Abstract
Brain-wide communication supporting flexible behavior requires coordination between sensory and associative regions but how brain networks route sensory information at fast timescales to guide action remains unclear. Using human intracranial electrophysiology and spiking neural networks during spatial attention tasks, where participants detected targets at cued locations, we show that high-frequency activity bursts (HFAbs) mark temporal windows of elevated population firing that enable fast, long-range communications. HFAbs were evoked by sensory cues and targets, dynamically coupled to low-frequency rhythms. Notably, both the strength of cue-evoked HFAbs and their decoupling from slow rhythms predicted behavioral accuracy. HFAbs synchronized across the brain, revealing distinct cue- and target-activated subnetworks. These subnetworks exhibited lead–lag dynamics following target onset, with cue-activated subnetworks preceding target-activated subnetworks when cues were informative. Computational modeling suggested that HFAbs reflect transitions to population spiking, denoting temporal windows for network communications supporting attentional performance. These findings establish HFAbs as signatures of population state transitions, supporting information routing across distributed brain networks.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$32.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
Data availability
Preprocessed data used for generating these results are deposited at figshare at https://doi.org/10.6084/m9.figshare.30434683 (ref. 107). Source data are provided with this paper.
Code availability
Custom programming codes for analysis and modeling are available from https://github.com/banaiek/Attentional_Routing/.
References
Saalmann, Y. B., Pigarev, I. N. & Vidyasagar, T. R. Neural mechanisms of visual attention: how top-down feedback highlights relevant locations. Science 316, 1612–1615 (2007).
Gregoriou, G. G., Gotts, S. J., Zhou, H. & Desimone, R. High-frequency, long-range coupling between prefrontal and visual cortex during attention. Science 324, 1207–1210 (2009).
Szczepanski, S. M. et al. Dynamic changes in phase-amplitude coupling facilitate spatial attention control in fronto-parietal cortex. PLoS Biol. 12, e1001936 (2014).
Buschman, T. J. & Kastner, S. From behavior to neural dynamics: an integrated theory of attention. Neuron 88, 127–144 (2015).
Fiebelkorn, I. C. & Kastner, S. Functional specialization in the attention network. Annu. Rev. Psychol. 71, 221–249 (2020).
Banaie Boroujeni, K. & Womelsdorf, T. Routing states transition during oscillatory bursts and attentional selection. Neuron https://doi.org/10.1016/j.neuron.2023.06.012 (2023).
Womelsdorf, T. & Fries, P. Neuronal coherence during selective attentional processing and sensory–motor integration. J. Physiol. 100, 182–193 (2006).
Lakatos, P., Karmos, G., Mehta, A. D., Ulbert, I. & Schroeder, C. E. Entrainment of neuronal oscillations as a mechanism of attentional selection. Science 320, 110–113 (2008).
Siegel, M., Donner, T. H., Oostenveld, R., Fries, P. & Engel, A. K. Neuronal synchronization along the dorsal visual pathway reflects the focus of spatial attention. Neuron 60, 709–719 (2008).
Buschman, T. J. & Miller, E. K. Top-down versus bottom-up control of attention in the prefrontal and posterior parietal cortices. Science 315, 1860–1862 (2007).
Siegel, M., Buschman, T. J. & Miller, E. K. Cortical information flow during flexible sensorimotor decisions. Science 348, 1352–1355 (2015).
Canolty, R. T. et al. Oscillatory phase coupling coordinates anatomically dispersed functional cell assemblies. PNAS 107, 17356–17361 (2010).
Braun, U. et al. Dynamic reconfiguration of frontal brain networks during executive cognition in humans. Proc. Natl Acad. Sci. USA 112, 11678–11683 (2015).
Bassett, D. S. & Sporns, O. Network neuroscience. Nat. Neurosci. 20, 353–364 (2017).
Avena-Koenigsberger, A., Misic, B. & Sporns, O. Communication dynamics in complex brain networks. Nat. Rev. Neurosci. 19, 17–33 (2018).
Brookes, M. J. et al. Measuring functional connectivity using MEG: methodology and comparison with fcMRI. NeuroImage 56, 1082–1104 (2011).
Hari, R. & Parkkonen, L. The brain timewise: how timing shapes and supports brain function. Philos. Trans. R. Soc. B 370, 20140170 (2015).
Higley, M. J. & Cardin, J. A. Spatiotemporal dynamics in large-scale cortical networks. Curr. Opin. Neurobiol. 77, 102627 (2022).
Lachaux, J.-P., Axmacher, N., Mormann, F., Halgren, E. & Crone, N. E. High-frequency neural activity and human cognition: past, present and possible future of intracranial EEG research. Prog. Neurobiol. 98, 279–301 (2012).
Parvizi, J. & Kastner, S. Human intracranial EEG: promises and limitations. Nat. Neurosci. 21, 474–483 (2018).
Voytek, B. et al. Oscillatory dynamics coordinating human frontal networks in support of goal maintenance. Nat. Neurosci. 18, 1318–1324 (2015).
Haller, M. et al. Persistent neuronal activity in human prefrontal cortex links perception and action. Nat. Hum. Behav. 2, 80–91 (2018).
Helfrich, R. F. et al. Neural mechanisms of sustained attention are rhythmic. Neuron 99, 854–865.e5 (2018).
Martin, A. B. et al. Temporal dynamics and response modulation across the human visual system in a spatial attention task: an ECoG study. J. Neurosci. 39, 333–352 (2019).
Ray, S. & Maunsell, J. H. R. Different origins of γ rhythm and high-γ activity in macaque visual cortex. PLoS Biol. 9, e1000610 (2011).
Banaie Boroujeni, K., Tiesinga, P. & Womelsdorf, T. Adaptive spike-artifact removal from local field potentials uncovers prominent β and γ band neuronal synchronization. J. Neurosci. Methods 330, 108485 (2020).
Leszczyński, M. et al. Dissociation of broadband high-frequency activity and neuronal firing in the neocortex. Sci. Adv. 6, eabb0977 (2020).
Leonard, M. K. et al. Large-scale single-neuron speech sound encoding across the depth of human cortex. Nature 626, 593–602 (2024).
Zolnik, T. A. et al. Layer 6b controls brain state via apical dendrites and the higher-order thalamocortical system. Neuron 112, 805–820.e4 (2024).
Canolty, R. T. & Knight, R. T. The functional role of cross-frequency coupling. Trends Cogn. Sci. 14, 506–515 (2010).
Arnulfo, G. et al. Long-range phase synchronization of high-frequency oscillations in human cortex. Nat. Commun. 11, 5363 (2020).
Williams, N. et al. Modules in connectomes of phase-synchronization comprise anatomically contiguous, functionally related regions. NeuroImage 272, 120036 (2023).
Garrett, J. C. et al. Binding of cortical functional modules by synchronous high-frequency oscillations. Nat. Hum. Behav. https://doi.org/10.1038/s41562-024-01952-2 (2024).
Jensen, O. & Colgin, L. L. Cross-frequency coupling between neuronal oscillations. Trends Cogn. Sci. 11, 267–269 (2007).
Jiang, H., Bahramisharif, A., van Gerven, M. A. J. & Jensen, O. Measuring directionality between neuronal oscillations of different frequencies. NeuroImage 118, 359–367 (2015).
Kam, J. W. Y. et al. Default network and frontoparietal control network theta connectivity supports internal attention. Nat. Hum. Behav. 3, 1263–1270 (2019).
Johnson, E. L. et al. A rapid theta network mechanism for flexible information encoding. Nat. Commun. 14, 2872 (2023).
Tong, A. P. S., Vaz, A. P., Wittig, J. H., Inati, S. K. & Zaghloul, K. A. Ripples reflect a spectrum of synchronous spiking activity in human anterior temporal lobe. eLife 10, e68401 (2021).
Onslow, A. C. E., Jones, M. W. & Bogacz, R. A canonical circuit for generating phase-amplitude coupling. PLoS ONE 9, e102591 (2014).
Colgin, L. L. et al. Frequency of γ oscillations routes flow of information in the hippocampus. Nature 462, 353–357 (2009).
Akam, T. & Kullmann, D. M. Oscillations and filtering networks support flexible routing of information. Neuron 67, 308–320 (2010).
Palmigiano, A., Geisel, T., Wolf, F. & Battaglia, D. Flexible information routing by transient synchrony. Nat. Neurosci. 20, 1014–1022 (2017).
Lundqvist, M., Herman, P., Warden, M. R., Brincat, S. L. & Miller, E. K. γ and β bursts during working memory readout suggest roles in its volitional control. Nat. Commun. 9, 394 (2018).
Hahn, G., Ponce-Alvarez, A., Deco, G., Aertsen, A. & Kumar, A. Portraits of communication in neuronal networks. Nat. Rev. Neurosci. 20, 117–127 (2019).
Corbetta, M. & Shulman, G. L. Spatial neglect and attention networks. Annu. Rev. Neurosci. 34, 569–599 (2011).
Chica, A. B., Bartolomeo, P. & Lupiáñez, J. Two cognitive and neural systems for endogenous and exogenous spatial attention. Behav. Brain Res. 237, 107–123 (2013).
Katsuki, F. & Constantinidis, C. Bottom-up and top-down attention: different processes and overlapping neural systems. Neuroscientist 20, 509–521 (2014).
Vossel, S., Geng, J. J. & Fink, G. R. Dorsal and ventral attention systems: distinct neural circuits but collaborative roles. Neuroscientist 20, 150–159 (2014).
König, P., Engel, A. K. & Singer, W. Relation between oscillatory activity and long-range synchronization in cat visual cortex. Proc. Natl Acad. Sci. USA 92, 290–294 (1995).
Roelfsema, P. R., Engel, A. K., König, P. & Singer, W. Visuomotor integration is associated with zero time-lag synchronization among cortical areas. Nature 385, 157–161 (1997).
Uhlhaas, P. J. et al. Neural synchrony in cortical networks: history, concept and current status. Front. Integr. Neurosci. 3, 17 (2009).
Gollo, L. L., Mirasso, C. & Villa, A. E. P. Dynamic control for synchronization of separated cortical areas through thalamic relay. NeuroImage 52, 947–955 (2010).
Gollo, L. L., Mirasso, C. R., Atienza, M., Crespo-Garcia, M. & Cantero, J. L. Theta band zero-lag long-range cortical synchronization via hippocampal dynamical relaying. PLoS ONE 6, e17756 (2011).
Saalmann, Y. B. & Kastner, S. Cognitive and perceptual functions of the visual thalamus. Neuron 71, 209–223 (2011).
Logothetis, N. K. et al. Hippocampal–cortical interaction during periods of subcortical silence. Nature 491, 547–553 (2012).
Saalmann, Y. B., Pinsk, M. A., Wang, L., Li, X. & Kastner, S. The pulvinar regulates information transmission between cortical areas based on attention demands. Science https://doi.org/10.1126/science.1223082 (2012).
Vijayan, S. & Kopell, N. J. Thalamic model of awake α oscillations and implications for stimulus processing. Proc. Natl Acad. Sci. USA 109, 18553–18558 (2012).
Fiebelkorn, I. C., Pinsk, M. A. & Kastner, S. The mediodorsal pulvinar coordinates the macaque fronto-parietal network during rhythmic spatial attention. Nat. Commun. 10, 215 (2019).
Gollo, L. L., Mirasso, C., Sporns, O. & Breakspear, M. Mechanisms of zero-lag synchronization in cortical motifs. PLoS Comput. Biol. 10, e1003548 (2014).
Zolnik, T. A. et al. Layer 6b is driven by intracortical long-range projection neurons. Cell Rep. 30, 3492–3505.e5 (2020).
Menara, T., Qin, Y., Bassett, D. S. & Pasqualetti, F. Relay interactions enable remote synchronization in networks of phase oscillators. IEEE Control Syst. Lett. 6, 500–505 (2022).
Smith, S. M. et al. Correspondence of the brain’s functional architecture during activation and rest. Proc. Natl Acad. Sci. USA 106, 13040–13045 (2009).
Srivastava, P. et al. Models of communication and control for brain networks: distinctions, convergence, and future outlook. Netw. Neurosci. 4, 1122–1159 (2020).
Engel, T. A., Schölvinck, M. L. & Lewis, C. M. The diversity and specificity of functional connectivity across spatial and temporal scales. NeuroImage 245, 118692 (2021).
Battiston, F. et al. Networks beyond pairwise interactions: structure and dynamics. Phys. Rep. 874, 1–92 (2020).
Jensen, O., Gips, B., Bergmann, T. O. & Bonnefond, M. Temporal coding organized by coupled α and γ oscillations prioritize visual processing. Trends Neurosci. 37, 357–369 (2014).
Chapeton, J. I., Haque, R., Wittig, J. H., Inati, S. K. & Zaghloul, K. A. Large-scale communication in the human brain is rhythmically modulated through α coherence. Curr. Biol. 29, 2801–2811.e5 (2019).
Harris, K. D. & Thiele, A. Cortical state and attention. Nat. Rev. Neurosci. 12, 509–523 (2011).
Hanslmayr, S., Staudigl, T. & Fellner, M.-C. Oscillatory power decreases and long-term memory: the information via desynchronization hypothesis. Front. Hum. Neurosci. 6, (2012).
Hanslmayr, S., Staresina, B. P. & Bowman, H. Oscillations and episodic memory: addressing the synchronization/desynchronization conundrum. Trends Neurosci. 39, 16–25 (2016).
Beaman, C. B., Eagleman, S. L. & Dragoi, V. Sensory coding accuracy and perceptual performance are improved during the desynchronized cortical state. Nat. Commun. 8, 1308 (2017).
Dickey, C. W. et al. Widespread ripples synchronize human cortical activity during sleep, waking, and memory recall. Proc. Natl Acad. Sci. USA 119, e2107797119 (2022).
Verzhbinsky, I. A. et al. Co-occurring ripple oscillations facilitate neuronal interactions between cortical locations in humans. Proc. Natl Acad. Sci. USA 121, e2312204121 (2024).
Egly, R., Driver, J. & Rafal, R. D. Shifting visual attention between objects and locations: evidence from normal and parietal lesion subjects. J. Exp. Psychol.: Gen. 123, 161–177 (1994).
Fiebelkorn, I. C., Saalmann, Y. B. & Kastner, S. Rhythmic sampling within and between objects despite sustained attention at a cued location. Curr. Biol. 23, 2553–2558 (2013).
Wang, L., Mruczek, R. E. B., Arcaro, M. J. & Kastner, S. Probabilistic maps of visual topography in human cortex. Cereb. Cortex 25, 3911–3931 (2015).
Lancaster, J. L. et al. Automated labeling of the human brain: a preliminary report on the development and evaluation of a forward-transform method. Hum. Brain Mapp. 5, 238–242 (1997).
Oostenveld, R., Fries, P., Maris, E. & Schoffelen, J.-M. FieldTrip: open source software for advanced analysis of MEG, EEG, and invasive electrophysiological data. Comput. Intell. Neurosci. 2011, 156869 (2011).
Boashash, B. Estimating and interpreting the instantaneous frequency of a signal. I: fundamentals. Proc. IEEE 80: 520–538 (1992).
Hill, M. R. H., Fried, I. & Koch, C. Quantification and classification of neuronal responses in kernel-smoothed peristimulus time histograms. J. Neurophysiol. 113, 1260–1274 (2015).
Dunn, O. J. Multiple comparisons among means. J. Am. Stat. Assoc. 56, 52–64 (1961).
Benjamini, Y. & Yekutieli, D. False discovery rate–adjusted multiple confidence intervals for selected parameters. J. Am. Stat. Assoc. 100, 71–81 (2005).
Vinck, M., van Wingerden, M., Womelsdorf, T., Fries, P. & Pennartz, C. M. A. The pairwise phase consistency: a bias-free measure of rhythmic neuronal synchronization. NeuroImage 51, 112–122 (2010).
Milligan, G. W. & Cooper, M. C. An examination of procedures for determining the number of clusters in a data set. Psychometrika 50, 159–179 (1985).
Jain, A. K. & Dubes, R. C. Algorithms for Clustering Data (Prentice-Hall, 1988).
Halkidi, M., Batistakis, Y. & Vazirgiannis, M. On clustering validation techniques. J. Intell. Inf. Syst. 17, 107–145 (2001).
Vendramin, L., Campello, R. J. G. B. & Hruschka, E. R. Relative clustering validity criteria: a comparative overview. Stat. Anal. Data Min. 3, 209–235 (2010).
Freedman, D. & Diaconis, P. On the histogram as a density estimator: L2 theory. Z. Wahrscheinlichkeitstheorie verw Geb. 57, 453–476 (1981).
Billeh, Y. N. et al. Systematic integration of structural and functional data into multi-scale models of mouse primary visual cortex. Neuron 106, 388–403.e18 (2020).
Potjans, T. C. & Diesmann, M. The Cell-Type Specific Cortical Microcircuit: Relating Structure and Activity in a Full-Scale Spiking Network Model. Cereb. Cortex 24, 785–806 (2014).
Barral, J. & D. Reyes, A. Synaptic scaling rule preserves excitatory–inhibitory balance and salient neuronal network dynamics. Nat. Neurosci. 19, 1690–1696 (2016).
Izhikevich, E. M. Which model to use for cortical spiking neurons? IEEE Trans. Neural Netw. 15, 1063–1070 (2004).
Izhikevich, E. M. Simple model of spiking neurons. IEEE Trans. Neural Netw. 14, 1569–1572 (2003).
Pfeffer, C. K., Xue, M., He, M., Huang, Z. J. & Scanziani, M. Inhibition of inhibition in visual cortex: the logic of connections between molecularly distinct interneurons. Nat. Neurosci. 16, 1068–1076 (2013).
Karnani, M. M. et al. Cooperative subnetworks of molecularly similar interneurons in mouse neocortex. Neuron 90, 86–100 (2016).
Medalla, M., Gilman, J. P., Wang, J.-Y. & Luebke, J. I. Strength and diversity of inhibitory signaling differentiates primate anterior cingulate from lateral prefrontal cortex. J. Neurosci. 37, 4717–4734 (2017).
Gilman, J. P., Medalla, M. & Luebke, J. I. Area-specific features of pyramidal neurons—a comparative study in mouse and rhesus monkey. Cereb. Cortex 27, 2078–2094 (2017).
Medalla, M. & Luebke, J. I. Diversity of glutamatergic synaptic strength in lateral prefrontal versus primary visual cortices in the rhesus monkey. J. Neurosci. 35, 112–127 (2015).
Amatrudo, J. M. et al. Influence of highly distinctive structural properties on the excitability of pyramidal neurons in monkey visual and prefrontal cortices. J. Neurosci. 32, 13644–13660 (2012).
Roxin, A., Brunel, N., Hansel, D., Mongillo, G. & van Vreeswijk, C. On the distribution of firing rates in networks of cortical neurons. J. Neurosci. 31, 16217–16226 (2011).
Milstein, J., Mormann, F., Fried, I. & Koch, C. Neuronal shot noise and Brownian 1/f2 behavior in the local field potential. PLoS ONE 4, e4338 (2009).
Bédard, C., Kröger, H. & Destexhe, A. Modeling extracellular field potentials and the frequency-filtering properties of extracellular space. Biophys. J. 86, 1829–1842 (2004).
Deco, G. & Rolls, E. T. Neurodynamics of biased competition and cooperation for attention: a model with spiking neurons. J. Neurophysiol. 94, 295–313 (2005).
Markov, N. T. et al. A weighted and directed interareal connectivity matrix for macaque cerebral cortex. Cereb. Cortex 24, 17–36 (2014).
Zhou, H., Schafer, R. J. & Desimone, R. Pulvinar-cortex interactions in vision and attention. Neuron 89, 209–220 (2016).
Buffalo, E. A., Fries, P., Landman, R., Liang, H. & Desimone, R. A backward progression of attentional effects in the ventral stream. Proc. Natl Acad. Sci. USA 107, 361–365 (2010).
Banaie Boroujeni, K. et al. Preprocessed burst-triggered HFA datasets for individual subjects. figshare https://doi.org/10.6084/m9.figshare.30434683 (2025).
Acknowledgements
This work was supported by the C.V. Starr Fellowship (K.B.B.), German Research Foundation, Emmy Noether Program (DFG HE8329/2-1; R.F.H.), National Institute of Biomedical Imaging and Bioengineering (P41-EB018783; P.B.), National Institute of Neurological Disorders and Stroke (R01NS021135; R.T.K.), National Eye Institute (2R01EY017699; S.K.), National Institutes of Health (1R01MH137624; SK) and National Institute of Mental Health (2R01MH064043, P50MH132642; S.K.). The funders had no role in study design, data collection and analysis, decision to publish or preparation of the manuscript.
Author information
Authors and Affiliations
Contributions
Conceptualization, K.B.B. and S.K.; Methodology, K.B.B.; Formal Analysis, K.B.B.; Modeling, K.B.B.; Investigation, R.F.H., R.T.K. and J.J.L.; Software, R.F.H. and I.C.F.; Visualization, K.B.B; Writing—Original Draft, K.B.B.; Writing—Review and Editing, K.B.B., R.F.H., I.C.F., J.N.B., P.B., J.J.L., R.T.K. and S.K.; and Supervision, S.K.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Neuroscience thanks the anonymous reviewers for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data
Extended Data Fig. 1 Behavioral performance and HFAb activation patterns across experiments.
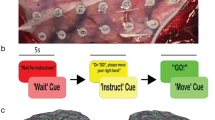
a, Task structure of experiment 2. Subjects hold their gaze fixation to the center of a screen (a white plus sign) with red circles turning on and off. A spatial cue endogenously cues subject’s attention to a hemifield. A target appears at one hemifield and subjects should report whether the target was seen in the cued hemifield. The brain shows the localization of electrodes across all subjects. b, Reaction times (top) and accuracy (bottom) for individual subjects (n = 7 in experiment 1, and n = 5 in experiment 2) across valid, invalid, and catch trials (trials with no targets in experiment 1). Box plots show median (center line), interquartile range (box bounds = 25th–75th percentile), and whiskers extending to non-outlier minima and maxima (values within 1.5×IQR). c, Population-averaged HFAb density around cue (top) and target (bottom) onsets, similar to Fig. 1b in experiment 2 (mean ± SEM). d, HFAb responses grouped by trial outcomes (correct hit, correct reject, miss, false alarm) for cue (top) and target (bottom) epochs [median ± SEM], (n = 5). Asterisks indicate significant differences (Kruskal–Wallis test p = 0.003 for cue, and p < 0.001 for target response; followed by Dunn’s test, p < 0.05). e, Group topography of cue (left) and target (right) response for incorrect trials. f, Individual subject topographies for correct and incorrect trials, similar to Fig. 1h. g,h, HFAb responses comparing valid/invalid cues and contra/ipsi laterality conditions for experiments 1 (g, n = 6, GLME, n.s.) and 2 (h, n = 5), similar to Fig. 1i,j (median ± SEM). Horizontal lines indicate a significant main effect of validity on target (GLME, t = 5.03 p < 0.001), and laterality on target (GLME, t = 2.23, p = 0.025). i, Classifier accuracy predicting trial outcomes for cue-responsive (Cue + ) and cue-unresponsive (Cue-) in experiment 2 (error bars: SEM across realizations and cross-validations). Red line indicates significant prediction above baseline and chance (two-sided binomial test, p < 0.05, FDR-corrected for multiple dependent comparisons).
Extended Data Fig. 2 HFAb phase-locking to low-frequency rhythms across subjects.
a, Individual examples showing HFAb phase distributions at the peak (left), phase-locking value (PLV, middle), and HFAb-triggered spectrum (right) from different subjects. Peak frequencies noted for each example in red and blue circles. b, Phase histograms of HFAbs at theta/alpha (8 Hz, purple) and beta (20 Hz, green) frequencies for all subjects. Orange and red lines indicate circular means. c, Distribution of peak phases across all electrodes. d, Distribution of phase-locking peak widths (mean: 7.3 ± 0.3 Hz). e, Relationship between peak frequency and power ratio (HFAb-triggered/baseline), colored by phase value. f, Distribution of power enhancement ratios at phase-locking frequencies (median: 2.1-fold increase), dashed line shows 1.
Extended Data Fig. 3 HFAb synchronization with low-frequency activity in experiment 1 and 2.
a, Group-average spatial pattern of observed frequency peak of HFAb phase-locking to LFP. b, Individual examples showing the spatial pattern of the observed frequency peaks of the HFAb phase-locking to LFP. c, Corresponding to Fig. 2e, showing the proportion of time-frequency points where phase-locking was significantly lower than baseline (p < 0.05, two-sided random permutation test). d, Similar to Fig. 2e, but after removing event-related potential from the LFP (see Methods). e, Group-average heatmaps of pairwise phase consistency (PPC) as a measure for HFAb synchronization to LFPs, Similar to Fig. 2e. f, Examples of the coupling ratio between HFAb and low-frequency (4–25 Hz) LFP following cue and target onsets in correct and incorrect trials. g, Similar to Fig. 2e for experiment 2. h,i, Regression plots showing correlation of coupling ratios following cue onset with target responses (green, h), and coupling ratios following target onset with cue responses (purple, i). Scatter points denote electrodes, lines indicate individual subjects with orange line showing the regression across all subjects.
Extended Data Fig. 4 A network clustering approach based on the synchronization of HFAbs between electrodes.
a, The average HFAb-triggered HFA for individuals in experiment 2. b, The normalized PSD for HFAb-triggered HFA in experiment 2. c, HFAb-triggered HFA for electrodes distanced in 4 different quantiles (25,50, 75, 100 mm), ranging from green (short) to red (long) in experiment 2, similar to Fig. 3c. d, The first and second principal components of HFAb-triggered HFA for individual subjects in experiment 2. e, A schematic demonstration of network clustering algorithm. We used HFAbs outside of cue/delay and target/response periods. The network synchrony matrix shows the loading values for each electrode pair on the synchronized component. A K-means clustering was performed on randomly selected electrode samples for different cluster numbers (K = 2 to 8). We calculated a pairwise grouping probability matrix in which each element indicates how likely it is that two electrodes will be grouped together. The next step was clustering with network subsampling, similar to the previous step but based on the pairwise grouping likelihood matrix. The final clustering of the pairwise grouping likelihood results indicated stable clusters for each K (see Methods).
Extended Data Fig. 5 Organization of clusters based on cluster numbers.
Columns from left to right show the results for cluster numbers K = 2–8. The cluster IDs are sorted by cluster stability.
Extended Data Fig. 6 Clustering results for individual subjects.
a, Color-coded loading values on the synchronized PC (left) and the pairwise grouping probability (right) for each subject. b, Electrode distributions for optimal cluster numbers. c, Cluster number selection using four metrics with nonparametric voting rank (black). The accuracy is determined by the median diagonals, the confusion by the median nondiagonal, selectivity by the relative rank of the diagonal over the highest nondiagonal rank, and stability by the relative rank of the diagonal over the nondiagonal rank. d, HFAb density around cue and target onsets for each cluster, similar to Fig. 3f, across subjects. Shaded error bars indicate the standard error of the means, thicker lines indicate significant functional subnetworks. e, Leave-one-subject-out classifier analysis, similar to Fig. 3h. f, g, Classifier accuracy predicting correct trials (green), errors (brown), and overall accuracy (gray) for (f) experiment 1 and (g) experiment 2. h, outcome prediction by cue- and target-subnetworks following target onset. Shaded error bars show standard error of the mean. Thick lines in panels e-h indicate time points above baseline and chance (two-sided binomial test, p < 0.05, FDR-corrected for multiple dependent comparisons).
Extended Data Fig. 7 Temporal precession between cue and target-subnetworks.
a, HFAb-triggered HFA examples for individual subjects (mean ± SEM), similar to Fig. 4a. b, Top: Group-level average of HFAb-triggered HFA in experiment 2 (n = 3), similar to Fig. 4b. Shaded regions indicate significant lead-lag patterns (two-sided permutation test, p < 0.05; red: cue leads, [135-244] ms, blue: target leads, [-298 to -155] ms). Bottom: Peak time-lag distributions in experiment 2 (n = 3, medina ± SEM: 14.9 ± 4.7 ms, two-sided Wilcoxon rank-sum test between directions; *: p < 0.05, **: p < 0.01, ***: p < 0.001). c, HFAb-triggered HFA during cue/delay period, Similar to Fig. 4a. d, Lead-lag pattern topography for individual subjects, similar to Fig. 4c. e, DMI heatmaps for individual subjects, similar to Fig. 4d. f, DMI analysis around cue onset, similar to Fig. 4e. g, Group-average DMI in experiment 2 (n = 3), similar to Fig. 4d. DMI peaked 297.7 ± 19.6 ms after target onset with 125.6 ± 26.2 ms time-lag (n = 3). h, DMI peak distributions for invalid/valid trials in experiment 2 (n = 3), similar to Fig. 4e.
Extended Data Fig. 8 Computational modeling of iEEG signal.
a, Connection probability between neurons decreases with distance. b, Frequency-dependent attenuation factor for iEEG signals at the recording disks. c, A complete trial simulation example. Raster shows the activity of neurons in one network (bottom, blue and red show excitatory and inhibitory neurons, respectively). Raw and attenuated traces correspond to field dynamics of the same network. d, Network activity with coherent (top) and random (bottom) input phases. e, Connectivity matrix for feedforward network (N1 → N2, each scatter point indicates whether two neurons have excitatory (blue) or inhibitory (red) connections). f, An input design evaluating how a transient stimulus affects HFAb coherence with LFP in a network as shown in E (network 1 (top) receives an impulse input). g, PLV changes in both networks following transient input, similar to Fig. 5g. h In both networks 1 and 2, the PLV drops within 500 ms of stimulus onset (p < 0.001, two-sided Wilcoxon test). I, Normalized HFAb rate relative to stimulus onset. j, Four-network architecture and example raster activity during spatial attention task simulation. k, HFAb rate distributions in target networks for valid versus invalid trials. l, Normalized DMI between cue and target networks following cue onset, similar to Fig. 5l.
Supplementary information
Source data
Source Data Fig. 1 (download ZIP )
Zip file containing numerical csv files for the plots.
Source Data Fig. 2 (download ZIP )
Zip file containing numerical csv files for the plots.
Source Data Fig. 3 (download ZIP )
Zip file containing numerical csv files for the plots.
Source Data Fig. 4 (download ZIP )
Zip file containing numerical csv files for the plots.
Source Data Fig. 5 (download ZIP )
Zip file containing numerical csv files for the plots.
Source Data Extended Data Fig. 1 (download ZIP )
Zip file containing numerical csv files for the plots.
Source Data Extended Data Fig. 3 (download ZIP )
Zip file containing numerical csv files for the plots.
Source Data Extended Data Fig. 4 (download ZIP )
Zip file containing numerical csv files for the plots.
Source Data Extended Data Fig. 5 (download CSV )
csv file containing numerical values for the plots.
Source Data Extended Data Fig. 6 (download ZIP )
Zip file containing numerical csv files for the plots.
Source Data Extended Data Fig. 7 (download ZIP )
Zip file containing numerical csv files for the plots.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Banaie Boroujeni, K., Helfrich, R.F., Fiebelkorn, I.C. et al. High-frequency bursts facilitate fast communication for human spatial attention. Nat Neurosci 29, 435–444 (2026). https://doi.org/10.1038/s41593-025-02160-5
Received:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41593-025-02160-5