Abstract
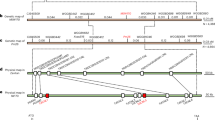
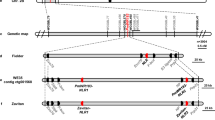
Wheat stripe rust poses a major threat to global food security. Discovery of disease resistance genes from wild relatives enables multigene stacking that could enhance durability. Here we use map-based cloning and long-read sequencing to isolate two adjacent nucleotide-binding and leucine-rich repeat (NLR) receptors from wild emmer wheat. Using mutagenesis, gene silencing and genetic transformation, we show that the genes TdNLR1 and TdNLR2 oriented head-to-head are both required for YrTD121-mediated stripe rust resistance. TdNLR1 encodes a canonical NLR (CC-NB-ARC-LRR) protein, whereas TdNLR2 encodes an atypical one (NB-ARC-LRR). Both genes lack an integrated domain previously associated with effector perception, representing an uncommon architecture for paired NLRs in plants. The coiled coil domain of TdNLR1 triggers cell death and self-associates in planta. YrTD121 was present in wild emmer but absent in all other Triticum species examined. Our work sheds light on the function of paired NLRs in conferring disease resistance and facilitates breeding for resistance.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$32.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
Data availability
Data supporting the findings of this work are available in the article and its supplementary information. The wild emmer reference genome assembly WEW_v2.1 was collected from the NCBI database under BioProject no. PRJNA310175 (ref. 52). The transcriptome raw data are available under BioProject no. PRJNA1221773. The genomic sequence of the ptg005881l contig for the YrTD121 region is available under accession no. PV110151.1. The genomic DNA sequences of TdNLR1 and TdNLR2 from TD121 are accessible under GenBank accession nos. OP948046.1 and PP874936.1. The genomic DNA sequences of other TdNLR haplotypes in this study are available under accession nos. OP948047.1–OP948060.1, accession no. OR472555.1 for TdNLR1, and accession nos. PP874937.1–PP874945.1 and PQ412542.1 for TdNLR2. Source data are provided with this paper.
Code availability
All software packages used in the study are publicly available and as described in Methods and Reporting Summary.
References
Kourelis, J. & van der Hoorn, R. A. L. Defended to the nines: 25 years of resistance gene cloning identifies nine mechanisms for R protein function. Plant Cell 30, 285–299 (2018).
Shao, Z. Q. et al. Large-scale analyses of angiosperm nucleotide-binding site-leucine-rich repeat genes reveal three anciently diverged classes with distinct evolutionary patterns. Plant Physiol. 170, 2095–2109 (2016).
Tamborski, J. & Krasileva, K. V. Evolution of plant NLRs: from natural history to precise modifications. Annu. Rev. Plant Biol. 71, 355–378 (2020).
Takken, F. L., Albrecht, M. & Tameling, W. I. Resistance proteins: molecular switches of plant defence. Curr. Opin. Plant Biol. 9, 383–390 (2006).
Chia, K. S. et al. The N-terminal domains of NLR immune receptors exhibit structural and functional similarities across divergent plant lineages. Plant Cell 36, 2491–2511 (2024).
Marchal, C., Michalopoulou, V. A., Zou, Z., Cevik, V. & Sarris, P. F. Show me your ID: NLR immune receptors with integrated domains in plants. Essays Biochem. 66, 527–539 (2022).
Contreras, M. P. et al. Sensor NLR immune proteins activate oligomerization of their NRC helpers in response to plant pathogens. EMBO J. 42, e111519 (2023).
Adachi, H., Derevnina, L. & Kamoun, S. NLR singletons, pairs, and networks: evolution, assembly, and regulation of the intracellular immunoreceptor circuitry of plants. Curr. Opin. Plant Biol. 50, 121–131 (2019).
Césari, S. et al. The NB-LRR proteins RGA4 and RGA5 interact functionally and physically to confer disease resistance. EMBO J. 33, 1941–1959 (2014).
Loutre, C. et al. Two different CC-NBS-LRR genes are required for Lr10-mediated leaf rust resistance in tetraploid and hexaploid wheat. Plant J. 60, 1043–1054 (2009).
Zdrzałek, R., Kamoun, S., Terauchi, R., Saitoh, H. & Banfield, M. J. The rice NLR pair Pikp-1/Pikp-2 initiates cell death through receptor cooperation rather than negative regulation. PLoS ONE 15, e0238616 (2020).
Huh, S. U. et al. Protein–protein interactions in the RPS4/RRS1 immune receptor complex. PLoS Pathog. 13, e1006376 (2017).
Wu, C. H. et al. NLR network mediates immunity to diverse plant pathogens. Proc. Natl Acad. Sci. USA 114, 8113–8118 (2017).
Wu, C. H., Derevnina, L. & Kamoun, S. Receptor networks underpin plant immunity. Science 360, 1300–1301 (2018).
Cesari, S., Bernoux, M., Moncuquet, P., Kroj, T. & Dodds, P. N. A novel conserved mechanism for plant NLR protein pairs: the ‘integrated decoy’ hypothesis. Front. Plant Sci. 5, 606 (2014).
Shewry, P. R. & Hey, S. J. The contribution of wheat to human diet and health. Food Energy Secur. 4, 178–202 (2015).
McIntosh, R. A. et al. Catalogue of gene symbols for wheat: 2021 supplement. Annu. Wheat Newsl. 67, 104–113 (2021).
Klymiuk, V. et al. Discovery of stripe rust resistance with incomplete dominance in wild emmer wheat using bulked segregant analysis sequencing. Commun. Biol. 5, 826 (2022).
Sharma, D. et al. A single NLR gene confers resistance to leaf and stripe rust in wheat. Nat. Commun. 15, 9925 (2024).
Hafeez, A. N. et al. Creation and judicious application of a wheat resistance gene atlas. Mol. Plant 14, 1053–1070 (2021).
Dibley, K., Jost, M., McIntosh, R., Lagudah, E. & Zhang, P. The wheat stripe rust resistance gene YrNAM is Yr10.Nat. Commun. 15, 3291 (2024).
Athiyannan, N. et al. Long-read genome sequencing of bread wheat facilitates disease resistance gene cloning. Nat. Genet. 54, 227–231 (2022).
Huang, L. et al. Evolution and adaptation of wild emmer wheat populations to biotic and abiotic stresses. Annu. Rev. Phytopathol. 54, 279–301 (2016).
Li, Y. et al. Dissection of a rapidly evolving wheat resistance gene cluster by long-read genome sequencing accelerated the cloning of Pm69. Plant Commun. 5, 100646 (2024).
Li, M. et al. A membrane associated tandem kinase from wild emmer wheat confers broad-spectrum resistance to powdery mildew. Nat. Commun. 15, 3124 (2024).
Li, M. et al. A CNL protein in wild emmer wheat confers powdery mildew resistance. New Phytol. 228, 1027–1037 (2020).
Walkowiak, S. et al. Multiple wheat genomes reveal global variation in modern breeding. Nature 588, 277–283 (2020).
Avni, R. et al. Wild emmer genome architecture and diversity elucidate wheat evolution and domestication. Science 357, 93–96 (2017).
Wenger, A. M. et al. Accurate circular consensus long-read sequencing improves variant detection and assembly of a human genome. Nat. Biotechnol. 37, 1155–1162 (2019).
Huang, L. et al. Distribution and haplotype diversity of WKS resistance genes in wild emmer wheat natural populations. Theor. Appl. Genet. 129, 921–934 (2016).
Trick, M. et al. Combining SNP discovery from next-generation sequencing data with bulked segregant analysis (BSA) to fine-map genes in polyploid wheat. BMC Plant Biol. 12, 14 (2012).
Zhai, C. et al. The isolation and characterization of Pik, a rice blast resistance gene which emerged after rice domestication. New Phytol. 189, 321–334 (2011).
Cesari, S. et al. Cytosolic activation of cell death and stem rust resistance by cereal MLA-family CC-NLR proteins. Proc. Natl Acad. Sci. USA 113, 10204–10209 (2016).
Klymiuk, V. et al. Cloning of the wheat Yr15 resistance gene sheds light on the plant tandem kinase-pseudokinase family. Nat. Commun. 9, 3735 (2018).
Fu, D. et al. A kinase-START gene confers temperature-dependent resistance to wheat stripe rust. Science 323, 1357–1360 (2009).
Zhu, K. Y. et al. An atypical NLR pair TdCNL1/TdCNL5 from wild emmer confers powdery mildew resistance in wheat. Nat. Genet. (in the press).
Li, Y. et al. TdPm60 identified in wild emmer wheat is an ortholog of Pm60 and constitutes a strong candidate for PmG16 powdery mildew resistance. Theor. Appl. Genet. 134, 2777–2793 (2021).
Klymiuk, V. et al. Coordinated function of paired NLRs confers Yr84-mediated stripe rust resistance in wheat. Nat. Genet. (in the press).
Li, L. F. et al. Genome sequences of five Sitopsis species of Aegilops and the origin of polyploid wheat B subgenome.Mol. Plant 15, 488–503 (2022).
van Wersch, S. & Li, X. Stronger when together: clustering of plant NLR disease resistance genes. Trends Plant Sci. 24, 688–699 (2019).
Chen, J. et al. Loss of AvrSr50 by somatic exchange in stem rust leads to virulence for Sr50 resistance in wheat. Science 358, 1607–1610 (2017).
Wang, J. et al. Reconstitution and structure of a plant NLR resistosome conferring immunity. Science 364, eaav5870 (2019).
Yang, Y. et al. Allelic variation in the Arabidopsis TNL CHS3/CSA1 immune receptor pair reveals two functional cell-death regulatory modes. Cell Host Microbe 30, 1701–1706 (2022).
Contreras, M. P., Lüdke, D., Pai, H., Toghani, A. & Kamoun, S. NLR receptors in plant immunity: making sense of the alphabet soup. EMBO Rep. 24, e57495 (2023).
Liang, W., van Wersch, S., Tong, M. & Li, X. TIR-NB-LRR immune receptor SOC3 pairs with truncated TIR-NB protein CHS1 or TN2 to monitor the homeostasis of E3 ligase SAUL1. New Phytol. 221, 2054–2066 (2019).
Wang, F. et al. Pyramiding of adult-plant resistance genes enhances all-stage resistance to wheat stripe rust. Plant Dis. 107, 879–885 (2023).
Huerta-Espino, J. et al. Adult plant slow rusting genes confer high levels of resistance to rusts in bread wheat cultivars from Mexico. Front. Plant Sci. 11, 824 (2020).
Krattinger, S. G. et al. A putative ABC transporter confers durable resistance to multiple fungal pathogens in wheat. Science 323, 1360–1363 (2009).
Moore, J. W. et al. A recently evolved hexose transporter variant confers resistance to multiple pathogens in wheat. Nat. Genet. 47, 1494–1498 (2015).
Line, R. F. & Qayoum, A. Virulence, aggressiveness, evolution, and distribution of races of Puccinia striiformis (the cause of stripe rust of wheat) in North America. U.S. Dep. Agric. Tech. Bull. 1788, 1968–1987 (1992).
Bolger, A. M., Lohse, M. & Usadel, B. Trimmomatic: a flexible trimmer for Illumina sequence data. Bioinformatics 30, 2114–2120 (2014).
Zhu, T. et al. Improved genome sequence of wild emmer wheat Zavitan with the aid of optical maps. G3 9, 619–624 (2019).
Dobin, A. et al. STAR: ultrafast universal RNA-seq aligner. Bioinformatics 29, 15–21 (2013).
McKenna, A. et al. The Genome Analysis Toolkit: a MapReduce framework for analyzing next-generation DNA sequencing data. Genome Res. 20, 1297–1303 (2010).
Wang, Y. et al. Mapping stripe rust resistance gene YrZH22 in Chinese wheat cultivar Zhoumai 22 by bulked segregant RNA-Seq (BSR-Seq) and comparative genomics analyses. Theor. Appl. Genet. 130, 2191–2201 (2017).
Rogers, S. O. & Bendich, A. J. Extraction of DNA from milligram amounts of fresh, herbarium and mummified plant tissues. Plant Mol. Biol. 5, 69–76 (1985).
Langmead, B. & Salzberg, S. L. Fast gapped-read alignment with Bowtie 2. Nat. Methods 9, 357–359 (2012).
Robinson, J. T. et al. Integrative genomics viewer. Nat. Biotechnol. 29, 24–26 (2011).
Tsushima, A., Gan, P. & Shirasu, K. Method for assessing virulence of Colletotrichum higginsianum on Arabidopsis thaliana leaves using automated lesion area detection and measurement. Bio. Protoc. 9, e3434 (2019).
Liu, H. et al. Efficient induction of haploid plants in wheat by editing of TaMTL using an optimized Agrobacterium-mediated CRISPR system. J. Exp. Bot. 71, 1337–1349 (2020).
Wang, K., Liu, H., Du, L. & Ye, X. Generation of marker-free transgenic hexaploid wheat via an Agrobacterium-mediated co-transformation strategy in commercial Chinese wheat varieties. Plant Biotechnol. J. 15, 614–623 (2017).
Pearce, S., Vanzetti, L. S. & Dubcovsky, J. Exogenous gibberellins induce wheat spike development under short days only in the presence of VERNALIZATION1. Plant Physiol. 163, 1433–1445 (2013).
Hall, T. BioEdit, Version 7.0.9. Computer Program and Documentation (lbis Biosciences, 2007).
Ma, S. et al. WheatOmics: a platform combining multiple omics data to accelerate functional genomics studies in wheat. Mol. Plant 14, 1965–1968 (2021).
Lin, J. S. & Lai, E. M. Protein–protein interactions: yeast two-hybrid system. Methods Mol. Biol. 1615, 177–187 (2017).
Chen, H. et al. Firefly luciferase complementation imaging assay for protein–protein interactions in plants. Plant Physiol. 146, 368–376 (2008).
Acknowledgements
We thank R. A. McIntosh (Plant Breeding Institute, University of Sydney) for the critical review of this paper; The Lady Barbara Davis Wild Cereal Gene bank, the Institute of Evolution, at the University of Haifa and Y. X. Liu for providing the seeds of wild emmer accessions TD121 (TD104059) and PI 487260, respectively; and L. L. Dong and D. D. Wu for assistance with the bioinformatics analysis. This research was supported by the National Natural Science Foundation of China (grant nos. 32272068 and 32341035 to L.H.), the National Key Research and Development Program of China (grant no. 2023YFD1200402 to Z.Y.L. and L.H.), the Key Research and Development Program of Sichuan Province (grant nos. 2021YFYZ0002 to D.C.L. and 2024YFNH0024 to L.H.), the Sichuan Provincial Agricultural Department Innovative Research Team (grant no. SCCXTD-2024-11 to D.C.L.), the State Key Laboratory of Crop Gene Exploration and Utilization in Southwest China (grant nos. SKL-ZD202206 to L.Q.Z. and SKL-KF202322 to L.H.) and the United States-Israel Binational Agricultural Research and Development Fund (nos. IS-4628-13 and US-5515-22C to T.F.).
Author information
Authors and Affiliations
Contributions
B.H.W., Z.Y.L., T.F., D.C.L. and L.H. conceived and designed the research. Y.L.H., L.H.F. and L.H. developed the mapping population. Y.L.H., Y.Q.L., S.Z.N., M.H. and L.Q.Z. contributed to the development of the mutant lines. Y.L.H., Y.Q.L. and R.J.X. conducted the VIGS. Y.L.H., M.M.L., L.L.D. and R.J.X. performed the gene cloning and expression analysis. K.W. performed the gene editing work. M.M.L. and Z.Y.L. performed some of the transgenic experiments. Y.L.H., M.M.L., L.H.F., B.J., Z.W.Y. and M.H. performed the field stripe rust tests and haplotype analysis of NLRs. Y.L.H., Y.Q.L. and Y.Y.H. contributed to the cell death assay and protein–protein interaction experiments. Y.L.H., M.M.L., S.S.C., B.Y.X. and C.J.X. performed the stripe rust tests in the growth chambers. F.N. and Y.F.J. performed the PacBio sequence assembly and transcriptome analysis. Y.L.H. and Y.H.L. contributed to the phylogenetic analysis. Y.L.H., L.Q.Z. and L.L.D. developed the introgression lines. Y.L.H., B.H.W., Z.Y.L., T.F., D.C.L. and L.H. wrote the paper with input from all coauthors.
Corresponding authors
Ethics declarations
Competing interests
L.H., Y.L.H., D.C.L., B.H.W., L.H.F., L.Q.Z., Z.W.Y., S.Z.N. and M.H. declare using the TdNLR sequences under a China patent filing no. 202211502138.2, which are based on the results described. L.H., Y.L.H., D.C.L., B.H.W., L.Q.Z., Z.W.Y., S.Z.N. and M.H. declare competing interest in the use of the markers linked to YrTD121 under a China patent filing no. 202110932684.9. The other authors declare no competing interests.
Peer review
Peer review information
Nature Genetics thanks Matthew Rouse and the other, anonymous, reviewer(s) for their contribution to the peer review of this work. Peer reviewer reports are available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information (download PDF )
Supplementary Figs. 1–25 and supporting data for Supplementary Figs. 1, 18 and 20.
Supplementary Data (download XLSX )
Supporting data for Supplementary Figs. 5–7 and 9.
Source data
Source Data Fig. 2 (download XLSX )
Statistical source data.
Source Data Fig. 5 (download PDF )
Unprocessed immunoblots.
Source Data Fig. 6 (download XLSX )
Statistical source data.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Hu, Y., Li, M., Li, Y. et al. A head-to-head NLR gene pair from wild emmer confers stripe rust resistance in wheat. Nat Genet 57, 1543–1552 (2025). https://doi.org/10.1038/s41588-025-02207-0
Received:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41588-025-02207-0
This article is cited by
-
Identification of a novel stripe rust resistance locus in a Chinese wheat landrace by bulked segregant analysis of recombinant inbred lines combined with the 660 K SNP array
Euphytica (2026)
-
Integrated genome-wide association study (GWAS) and metabolomics identify genetic and metabolic drivers of stripe rust resistance in wheat from the Western Himalayas
Functional & Integrative Genomics (2026)
-
Coordinated function of paired NLRs confers Yr84-mediated stripe rust resistance in wheat
Nature Genetics (2025)
-
Two complementary NLRs from wild emmer wheat confer powdery mildew resistance
Nature Communications (2025)
-
Paired NLRs originated from Triticum dicoccoides coordinately confer resistance to powdery mildew in wheat
Nature Communications (2025)