Abstract
The association between obstructive sleep apnea (OSA) and bone mineral density (BMD) is poorly elucidated and has contradictory findings. Abdominal computed tomography (CT) for other indications can provide a valuable opportunity for osteoporosis screening. Thus, we retrospectively explored the association between OSA and BMD by examining abdominal CT vertebrae images for a multitude of conditions and indications. We included 315 subjects (174 with OSA and 141 without OSA) who performed at least two CT scans (under similar settings). Both groups had a similar duration between the first and second CT scans of 3.6 years. BMD decreased in those with OSA and increased age. A multivariate linear regression indicated that OSA is associated with BMD alterations after controlling for age, gender, and cardiovascular diseases. Here, we report that OSA is associated with BMD alterations. Further studies are required to untangle the complex affect of OSA on BMD and the possible clinical implications of vertebra-depressed or femoral neck fractures.
Similar content being viewed by others
Introduction
Obstructive sleep apnea (OSA) is a common disorder that is associated with recurrent episodes of airway obstruction during sleep1,2. It affects more than one in seven adults1,2, many of who are undiagnosed3,4. This disorder is associated with a large body mass index (overweight and obesity), metabolic abnormalities, and cardiovascular diseases1. OSA has been associated with impaired motor function, increased risk for accidents, low bone mineral density (BMD), and fractures3,5,6,7,8,9,10.
Radiological interpretations of spine computed tomography (CT) scans for other clinical indications are the first to identify low bone density and osteoporosis, with no additional cost or exposure to unnecessary radiation11,12,13,14. CT examination allows BMD to be measured independent of large body mass index15,16. To the best of our knowledge, only one study investigated the association between OSA and CT-derived BMD alterations17; Hamada et al.17 found that males with severe OSA had alterations in lumber vertebra BMD.
Little information is available on the association between OSA and the attenuation of BMD using clinical CT scans. Earlier studies reported considerable variability in BMD between patients16, and it is possible that longitudinal assessment of BMD changes could overcome this limitation. Here, we performed a comparison study to explore alterations (in Hounsfield units; HU) in longitudinal abdominal CTs for other indications in OSA patients and no-OSA participants. The study was conducted with enrollees at the Soroka University Medical Center (SMC) from the Clalit Health Services (CHS) group, the largest health maintenance organization in Israel that keeps strict electronic registry of patient records3,4,18.
Results
Participant characteristics
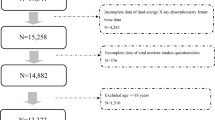
174 OSA patients (apnea–hypopnea index, AHI ≥ 5 events/hr) (60.9% men) (Fig. 1) and 141 participants without OSA (79, 56% men) were recruited. No significant differences were found in gender distribution (men/women, 79/62 and 106/68) between the no-OSA and OSA groups, respectively. The prevalence of hypertension and cardiovascular diseases was significantly higher in the OSA group (Table 1, p < 0.001). Table 2 summarizes patient characteristics according to OSA severity. Moderate/severe OSA versus mild OSA patients were older (p = 0.007), had a higher arousal and awakening index (p < 0.05), and a higher ODI (≥ 4%) index (p < 0.001). Women with OSA versus no OSA were about 5 years older than women with no OSA (Table 3, p < 0.05). Both groups had a similar duration and duration range between their first and second CT scans (Table 3, p = 0.903). The 12 thoracic vertebra BMD correlated with the first lumbar vertebra BMD on the first r = 0.863, (p < 0.001) and the second CT scan r = 0.868 (p < 0.001), respectively (Supplementary Fig. 1).
Flow chart of study participants.
Analysis of BMD including subjects who received a contrast agent
174 OSA patients and 141 participants without OSA were analyzed. Of them n = 55 OSA and n = 40 non-OSA patients, received a contrast agent. The BMD of OSA versus no OSA was significantly decreased (F = 5.9, p < 0.05), and in both groups, BMD significantly decreased over time (F = 10.67, p < 0.001) (Supplementary Table S1). The difference in BMD (i.e., the BMD in the second scan minus the BMD in the first scan) was significantly attenuated by about 6.6 HU in the OSA versus no-OSA group (Supplementary Table S1, p < 0.01). Women with OSA compared to those without OSA had a low BMD of about 11–17 HU (F = 6.84, p < 0.01), and over time, the BMD was attenuated in both groups (F = 4.23, p < 0.05). Over time, the BMD in men was significantly attenuated in both groups (F = 6.49, p < 0.05). No significant changes were found in the BMD between groups in men (F = 0.59, p = 0.44). A BMD difference was attenuated significantly more in men with OSA versus the no-OSA group by 7 HU (Supplementary Table S1, p < 0.01).
Analysis of BMD excluding subjects who received a contrast agent
In all subjects (n = 119 OSA and n = 101 non-OSA), BMD was significantly attenuated over time in both groups (Table 3, F = 11.03, p < 0.001). BMD was significantly attenuated in the OSA versus no-OSA group (Fig. 2, F = 5.54, p < 0.05), and the BMD difference decreased by 5.3 HU more in the OSA group (p = 0.01). Decreased BMD was found over time in women from both groups (F = 5.21, p < 0.05), and it was attenuated in the OSA versus no-OSA group (F = 7.50, p < 0.01). Men with OSA BMD measurements that were similar to the no-OSA group (F = 0.54, p = 0.46), and over time, BMD was attenuated in both groups (F = 5.83, p < 0.05).
Vertebrae bone mineral density. (A) Vertebra BMD for the entire group, (B) women’s BMD, (C) men’s BMD, (D) DIFF for the entire group, (E) DIFF for women, (F) DIFF for men. Data showing mean BMD of the 12 thoracic vertebrae and first lumbar vertebra; BMD bone mineral density, DIFF difference in HU between the first and second CT scans. HU Hounsfield units, OSA obstructive sleep apnea. #p < 0.05, ##p < 0.01 first scan vs. second scan BMD. Statistical differences were determined by a two-tailed t test. +p < 0.01, no-OSA vs. OSA, statistical differences were determined by a 2-way repeated measures ANOVA.
Factors associated with BMD alterations
BMD was negatively correlated with age r = − 0.49 (p < 0.01) and r = − 0.43 (p < 0.01) for OSA and no-OSA, respectively. No correlation was found between BMD (or a BMD difference) and polysomnography (PSG) findings such as in the ODI, apnea–hypopnea index, and arousal and awakening indices. The presence of cardiovascular diseases in the OSA versus non-OSA group was associated with lower BMD 127.4 ± 4.7 (HU) versus 139.8 ± 4.2 (HU) (p = 0.04) and 125.2 ± 3.9 (HU) versus 142.2 ± 7.9 (HU) (p = 0.01), respectively. Hypertension in the OSA versus no-OSA groups was associated with low BMD 126.9 ± 4.3 (HU) versus 142.2 ± 4.6 (HU) (p = 0.01) and 129.7 ± 5.4 (HU) versus 144.4 ± 5.3 (HU) (p = 0.05), respectively. Neither cardiovascular diseases nor hypertension affected the BMD difference (data not shown).
Supplementary Table S2 shows the multivariate linear regression of determinants of BMD and BMD difference alterations, including subjects who were administered an enhancement agent. Age (β = -1.8 HU, p < 0.05) and the presence of OSA (β = − 7.7 HU, p < 0.029) were associated with BMD alteration after controlling for the covariates of gender, enhancement agent, and cardiovascular diseases. Age (β = 0.15 HU, p < 0.05), OSA (β = − 5.99 HU, p < 0.01), and enhancement agent (β = -9.07 HU, p < 0.01) were associated with alterations in BMD differences after controlling for the covariates of gender and cardiovascular disease.
Tables 4 exhibits the univariate and multivariate linear regression of determinants of BMD and BMD difference alterations, excluding subjects who received an enhancement agent. Univariate regression indicated that age and cardiovascular diseases (p < 0.01) were associated with BMD alterations, and OSA was associated with BMD difference alterations (p < 0.01). A multivariate regression indicated that age (p < 0.05) and OSA (p < 0.01) were associated with alterations in BMD after controlling for gender and cardiovascular diseases. Age (p < 0.01), OSA (p < 0.05), and cardiovascular diseases (p = 0.05) were associated with alterations in BMD differences after controlling for gender.
Discussion
A limited number of studies have explored the association between OSA and vertebra BMD using clinical CT scans. To the best of our knowledge, this is the first longitudinal study reporting BMD alterations in OSA using real-life clinical records. OSA is associated with BMD alterations and differences in this alteration independent of age, cardiovascular disease, and/or contrast agent. Due to the retrospective nature of our study, we do not know the causality or mechanism between OSA and BMD. Further exploration is required to explore the mechanism that leads to BMD loss in OSA and its clinical implications, such as vertebra-depressed or femoral neck fractures.
We analyzed BMD following statistical adjustments or exclusion of participants who were administered a contrast agent. BMD decreased more in the OSA group relative to the no-OSA group. Administration of a contrast agent to the same patients in a single session may enhance the trabecular space and affect HU in the range of 11–18; however, considerable variability between patients was observed19. Recently, after analysis of a large retrospective cohort of 20,374 CT scans, Jang et al.16 concluded that it is debatable whether such differences in HU would be meaningful enough in the setting of optimistic screening.
We found lower BMD in our OSA patients, suggesting a higher risk for osteoporosis. Several clinical studies support our findings that OSA is associated with low bone BMD and fractures5,6,9. Interestingly, we found no association between BMD and the oxygen desaturation index (ODI), arousals and the awakenings index, or the apnea–hypopnea index. This finding suggests that classical PSG parameters cannot predict BMD alterations in OSA. Further exploration is required to explore serum markers of bone resorption and sympathetic activity associated with BMD loss in OSA7,8. Supporting our findings, in a cross-sectional study of 115 obese men and women with OSA, Mariani et al.20 also found a lack of association between the AHI and BMD. Hamada et al.17 found that only an alveolar–arterial oxygen pressure difference in OSA was associated with alterations in BMD and that these alterations were not associated with the apnea–hypopnea index, oxygen saturation, or arousals and the awakenings index. Cohort studies found that hypoxia—the hallmark of OSA—was associated with an increased risk for falls, incidents of fractures21, lower back pain associated with lumbar spondylosis22, and osteoporosis23. Sleep fragmentation can affect bone resorption via increased sympathetic tone and/or hormonal factors7,24,25,26,27. In the current study, cardiovascular diseases and/or hypertension negatively affected BMD and BMD differences in both groups. It is possible that increased age accompanied with cardiovascular diseases, and possibly undiagnosed OSA, could have contributed to BMD in the controls7,8. It is also possible that the sympathetic overdrive associated with cardiovascular diseases28,29 contributed to MBD loss in both of our study groups, as orexin plays a key role in sleep homeostasis and sympathetic activity, as well as through orexin receptor 1 on bone mass30. We recently found that enhanced orexin in chronic upper airway obstruction (in a rat model for OSA) can contribute to inadequate sleep and bone mass loss32,32.
Measurement of BMD by dual-energy x-ray absorptiometry revealed an association between OSA and alterations in bone metabolism/architecture21,33,34,35,36,37,38,39. However, such absorptiometry is limited in measuring alterations in a large body mass index40,41,42. Moreover, confounding information is available regarding the association between OSA and bone health using this type of scanning20,34,35,36,38,39,40. Because OSA is associated with overweight and obesity1, BMD values determined by dual-energy x-ray absorptiometry may be misleading (5–50% in several studies)41,42. Thus, further studies are required to reinforce our findings by measuring dual-energy x-ray absorptiometry and bicameral markers at the same time as CT scans are performed.
Study strengths and limitations
One of the strengths of our study is its “real-life” conditions with no research intervention. We analyzed CT scans for a multitude of conditions and indications not related to OSA of adults referred for OSA diagnosis. All enrollees had free access to medical services43. We included enrollees of the largest health maintenance organization in Israel (about 53% of the Israeli population) that keeps strict electronic registry of patient records3,4,18. Since physicians are paid per patient by a capitation fee once every three months, they have no economic incentives to prevent or deter patients from medical services3,4,18,44. Our study does have some limitations due to its retrospective single-center nature. The non-OSA group could not be considered “normal healthy” since the participants in this group were randomly selected from a large database (of over one million patients during the study period) of ambulatory CT scans in a medical arena. We also were not permitted to contact our participants to obtain additional medical information regarding their BMI, type 2 diabetes, and snoring due to legislation protecting patient confidentiality43, limiting the ability to adjust our model to this covariate. According to our medical registry, the no-OSA participants did not have a PSG study history; however, due to poor awareness of sleep-disordered breathing, we cannot rule out the possibility that some may have had unrecognized OSA45.
Conclusions
To the best of our knowledge, this is the first longitudinal study that reports BMD alterations in OSA using real-life clinical records. OSA was associated with alterations in BMD after controlling for age, gender, and cardiovascular diseases. Further studies are needed regarding clinical implications of BMD alterations in OSA, such as vertebra-depressed or femoral neck fractures.
Methods
Setting
A retrospective comparison study was conducted at the Sleep–Wake Disorders Center and Imaging Institute at SMC, in which all patients were enrollees of Clalit Health Care Services (CHS). According to the Israeli National Health Insurance Law43, all enrollees had free access to medical services, and physicians had no economic incentives to prevent or deter patients from either a PSG study or CT examination3,4,18.
This was a retrospective study, and a waiver for informed consent was obtained from the Institutional Review Committee of the SMC. All methods were performed in accordance with Israeli regulations. The Institutional Review Committee of the SMC approved the study protocol (protocol number SOR-20-0250).
Study protocol
We retrospectively searched the SMC database from June 2010 until September 2020 for all adult patients who underwent an overnight PSG, and performed at least two abdominal CTs for a multitude of conditions and indications not related to OSA. The two CT scans of each participant were done under similar settings. Prior to the PSG, all patients completed a self-administered questionnaire that included sleep habits, clinical history, and the Epworth Sleepiness Scale questionnaire46. On the study day, participants were advised to maintain their sleep–wake routine and to avoid consumption of caffeine and soft drinks. The comparison group (no-OSA) were selected randomly from the SMC database that contained over one million abdominal CT scans (during the study period) and were matched by gender with OSA patients.
Study groups
We identified 259 patients (aged 24 through 85 years) who had undergone an overnight PSG and had at least two ambulatory abdominal CT examinations for conditions not related to OSA. All subjects had “typical” symptoms of OSA and had been referred by an otolaryngology surgeon, pulmonologist, or neurologist. According to our database, none of the no-OSA group had a history of sleep problems and nor had done a PSG study.
Excluded from both groups were patients with any disease that influenced bone metabolism or who were receiving medicine that could influence bone metabolism (n = 41, Fig. 1) such as: chronic obstructive pulmonary disease, genetic disorders, cancer, autoimmune disorders, chronic liver disease, chronic renal insufficiency, musculoskeletal and connective tissue disorders, lumbar surgery, malabsorption disease, fibromatosis, unspecified anticonvulsants, and endocrine disorders. Also, excluded were those undertaking supplemental therapy with calcium or vitamin D or who needed a wheelchair for mobility. We further excluded patients receiving the following medications: oral corticoids, hormone replacement or osteoporosis therapy, proton pump inhibitors, and anticonvulsant and anticoagulant drugs. Of the remaining 280 patients, two subjects had missing PSG data, and in 15 patients, CT data could not be analyzed (the scan was not performed at 120 kV). 201 patients were included in the analysis, and of these, 27 did not have OSA (an AHI < 5 events/hr). Therefore, the OSA group comprised 174 patients.
CT examinations of 124 non-OSA patients were analyzed and, of these, data from 10 CTs could not be analyzed (examination not performed at 120 kV). We grouped 27 PSG patients who tested negative to OSA (AHI < 5 events/h) with the 114 subjects in the comparison group; the no-OSA group included 141 subjects.
Medical diagnoses retrieved from the SMC database are documented only by physicians using the International Classification of Diseases, Ninth Revision (ICD-9) code. This database contains > 99% of all patient diagnoses. We reviewed the following diagnosed cardiovascular diseases [codes 410–414, 426–438, 443] and hypertension [codes 401–405]. A self-administered questionnaire assessed the Epworth Sleepiness Scale, in which a higher score indicates a higher level of sleepiness46.
PSG study
Data were acquired using a commercially available sleep monitoring system (Viasys, SomnoStar Pro; Yorba Linda, CA, USA or SomniPro 19 PSG; Deymed Diagnostic, Hronov, Czech Republic), as previously described by our laboratory. The overnight PSG included recordings of an EEG (C3/A2, C4/A1, and O2/A1, O1/A2, according to the international 10–20 system), EOG, EMG, and ECG electrode abdomen and chest effort belts to measure respiratory activity, and an oxygen saturation sensor (SomniPro 19 PSG, Deymed Diagnostic, Hronov, Czech Republic). Scoring was done by a trained technologist and reviewed by one of the investigators (A.T.). Arousals and awakenings were scored using the American Sleep Disorders Association (ASDA) assessment47. The AHI was defined as the sum of all obstructive and mixed apneas, plus hypopneas associated with a ≥ 30% reduction in airflow and either ≥ 4% oxygen desaturation or electroencephalographic arousal, divided by the hours of total sleep time47. In addition, the percent of sleeping time in which oxygen saturation was below 90% (T90) was calculated. OSA severity was defined as AHI of 1–4.9 events/h, AHI of 5.0–14.9 events/h, or AHI ≥ 15 events/h were considered as no-OSA, mild OSA or moderate/severe OSA, respectively.
BMD measurement of the vertebral bone
We used 1-mm thick abdominal sections from CT examinations, obtained during ambulatory or emergency room visits, using a Siemens SOMATOM Definition AS + Scanner (Siemens Healthcare GmbH, Erlangen Germany) or a Philips Brilliance ICT scanner (Haifa, Israel). We retrospectively accessed the CT examinations at a constant peak voltage of 120 kV with a variable mAs tube. We included CT scans with or without a contrast agent (n = 76 no-OSA and n = 90 OSA), and evaluated vertebral BMD on a standard radiology picture archiving and communication (PACS) system workstation, with images viewed in bone windows, i.e., gray-scale assignment of the image display, to emphasize bone without the influence of alteration/BMD values (Fig. 3)16,48. We included examinations either with or without an intravenous enhancement agent. We assessed vertebral BMD by placing a single oval click-and-drag region of interest (ROI) in an axial and sagittal slice over an area of vertebral body trabecular bone and then measured CT alteration in HU, with lower HU (lower alteration) representing less-dense bone, at each of the T12 and L1 levels. On the axial images of the selected slice—the superior part of the vertebra—the elliptical ROI was encompassed manually as the largest possible area at the anterior portion of each vertebral body, and in the sagittal plane, we focused on the upper anterior part of the vertebra in order to avoid the Dense Tracecular zone. The mean CT scan density of the ROI was measured. We avoided placing the ROI near areas that would distort the BMD measurement (focal heterogeneity or lesion, posterior venous plexus, compression fracture, and artifacts). The two CT scans of each participant were performed under similar settings, and a CHS engineer from the Biomedical Engineering Department calibrated the CT scanners routinely according to the manufacturer’s instructions using an American College of Radiology-accredited phantom. BMD was expressed in milligrams per milliliter of hydroxyapatite16.
Example of unenhanced computed tomography (CT) images of the spine of a 59-year-old woman. (A) Bone window showing the first lumbar vertebra level (arrow) and oval yellow ring shows the ROI, (B) Magnified view of the first lumbar vertebra and region of interest. The yellow midline line matches the automated level; the white line reflects the standard manual level. (C) First lumbar vertebra axial image; oval yellow ring shows the region of interest. CT examination was performed at a constant peak voltage of 120 kV.
Statistical analysis
Statistical analyses were performed using R Statistical Software, version 3.5.2 (Foundation for Statistical Computing, Vienna, Austria). We compared the proportion of cardiovascular diseases and hypertension between patients with and without OSA using a Pearson chi-square test. Age, body mass index, the Epworth Sleepiness Scale, the arousal and awakening index, sleep efficiency, oxygen desaturation index (> 4%), and the percent of sleeping time in which oxygen saturation was below 90% were compared between patients with mild and moderate/severe OSA using a Student's t-test. The first and second BMD measurements and the difference between the two BMD (i.e., BMD on the second scan minus BMD on the first scan) measurements were compared between the OSA and no-OSA groups using a Student's t-test. We further evaluated the difference within the two measurements between the OSA and no-OSA groups using a two-way repeated measurements ANOVA. We calculated the study power for two different sample sizes: the first- all patients in the study, including patients who received contrast agents on imaging (OSA patients- 174, non-OSA patients-141; OSA vertebrae- 348, non-OSA vertebrae- 282). The second—patients who did not receive contrast agents on imaging only (OSA patients- 119, non-OSA patients -101, OSA vertebrae- 238, non-OSA vertebrae- 202). The calculated power to detect a difference of 1 HU in BMD between vertebrae of all patients with and without OSA with a basic mean BMD of 131 HU, a standard deviation of 2.6 HU and α = 0.05 was 99.9%. Furthermore, the calculated power to detect a difference of 1 HU in BMD between vertebrae of patients with and without OSA for which contrast enhancement was not used with a basic mean BMD of 129 HU, a standard deviation of 3.1 HU and α = 0.05 was also 99.9%. To assess the independent association between OSA and the first measurement BMD and the BMD difference between the two measurements, we used a multivariate linear regression adjusted for patient age, gender, and diagnosis of cardiovascular diseases. The beta values, including the 95% Confidence Interval (CI) and p values for each variable, were calculated. Null hypotheses were rejected at the 5% level.
References
Gottlieb, D. J. & Punjabi, N. M. Diagnosis and management of obstructive sleep apnea: A review. JAMA 323(14), 1389–1400 (2020).
Young, T. et al. Burden of sleep apnea: Rationale design major findings of the Wisconsin Sleep Cohort study. WMJ 108(5), 246–249 (2009).
Tarasiuk, A. & Reuveni, H. The economic impact of obstructive sleep apnea. Curr. Opin Pulm. Med. 19(6), 639–644 (2013).
Reuveni, H., Greenberg-Dotan, S., Simon-Tuval, T., Oksenberg, A. & Tarasiuk, A. Elevated healthcare utilisation in young adult males with obstructive sleep apnoea. Eur. Respir. J. 31(2), 273–279 (2008).
Wright, N. C. et al. The recent prevalence of osteoporosis and low bone mass in the United States based on bone mineral density at the femoral neck or lumbar spine. J Bone Miner. Res. 29(11), 2520–2526 (2014).
Sadaf, S., Shameem, M., Siddiqi, S. S., Anwar, S. & Mohd, S. effect of obstructive sleep apnea on bone mineral density. Turk. Thorac. J. 22(4), 301–331 (2021).
Swanson, C. M. et al. Obstructive sleep apnea and metabolic bone disease: Insights into the relationship between bone and sleep. J. Bone Miner. Res. 30(2), 199–211 (2015).
Swanson, C. M. et al. The importance of the circadian system & sleep for bone health. Metabolism 84, 28–43 (2018).
Sin, D. D., Man, J. P. & Man, S. F. The risk of osteoporosis in Caucasian men and women with obstructive airways disease. Am. J. Med. 114(1), 10–14 (2003).
Huang, T., Tworogerm, S., Redline, S., Curhan, G. C. & Paik, J. M. Obstructive sleep apnea and risk for incident vertebral and hip fracture in women. J. Bone Miner. Res. 35(11), 2143–2150 (2020).
Johannesdottir, F., Allaire, B. & Bouxsein, M. L. Fracture prediction by computed tomography and finite element analysis: Current and future perspectives. Curr. Osteoporos. Rep. 16(4), 411–422 (2018).
Lee, S. J. & Pickhardt, P. J. Opportunistic screening for osteoporosis using body CT scans obtained for other indications: The UW experience. Clin. Rev. Bone. Miner. Metab. 15(3), 128–137 (2017).
Jain, R. K. et al. Using opportunistic screening with abdominal CT to identify osteoporosis and osteopenia in patients with diabetes. Osteoporos. Int. 31(11), 2189–2196 (2020).
Romme, E. A. et al. Bone alteration on routine chest CT correlates with bone mineral density on DXA in patients with COPD. J. Bone Miner. Res. 27(11), 2338–2343 (2012).
Yu, W. et al. Spinal bone mineral assessment in postmeno pausal women: A comparison between dual X-ray absorptiometry and quantitative computed tomography. Osteoporos. Int. 5(6), 433–439 (1995).
Jang, S. et al. Opportunistic osteoporosis screening at routine abdominal and thoracic CT: Normative L1 trabecular alteration values in more than 20 000 adults. Radiology 291(2), 360–367 (2019).
Hamada, S. et al. Evaluation of bone mineral density by computed tomography in patients with obstructive sleep apnea. J. Clin. Sleep Med. 12(1), 25–34 (2016).
Dagan, N. et al. BNT162b2 mRNA Covid-19 vaccine in a nationwide mass vaccination setting. N. Engl. J. Med. 384(15), 1412–1423 (2021).
Pompe, E. et al. Intravenous contrast injection significantly affects bone mineral density measured on CT. Eur Radiol. 25(2), 283–289 (2015).
Mariani, S. et al. Obstructive sleep apnea and bone mineral density in obese patients. Diabetes. Metab. Syndr. Obes. 5, 395–401 (2012).
Cauley, J. A. et al. Osteoporotic Fractures in Men Study. Hypoxia during sleep and the risk of falls and fractures in older men: The Osteoporotic Fractures in Men Sleep Study. J. Am. Geriatr. Soc. 62(10), 1853–1859 (2014).
Tarnoki, A. D. et al. Lumbar spine abnormalities in patients with obstructive sleep apnoea. Sci. Rep. 11(1), 16233 (2021).
Upala, S., Sanguankeo, A. & Congrete, S. Association between obstructive sleep apnea and osteoporosis: A systematic review and meta-analysis. Int. J. Endocrinol Metab. 14(3), e36317 (2016).
Almendros, I. et al. Potential role of adult stem cells in obstructive sleep apnea. Front. Neurol. 3, 112 (2012).
Oishi, S. et al. Intermittent hypoxia influences alveolar bone proper microstructure via hypoxia-inducible factor and VEGF expression in periodontal ligaments of growing rats. Front. Physiol. 7, 416 (2016).
Elefteriou, F., Campbell, P. & Ma, Y. Control of bone remodeling by the peripheral sympathetic nervous system. Calcif. Tissue Int. 94(1), 140–151 (2014).
Tarasiuk, A. & Segev, Y. Abnormal growth and feeding behavior in upper airway obstruction in rats. Front. Endocrinol. 9, 298 (2018).
Grassi, G., Seravalle, G. & Mancia, G. Sympathetic activation in cardiovascular disease: Evidence, clinical impact and therapeutic implications. Eur. J. Clin. Invest. 45(12), 1367–1375 (2015).
Farhat, G. N. & Cauley, J. A. The link between osteoporosis and cardiovascular disease. Clin. Cases Miner. Bone Metab. 5(1), 19–34 (2008).
Wei, W. et al. Orexin regulates bone remodeling via a dominant positive central action and a subordinate negative peripheral action. Cell Metab. 19(6), 927–940 (2014).
Assadi, M. H., Segev, Y. & Tarasiuk, A. Upper airway obstruction elicited energy imbalance leads to growth retardation that persists after the obstruction removal. Sci. Rep. 10(1), 3206 (2020).
Tarasiuk, A., Levi, A., Assadi, M. H., Troib, A. & Segev, Y. Orexin plays a role in growth impediment induced by obstructive sleep-breathing in rats. Sleep 39(4), 887–897 (2016).
Blake, G. M. & Fogelman, I. The role of DXA bone density scans in the diagnosis and treatment of osteoporosis. Postgrad. Med. J. 83(982), 509–517 (2007).
Chen, Y. L. et al. Obstructive sleep apnea and risk of osteoporosis: A population-based cohort study in Taiwan. J. Clin. Endocrinol. Metab. 99(7), 2441–2447 (2014).
Tomiyama, H. Link between obstructive sleep apnea and increased bone resorption in men. Osteoporos Int. 19(8), 1185–1192 (2008).
Uzkeser, H. et al. Bone mineral density in patients with obstructive sleep apnea syndrome. Sleep Breath. 17(1), 339–342 (2013).
Mariani, S. et al. Obstructive sleep apnea and bone mineral density in obese patients. Diabetes Metab. Syndr. Obes. 5, 395–401 (2012).
Sforza, E., Thomas, T., Barthélémy, J. C., Collet, P. & Roche, F. Obstructive sleep apnea is associated with preserved bone mineral density in healthy elderly subjects. Sleep 36(10), 1509–1515 (2013).
Vilovic, M. et al. Bone metabolism parameters and inactive matrix Gla protein in patients with obstructive sleep apnea. Sleep 43(3), zsz243 (2020).
Link, T. M. Osteoporosis imaging: State of the art and advanced imaging. Radiology 263(1), 3–17 (2012).
Ho, C. P., Kim, R. W., Schaffler, M. B. & Sartoris, D. J. Accuracy of dual-energy radiographic absorptiometry of the lumbar spine: Cadaver study. Radiology 176(1), 171–173 (1990).
Bolotin, H. H. DXA in vivo BMD methodology: An erroneous and misleading research and clinical gauge of bone mineral status, bone fragility, and bone remodelling. Bone 41(1), 138–154 (2007).
National Health Insurance Law. Clause 3(d). Israeli Book of Laws 5754. No. 1469. 1994a.
Tarasiuk, A., Reznor, G., Greenberg-Dotan, S. & Reuveni, H. Financial incentive increases CPAP acceptance in patients from low socioeconomic background. PLoS ONE 7(3), e33178 (2012).
Reuveni, H. et al. Awareness level of obstructive sleep apnea syndrome during routine unstructured interviews of a standardized patient by primary care physicians. Sleep 27(8), 1518–1525 (2004).
Johns, M. W. A new method for measuring daytime sleepiness: The Epworth sleepiness scale. Sleep 14(6), 540–544 (1991).
Ilber, C., Ancoli-Israel, S., Chesson, A. & Quan, S. F. The AASM Manual for the Scoring of Sleep and Associated Events: Rules, Terminology, and Technical Specifications (American Academy of Sleep Medicine, 2007).
Pickhardt, P. J. et al. Simultaneous screening for osteoporosis at CT colonography: Bone mineral density assessment using MDCT alteration techniques compared with the DXA reference standard. J. Bone Miner. Res. 26(9), 2194–2203 (2011).
Acknowledgments
This study was supported by the Israel Science Foundation, Grant no. 164/2018.
Author information
Authors and Affiliations
Contributions
D.S., I.S., and A.T., conceived and designed the experiments; Y.C.F analyzed the CT scans; S.D. and A.T. analyzed the data; A.T. wrote the final version of the paper and recruited funds.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Daniel, S., Cohen-Freud, Y., Shelef, I. et al. Bone mineral density alteration in obstructive sleep apnea by derived computed tomography screening. Sci Rep 12, 6462 (2022). https://doi.org/10.1038/s41598-022-10313-w
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-022-10313-w
This article is cited by
-
High prevalence of vertebral fractures associated with apnea-hypopnea index in patients with recent diagnosis of obstructive sleep apnea
BMC Musculoskeletal Disorders (2025)
-
The joint effects of physical activity and sleep duration on risk of osteoporosis in Taiwanese adult population: The Taiwan Biobank Study
Osteoporosis International (2024)