Abstract
Peptide cyclization is a defining feature of many bioactive molecules, particularly in the ribosomally synthesized and post-translationally modified peptide (RiPP) family of natural products. Although enzymes responsible for N- to C-terminal macrocyclization, lanthipeptide formation or heterocycle installation have been well documented, a diverse array of cyclases have been discovered that perform crosslinking of aromatic side chains. These enzymes form either biaryl linkages between two aromatic amino acids or a crosslink between one aliphatic amino acid and one aromatic amino acid. Incredibly, nature has evolved multiple routes to install these crosslinks. While enzymes such as cytochromes P450 and radical S-adenosylmethionine (rSAM) enzymes are well known from other pathways, this role in RiPP biosynthesis has only recently been appreciated. Others, such as burpitide cyclases and DUF3328 (UstY) family proteins, come from eukaryotes and are relatively uncharacterized enzyme classes. This Review covers the emerging theme of aromatic amino acid side-chain crosslinking in RiPPs by focusing on the newly discovered enzymes responsible for catalyzing these challenging reactions.

This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$32.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
Change history
23 January 2025
In the version of this article initially published, the name of peer reviewer A. James Link was listed incompletely and is now amended in the HTML and PDF versions of the article.
References
Lai, Z. et al. Strategies employed in the design of antimicrobial peptides with enhanced proteolytic stability. Biotechnol. Adv. 59, 107962 (2022).
Walensky, L. D. & Bird, G. H. Hydrocarbon-stapled peptides: principles, practice, and progress. J. Med. Chem. 57, 6275–6288 (2014).
Muttenthaler, M., King, G. F., Adams, D. J. & Alewood, P. F. Trends in peptide drug discovery. Nat. Rev. Drug. Discov. 20, 309–325 (2021).
Martin, R. & Buchwald, S. L. Palladium-catalyzed Suzuki−Miyaura cross-coupling reactions employing dialkylbiaryl phosphine ligands. Acc. Chem. Res. 41, 1461–1473 (2008).
Willemse, T., Schepens, W., van Vlijmen, H. W. T., Maes, B. U. W. & Ballet, S. The Suzuki–Miyaura cross-coupling as a versatile tool for peptide diversification and cyclization. Catalysts 7, 74 (2017).
Yim, G., Thaker, M. N., Koteva, K. & Wright, G. Glycopeptide antibiotic biosynthesis. J. Antibiot. 67, 31–41 (2014).
Zhu, W. & Klinman, J. P. Biogenesis of the peptide-derived redox cofactor pyrroloquinoline quinone. Curr. Opin. Chem. Biol. 59, 93–103 (2020).
Pommerehne, K., Walisko, J., Ebersbach, A. & Krull, R. The antitumor antibiotic rebeccamycin—challenges and advanced approaches in production processes. Appl. Microbiol. Biotechnol. 103, 3627–3636 (2019).
Das, T. K., Pecoraro, C., Tomson, F. L., Gennis, R. B. & Rousseau, D. L. The post-translational modification in cytochrome c oxidase is required to establish a functional environment of the catalytic site. Biochemistry 37, 14471–14476 (1998).
Montalbán-López, M. et al. New developments in RiPP discovery, enzymology and engineering. Nat. Prod. Rep. 38, 130–239 (2021).
Richter, D. & Piel, J. Novel types of RiPP-modifying enzymes. Curr. Opin. Chem. Biol. 80, 102463 (2024).
Repka, L. M., Chekan, J. R., Nair, S. K. & van der Donk, W. A. Mechanistic understanding of lanthipeptide biosynthetic enzymes. Chem. Rev. 117, 5457–5520 (2017).
de Veer, S. J., Kan, M.-W. & Craik, D. J. Cyclotides: from structure to function. Chem. Rev. 119, 12375–12421 (2019).
Gu, W., Dong, S.-H., Sarkar, S., Nair, S. K. & Schmidt, E. W. The biochemistry and structural biology of cyanobactin pathways: enabling combinatorial biosynthesis. Methods Enzymol. 604, 113–163 (2018).
Sofia, H. J., Chen, G., Hetzler, B. G., Reyes-Spindola, J. F. & Miller, N. E. Radical SAM, a novel protein superfamily linking unresolved steps in familiar biosynthetic pathways with radical mechanisms: functional characterization using new analysis and information visualization methods. Nucleic Acids Res. 29, 1097–1106 (2001).
Broderick, J. B., Broderick, W. E. & Hoffman, B. M. Radical SAM enzymes: nature’s choice for radical reactions. FEBS Lett. 597, 92–101 (2023).
Broderick, J. B., Duffus, B. R., Duschene, K. S. & Shepard, E. M. Radical S-adenosylmethionine enzymes. Chem. Rev. 114, 4229–4317 (2014).
Grell, T. A. J., Goldman, P. J. & Drennan, C. L. SPASM and Twitch domains in S-adenosylmethionine (SAM) radical enzymes. J. Biol. Chem. 290, 3964–3971 (2015).
Zhu, W. et al. Structural properties and catalytic implications of the SPASM domain iron–sulfur clusters in Methylorubrum extorquens PqqE. J. Am. Chem. Soc. 142, 12620–12634 (2020).
Davis, K. M. et al. Structures of the peptide-modifying radical SAM enzyme SuiB elucidate the basis of substrate recognition. Proc. Natl Acad. Sci. USA 114, 10420–10425 (2017).
Arnison, P. G. et al. Ribosomally synthesized and post-translationally modified peptide natural products: overview and recommendations for a universal nomenclature. Nat. Prod. Rep. 30, 108–160 (2013).
Phan, C. S. & Morinaka, B. I. Bacterial cyclophane-containing RiPPs from radical SAM enzymes. Nat. Prod. Rep. 41, 708–720 (2024).
Clark, K. A., Bushin, L. B. & Seyedsayamdost, M. R. RaS-RiPPs in streptococci and the human microbiome. ACS Bio Med Chem Au 2, 328–339 (2022).
Ibrahim, M. et al. Control of the transcription of a short gene encoding a cyclic peptide in Streptococcus thermophilus: a new quorum-sensing system? J. Bacteriol. 189, 8845–8854 (2007).
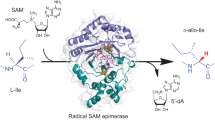
Schramma, K. R., Bushin, L. B. & Seyedsayamdost, M. R. Structure and biosynthesis of a macrocyclic peptide containing an unprecedented lysine-to-tryptophan crosslink. Nat. Chem. 7, 431–437 (2015). This study determined the structure and biosynthesis of streptide, the first of a large family of aromatic side-chain-crosslinked peptides installed by rSAM enzymes in Streptococci.
Schramma, K. R., Forneris, C. C., Caruso, A. & Seyedsayamdost, M. R. Mechanistic investigations of lysine–tryptophan cross-link formation catalyzed by streptococcal radical S-adenosylmethionine enzymes. Biochemistry 57, 461–468 (2018).
Balo, A. R. et al. Trapping a cross-linked lysine-tryptophan radical in the catalytic cycle of the radical SAM enzyme SuiB. Proc. Natl Acad. Sci. USA 118, e2101571118 (2021).
Bushin, L. B., Clark, K. A., Pelczer, I. & Seyedsayamdost, M. R. Charting an unexplored streptococcal biosynthetic landscape reveals a unique peptide cyclization motif. J. Am. Chem. Soc. 140, 17674–17684 (2018).
Rued, B. E. et al. Quorum sensing in Streptococcus mutans regulates production of tryglysin, a novel Ras-Ripp antimicrobial compound. mBio 12, e02688-20 (2021).
Caruso, A., Martinie, R. J., Bushin, L. B. & Seyedsayamdost, M. R. Macrocyclization via an arginine–tyrosine crosslink broadens the reaction scope of radical S-adenosylmethionine enzymes. J. Am. Chem. Soc. 141, 16610–16614 (2019).
Clark, K. A. & Seyedsayamdost, M. R. Bioinformatic atlas of radical SAM enzyme-modified RiPP natural products reveals an isoleucine-tryptophan crosslink. J. Am. Chem. Soc. 144, 17876–17888 (2022).
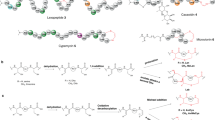
Nguyen, T. Q. N. et al. Post-translational formation of strained cyclophanes in bacteria. Nat. Chem. 12, 1042–1053 (2020). An extensive study that discovered the triceptide family of RiPPs and their rSAM biosynthetic enzymes 3-CyFEs.
Han, Y., Ma, S. & Zhang, Q. Substrate specificity and reaction directionality of a three-residue cyclophane forming enzyme PauB. Chin. Chem. Lett. 34, 107589 (2023).
Phan, C.-S. et al. Substrate promiscuity of the triceptide maturase XncB leads to incorporation of various amino acids and detection of oxygenated products. ACS Chem. Biol. 19, 855–860 (2024).
Purushothaman, M., Chang, L., Zhong, R. J. & Morinaka, B. I. The triceptide maturase OscB catalyzes uniform cyclophane topology and accepts diverse Gly-rich precursor peptides. ACS Chem. Biol. 19, 1229–1236 (2024).
Ma, S. et al. Post-translational formation of aminomalonate by a promiscuous peptide-modifying radical SAM enzyme. Angew. Chem. Int. Ed. Engl. 60, 19957–19964 (2021).
Sugiyama, R. et al. The biosynthetic landscape of triceptides reveals radical SAM enzymes that catalyze cyclophane formation on Tyr- and His-containing motifs. J. Am. Chem. Soc. 144, 11580–11593 (2022).
He, B.-B. et al. Expanded sequence space of radical S-adenosylmethionine-dependent enzymes involved in post-translational macrocyclization. Angew. Chem. Int. Ed. Engl. 61, e202212447 (2022).
Suarez, A. F. L. et al. Functional and promiscuity studies of three-residue cyclophane forming enzymes show nonnative C–C cross-linked products and leader-dependent cyclization. ACS Chem. Biol. 19, 774–783 (2024).
Morishita, Y. et al. Fused radical SAM and αKG-HExxH domain proteins contain a distinct structural fold and catalyse cyclophane formation and β-hydroxylation. Nat. Chem. 16, 1882–1893 (2024).
Imai, Y. et al. A new antibiotic selectively kills Gram-negative pathogens. Nature 576, 459–464 (2019).
Kaur, H. et al. The antibiotic darobactin mimics a β-strand to inhibit outer membrane insertase. Nature 593, 125–129 (2021).
Wuisan, Z. G., Kresna, I. D. M., Böhringer, N., Lewis, K. & Schäberle, T. F. Optimization of heterologous darobactin A expression and identification of the minimal biosynthetic gene cluster. Metab. Eng. 66, 123–136 (2021).
Guo, S. et al. Radical SAM-dependent ether crosslink in daropeptide biosynthesis. Nat. Commun. 13, 2361 (2022).
Groß, S. et al. Improved broad-spectrum antibiotics against gram-negative pathogens via darobactin biosynthetic pathway engineering. Chem. Sci. 12, 11882–11893 (2021).
Nguyen, H. et al. Characterization of a radical SAM oxygenase for the ether crosslinking in darobactin biosynthesis. J. Am. Chem. Soc. 144, 18876–18886 (2022). This study demonstrated the oxygen dependence of the rSAM DarE, the enzyme involved in the crosslink formation of darobactin biosynthesis, through in vitro studies.
Woodard, A. M., Peccati, F., Navo, C. D., Jiménez-Osés, G. & Mitchell, D. A. Darobactin substrate engineering and computation show radical stability governs ether versus C–C bond formation. J. Am. Chem. Soc. 146, 14328–14340 (2024).
Ma, S. et al. Substrate-controlled catalysis in the ether cross-link-forming radical SAM enzymes. J. Am. Chem. Soc. 145, 22945–22953 (2023).
Böhringer, N. et al. Mutasynthetic production and antimicrobial characterization of darobactin analogs. Microbiol. Spectr. 9, e01535-21 (2021).
Seyfert, C. E. et al. Darobactins exhibiting superior antibiotic activity by cryo-EM structure guided biosynthetic engineering. Angew. Chem. Int. Ed. Engl. 62, e202214094 (2023).
Miller, R. D. et al. Computational identification of a systemic antibiotic for Gram-negative bacteria. Nat. Microbiol. 7, 1661–1672 (2022).
Nguyen, B. X., Gurusinga, F. F., Mettalm, U., Schäberle, T. F. & Yokoyama, K. Radical-mediated nucleophilic peptide cross-linking in dynobactin biosynthesis. J. Am. Chem. Soc. 146, 31715–31732 (2024).
Han, Y., Li, H., Li, X., Ding, W. & Zhang, Q. Radical-mediated N–C crosslinking in the biosynthesis of dynobactin A, a ribosomally-derived peptide targeting Gram-negative bacteria. CCS Chem. https://doi.org/10.31635/ccschem.024.202404983 (2024).
Duine, J. A. The PQQ story. J. Biosci. Bioeng. 88, 231–236 (1999).
Latham, J. A., Barr, I. & Klinman, J. P. At the confluence of ribosomally synthesized peptide modification and radical S-adenosylmethionine (SAM) enzymology. J. Biol. Chem. 292, 16397–16405 (2017).
Zhu, W., Iavarone, A. T. & Klinman, J. P. Hydrogen–deuterium exchange mass spectrometry identifies local and long-distance interactions within the multicomponent radical SAM enzyme, PqqE. ACS Cent. Sci. 10, 251–263 (2024).
Nelson, D. R. Cytochrome P450 diversity in the tree of life. Biochim. Biophys. Acta Proteins Proteom. 1866, 141–154 (2018).
Reisberg, S. H. et al. Total synthesis reveals atypical atropisomerism in a small-molecule natural product, tryptorubin A. Science 367, 458–463 (2020).
Nguy, A. K. L., Martinie, R. J., Cai, A. & Seyedsayamdost, M. R. Detection of a kinetically competent compound-I intermediate in the vancomycin biosynthetic enzyme OxyB. J. Am. Chem. Soc. 146, 19629–19634 (2024).
Belin, P. et al. Identification and structural basis of the reaction catalyzed by CYP121, an essential cytochrome P450 in Mycobacterium tuberculosis. Proc. Natl Acad. Sci. USA 106, 7426–7431 (2009).
Guengerich, F. P. Mechanisms of cytochrome P450-catalyzed oxidations. ACS Catal. 8, 10964–10976 (2018).
Ali, H. S., Henchman, R. H. & de Visser, S. P. Cross-linking of aromatic phenolate groups by cytochrome P450 enzymes: a model for the biosynthesis of vancomycin by OxyB. Org. Biomol. Chem. 18, 4610–4618 (2020).
Nam, H. et al. Exploring the diverse landscape of biaryl-containing peptides generated by cytochrome P450 macrocyclases. J. Am. Chem. Soc. 145, 22047–22057 (2023).
Hu, Y. L. et al. P450-modified ribosomally synthesized peptides with aromatic cross-links. J. Am. Chem. Soc. 145, 27325–27335 (2023).
Liu, C. L. et al. P450-modified multicyclic cyclophane-containing ribosomally synthesized and post-translationally modified peptides. Angew. Chem. Int. Ed. Engl. 63, e202314046 (2024).
Wyche, T. P., Ruzzini, A. C., Schwab, L., Currie, C. R. & Clardy, J. Tryptorubin A: a polycyclic peptide from a fungus-derived streptomycete. J. Am. Chem. Soc. 139, 12899–12902 (2017).
Nanudorn, P. et al. Atropopeptides are a novel family of ribosomally synthesized and posttranslationally modified peptides with a complex molecular shape. Angew. Chem. Int. Ed. Engl. 61, e202208361 (2022).
He, B.-B. et al. Bacterial cytochrome P450 catalyzed post-translational macrocyclization of ribosomal peptides. Angew. Chem. Int. Ed. Engl. 62, e202311533 (2023).
An, J. S. et al. Discovery and biosynthesis of cihunamides, macrocyclic antibacterial RiPPs with a unique C–N linkage formed by CYP450 catalysis. Angew. Chem. Int. Ed. Engl. 62, e202300998 (2023).
Zdouc, M. M. et al. A biaryl-linked tripeptide from Planomonospora reveals a widespread class of minimal RiPP gene clusters. Cell Chem. Biol. 28, 733–739 (2021).
Hug, J. J. et al. Genome-guided discovery of the first myxobacterial biarylitide myxarylin reveals distinct C–N biaryl crosslinking in RiPP biosynthesis. Molecules 26, 7483 (2021).
Zhao, Y. et al. Cytochrome P450Blt enables versatile peptide cyclisation to generate histidine- and tyrosine-containing crosslinked tripeptide building blocks. Angew. Chem. Int. Ed. Engl. 61, e202204957 (2022). An in vitro study of P450Blt developed an important model system that has served as the basis for both engineering and structural work.
Coe, L. J. et al. Reassignment of the structure of a tryptophan-containing cyclic tripeptide produced by the biarylitide crosslinking cytochrome P450blt. Chem. Eur. J. 30, e202400988 (2024).
Hansen, M. H. et al. Structural insights into a side chain cross-linking biarylitide P450 from RiPP biosynthesis. ACS Catal. 14, 812–826 (2024).
Treisman, M. et al. An engineered biarylitide cross-linking P450 from RiPP biosynthesis generates alternative cyclic peptides. Org. Lett. 26, 1828–1833 (2024).
Hug, J. J. et al. Biosynthesis of cittilins, unusual ribosomally synthesized and post-translationally modified peptides from Myxococcus xanthus. ACS Chem. Biol. 15, 2221–2231 (2020).
Saad, H. et al. Bioinformatics-guided discovery of biaryl-linked lasso peptides. Chem. Sci. 14, 13176–13183 (2023).
Nguyen, D. T. et al. Biosynthesis of macrocyclic peptides with C-terminal β-amino-α-keto acid groups by three different metalloenzymes. ACS Cent. Sci 10, 1022–1032 (2024).
Maksimov, M. O., Pan, S. J. & Link, A. J. Lasso peptides: structure, function, biosynthesis, and engineering. Nat. Prod. Rep. 29, 996–1006 (2012).
Noyon, M. R. O. K. & Hematian, S. Peptide crosslinking by a class of plant copper enzymes. Trends Chem. 6, 649–655 (2024).
Chekan, J. R., Mydy, L. S., Pasquale, M. A. & Kersten, R. D. Plant peptides — redefining an area of ribosomally synthesized and post-translationally modified peptides. Nat. Prod. Rep. 41, 1020–1059 (2024).
Hattori, J., Boutilier, K. A., Campagne, M. M. L. & Miki, B. L. A conserved BURP domain defines a novel group of plant proteins with unusual primary structures. Mol. Gen. Genet. 259, 424–428 (1998).
Mydy, L. S. et al. An intramolecular macrocyclase in plant ribosomal peptide biosynthesis. Nat. Chem. Biol. 20, 530–540 (2024).
Kersten, R. D. & Weng, J. K. Gene-guided discovery and engineering of branched cyclic peptides in plants. Proc. Natl Acad. Sci. USA 115, E10961–E10969 (2018).
Lee, Y.-Y. et al. HypoRiPPAtlas as an atlas of hypothetical natural products for mass spectrometry database search. Nat. Commun. 14, 4219 (2023).
Chigumba, D. N. et al. Discovery and biosynthesis of cyclic plant peptides via autocatalytic cyclases. Nat. Chem. Biol. 18, 18–28 (2022). This study showed burpitide cyclase activity for the first time and established that these enzymes are copper-dependent peptide cyclases.
Kersten, R. D. et al. Gene-guided discovery and ribosomal biosynthesis of moroidin peptides. J. Am. Chem. Soc. 144, 7686–7692 (2022).
Lima, S. T. et al. A widely distributed biosynthetic cassette is responsible for diverse plant side chain cross-linked cyclopeptides. Angew. Chem. Int. Ed. Engl. 62, e202218082 (2023). This study first identified split burpitide cyclases and demonstrated their in vitro peptide cyclization activity.
Kriger, D., Pasquale, M. A., Ampolini, B. G. & Chekan, J. R. Mining raw plant transcriptomic data for new cyclopeptide alkaloids. Beilstein J. Org. Chem. 20, 1548–1559 (2024).
Nagano, N. et al. Class of cyclic ribosomal peptide synthetic genes in filamentous fungi. Fungal Genet. Biol. 86, 58–70 (2016).
Kessler, S. C. et al. Victorin, the host-selective cyclic peptide toxin from the oat pathogen Cochliobolus victoriae, is ribosomally encoded. Proc. Natl Acad. Sci. USA 117, 24243–24250 (2020).
Sogahata, K. et al. Biosynthetic studies of phomopsins unveil posttranslational installation of dehydroamino acids by UstYa family proteins. Angew. Chem. Int. Ed. Engl. 60, 25729–25734 (2021).
Kessler, S. C. & Chooi, Y.-H. Out for a RiPP: challenges and advances in genome mining of ribosomal peptides from fungi. Nat. Prod. Rep. 39, 222–230 (2022).
Jiang, Y. et al. Biosynthesis of cyclochlorotine: identification of the genes involved in oxidative transformations and intramolecular O,N-transacylation. Org. Lett. 23, 2616–2620 (2021).
Ye, Y. et al. Heterologous production of asperipin-2a: proposal for sequential oxidative macrocyclization by a fungi-specific DUF3328 oxidase. Org. Biomol. Chem. 17, 39–43 (2019).
Umemura, M. et al. Characterization of the biosynthetic gene cluster for the ribosomally synthesized cyclic peptide ustiloxin B in Aspergillus flavus. Fungal Genet. Biol. 68, 23–30 (2014).
Ye, Y. et al. Unveiling the biosynthetic pathway of the ribosomally synthesized and post-translationally modified peptide ustiloxin B in filamentous fungi. Angew. Chem. Int. Ed. Engl. 55, 8072–8075 (2016).
Ding, W. et al. Biosynthetic investigation of phomopsins reveals a widespread pathway for ribosomal natural products in ascomycetes. Proc. Natl Acad. Sci. USA 113, 3521–3526 (2016).
Zhang, W. et al. Characterization of the biosynthetic gene cluster for the ribosomally synthesized cyclic peptide epichloëcyclins in Epichloë festucae. J. Agric. Food Chem. 71, 13965–13978 (2023).
Johnson, R. D. et al. A novel family of cyclic oligopeptides derived from ribosomal peptide synthesis of an in planta-induced gene, gigA, in Epichloë endophytes of grasses. Fungal Genet. Biol. 85, 14–24 (2015).
May, J. P. & Perrin, D. M. Tryptathionine bridges in peptide synthesis. Biopolymers 88, 714–724 (2007).
Pulman, J. A., Childs, K. L., Sgambelluri, R. M. & Walton, J. D. Expansion and diversification of the MSDIN family of cyclic peptide genes in the poisonous agarics Amanita phalloides and A. bisporigera. BMC Genomics 17, 1038 (2016).
Hallen, H. E., Luo, H., Scott-Craig, J. S. & Walton, J. D. Gene family encoding the major toxins of lethal Amanita mushrooms. Proc. Natl Acad. Sci. USA 104, 19097–19101 (2007).
Luo, H. et al. Genes and evolutionary fates of the amanitin biosynthesis pathway in poisonous mushrooms. Proc. Natl Acad. Sci. USA 119, e2201113119 (2022).
Adam, S. et al. Unusual peptide-binding proteins guide pyrroloindoline alkaloid formation in crocagin biosynthesis. Nat. Chem. 15, 560–568 (2023).
Li, Y. et al. Discovery and biosynthesis of tricyclic copper-binding ribosomal peptides containing histidine-to-butyrine crosslinks. Nat. Commun. 14, 2944 (2023).
Fleming, M. C., Bowler, M. M., Park, R., Popov, K. I. & Bowers, A. A. Tyrosinase-catalyzed peptide macrocyclization for mRNA display. J. Am. Chem. Soc. 145, 10445–10450 (2023).
Seyfert, C. E. et al. New genetically engineered derivatives of antibacterial darobactins underpin their potential for antibiotic development. J. Med. Chem. 66, 16330–16341 (2023).
Eastman, K. A. S., Kincannon, W. M. & Bandarian, V. Leveraging substrate promiscuity of a radical S-adenosyl-l-methionine RiPP maturase toward intramolecular peptide cross-linking applications. ACS Cent. Sci. 8, 1209–1217 (2022).
Eastman, K. A. S., Mifflin, M. C., Oblad, P. F., Roberts, A. G. & Bandarian, V. A promiscuous rSAM enzyme enables diverse peptide cross-linking. ACS Bio Med Chem Au 3, 480–493 (2023).
Acknowledgements
This work was supported by the National Institute of General Medical Sciences of the National Institutes of Health (R35GM147439 to J.R.C.) and the National Center for Complementary and Integrative Health (T32AT008938 to M.A.P.).
Author information
Authors and Affiliations
Contributions
S.K.K., M.A.P. and J.R.C. contributed to discussions and wrote the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Chemical Biology thanks A. James Link, Huan Wang and the other, anonymous reviewer(s) for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Kandy, S.K., Pasquale, M.A. & Chekan, J.R. Aromatic side-chain crosslinking in RiPP biosynthesis. Nat Chem Biol 21, 168–181 (2025). https://doi.org/10.1038/s41589-024-01795-y
Received:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41589-024-01795-y
This article is cited by
-
S9 protease WprP2 catalyzes uniform cleavage on the precursor peptide in RiPP biosynthesis
Communications Chemistry (2026)