Abstract
D3S-001 is a next-generation KRAS-G12C inhibitor (G12Ci) designed to enhance target engagement efficiency and overcome growth factor-induced nucleotide exchange. D3S-001 was evaluated in a phase 1a dose-escalation study in patients with advanced solid tumors harboring KRASG12C mutation (N = 42) and a phase 1b expansion cohort of patients with non-small-cell lung cancer (NSCLC) whose disease progressed after prior G12Ci therapy (N = 20). The primary endpoints were safety and determination of the maximum tolerated dose. Secondary endpoints included pharmacokinetics, confirmed objective response rate (ORR) and disease control rate. D3S-001 demonstrated dose-dependent pharmacokinetics and no dose-limiting toxicities, and the maximum tolerated dose was not reached. Grade 3 treatment-related adverse events were reported in seven patients (16.7%) in the G12Ci-naive dose-escalation cohort and two patients (10.0%) in the G12Ci-pretreated NSCLC expansion cohort. There were no grade 4 or 5 treatment-related adverse events. D3S-001 600 mg was selected as the dose for further investigation based on pharmacokinetics. Confirmed ORR in the G12Ci-naive population was 73.5% overall (25 of 34), and 66.7% (14 of 21), 88.9% (8 of 9) and 75.0% (3 of 4) in patients with NSCLC, colorectal cancer and pancreatic ductal adenocarcinoma, respectively. Among patients with G12Ci-pretreated NSCLC, ORR was 30.0% (6 of 20) and disease control rate was 80.0% (16 of 20). This study demonstrates the safety and tolerability of D3S-001 monotherapy with promising antitumor activity. The phase 1b expansion phase is ongoing. ClinicalTrials.gov identifier: NCT05410145.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$32.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
Data availability
Deidentified participant data that underlie the results reported in this article, the protocol and statistical analysis plan may be available upon reasonable request to the corresponding author from qualified researchers following completion of the study when all dose-expansion cohorts are completed and the clinical study reports have been finalized. Data are only available upon request to protect the privacy of the company and clinical trial participants. Qualified researchers may email Publication@d3bio.com to request data. A response will be provided within 2 months of the request depending on the extent of the request. Databases used in this study were Exome Aggregation Consortium (http://exac.broadinstitute.org), 1000 Genomes Project (https://www.internationalgenome.org/), dbSNP (https://www.ncbi.nlm.nih.gov/snp/) and ESP6500SI-V2 (https://genome.ucsc.edu/).
References
Zehir, A. et al. Mutational landscape of metastatic cancer revealed from prospective clinical sequencing of 10,000 patients. Nat. Med. 23, 703–713 (2017).
Prior, I. A., Lewis, P. D. & Mattos, C. A comprehensive survey of Ras mutations in cancer. Cancer Res. 72, 2457–2467 (2012).
Nassar, A. H., Adib, E. & Kwiatkowski, D. J. Distribution of KRAS (G12C) somatic mutations across race, sex, and cancer type. N. Engl. J. Med. 384, 185–187 (2021).
Frost, M. G. et al. KRAS G12C mutated advanced non-small cell lung cancer (NSCLC): characteristics, treatment patterns and overall survival from a Danish nationwide observational register study. Lung Cancer 178, 172–182 (2023).
Sebastian, M. et al. KRAS G12C-mutated advanced non-small cell lung cancer: a real-world cohort from the German prospective, observational, nation-wide CRISP Registry (AIO-TRK-0315). Lung Cancer 154, 51–61 (2021).
Salem, M. et al. O-3 characterization of KRAS mutation variants and prevalence of KRAS-G12C in gastrointestinal malignancies. Ann. Oncol. 32, S218 (2021).
Kolch, W., Berta, D. & Rosta, E. Dynamic regulation of RAS and RAS signaling. Biochem. J. 480, 1–23 (2023).
Huang, L., Guo, Z., Wang, F. & Fu, L. KRAS mutation: from undruggable to druggable in cancer. Signal Transduct. Target. Ther. 6, 386 (2021).
Hunter, J. C. et al. Biochemical and structural analysis of common cancer-associated KRAS mutations. Mol. Cancer Res. 13, 1325–1335 (2015).
Patricelli, M. P. et al. Selective inhibition of oncogenic KRAS output with small molecules targeting the inactive state. Cancer Discov. 6, 316–329 (2016).
Ostrem, J. M., Peters, U., Sos, M. L., Wells, J. A. & Shokat, K. M. K-Ras(G12C) inhibitors allosterically control GTP affinity and effector interactions. Nature 503, 548–551 (2013).
Janes, M. R. et al. Targeting KRAS mutant cancers with a covalent G12C-specific inhibitor. Cell 172, 578–589.e517 (2018).
Mok, T. S. K. et al. KRYSTAL-12: phase 3 study of adagrasib versus docetaxel in patients with previously treated advanced/metastatic non-small cell lung cancer (NSCLC) harboring a KRASG12C mutation. J. Clin. Oncol. 42, LBA8509 (2024).
de Langen, A. J. et al. Sotorasib versus docetaxel for previously treated non-small-cell lung cancer with KRAS(G12C) mutation: a randomised, open-label, phase 3 trial. Lancet 401, 733–746 (2023).
Fakih, M. G. et al. Sotorasib for previously treated colorectal cancers with KRAS(G12C) mutation (CodeBreaK100): a prespecified analysis of a single-arm, phase 2 trial. Lancet Oncol. 23, 115–124 (2022).
Yaeger, R. et al. Adagrasib with or without cetuximab in colorectal cancer with mutated KRAS G12C. N. Engl. J. Med. 388, 44–54 (2023).
Fakih, M. G. et al. Sotorasib plus panitumumab in refractory colorectal cancer with mutated KRAS G12C. N. Engl. J. Med. 389, 2125–2139 (2023).
Yaeger, R. et al. Efficacy and safety of adagrasib plus cetuximab in patients with KRASG12C-mutated metastatic colorectal cancer. Cancer Discov. 14, 982–993 (2024).
Zhang, J. et al. D3S-001, a KRAS G12C inhibitor with rapid target engagement kinetics, overcomes nucleotide cycling, and demonstrates robust preclinical and clinical activities. Cancer Discov. 14, 1675–1698 (2024).
Ryan, M. B. et al. KRAS(G12C)-independent feedback activation of wild-type RAS constrains KRAS(G12C) inhibitor efficacy. Cell Rep. 39, 110993 (2022).
Xue, J. Y. et al. Rapid non-uniform adaptation to conformation-specific KRAS(G12C) inhibition. Nature 577, 421–425 (2020).
Amodio, V. et al. EGFR blockade reverts resistance to KRAS(G12C) inhibition in colorectal cancer. Cancer Discov. 10, 1129–1139 (2020).
Jänne, P. A. et al. Adagrasib in non-small-cell lung cancer harboring a KRAS(G12C) mutation. N. Engl. J. Med. 387, 120–131 (2022).
Skoulidis, F. et al. Sotorasib for lung cancers with KRAS p.G12C mutation. N. Engl. J. Med. 384, 2371–2381 (2021).
Shi, Y. et al. Glecirasib in KRAS(G12C)-mutated nonsmall-cell lung cancer: a phase 2b trial. Nat. Med. 31, 894–900 (2025).
Burns, T. F. et al. Efficacy and safety of olomorasib (LY3537982), a second-generation KRAS G12C inhibitor (G12Ci), in combination with pembrolizumab in patients with KRAS G12C-mutant advanced NSCLC. J. Clin. Oncol. 42(Suppl. 16), abstr 8510 (2024).
Sacher, A. et al. Single-agent divarasib (GDC-6036) in solid tumors with a KRAS G12C mutation. N. Engl. J. Med. 389, 710–721 (2023).
Jänne, P. A. et al. Abstract PR014: preliminary safety and anti-tumor activity of RMC-6291, a first-in-class, tri-complex KRASG12C(ON) inhibitor, in patients with or without prior KRASG12C(OFF) inhibitor treatment. Mol. Cancer Ther. 22, PR014 (2023).
Nokin, M. J. et al. RAS-ON inhibition overcomes clinical resistance to KRAS G12C-OFF covalent blockade. Nat. Commun. 15, 7554 (2024).
Lu, J. et al. D3S-001, a highly potent, selective, and differentiated covalent inhibitor of KRAS G12C: human dose prediction and first-in-human (FIH) trial design. J. Clin. Oncol. 40, e15087 (2022).
Lee, A. T. M. & Nagasaka, M. Adagrasib in KRYSTAL-12 has not broken the KRAS G12C enigma code of the unspoken 6-month PFS barrier in NSCLC. Lung Cancer (Auckl.) 15, 169–176 (2024).
Awad, M. M. et al. Acquired resistance to KRAS(G12C) inhibition in cancer. N. Engl. J. Med. 384, 2382–2393 (2021).
Zhao, Y. et al. Diverse alterations associated with resistance to KRAS(G12C) inhibition. Nature 599, 679–683 (2021).
Brazel, D. & Nagasaka, M. Divarasib in the evolving landscape of KRAS G12C inhibitors for NSCLC. Target. Oncol. 19, 297–301 (2024).
Odegaard, J. I. et al. Validation of a plasma-based comprehensive cancer genotyping assay utilizing orthogonal tissue- and plasma-based methodologies. Clin. Cancer Res. 24, 3539–3549 (2018).
Lanman, R. B. et al. Analytical and clinical validation of a digital sequencing panel for quantitative, highly accurate evaluation of cell-free circulating tumor DNA. PLoS ONE 10, e0140712 (2015).
Willis, J. et al. Validation of microsatellite instability detection using a comprehensive plasma-based genotyping panel. Clin. Cancer Res. 25, 7035–7045 (2019).
Mao, X. et al. Capture-based targeted ultradeep sequencing in paired tissue and plasma samples demonstrates differential subclonal ctDNA-releasing capability in advanced lung cancer. J. Thorac. Oncol. 12, 663–672 (2017).
Li, Y. S. et al. Unique genetic profiles from cerebrospinal fluid cell-free DNA in leptomeningeal metastases of EGFR-mutant non-small-cell lung cancer: a new medium of liquid biopsy. Ann. Oncol. 29, 945–952 (2018).
Tao, Z. et al. Profiling receptor tyrosine kinase fusions in Chinese breast cancers. Front. Oncol. 11, 741142 (2021).
Acknowledgements
Funding for this research was provided by D3 Bio. We thank the patients who participated in this trial, as well as their families and caregivers, and the staff and investigators at all the trial sites. Medical writing assistance was provided by M. Sweetlove and S. Duggan of ApotheCom, and was funded by D3 Bio.
Author information
Authors and Affiliations
Contributions
B.C.C., S.L., T.M., Z.J.C., C.C. and J.W. were responsible for the study conception or design. B.C.C., S.L., M.A.L., Z.S., J.J.P., S.M.L., Z.L., J. Zhao, G.R., Y.Z., Jun Zhang, A.L., H.H.L., C.C., J.W., Y.S., Z.F., Q.C., H.W., Jing Zhang, Z.J.C., M.L.J. and T.M. conducted the study and were involved in acquisition, analysis or interpretation of data. J.W. performed statistical analysis and Y.S. verified the biomarker data. B.C.C., S.L., M.A.L., Z.S., J.J.P., S.M.L., Z.L., J. Zhao, G.R., Y.Z., Jun Zhang, A.L., H.H.L., C.C., J.W., Y.S., Z.F., Q.C., H.W., Jing Zhang, Z.J.C., M.L.J. and T.M. drafted the work or reviewed it critically for important intellectual content. B.C.C., S.L., M.A.L., Z.S., J.J.P., S.M.L., Z.L., J. Zhao, G.R., Y.Z., Jun Zhang, A.L., H.H.L., C.C., J.W., Y.S., Z.F., Q.C., H.W., Jing Zhang, Z.J.C., M.L.J. and T.M. approved the final version for publication and agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Corresponding author
Ethics declarations
Competing interests
B.C.C. reports research funding from GIInovation, AstraZeneca, Champions Oncology, CJ Bioscience, Cyrus, Janssen, Merck Sharp & Dohme, Dong-A ST, Yuhan, ImmuneOncia, Therapex, JINTSbio and Vertical Bio AG; royalties from Champions Oncology, Crown Bioscience, Imagen and PearlRiver Bio GmbH; consulting fees from BeiGene, Novartis, AstraZeneca, Boehringer Ingelheim, Roche, Bristol Myers Squibb, CJ, Cyrus Therapeutics, Ono, Yuhan, Pfizer, Eli Lilly, Janssen, Takeda, Merck Sharp & Dohme, Gilead, Amgen, Daiichi Sankyo, Regeneron, Sanofi, AnHeart Therapeutics, Seagen, Harpoon Therapeutics, GlaxoSmithKline and ArriVent; honoraria as an invited speaker from ASCO, AstraZeneca, Guardant, Roche, ESMO, IASLC, Korean Cancer Association, Korean Society of Medical Oncology, Korean Society of Thyroid-Head and Neck Surgery, Korean Cancer Study Group, Novartis, Merck Sharp & Dohme, the Chinese Thoracic Oncology Society, Pfizer and ZaiLab; participation on scientific advisory boards for KANAPH Therapeutic Inc., Bridgebio Therapeutics, Cyrus Therapeutics, Guardant Health, J INTS Bio and Therapex Co. Ltd; stock ownership with TheraCanVac Inc., Gencurix Inc., Bridgebio Therapeutics, KANPH Therapeutic Inc., Cyrus Therapeutics, Interpark Bio Convergence Corp. and J INTS Bio; and other financial or nonfinancial interests with Yonsei University Health System (employment), DAAN Biotherapeutics (founder) and J INTS BIO (member of the board of directors). S.L. reports research funding from AstraZeneca, Hutchison, Bristol Myers Squibb, Hengrui Therapeutics, BeiGene, Roche and Hansoh; consulting fees from AstraZeneca, Hutchison, Simcere, ZaiLab and Yuhan Corporation; honoraria from AstraZeneca, Roche, Hansoh and Hengrui Therapeutics; participation on advisory boards for AstraZeneca, Yuhan Corporation, Inventis Bio, Merck Sharp & Dohme, Simcere Zaiming Pharmaceutical, Shanghai Fosun Pharmaceutical and Phase Therapeutics; and a leadership role with Innovent Biologics. Z.L. reports honoraria from AstraZeneca, Roche, Hansoh and Pfizer. G.R. reports research funding from Bristol Myers Squibb, Roche/Genentech, AstraZeneca, Merck, Takeda, BeiGene, Pfizer, CBT Pharmaceuticals, Corvus Pharmaceuticals, Novotech, Shanghai Fosun Pharmaceutical Development, Shanghai Henlius Biotech, Five Prime Therapeutics, Suzhou Alphamab, Boehringer Ingelheim, Adagene Inc., Bio-Thera Solutions, ChemoCentryx, Curon Biopharmaceutical, D3 Bio, Inventis Bio, Senz Oncology, Genfleet Therapeutics, GeneQuantum Healthcare, Keythera Pharmaceuticals, LaNova Australia, Medicenna Therapeutics, Minghui Pharmaceutical, Neoleukin Therapeutics, PharmAbcine Australia, RemeGen, Seagen, Surface Oncology, Eucure Biopharma, Janssen Oncology, ImmuGen, Imugene, Therapim, Zentalis and Agenus. Jun Zhang reports participation on advisory boards for Astellas and Shanghai HengRui. H.H.L. reports research funding from MSD, MundiPharma and Novartis; consulting fees from Boehringer Ingelheim, Celgene, Eli Lilly, Illumina, Novartis, Merck Serono, Takeda and George Clinical; honoraria from AbbVie, Bayer, Eisai, Eli Lilly, Guardant Health and Novartis; and support for meeting attendance and/or travel from Bayer, Boehringer Ingelheim, Merck Sharp & Dohme, Novartis and Pfizer. C.C., J.W., Y.S., Z.F., Q.C., H.W., Jing Zhang and Z.J.C. report employment and stock or stock options with D3 Bio. M.L.J. reports funding to the institution from D3 Bio for the current work; grants to the institution from AbbVie, Adaptimmune, Amgen, Arcus Biosciences, Array BioPharma, ArriVent BioPharma, Artios Pharma, AstraZeneca, Bayer, BeiGene, BerGenBio, BioAtla, Black Diamond, Boehringer Ingelheim, Bristol Myers Squibb, Calithera Biosciences, Carisma Therapeutics, City of Hope National Medical Center, Conjupro Biotherapeutics, Corvus Pharmaceuticals, Curis, CytomX, Daiichi Sankyo, Dracen Pharmaceuticals, Lilly, Elicio Therapeutics, EMD Serono, EQRx, Erasca, Exelixis, Fate Therapeutics, Genentech/Roche, Genmab, Genocea Biosciences, GlaxoSmithKline, Gritstone Oncology, Harpoon, Helsinn Healthcare SA, Hengrui Therapeutics, Hutchinson MediPharma, IDEAYA Biosciences, IGM Biosciences, Immuneering Corporation, Immunitas Therapeutics, Immunocore, Impact Therapeutics, Incyte, Janssen, Kartos Therapeutics, LockBody Therapeutics, Loxo Oncology, Memorial Sloan Kettering, Merck, Merus, Mirati Therapeutics, Mythic Therapeutics, NeoImmune Tech, Neovia Oncology, NextPoint Therapeutics, Novartis, Numab Therapeutics, Nuvalent, OncoC4, Palleon Pharmaceuticals, Pfizer, PMV Pharmaceuticals, Rain Therapeutics, RasCal Therapeutics, Regeneron Pharmaceuticals, Relay Therapeutics, Revolution Medicines, Ribon Therapeutics, Rubius Therapeutics, Sanofi, Seven and Eight Biopharmaceuticals/Birdie Pharmaceuticals, Shattuck Laboratories, Silicon Therapeutics, Systimmune, Taiho Oncology, Takeda Pharmaceuticals, TCR2 Therapeutics, Tempest Therapeutics, TheRas, Tizona Therapeutics, TMUNITY Therapeutics, Turning Point Therapeutics, Vividion, Vyriad and Y-mAbs Therapeutics; consulting fees from AbbVie, Alentis Therapeutics, Amgen, Arcus Biosciences, AstraZeneca, Biohaven Pharmaceuticals, Boehringer Ingelheim, Bristol Myers Squibb, D3 Bio Limited, Daiichi Sankyo, Fate Therapeutics, Genentech/Roche, Gilead Sciences, GlaxoSmithKline, Gritstone Oncology, Hookipa Biotech, Immunocore, Janssen Pharmaceuticals, Lilly, Merck, Mirati Therapeutics, ModeX Therapeutics, Normunity, Novartis, Novocure, Pfizer, Regneron Pharmaceuticals, Revolution Medicines, Sanofi-Aventis, SeaGen, Synthekine, Takeda Pharmaceuticals and Zai Laboratory. T.M. reports research funding from AstraZeneca, Bristol Myers Squibb, G1 Therapeutics, Merck Sharp & Dohme, Merck Serono, Novartis, Pfizer, Roche, SFJ, Takeda and XCovery; consulting fees from AbbVie, ACEA Pharma, Adagen, Alentis Therapeutics, Alpha Biopharma, Amgen, Amoy Diagnostics Co, AnHeart Therapeutics, AVEO Pharmaceuticals, Bayer Healthcare Pharmaceuticals, BeiGene, BerGenBio ASA, Berry Oncology, Boehringer Ingelheim, Blueprint Medicines Corporation, BridgeBio, Bristol Myers Squibb, Bowtie Life Insurance Company Limited, Bridge Biotherapeutics Inc., Covidien LP, C4 Therapeutics Inc., Cirina, CStone Pharmaceuticals, Curio Science, D3 Bio, Da Volterra, Daiichi Sankyo, Eisai, Elevation Oncology, Erasca, F. Hoffmann–La Roche/Genentech, Fishawack Facilitate, G1 Therapeutics, geneDecode, Gilead Sciences, GLG’s Healthcare, Gritstone Oncology, Guardant Health, HengRui Therapeutics, HiberCell, HutchMed, Ignyta, Illumina, Imagene AI, Incyte Corporation, Inivata, InxMed, IQVIA, Janssen, Lakeshore Biotech, Lilly, Lunit USA, Loxo Oncology, Lucence Health, Medscape/WebMD, Medtronic, Merck Serono, Merck Sharp & Dohme, Mirati Therapeutics, MiRXES, MoreHealth, Novartis, Novocure GmbH, Ningbo NewBay Technology Development, Omega Therapeutics, OrigiMed, OSE Immunotherapeutics, PeerVoice, Phanes Therapeutics, Pfizer, PrIME Oncology, Prenetics Global, Puma Biotechnology, Qiming Development (HK), Regen Medtech Holdings, Regeneron Pharmaceuticals, Roche Pharmaceuticals/Diagnostics/Foundation One, Sanofi-Aventis, Schrödinger, Seagen International GmbH, SFJ Pharmaceutical, Simcere of America, Synergy Research, Summit Therapeutics, Takeda Pharmaceuticals HK, Tigermed, Vertex Pharmaceuticals, Virtus Medical Group, Xencor and Yuhan Corporation; honoraria from ACEA Pharma, Alpha Biopharma, Amgen, Amoy Diagnostics, AstraZeneca, BeiGene, Boehringer Ingelheim, Bristol Myers Squibb, Daiichi Sankyo, Daz Group, Fishawack Facilitate, InMed Medical Communication, Janssen Pharmaceutica NV, Jiahui Holdings, LiangYiHui Healthcare, Lilly, Lucence Health, MD Health Brazil, Medscape, Merck Pharmaceuticals HK, Merck Sharp & Dohme, MiRXES, Novartis, OrigiMed, P. Permanyer SL, PeerVoice, Physicians’ Education Resource, Pfizer, PrIME Oncology, Research to Practice, Roche Pharmaceuticals/Diagnostics/Foundation One, Sanofi-Aventis, Shanghai BeBirds Translation & Consulting, Taiho Pharmaceutical, Takeda Oncology, and Touch Independent Medical Education; support for meeting attendance and/or travel from Novartis, Roche, Pfizer, AstraZeneca, Daiichi Sankyo, Boehringer Ingelheim, MiRXES, Bristol Myers Squibb, Merck Sharp & Dohme, AbbVie, ZaiLab and Liangyihui; participation in advisory boards for AbbVie, ACEA Pharma, Amgen, AstraZeneca, Alentis Therapeutics AG, BerGenBio ASA, Berry Oncology, Blueprint Medicines Corporation, Boehringer Ingelheim, Bowtie Life Insurance, Bristol Myers Squibb, C4 Therapeutics, Covidien LP, CStone Pharmaceuticals, Curio Science, D3 Bio, Daiichi Sankyo, Eisai, Erasca, Fishawack Facilitate, G1 Therapeutics, Gilead Sciences, Gritstone Oncology, Guardant Health, geneDecode (uncompensated), Hengrui Therapeutics, HutchMed, Ignyta, Incyte Corporation, Imagene AI, Inivata, IQVIA, Janssen, Lakeshore Biotech, Lilly, Loxo Oncology, Lunit, Merck Serono, Merck Sharp & Dohme, Mirati Therapeutics, MiRXES Group, Novartis, OrigiMed, Phanes Therapeutics, Pfizer, Prenetics Global, Puma Biotechnology, Roche/Genentech, Regeneron Pharmaceuticals, Sanofi-Aventis R&D, SFJ Pharmaceutical, Simcere of America, Simcere Zaiming, Takeda, Vertex Pharmaceuticals, Virtus Medical Group, Xencor and Yuhan Corporation; leadership role for AstraZeneca, HutchMed, Aurora, Epoch Biosciences and Insighta; stock or stock options with AstraZeneca, Aurora Tele-Oncology, Biolidics, HutchMed, Prenetics Global, D3 Bio, Lunit, Bowtie Life Insurance, Lakeshore Biotech, Loxo Oncology, Virtus Medical Group, Yinson Capital, Phanes Therapeutics, Insighta and Alentis Therapeutics AG. The other authors declare no competing interests.
Peer review
Peer review information
Nature Medicine thanks Benjamin Besse, David Hong and the other, anonymous, reviewer(s) for their contribution to the peer review of this work. Primary Handling Editor: Saheli Sadanand, in collaboration with the Nature Medicine team.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data
Extended Data Fig. 1 D3S-001 pharmacokinetics at steady state in part 1 (phase 1a dose escalation).
(A) Geometric mean steady-state plasma concentrations; error bars represent geometric standard error. (B) Individual steady-state plasma AUCtau,total. The sample size for each dose group is: 50 mg (n = 2); 100 mg (n = 7); 200 mg (n = 4); 400 mg (n = 9); 600 mg (n = 8); 900 mg (n = 8). AUC, area under the concentration time curve; PK, pharmacokinetics.
Extended Data Fig. 2 Magnetic resonance imaging scans of brain and computed tomography scans of liver metastatic lesions following treatment with D3S-001.
Magnetic resonance imaging scans of the brain and computed tomography scans of the liver are shown for a patient with NSCLC who had prior treatment with platinum doublet chemotherapy and immunotherapy (atezolizumab). The patient had previously treated/stable brain metastatic lesion at baseline (received Gamma Knife treatment 1.5 years before starting study treatment with D3S-001 during which time no postradiation shrinkage was observed). Following D3S-001, the patient had confirmed partial response, and shrinkage of the brain metastatic lesion was observed following the first dose (50 mg) with further shrinkage observed in parallel with shrinkage of liver metastatic lesion at higher doses during intra-patient dose escalation. Treatment with D3S-001 is ongoing ( > 22 months).
Extended Data Fig. 3 D3S-001 pharmacokinetics at steady state in part 2 (phase 1b dose-expansion cohort).
Geometric mean steady-state plasma concentrations (n = 17); error bars represent geometric standard error.
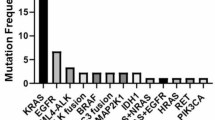
Extended Data Fig. 4 Baseline somatic mutation profile of KRAS G12C inhibitor pretreated patients before D3S-001 treatment in Part 2.
BOR, best overall response; PD progressive disease; PR, partial response; SD, stable disease.
Supplementary information
Supplementary Information (download PDF )
Supplementary Tables 1 and 2.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Cho, B.C., Lu, S., Lee, M.A. et al. D3S-001 in advanced solid tumors with KRASG12C mutations: a phase 1 trial. Nat Med 31, 2768–2777 (2025). https://doi.org/10.1038/s41591-025-03688-6
Received:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41591-025-03688-6
This article is cited by
-
Therapeutic advances with KRASG12C inhibitors and combination strategies in non-small cell lung cancer brain metastases
Cancer Gene Therapy (2026)
-
Evolution of direct RAS inhibitors: from undruggable target to clinical breakthroughs
Molecular Cancer (2025)
-
The evolving landscape of RAS-targeted therapies
Nature Cancer (2025)
-
Next-generation KRAS-G12C inhibitor D3S-001 shows promise
Nature Reviews Clinical Oncology (2025)
-
Tissue-agnostic biomarkers in solid tumors: current approvals and emerging candidates
Cancer and Metastasis Reviews (2025)