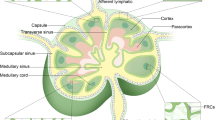
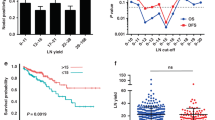
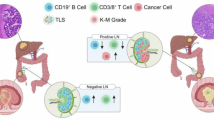
Abstract
Lymph node (LN) metastasis is a conserved feature across most solid organ malignancies and portends worse prognoses. Functionally, LN metastases induce systemic tumor-specific immune tolerance and may serve as a reservoir for distant metastases. Nonetheless, there are relatively few preclinical models for interrogating the biology of LN metastasis and its systemic effects at various stages of metastatic progression. We describe a method for modeling LN metastasis of melanoma tumors in mice that enables assessment of tumor and immune cell phenotypes and the functional roles of nodal involvement on distant metastasis. Our model comprises a family of transplantable syngeneic melanoma tumor cell lines evolved to exhibit enhanced LN metastatic potential, which can be used to probe cancer–immune interactions and test new therapeutics. We present both (i) a spontaneous LN metastasis model involving primary tumor implantation and assessment of LN colonization 21–28 d later and (ii) an experimental metastasis model involving implantation of primary tumors followed by direct intra-LN injections of tumor cells. Both models can be extended to assess the impact of LN metastasis on the development of distant metastases through asynchronous intravenous injections of tumors. Finally, we discuss experimental design considerations including when to use spontaneous or experimental models and troubleshooting consistent LN metastasis, making this model accessible for researchers with basic mouse survival-surgery skills. We highlight how LN metastasis models can be used to profile metastatic immune reprogramming, measure the impact of nodal metastases on distant metastases and assess novel anti-metastatic therapeutics.
Key points
-
The procedure covers two models of LN metastasis. The spontaneous model recapitulates the metastatic cascade and can be used for mechanistic studies of the metastatic processes via the subcutaneous implantation of LN metastatic cells in mice.
-
The experimental model of LN metastasis is instead preferred for studying systemic alterations in the host during the metastatic process via the administration of a subcutaneous parental non-metastatic tumor followed by intra-LN implantation of metastatic cells.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$32.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
Data availability
Cell line bulk RNA-seq data have been deposited at the Gene Expression Omnibus with accession code GSE117529.
References
Gerstberger, S., Jiang, Q. & Ganesh, K. Metastasis. Cell 186, 1564–1579 (2023).
Amin, M. B. et al. The Eighth Edition AJCC Cancer Staging Manual: continuing to build a bridge from a population‐based to a more “personalized” approach to cancer staging. CA Cancer J. Clin. 67, 93–99 (2017).
Leong, S. P. L. et al. Impact of nodal status and tumor burden in sentinel lymph nodes on the clinical outcomes of cancer patients. J. Surg. Oncol. 103, 518–530 (2011).
von Andrian, U. H. & Mempel, T. R. Homing and cellular traffic in lymph nodes. Nat. Rev. Immunol. 3, 867–878 (2003).
Qi, H., Kastenmüller, W. & Germain, R. N. Spatiotemporal basis of innate and adaptive immunity in secondary lymphoid tissue. Annu. Rev. Cell Dev. Biol. 30, 1–27 (2014).
Spitzer, M. H. et al. Systemic immunity is required for effective cancer immunotherapy. Cell 168, 487–502.e15 (2017).
Prokhnevska, N. et al. CD8+ T cell activation in cancer comprises an initial activation phase in lymph nodes followed by effector differentiation within the tumor. Immunity 56, 107–124.e5 (2023).
Connolly, K. A. et al. A reservoir of stem-like CD8+ T cells in the tumor-draining lymph node preserves the ongoing anti-tumor immune response. Sci. Immunol. 6, eabg7836 (2021).
Fransen, M. F. et al. Tumor-draining lymph nodes are pivotal in PD-1/PD-L1 checkpoint therapy. JCI Insight 3, e124507 (2018).
Dammeijer, F. et al. The PD-1/PD-L1-checkpoint restrains T cell immunity in tumor-draining lymph nodes. Cancer Cell 38, 685–700.e8 (2020).
Huang, Q. et al. The primordial differentiation of tumor-specific memory CD8+ T cells as bona fide responders to PD-1/PD-L1 blockade in draining lymph nodes. Cell 185, 4049–4066.e25 (2022).
Francia, G., Cruz-Munoz, W., Man, S., Xu, P. & Kerbel, R. S. Mouse models of advanced spontaneous metastasis for experimental therapeutics. Nat. Rev. Cancer 11, 135–141 (2011).
Hebert, J. D., Neal, J. W. & Winslow, M. M. Dissecting metastasis using preclinical models and methods. Nat. Rev. Cancer 23, 391–407 (2023).
Speak, A. O. et al. A high-throughput in vivo screening method in the mouse for identifying regulators of metastatic colonization. Nat. Protoc. 12, 2465–2477 (2017).
Gupta, G. P. & Massagué, J. Cancer metastasis: building a framework. Cell 127, 679–695 (2006).
Pereira, E. R. et al. Lymph node metastases can invade local blood vessels, exit the node, and colonize distant organs in mice. Science 359, 1403–1407 (2018).
Aslakson, C. J. & Miller, F. R. Selective events in the metastatic process defined by analysis of the sequential dissemination of subpopulations of a mouse mammary tumor. Cancer Res. 52, 1399–1405 (1992).
Green, E. L. (ed.) Handbook of Genetically Standardized JAX Mice (The Jackson Laboratory, 1962).
Bertalanffy, F. D. & McAskill, C. Rate of cell division of malignant mouse melanoma B16. J. Natl Cancer Inst. 32, 535–545 (1964).
Fidler, I. J. Selection of successive tumour lines for metastasis. Nat. N. Biol. 242, 148–149 (1973).
Fidler, I. J. Biological behavior of malignant melanoma cells correlated to their survival in vivo. Cancer Res. 35, 218–224 (1975).
Gengenbacher, N., Singhal, M. & Augustin, H. G. Preclinical mouse solid tumour models: status quo, challenges and perspectives. Nat. Rev. Cancer 17, 751–765 (2017).
Westcott, P. M. K. et al. The mutational landscapes of genetic and chemical models of Kras-driven lung cancer. Nature 517, 489–492 (2015).
McFadden, D. G. et al. Mutational landscape of EGFR-, MYC-, and Kras-driven genetically engineered mouse models of lung adenocarcinoma. Proc. Natl Acad. Sci. USA 113, E6409–E6417 (2016).
Reticker-Flynn, N. E. et al. Lymph node colonization induces tumor-immune tolerance to promote distant metastasis. Cell 185, 1924–1942.e23 (2022).
Zhang, W. et al. Identification of cell types in multiplexed in situ images by combining protein expression and spatial information using CELESTA. Nat. Methods 19, 759–769 (2022).
Valastyan, S. & Weinberg, R. A. Tumor metastasis: molecular insights and evolving paradigms. Cell 147, 275–292 (2011).
Clark, E. A., Golub, T. R., Lander, E. S. & Hynes, R. O. Genomic analysis of metastasis reveals an essential role for RhoC. Nature 406, 532–535 (2000).
Kang, Y. et al. A multigenic program mediating breast cancer metastasis to bone. Cancer Cell 3, 537–549 (2003).
Bos, P. D. et al. Genes that mediate breast cancer metastasis to the brain. Nature 459, 1005–1009 (2009).
Minn, A. J. et al. Genes that mediate breast cancer metastasis to lung. Nature 436, 518–524 (2005).
Nguyen, D. X., Bos, P. D. & Massagué, J. Metastasis: from dissemination to organ-specific colonization. Nat. Rev. Cancer 9, 274–284 (2009).
Tamada, Y., Aoki, D., Nozawa, S. & Irimura, T. Model for paraaortic lymph node metastasis produced by orthotopic implantation of ovarian carcinoma cells in athymic nude mice. Eur. J. Cancer 40, 158–163 (2004).
Takahashi, K. et al. Development of a mouse model for lymph node metastasis with endometrial cancer. Cancer Sci. 102, 2272–2277 (2011).
Lund, A. W. et al. VEGF-C promotes immune tolerance in B16 melanomas and cross-presentation of tumor antigen by lymph node lymphatics. Cell Rep. 1, 191–199 (2012).
Carr, I. & Carr, J. in Tumor Invasion and Metastasis. Developments in Oncology Vol. 7 (eds. Liotta, L. A. & Hart, I. R.) 189–205 (Springer, 1982).
Li, L., Mori, S., Sakamoto, M., Takahashi, S. & Kodama, T. Mouse model of lymph node metastasis via afferent lymphatic vessels for development of imaging modalities. PLoS ONE 8, e55797 (2013).
Pulaski, B. A. & Ostrand‐Rosenberg, S. Mouse 4T1 breast tumor model. Curr. Protoc. Immunol. 39, 20.2.1–20.2.16 (2000).
Banan, B. et al. Development of a novel murine model of lymphatic metastasis. Clin. Exp. Metastasis 37, 247–255 (2020).
Zawieja, D. C. et al. Lymphatic cannulation for lymph sampling and molecular delivery. J. Immunol. 203, 2339–2350 (2019).
Brown, M. et al. Lymph node blood vessels provide exit routes for metastatic tumor cell dissemination in mice. Science 359, 1408–1411 (2018).
Ubellacker, J. M. et al. Lymph protects metastasizing melanoma cells from ferroptosis. Nature 585, 113–118 (2020).
Smith, A. J., Clutton, R. E., Lilley, E., Hansen, K. E. A. & Brattelid, T. PREPARE: guidelines for planning animal research and testing. Lab. Anim. 52, 135–141 (2017).
Vleeschauwer, S. I. D. et al. OBSERVE: guidelines for the refinement of rodent cancer models. Nat. Protoc. 19, 2571–2596 (2024).
Sert, N. Pdu et al. The ARRIVE guidelines 2.0: updated guidelines for reporting animal research. PLoS Biol. 18, e3000410 (2020).
Liu, S., Shen, M., Le, K., Hartono, A. B. & Stoyanova, T. Protocol for establishing spontaneous metastasis in mice using a subcutaneous tumor model. STAR Protoc. 5, 103239 (2024).
Duyverman, A. M. M. J., Steller, E. J. A., Fukumura, D., Jain, R. K. & Duda, D. G. Studying primary tumor–associated fibroblast involvement in cancer metastasis in mice. Nat. Protoc. 7, 756–762 (2012).
McLane, L. M., Abdel-Hakeem, M. S. & Wherry, E. J. CD8 T cell exhaustion during chronic viral infection and cancer. Annu. Rev. Immunol. 37, 1–39 (2015).
Acknowledgements
This work was supported by NIH grant DP2 AI177915 (to N.E.R.-F.), ARPA-H contract 1AY1AX000003 (to N.E.R.-F.). K.C.G. is an Investigator of the Howard Hughes Medical Institute. C.B.B. was supported by an Arc Institute Fellowship (Breuer). G.E.R. was supported by an NSF Graduate Research Fellowship and a Stanford Graduate Fellowship in Science & Engineering. Cell sorting/flow cytometry analysis for this project was performed on instruments in the Stanford Shared FACS Facility.
Author information
Authors and Affiliations
Contributions
C.B.B. and N.E.R.-F. conceived the study and wrote the manuscript. All authors reviewed and edited the manuscript. N.E.R.-F. derived the cell lines and established the protocol. C.B.B. refined the protocol and performed all experiments for anticipated results. Z.X. and A.W. assisted with flow cytometry experiments. G.E.R. and G.C.A. assisted with therapeutic testing of anticipated results. K.C.G. and N.E.R.-F. supervised the study.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Protocols thanks the anonymous reviewers for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Key reference
Reticker-Flynn, N. E. et al. Cell 185, 1924–1942.e23 (2022): https://doi.org/10.1016/j.cell.2022.04.019
Supplementary information
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Breuer, C.B., Xiong, Z., Wang, A. et al. Spontaneous and experimental models of lymph node metastasis. Nat Protoc 20, 3170–3187 (2025). https://doi.org/10.1038/s41596-025-01200-5
Received:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41596-025-01200-5
This article is cited by
-
Lymph node environment drives FSP1 targetability in metastasizing melanoma
Nature (2026)
-
Modeling lymph node metastases in vivo
Nature Protocols (2025)