluidics and serpentine microchannels for human motion detection. Microelectron. Eng. 231, 111402 (2020).
Brooks, A. K., Chakravarty, S., Ali, M. & Yadavalli, V. K. Kirigami-inspired biodesign for applications in healthcare. Adv. Mater. 34, e2109550 (2022).
Xu, R. et al. Kirigami-inspired, highly stretchable micro-supercapacitor patches fabricated by laser conversion and cutting. Microsyst. Nanoeng. 4, 36 (2018).
Tu, J. et al. Stressomic: a wearable microfluidic biosensor for dynamic profiling of multiple stress hormones in sweat. Sci. Adv. 11, eadx6491 (2025). This article introduces a multiplexed microfluidic biosensor integrating iontophoresis-driven sweat extraction and picomolar-sensitivity electrochemical detection to continuously monitor cortisol, epinephrine and norepinephrine, revealing distinct temporal patterns of stress hormone dynamics across multiple stressors.
Cui, F., Yue, Y., Zhang, Y., Zhang, Z. & Zhou, H. S. Advancing biosensors with machine learning. ACS Sens. 5, 3346–3364 (2020).
Abdigazy, A. et al. End-to-end design of ingestible electronics. Nat. Electron. 7, 102–118 (2024).
Sempionatto, J. R., Lasalde-Ramírez, J. A., Mahato, K., Wang, J. & Gao, W. Wearable chemical sensors for biomarker discovery in the omics era. Nat. Rev. Chem. 6, 899–915 (2022). This review outlines criteria, strategies and technologies for biomarker discovery using wearable chemical sensors, highlighting how continuous temporal data can reveal new biomarkers for precision medicine.
Cho, S. et al. A skin-interfaced microfluidic platform supports dynamic sweat biochemical analysis during human exercise. Sci. Transl. Med. 16, eado5366 (2024).
Chen, Y. et al. A biochemical sensor with continuous extended stability in vivo. Nat. Biomed. Eng. 9, 1517–1530 (2025). This article introduces a mucosa-inspired biosensor with a nanoporous, polymer-coated aptamer interface that resists fouling and degradation, enabling stable, continuous in vivo monitoring and real-time pharmacokinetic tracking in freely moving rats.
Inda-Webb, M. E. et al. Sub-1.4 cm3 capsule for detecting labile inflammatory biomarkers in situ. Nature 620, 386–392 (2023).
Sabaté del Río, J., Henry, O. Y., Jolly, P. & Ingber, D. E. An antifouling coating that enables affinity-based electrochemical biosensing in complex biological fluids. Nat. Nanotechnol. 14, 1143–1149 (2019).
Zargartalebi, H. et al. Active-reset protein sensors enable continuous in vivo monitoring of inflammation. Science 386, 1146–1153 (2024). This article introduces a high-frequency potential oscillation strategy to rapidly dissociate protein analytes from aptamer-based biosensors, enabling continuous in vivo monitoring of inflammatory protein biomarkers.
Nguyen, P. Q. et al. Wearable materials with embedded synthetic biology sensors for biomolecule detection. Nat. Biotechnol. 39, 1366–1374 (2021).
Nakata, S. et al. A wearable pH sensor with high sensitivity based on a flexible charge-coupled device. Nat. Electron. 1, 596–603 (2018).
Arwani, R. T. et al. Stretchable ionic–electronic bilayer hydrogel electronics enable in situ detection of solid-state epidermal biomarkers. Nat. Mater. 23, 1115–1122 (2024).
Zhang, B. et al. A three-dimensional liquid diode for soft, integrated permeable electronics. Nature 628, 84–92 (2024).
Tehrani, F. et al. An integrated wearable microneedle array for the continuous monitoring of multiple biomarkers in interstitial fluid. Nat. Biomed. Eng. 6, 1214–1224 (2022).
Kim, J., Ghaffari, R. & Kim, D.-H. The quest for miniaturized soft bioelectronic devices. Nat. Biomed. Eng. 1, 0049 (2017).
Ye, C. et al. A wearable aptamer nanobiosensor for non-invasive female hormone monitoring. Nat. Nanotechnol. 19, 330–337 (2024).
Li, X. et al. Rapid and on-site wireless immunoassay of respiratory virus aerosols via hydrogel-modulated resonators. Nat. Commun. 15, 4035 (2024).
Min, J. et al. An autonomous wearable biosensor powered by a perovskite solar cell. Nat. Electron. 6, 630–641 (2023).
De la Paz, E. et al. A self-powered ingestible wireless biosensing system for real-time in situ monitoring of gastrointestinal tract metabolites. Nat. Commun. 13, 7405 (2022).
ICH. Validation of analytical procedures Q2(R2). https://database.ich.org/sites/default/files/ICH_Q2-R2_Document_Step2_Guideline_2022_0324.pdf (2022).
U.S. Department of Health and Human Services, FDA, CDER & CVM. Bioanalytical method validation guidance for industry. https://www.fda.gov/files/drugs/published/Bioanalytical-Method-Validation-Guidance-for-Industry.pdf (2018).
Xuan, X. et al. Fully integrated wearable device for continuous sweat lactate monitoring in sports. ACS Sens. 8, 2401–2409 (2023).
Brasier, N. et al. The molecular signature of heat stress in sweat reveals non-invasive biomarker candidates for health monitoring. Commun. Biol. 8, 650 (2025).
Ursem, R. F. R. et al. Worth your sweat: wearable microfluidic flow rate sensors for meaningful sweat analytics. Lab Chip 25, 1296–1315 (2025).
Xiao, H. & Wong, D. T. Method development for proteome stabilization in human saliva. Anal. Chim. Acta 722, 63–69 (2012).
Ding, S., Tang, S. & Wang, J. Interference-resistant wearables for long-term hyperuricemia monitoring. Device 3, 100782 (2025).
Wang, M. et al. A wearable electrochemical biosensor for the monitoring of metabolites and nutrients. Nat. Biomed. Eng. 6, 1225–1235 (2022).
Yang, P.-J., Hsu, Y.-C., Li, J.-R. & Luo, S.-C. Quantitatively elucidating the trade-off between zwitterionic antifouling surfaces and bioconjugation performance. Langmuir 40, 26259–26266 (2024).
Bariya, M., Nyein, H. Y. Y. & Javey, A. Wearable sweat sensors. Nat. Electron. 1, 160–171 (2018).
Roehrich, B. et al. Calibration-free, seconds-resolved in vivo molecular measurements using fourier-transform impedance spectroscopy interrogation of electrochemical aptamer sensors. ACS Sens. 8, 3051–3059 (2023).
Mahato, K. et al. Hybrid multimodal wearable sensors for comprehensive health monitoring. Nat. Electron. 7, 735–750 (2024).
Camera, E. et al. Use of lipidomics to investigate sebum dysfunction in juvenile acne. J. Lipid Res. 57, 1051–1058 (2016).
Lu, Y. et al. Volatile organic compounds profile of sebum from patients with Parkinson’s disease by gas chromatography-ion mobility spectrometry. Microchem. J. 208, 112381 (2025).
Coon, A. M., Setzen, G. & Musah, R. A. Mass spectrometric interrogation of earwax: toward the detection of Meniere’s disease. ACS Omega 8, 27010–27023 (2023).
Isom, M. & Desaire, H. Skin surface sebum analysis by ESI-MS. Biomolecules 14, 790 (2024).
Feig, M. A., Hammer, E., Völker, U. & Jehmlich, N. In-depth proteomic analysis of the human cerumen—a potential novel diagnostically relevant biofluid. J. Proteomics 83, 119–129 (2013).
Toma, K., Suzuki, S., Arakawa, T., Iwasaki, Y. & Mitsubayashi, K. External ears for non-invasive and stable monitoring of volatile organic compounds in human blood. Sci. Rep. 11, 10415 (2021).
Acknowledgements
This work was supported by the National Institutes of Health (grants R01HL155815 and R33DK132666), National Science Foundation (grants 2145802 and 2444815), Advanced Research Projects Agency for Health (grant ARPA-H-ICHUB-24-101-504), US Army Medical Research Acquisition Activity (grant HT9425-24-1-0249), Army Research Office (grant W911NF-23-1-0041), Office of Naval Research (grant N00014-25-1-2258) and Heritage Medical Research Institute. M.-J.K. was partially supported by the Basic Science Research Program through the National Research Foundation of Korea, supported by the Ministry of Education (RS-2023-00241415).
Author information
Authors and Affiliations
Contributions
M.-J.K. and W.G. conceived the study and designed the outline and scope. M.-J.K., J.A.L.R., W.H. and W.G. wrote the manuscript.
Corresponding author
Ethics declarations
Competing interests
W.G. is a cofounder and advisor at Persperity Health. The other authors declare no competing interests.
Peer review
Peer review information
Nature Biotechnology thanks Eden Morales-Narvaez, who co-reviewed with Ivan A Lujan-Cabrera; Sihong Wang; and Tiannan Guo for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information (download PDF )
Supplementary Notes 1–3, Supplementary Figs. 1–3 and Supplementary Table 1.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Kim, MJ., Lasalde-Ramírez, J.A., Heng, W. et al. Biomolecular profiling for noninvasive health monitoring. Nat Biotechnol (2026). https://doi.org/10.1038/s41587-026-03050-2
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41587-026-03050-2