Abstract
Synthetic fertilizer is a potential source of uranium to natural waters, yet evidence is lacking. We analyzed dissolved uranium concentrations in lakes, reservoirs, and rivers in Ohio, USA during the summer of 2017. All water bodies drain areas of extensive agriculture where phosphate-rich fertilizer is applied. Uranium concentrations ranged from 0.3 to 3.9 µg L−1, with the lowest concentrations observed in the most offshore Lake Erie samples. These results, especially when placed in the context of previous work on both surface and groundwater, suggest that dissolved uranium concentrations in this water emanating from agricultural lands are higher than background, and uranium should be categorized similarly to nitrate and phosphate in that it originates in part from fertilizer application.
Similar content being viewed by others
Introduction
The impact of modern agricultural practices on water quality is well documented. The addition of N and P-rich fertilizer can increase the flux of these macronutrients into aquatic systems causing eutrophication and hypoxia1. The oxidation of ammonia rich fertilizer can produce acid, which ultimately increases chemical weathering in the soils2,3 and enhances the flux of Ca, Mg, and HCO3 into streams and rivers due to carbonate amendment4,5. Long term fertilizer and lime amendments increase P, Ca, Mg, and K to agricultural soils over decades6.
Another impact of the addition of P-rich fertilizer on agricultural lands is the introduction of various trace elements associated with the original phosphoritic materials that are subsequently transferred to final commercial products during processing. The total U concentrations in P-rich fertilizers derived from phosphorites are particularly high, ranging 10–60 µg g−1 7. The U concentrations in the water soluble fraction of synthetic fertilizers vary greatly, from <0.05 μg g−1 to 7.68 μg g−1 with NPK fertilizers having the highest concentrations7. Long-term studies have documented U increases in agricultural soils due to the addition of P-rich fertilizers8, including increases of 4.4 to 13.7% over a 60 year period in Germany9. An estimated ~14,000 tons of U have been added to agricultural lands globally since 1951, corresponding to an average increase of ~1 kg U per hectare9. The fate of this anthropogenically introduced U is important – does the majority remain associated with soil, or is a fraction solubilized through time and introduced to aquatic systems? This is a significant question for two reasons: (1) the biochemical toxicity of U has been estimated to be six orders of magnitude higher than its radiological toxicity, and it has been implicated in potential chronic disease, such as kidney malfunction10; and (2) under oxic conditions, U(VI) is readily soluble and has the potential to be transported from soils to surface or groundwater.
Limestone dissolution has been identified a primary control on U concentrations in rivers, and black shales and other U-bearing lithologic units can be additional important sources11. In its U(VI) oxidation state, U forms strong complexes with carbonates and other oxygenated ligands to form highly soluble species over a wide pH range12. The agricultural processes of carbonate amendment and nitrate addition can work together to increase U mobility through direct increases in alkalinity, abiotic nitrate oxidation of U(IV) in minerals to U(VI), and a variety of biotic oxidation reactions that both increase alkalinity and oxidize U(IV)13,14,15. The concentration of U in surface waters in agricultural watersheds represents a mixture of natural (geogenic) sources resulting from water-rock interaction, anthropogenic agriculture-related factors that increase U solubility, and possibly direct contaminants from fertilizer impurities.
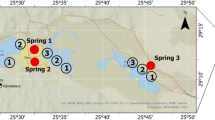
We present dissolved U concentrations from surface waters in central and northwestern Ohio, USA, that drain agricultural landscapes during a late spring–summer temporal cycle. We include data from two rivers draining into Lake Erie, western Lake Erie waters, two reservoirs that have recently suffered from harmful algal blooms promoted by excess nutrient input, and three reservoirs that provide potable water to Columbus, Ohio, which also drain primarily agricultural lands (Fig. 1). All these waters, except those from Lake Erie, are in the Eastern Corn Belt Eco region and sit upon glaciated till plains with primarily row crop land use (i.e., corn and soya) agriculture.
Water bodies and sampling locations in Ohio, United States.
Materials and Methods
Water samples were collected by hand in summer, 2017 using ultraclean techniques for low-level sampling of trace elements. Samples for uranium analysis were filtered within 12 hours of collection using pre-cleaned plastic syringes and 0.45 μm Whatman polypropylene syringe filters pre-cleaned with 10% HNO3 and rinsed multiple times with DI water. Aliquots for major ions and major nutrients were collected similarly, but filtered using plastic filter towers through 0.4 μm Nuclepore filters. Major cations were analyzed using a Dionex DX-120 ion chromatograph (IC), and major anions were analyzed using a Dionex ICS-2100 IC16. Bicarbonate concentrations were calculated by the difference between total cation and anion concentrations in equivalents. Uranium measurements were made using a ThermoFinnigan Element 2 ICP-SF-MS, with an internal Indium standard (10 µg L−1). Measurement accuracy was checked against the TMDA 64.2 standard (Trace Elements in Water, Environment Canada) diluted 200:1 to match sample concentrations, and was found to be within 3.7% of the reference value. Replicate determinations of standards yielded a precision of ±2.7%. All field blanks and filtration blanks (our best DI water processed as samples) were below the limits of detection.
Results and Discussion
Dissolved U concentrations range from 0.3 to 3.9 µg L−1, with the lowest values measured in offshore samples from Lake Erie, and the highest values in O’Shaughnessy and Griggs Reservoirs and the Maumee River (Table 1). U concentrations at the offshore (4 P) Lake Erie location remained relatively constant at 0.4 ± 0.07 µg L−1 while U in the southern reservoirs, two rivers and the O’Shaughnessy and Griggs Reservoirs varied considerably from the late spring through early summer (Table 1). Except for the offshore Lake Erie samples, all the samples were higher than the global mean river U concentration of 0.19 µg L−1 11. Uranium concentrations exhibited a positive relationship with bicarbonate concentrations (r2 = 0.44, p = 2 × 10−4) across the reservoir and stream data (Fig. 2), indicating that the U is likely complexed with carbonate species. However, this relationship is not present at the level of individual sites, suggesting that U in these surface water is not derived solely from carbonate lithologies.
Uranium concentrations vs. calculated bicarbonate concentrations in reservoirs and rivers in Ohio.
Although the subsurface lithologies of these watersheds consist of a wide range of Paleozoic sedimentary rocks, including carbonates and shales, they are mantled with an extensive drape of glacial tills. Much of the agricultural lands contain drainage tiles that can rapidly re-route surface runoff into local streams prior to recharging the deeper subsurface. Tile drains have been demonstrated to increase nutrient discharge to surface waters in Ohio17. Low groundwater recharge rates of 10–15 cm yr−1 are observed in the study area due to the presence of glacial tills18.
Watershed lithology can exert an influence on riverine U concentrations as waters draining the carbonate and shale bedrock can have higher U values19. The watersheds of all but Buckeye Lake and the Hoover Reservoir do have underlying Paleozoic carbonates which could be sources of dissolved U. The molar U:Ca of our water samples and the average molar U:Ca ratio (1.8 × 10−6) in limestone20 are plotted in Fig. 3. Stream and reservoir samples and Lake Erie samples from Maumee Bay plot above the mean limestone value while offshore Lake Erie samples plot on or near the average, indicating that U is either preferentially solubilized from the carbonates, the Ohio carbonates have higher U:Ca than other Paleozoic limestones, or there is another source of U to these systems. Previous work suggests that as carbonate rocks are weathered, some of the U present is retained in the soil profile, primarily adsorbed onto ferric oxyhydroxides21,22. Others have argued that U:Ca ratios in waters draining carbonate rocks are constrained by the initial U:Ca in the rocks23, thus the preferential solubilization of U over Ca in these Ohio carbonates seems unlikely. Another major natural source of U in these soils is from the Devonian Ohio Shale found in glacial drift throughout central and west central Ohio24. Soils in Ohio are elevated in U compared to average U.S. soils, with values in central and parts of eastern Ohio of 2.7–6.0 µg g−1 24, while the 50th percentile concentration of soils from the continuous US is 2.0 µg g−1 25. Although we cannot rule out the premise that these higher than mean global U fluvial concentrations are due, at least in part, to the dissolution of U-rich minerals in these Paleozoic rocks, we hypothesize that a significant portion of the dissolved U observed in these waters in Ohio is due to input from P-rich fertilizers.
Molar U/Ca ratios in Ohio surface waters during summer, 2017. ‘Mean limestone’ is the mean U/Ca molar ratio in limestone20. Error bars associated with analytical uncertainty are approximately the size of the symbols.
We are not the first to suggest the application of P-rich fertilizer on agricultural lands increases the dissolved U in surrounding surface waters. Rivers draining cotton agricultural lands in Texas were found to have higher U concentrations than forested watersheds26. Others have found soil waters in agricultural lands and agricultural drainage waters have high dissolved U concentrations27,28, with concentrations as high as 2.4 µg L−1 in agricultural drainage waters in the Florida Everglades (compared to background values of <0.1 µg L−1)29. Elevated dissolved U concentrations have been noted in agricultural watersheds in Germany30 and Brazil31.
High concentrations of dissolved U have been observed in groundwaters in the intensely agricultural lands of the High Plains and the Central Valley, California, USA, where 10% of wells exceeded the US Environmental Protection Agency maximum contaminant level (MCL) of 30 µg L−1, with concentrations as high as 2674 and 5400 µg L−1 32. These authors found that nitrate concentrations near the MCL were correlated to U, especially in shallower wells, and surmised that the U was produced from aquifer materials through nitrate-mediated U solubilization. Another possible interpretation of these data is that the dissolved U actually originated from the same fertilizer source as the nitrate, thereby being a direct, rather than an indirect, product of fertilizer application.
Shiller (1997) measured dissolved U concentrations in the Mississippi River in the range of 0.29 to 2.5 µg L−1 with means in two different years of 1.1 and 1.6 µg L−1 33. The highest values are associated with the record 1993 flood on the lower Mississippi River. Shiller noted that unlike all the other metals measured, “hydrologic factors” were important for U, reflecting the importance of discharge in controlling its concentration.
The planting of row crops, especially soya, has greatly affected the hydrology of many parts of the U.S. Increased streamflow in Iowa has been attributed to the reduction of evapotranspiration in row crop agriculture and a subsequent increase in the shallow groundwater entering into streams34. Fifty percent of the increase in bicarbonate in the Mississippi river was attributed to this increase in agriculturally induced discharge, and any soluble element associated with agricultural practices is probably also affected by this process35. Tile drainage in Illinois increased nitrate fluxes 2.5 to 4 times above the non-tiled agricultural watersheds36. Enhanced artificial drainage could help fertilizer-added U bypass soil adhesion and discharge directly into surface waters.
We posit that enhanced fertilizer usage along with increased discharge through the vadose zone of agricultural lands has increased the U concentration, both in the shallow groundwater and surface runoff. In addition, there is a large amount of legacy fertilizer products still on the landscape, both in the soil and groundwater, that can serve as a long term continued source to surface waters in the future37. It should be strongly emphasized that the U concentrations observed in Ohio are far below the drinking water standard of 30 µg L−1. However, we do suggest that, like nitrate and phosphate, dissolved U should be monitored on a more regular basis in agricultural watersheds than it is currently.
References
Rabalais, N. N. et al. Dynamics and distribution of natural and human-caused hypoxia. Biogeosciences 7, 585–619 (2010).
Pierson-Wickmann, A. C., Aquilina, L., Weyer, C., Molénat, J. & Lischeid, G. Acidification processes and soil leaching influenced by agricultural practices revealed by strontium isotopic ratios. Geochim. Cosmochim. Acta 73, 4688–4704 (2009).
Aquilina, L. et al. Long-term effects of high nitrogen loads on cation and carbon riverine export in agricultural catchments. Environ. Sci. Technol. 46, 9447–9455 (2012).
West, T. O. & McBride, A. C. The contribution of agricultural lime to carbon dioxide emissions in the United States: Dissolution, transport, and net emissions. Agric. Ecosyst. Environ. 108, 145–154 (2005).
Hamilton, S. K., Kurzman, A. L., Arango, C., Jin, L. & Robertson, G. P. Evidence for carbon sequestration by agricultural liming. Global Biogeochem. Cycles 21, 1–12 (2007).
Zhuang, P. & Mcbride, M. B. Changes during a century in trace element and macronutrient concentrations of an agricultural soil. Soil Sci. 178, 105–108 (2013).
Otero, N., Vitòria, L., Soler, A. & Canals, A. Fertiliser characterisation: Major, trace and rare earth elements. Appl. Geochemistry 20, 1473–1488 (2005).
De Kok, L. J. & Schnug, E. Loads and Fate of fertilizer derived Uranium. (Backhuys Publishers, 2008).
Schnug, E. & Lottermoser, B. G. Fertilizer-derived uranium and its threat to human health. Environ. Sci. Technol. 47, 2433–2434 (2013).
Vicente-Vicente, L. et al. Nephrotoxicity of uranium: Pathophysiological, diagnostic and therapeutic perspectives. Toxicol. Sci. 118, 324–347 (2010).
Palmer, M. & Edmond, J. Uranium in river water. Geochim. Cosmochim. Acta 57, 4947–4955 (1993).
Langmuir, D. Uranium solution-mineral equilibria at low temperatures with applications to sedimentary ore deposits. Geochim. Cosmochim. Acta 42, 547–569 (1978).
Senko, J. M., Suflita, J. M. & Krumholz, L. R. Geochemical controls on microbial nitrate-dependent U(IV) oxidation. Geomicrobiol. J. 22, 371–378 (2005).
Abdelouas, A., Lutze, W. & Nuttall, H. E. Oxidative dissolution of uraninite precipitated on Navajo sandstone. J. Contam. Hydrol. 36, 353–375 (1999).
Weber, K. A., Cameron Thrash, J., Ian Van Trump, J., Achenbach, L. A. & Coates, J. D. Environmental and taxonomic bacterial diversity of anaerobic uranium(IV) bio-oxidation. Appl. Environ. Microbiol. 77, 4693–4696 (2011).
Lyons, W. B. et al. The Geochemistry of Englacial Brine From Taylor Glacier, Antarctica. J. Geophys. Res. Biogeosciences 124, 2018JG004411 (2019).
Mrdjen, I. et al. Tile Drainage and Anthropogenic Land Use Contribute to Harmful Algal Blooms and Microbiota Shifts in Inland Water Bodies. Environ. Sci. Technol. 52, 8215–8223 (2018).
Dumouchelle, D. H. & Schiefer, M. C. Use of Streamflow Records and Basin Characteristics to Estimate Ground-Water Rechrarge Rates in Ohio. (2002).
Gardner, C. B. et al. Molybdenum, vanadium, and uranium weathering in small mountainous rivers and rivers draining high-standing islands. Geochim. Cosmochim. Acta 219, 22–43 (2017).
Li, Y.-H. A Compendium of Geochemistry: From Solar Nebula to Human Brain. (Princeton University Press, 2000).
Milton, G. M. & Brown, R. M. Adsorption of uranium from groundwater by common fracture secondary minerals. Can. J. Earth Sci. 24, 1321–1328 (1987).
Greeman, D. J., Rose, A. W. & Jester, W. A. Form and Behavior of Radium, Uranium, and Thorium in Central Pennsylvania Soils Derived from Dolomite. Geophys. Res. Lett. 17, 833–836 (1990).
Singh, S. K., Dalai, T. K. & Krishnaswami, S. 238U series isotopes and 232Th in carbonates and black shales from the Lesser Himalaya: Implications to dissolved uranium abundances in Ganga-Indus source waters. J. Environ. Radioact. 67, 69–90 (2003).
Harrell, M., McKenna, J. P. & Kumar, A. Geologic controls on indoor radon in Ohio. Rep. Investig. No. 144 (1993).
Smith, D. B., Cannon, W. F., Woodruff, L. G., Solano, F. & Ellefsen, K. J. Geochemical and mineralogical maps for soils of the conterminous United States. Open-File Report https://doi.org/10.3133/ofr20141082 (2014).
Spalding, R. F. & Sackett, W. M. Uranium in Runoff from the Gulf of Mexico Distributive Province: Anomalous Concentrations. Science. 175, 629–631 (1972).
Barišić, D., Lulić, S. & Miletić, P. Radium and uranium in phosphate fertilizers and their impact on the radioactivity of waters. Water Res. 26, 607–611 (1992).
Taylor, M. & Kim, N. The Fate of Uranium Contaminants of Phosphate Fertilizer. in Loads and Fate of Fertilizer-derived Uranium (eds. De Kok, L. J. & Schnug, E.) 147–155 (Backhuys Publishers, 2008).
Zielinski, R. A., Simmons, K. R. & Orem, W. H. Use of 234 U and 238 U isotopes to identify fertilizer-derived uranium in the Florida Everglades. Appl. Geochemistry 15, 369–383 (2000).
Liesch, T., Hinrichsen, S. & Goldscheider, N. Uranium in groundwater - Fertilizers versus geogenic sources. Sci. Total Environ. 536, 981–995 (2015).
Conceição, F. T. & Bonotto, D. M. Use of U-isotope disequilibrium to evaluate the weathering rate and fertilizer-derived uranium in São Paulo state, Brazil. Environ. Geol. 44, 408–418 (2003).
Nolan, J. & Weber, K. A. Natural Uranium Contamination in Major USAquifers Linked to Nitrate. Environ. Sci. Technol. Lett. 2, 215–220 (2015).
Shiller, A. M. Dissolved trace elements in the Mississippi River: Seasonal, interannual, and decadal variability. Geochim. Cosmochim. Acta 61, 4321–4330 (1997).
Zhang, Y. K. & Schilling, K. E. Effects of land cover on water table, soil moisture, evapotranspiration, and groundwater recharge: A Field observation and analysis. J. Hydrol. 319, 328–338 (2006).
Raymond, P. A., Oh, N. H., Turner, R. E. & Broussard, W. Anthropogenically enhanced fluxes of water and carbon from the Mississippi River. Nature 451, 449–452 (2008).
McIsaac, G. F. & Hu, X. Net N input and riverine N export from Illinois agricultural watersheds with and without extensive tile drainage. Biogeochemistry 70, 251–271 (2004).
Van Meter, K. J., Van Cappellen, P. & Basu, N. B. Legacy nitrogen may prevent achievement of water quality goals in the Gulf of Mexico. Science. 360, 427–430 (2018).
Acknowledgements
We are extremely grateful to Ms. Sydney Olund and Dr. Thomas B. Bridgeman for helping collect and process the samples. This work was supported in part by the College of Arts and Sciences and the School of Earth Sciences at The Ohio State University. We appreciate the help and knowledge of Mr. Anthony Lutton and Dr. John Olesik of TERL, School of Earth Sciences.
Author information
Authors and Affiliations
Contributions
W.B.L. is the lead author, C.B.G. and S.A.W. contributed to writing, sample analysis and data interpretation, S.I. collected samples and contributed to sample analysis and data interpretation.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Lyons, W.B., Gardner, C.B., Welch, S.A. et al. Uranium in Ohio, USA Surface Waters: Implications for a Fertilizer Source in Waters Draining Agricultural lands. Sci Rep 10, 5151 (2020). https://doi.org/10.1038/s41598-020-61922-2
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-020-61922-2
This article is cited by
-
Recent Advances in Mitigation of Uranium Ions from Wastewater: A Critical Review
Water, Air, & Soil Pollution (2025)
-
Toxicology Risk Assessment of Uranium in Drinking water of Ganderbal and Budgam Districts of Jammu and Kashmir, India
Water, Air, & Soil Pollution (2024)
-
A comprehensive review of Uranium in the terrestrial and aquatic environment: bioavailability, immobilization, tolerance and remediation approaches
Plant and Soil (2023)
-
Measurement of uranium in phosphate fertilizers for groundwater contamination employing X-ray and γ-ray spectroscopic techniques
Journal of Radioanalytical and Nuclear Chemistry (2022)
-
Spatial controls on riverbed sediment chemistry in three anthropogenically modified tropical mountainous watersheds
International Journal of Environmental Science and Technology (2022)