Abstract
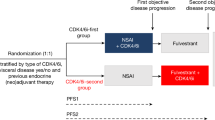
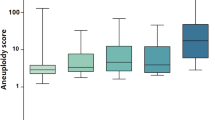
CDK4/6 inhibitors (CDK4/6i) improve outcome in patients with advanced estrogen receptor-positive, HER2− breast cancer. The phase 3 SONIA trial compared the addition of CDK4/6i to first- versus second-line endocrine therapy for time to disease progression after second-line treatment (progression-free survival after two lines of treatment (PFS2)), as well as for secondary outcomes overall survival, PFS after one line of treatment (PFS1), health-related quality of life (HRQOL), toxicity and cost-effectiveness. No significant difference in PFS2 was observed; however, on an individual patient level this may be different. Using prespecified circulating tumor DNA analyses, we performed an exploratory study to evaluate whether pretreatment circulating tumor DNA (ctDNA) levels in plasma can identify patients that benefit from CDK4/6i during their first-line treatment. Cell free DNA before start of first-line treatment from 409 female patients participating in SONIA was analyzed with the modified fast aneuploidy screening test-sequencing system. This assay yields a genome-wide aneuploidy score, indicative of ctDNA levels. Cox proportional hazard analyses for PFS1 and PFS2 were performed separately for the ctDNA high group (aneuploidy score ≥ 5) and the ctDNA low group (aneuploidy score < 5). In total, 141 of the 409 included patients had a high genome-wide aneuploidy score at baseline. PFS2 in the first- compared to the second-line CDK4/6i strategy showed hazard ratios of 0.58 (95% confidence interval 0.38–0.88) and 1.36 (95% confidence interval 0.95–1.96) in the high and low aneuploidy group, respectively. A significant interaction was demonstrated between treatment strategy and aneuploidy score for PFS2 (P = 0.004). In conclusion, this study demonstrated that pretreatment ctDNA levels can be used to identify patients that benefit from first-line CDK4/6i treatment. ClinicalTrials.gov registration: NCT03425838.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$32.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
Data availability
De-identified mFast-SeqS and patient clinical data underlying the results reported in this article will be made available to other researchers on reasonable request for academic use, within the limitations of the informed consent and the study’s consortium agreement. A detailed data proposal is required and will be considered on a case-by-case basis. Requests should be directed to BOOG study Center (info@boogstudycenter.nl) and will be reviewed by the study’s principal investigators. A response will be provided within 90 days. A signed data access agreement with the sponsor is required before accessing shared data.
References
Finn, R. S. et al. Palbociclib and letrozole in advanced breast cancer. N. Engl. J. Med. 375, 1925–1936 (2016).
Hortobagyi, G. N. et al. Updated results from MONALEESA-2, a phase III trial of first-line ribociclib plus letrozole versus placebo plus letrozole in hormone receptor-positive, HER2-negative advanced breast cancer. Ann. Oncol. 29, 1541–1547 (2018).
Johnston, S. et al. MONARCH 3 final PFS: a randomized study of abemaciclib as initial therapy for advanced breast cancer. npj Breast Cancer 5, 5 (2019).
Tripathy, D. et al. Ribociclib plus endocrine therapy for premenopausal women with hormone-receptor-positive, advanced breast cancer (MONALEESA-7): a randomised phase 3 trial. Lancet Oncol. 19, 904–915 (2018).
Cristofanilli, M. et al. Fulvestrant plus palbociclib versus fulvestrant plus placebo for treatment of hormone-receptor-positive, HER2-negative metastatic breast cancer that progressed on previous endocrine therapy (PALOMA-3): final analysis of the multicentre, double-blind, phase 3 randomised controlled trial. Lancet Oncol. 17, 425–439 (2016).
Slamon, D. J. et al. Phase III randomized study of ribociclib and fulvestrant in hormone receptor-positive, human epidermal growth factor receptor 2-negative advanced breast cancer: MONALEESA-3. J. Clin. Oncol. 36, 2465–2472 (2018).
Sledge, G. W. Jr. et al. MONARCH 2: abemaciclib in combination with fulvestrant in women with HR+/HER2− advanced breast cancer who had progressed while receiving endocrine therapy. J. Clin. Oncol. 35, 2875–2884 (2017).
Sledge, G. W. Jr. et al. The effect of abemaciclib plus fulvestrant on overall survival in hormone receptor-positive, ERBB2− negative breast cancer that progressed on endocrine therapy—MONARCH 2: a randomized clinical trial. JAMA Oncol. 6, 116–124 (2020).
Hortobagyi, G. N. et al. Overall survival with ribociclib plus letrozole in advanced breast cancer. N. Engl. J. Med. 386, 942–950 (2022).
Slamon, D. J. et al. Overall survival with ribociclib plus fulvestrant in advanced breast cancer. N. Engl. J. Med. 382, 514–524 (2020).
Lu, Y. S. et al. Updated overall survival of ribociclib plus endocrine therapy versus endocrine therapy alone in pre- and perimenopausal patients with HR+/HER2− advanced breast cancer in MONALEESA-7: a phase III randomized clinical trial. Clin. Cancer Res. 28, 851–859 (2022).
van Ommen-Nijhof, A. et al. Selecting the optimal position of CDK4/6 inhibitors in hormone receptor-positive advanced breast cancer—the SONIA study: study protocol for a randomized controlled trial. BMC Cancer 18, 1146 (2018).
Sonke, G. S. et al. Early versus deferred use of CDK4/6 inhibitors in advanced breast cancer. Nature 636, 474–480 (2024).
Wang, R. et al. Early evaluation of circulating tumor DNA as marker of therapeutic efficacy and prognosis in breast cancer patients during primary systemic therapy. Breast 76, 103738 (2024).
Abbosh, C. et al. Phylogenetic ctDNA analysis depicts early-stage lung cancer evolution. Nature 545, 446–451 (2017).
Belic, J. et al. Rapid identification of plasma DNA samples with increased ctDNA levels by a modified FAST-SeqS approach. Clin. Chem. 61, 838–849 (2015).
Verschoor, N. et al. Tumor-agnostic ctDNA levels by mFAST-SeqS in first-line HR-positive, HER2 negative metastatic breast cancer patients as a biomarker for survival. npj Breast Cancer 9, 61 (2023).
Mendelaar, P. A. J. et al. Genome-wide aneuploidy detected by mFast-SeqS in circulating cell-free DNA is associated with poor response to pembrolizumab in patients with advanced urothelial cancer. Mol. Oncol. 16, 2086–2097 (2022).
Isebia, K. T. et al. mFast-SeqS-based aneuploidy score in circulating cell-free DNA is a prognostic biomarker in prostate cancer. Mol. Oncol. 17, 1898–1907 (2023).
Reichert, Z. R. et al. Prognostic value of plasma circulating tumor DNA fraction across four common cancer types: a real-world outcomes study. Ann. Oncol. 34, 111–120 (2023).
Di Leo, A. et al. Prognostic characteristics in hormone receptor-positive advanced breast cancer and characterization of abemaciclib efficacy. npj Breast Cancer 4, 41 (2018).
Robertson, J. F. R. et al. Meta-analyses of visceral versus non-visceral metastatic hormone receptor-positive breast cancer treated by endocrine monotherapies. npj Breast Cancer 7, 11 (2021).
Heidary, M. et al. The dynamic range of circulating tumor DNA in metastatic breast cancer. Breast Cancer Res. 16, 421 (2014).
Liu, B. et al. The circulating tumor DNA (ctDNA) alteration level predicts therapeutic response in metastatic breast cancer: novel prognostic indexes based on ctDNA. Breast 65, 116–123 (2022).
Stover, D. G. et al. Association of cell-free DNA tumor fraction and somatic copy number alterations with survival in metastatic triple-negative breast cancer. J. Clin. Oncol. 36, 543–553 (2018).
Walter, C. B. et al. Characteristics and prognosis of patients with primary metastatic disease vs. recurrent HER2-negative, hormone receptor-positive advanced breast cancer. Breast 80, 104412 (2025).
de Maar, J. S. et al. Comparison between de novo and metachronous metastatic breast cancer: the presence of a primary tumour is not the only difference—a Dutch population-based study from 2008 to 2018. Breast Cancer Res. Treat. 198, 253–264 (2023).
Pernas, S., Tolaney, S. M., Winer, E. P. & Goel, S. CDK4/6 inhibition in breast cancer: current practice and future directions. Ther. Adv. Med. Oncol. 10, 1758835918786451 (2018).
Guo, L. et al. Breast cancer heterogeneity and its implication in personalized precision therapy. Exp. Hematol. Oncol. 12, 3 (2023).
Santonja, A. et al. Comparison of tumor-informed and tumor-naive sequencing assays for ctDNA detection in breast cancer. EMBO Mol. Med. 15, e16505 (2023).
Klocker, E. V. et al. Clinical applications of next-generation sequencing-based ctDNA analyses in breast cancer: defining treatment targets and dynamic changes during disease progression. Mol. Oncol. 19, 1897–1917 (2025).
van Dessel, L. F. et al. Application of circulating tumor DNA in prospective clinical oncology trials—standardization of preanalytical conditions. Mol. Oncol. 11, 295–304 (2017).
Eisenhauer, E. A. et al. New response evaluation criteria in solid tumours: revised RECIST guideline (version 1.1). Eur. J. Cancer 45, 228–247 (2009).
Acknowledgements
We thank all participating patients and their families as well as the entire SONIA Study Consortium and the Dutch Breast Cancer research Group (BOOG) for their support and sponsorship. This study was funded by the Dutch Cancer Society (KWF grant 12039; A.J.) and Breast Cancer Now’s Catalyst Program (grant reference number 2018NovPCC100; S.M.W.). The latter is supported by funding from Pfizer. The funders had no role in study design, data collection and analysis, decision to publish or preparation of the manuscript.
Author information
Authors and Affiliations
Contributions
E.M.J. and N.W. had full access to all the study data, and take responsibility for the integrity of the data and the accuracy of the data analyses. I.R.K., G.S.S., J.W.M.M., A.J. and S.M.W created the study concept and design. A.A.v.Z., A.v.d.P.-P., L.C.H., I.R.K., G.S.S. and A.J. were responsible for data acquisition. V.d.W., C.M.B., K.R.-R., M.N.V. and J.K. were responsible for administrative, technical or material support. E.M.J. and E.O.-d. H. carried out statistical analysis. The paper was drafted by E.M.J., N.W., J.W.M.M., A.J. and S.M.W and all authors were responsible for the critical revision of the paper.
Corresponding author
Ethics declarations
Competing interests
Institutional research grant support was received from the following: Novartis (I.R.K. and G.S.S.); Gilead (I.R.K.); Agendia and AstraZeneca (G.S.S. and A.J.); Merck (G.S.S. and J.W.M.M.); Roche and Seasgen (G.S.S.); Bayer, Menarini and Tzu Genomics (J.W.M.M.) and Pfizer (A.J. and S.M.W.). Consultancy was received from Biovica, Novaratis and Seagen (G.S.S.) and Novartis (J.W.M.M.). Participation on the advisory board of AstraZeneca (A.J.). The other authors declare no competing interests.
Peer review
Peer review information
Nature Medicine thanks Luc Cabel, Andrew Davis and the other, anonymous, reviewer(s) for their contribution to the peer review of this work. Primary Handling Editor: Ulrike Harjes, in collaboration with the Nature Medicine team.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data
Supplementary information
Supplementary Information (download PDF )
Supplementary Table 1.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Jongbloed, E.M., Wortelboer, N., de Weerd, V. et al. Early versus deferred use of CDK4/6 inhibitors in advanced breast cancer: circulating tumor DNA analysis of a randomized phase 3 trial. Nat Med 31, 3662–3667 (2025). https://doi.org/10.1038/s41591-025-03935-w
Received:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41591-025-03935-w