Abstract
To assess drug-resistant bacterial colonisation rates and associated risk factors in children with complex chronic conditions admitted to a national reference unit in Spain. Cross-sectional study that included all children admitted to our unit from September 2018 to July 2019. Rectal swabs were obtained to determine multidrug-resistant Gram-negative bacilli (MR-GNB) colonisation, and nasal swab to determine S. aureus and methicillin-resistant S. aureus (MRSA) colonisation. Medical records were reviewed. 100 children were included, with a median of four complex chronic conditions. Sixteen percent had S. aureus colonisation, including two MRSA. S. aureus colonisation was associated with technology-dependent children, while being on antibiotic prophylaxis or having undergone antibiotic therapy in the previous month were protective factors. The prevalence of MR-GNB colonisation was 27%, which was associated with immunosuppressive therapy (aOR 31; 2.02–47]; p = 0.01), antibiotic prophylaxis (aOR 4.56; 1.4–14.86; p = 0.012), previously treated skin-infections (aOR 2.9; 1.07–8.14; p = 0.03), surgery in the previous year (aOR 1.4; 1.06–1.8; p = 0.014), and hospital admission in the previous year (aOR 1.79; [1.26–2.56]; p = 0.001). The rate of S. aureus nasal colonisation in this series was not high despite the presence of chronic conditions, and few cases corresponded to MRSA. Antibiotic prophylaxis, immunosuppressive therapies, history of infections, previous surgeries, and length of admission in the previous year were risk factors for MR-GNB colonisation.
Similar content being viewed by others
Introduction
Drug-resistant bacteria are a public health challenge of growing concern, causing significant mortality and morbidity1,2,3 and with an increasing prevalence, both in healthcare settings and the community1,4. Colonisation by certain resistant bacteria might increase the risk of infections5, especially in patients undergoing invasive procedures, and poses additional challenges in terms of antimicrobial selection, potentially contributing to an increase in drug-resistant bacteria.
Among the first to garner attention, methicillin-resistant Staphylococcus aureus (MRSA) infections emerged in the 1960s, mainly in healthcare settings. Over the next decades, the number of community-associated MRSA (CA-MRSA) infections increased and spread worldwide, with significant differences among regions6.
Staphylococcus aureus colonizes 20–40% of healthy individuals7,8; however, data on paediatric cases are scarce. A previous study (COSACO) by our group found a prevalence of 33% for S. aureus nasal colonisation in the community among Spanish children, with a low prevalence of MRSA9.
However, there are scarce data regarding the prevalence of MRSA colonisation in children with complex chronic conditions (CCC)10, who are known to require frequent hospitalisations, numerous visits to healthcare settings, and the use of invasive medical devices11. Schlesinger et al. reported a 36.4% prevalence of S. aureus nasal colonisation in children in long-term healthcare institutions in Israel, 21% of whom had MRSA, both rates being significantly higher than the rates in healthy children7. The reported risk factors for MRSA colonisation in children include underlying health conditions, recent hospitalisation12,13, previous surgery, and neurological disease13.
These children can also be colonized by multidrug-resistant Gram-negative bacilli (MR-GNB), which produce extended-spectrum beta-lactamases (ESBL), Amp-C beta-lactamases and carbapenemases2. Genes encoding for these enzymes are usually located on plasmids, which contributes to their spread in healthcare settings and the community2,14. These plasmids frequently harbour multiple co-resistance genes to other antibiotics. As with MRSA, the prevalence of MR-GNB varies between regions15. In countries such as Italy and the US, a progressive increase in the incidence of infections and colonisation due to MR-GNB has been reported in children16,17. The risk factors for this colonisation include recent antibiotic use, previous hospitalisation, invasive procedures, and intrafamilial transmission14,18,19. In children, similar risk factors have been identified20,21,22,23, together with specific risk factors such as the presence of underlying neurological conditions22, prematurity, low birth weight, and vertical transmission from the mother in the case of neonates. Outbreaks in paediatric and neonatal intensive care units have been reported2,14.
In this study, we have analysed the colonization rates for both S. aureus and MRSA as well as for MR-GNB, in a unique population who is especially vulnerable to infections, such as children with multiple and severe CCC, trying identify the associated risk factors.
Materials and methods
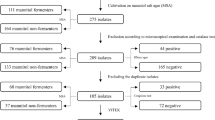
A cross-sectional study was performed at the national Spanish Referral Unit for Children with CCC at La Paz University Hospital in Madrid, Spain. Nasal and rectal swabs were obtained from patients younger than 20 years between September 2018 and July 2019. The Clinical Research Ethics Committee (PI18/3346) of La Paz University Hospital approved the study. Informed consent in accordance with the Declaration of Helsinki was obtained from the parents or guardians of all children before their inclusion, as well as from those patients aged 12 years or older, if appropriate.
Study participants
Children with two or more CCCs and those who were technology-dependent for at least 6 months and had one CCC were included11. CCC was defined based on the “Pediatric Medical Complexity Algorithm” from the Center of Excellence on Quality of Care Measures for Children with Complex Needs (COE4CCN), Seattle, Washington24. This algorithm defines CCC as: (a) any physical, mental, or developmental condition that can be expected to last at least a year, using healthcare resources above the level for a healthy child, requiring treatment to control. This can be expected to be episodically or continuously debilitating; (b) a progressive condition that is associated with deteriorating health with a decreased life expectancy in adulthood24.
All patients were screened at admission, following the Unit protocol of weekly screening, and they were included consecutively during the study period. No patient declined the inclusion in the study.
Patients with acute infections and intravenous antibiotic therapy at the time of the study were excluded. Clinical and epidemiological data were retrospectively collected for each patient through a review of the medical records (Supplementary File, supplementary Table S1).
Study samples for studying S. aureus colonisation
Nasal swabs to detect S. aureus and MRSA colonisation were collected during the first 48 h after hospital admission and analysed following the standardized protocol of the COSACO study9,25. Each sample was collected by inserting the swab tip into the two nostrils and rotating it for 5 s on each side, using ESwab™ flocked swabs with Liquid Amies Medium (Copan, Brescia, Italy)9. Methicillin resistance was detected by the disk diffusion method with cefoxitin (30 µg), as well as by susceptibility testing and disk diffusion to other antibiotics25. Minimum inhibitory concentrations for MRSA strains were studied with E-test® strips (bioMérieux). Antimicrobial susceptibility was interpreted in accordance with the European Committee on Antimicrobial Susceptibility Testing guidelines26. All isolates were screened for the presence of the mecA and mecC genes using primers by polymerase chain reaction (PCR). Strains in which the mecA or mecC gene was detected were considered MRSA. Eleven S. aureus strains were sent for molecular characterisation to the National Centre of Microbiology (Majadahonda, Spain) for further characterisation (spa typing and assessment of the following virulence factors by multiplex PCR reactions: pvl, tst, eta, etb and etd). In addition, MRSA isolates were characterised by pulsed field gel electrophoresis and staphylococcal cassette chromosome mec (SCCmec) typing.
Study samples for studying ESBL and/or carbapenemase-producing Enterobacteriaceae colonisation
Rectal swabs were collected by inserting swabs with Citoswab® semisolid Amies Transport Medium (Citotest Labware Manufacturing, Jiangsu, China) through the anal sphincter and rotating for 10 to 30 s. Samples were cultured on MacConkey agar plates containing 4 mg/L of cefotaxime, and strains were subsequently identified using a matrix-assisted laser desorption ionisation-time-of-flight system (Bruker Daltonics, Bremen, Germany). Phenotypic confirmation of ESBLs and carbapenemases was performed with the double-disk synergy method with cefotaxime, ceftazidime, cefotaxime/clavulanate, and ceftazidime/clavulanate and the modified Hodge test using imipenem, meropenem, and ertapenem discs27. PCRs with specific primers were used to detect blaKPC, blaVIM, blaIMP, blaNDM, and blaOXA-48 genes28.
Statistical analysis
The results are presented as absolute frequencies and/or percentages; the quantitative data are expressed as either a median and interquartile range or mean and standard deviation, according to the data distribution. Categorical variables were compared using the chi-squared and Fisher’s exact test, and continuous variables were compared with non-parametric tests. A consecutive multivariate analysis was performed to adjust for potential confounders using logistic regression models. The multivariate analysis included statistically significant variables from the univariate analysis and all variables considered clinically relevant. Nasal colonisation by S. aureus and colorectal colonisation by MR GNB were considered dependent variables in these models. A two-sided p-value ≤ 0.05 was considered statistically significant. We calculated the odds ratio (OR) and adjusted OR (aOR) and used them as the association measurement, with 95% confidence intervals (CI) to assess their precision. Stata v17.0 was used for the analysis.
Ethics approval
The Clinical Research Ethics Committee (PI18/3346) of La Paz University Hospital approved the study.
Consent to participate
Informed consent in accordance with the Declaration of Helsinki was obtained from the parents or guardians of all children before their inclusion, as well as from those patients aged 12 years or older, if appropriate.
Results
During the study period, 100 patients were screened (median age 2.3 years [IQR 0.8–5.6]; 60% male). The median number of CCCs per patient was four (IQR 3–5), and 80% were dependent on technology, being the most frequently required devices gastrostomy tube (32%), nasogastric tube (32%), home non-invasive ventilation (31%), home oxygen-therapy (31%), tracheostomy tube (17%) and central permanent central venous access (15%). Ninety-one percent of the patients had been hospitalized in a paediatric intensive care unit at least once in their life, 63% at least once in the last year; and 87% had undergone at least one surgery, with a median of 3 surgeries (1–5). The median intensive care stay during their lives was 30 days (IQR 8–96), and the median hospital stay was 77 days (IQR 30–140).
Staphylococcus aureus colonisation and associated risk factors
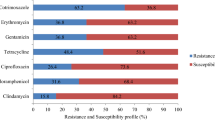
Staphylococcus aureus was isolated in 16 cases (16%). The study participants’ main characteristics according to S. aureus colonisation in the nostrils are summarized in Table 1. Among the 84 non-colonized children, 13 have been intranasally decolonized at least once in their lives with mupirocin, and 20 were receiving antibiotic prophylaxis (11 with cotrimoxazole, 7 with azithromycin and 2 with amoxicillin). None of the colonized patients were receiving antibiotic prophylaxis or have been previously decolonized.
In the univariate analysis, S. aureus colonisation was less common in the patients who were non-technology dependent, in those receiving antibiotic prophylaxis, and in those who had undergone antibiotic therapy the previous month (Table 1). A history of infections, hospital admissions, and recent surgery were not associated with a higher risk of S. aureus colonisation (Table 1). There were no differences when analysing the association between the number or type of CCC and S. aureus colonisation. Due to the small number of colonised patients, the OR could not be calculated for certain risk factors with no events in one of the comparison groups. As a result, multivariate models to predict the risk could not be fitted.
Eleven of these 16 strains were sent to the National Centre of Microbiology to assess the virulence factors and spa typing (Supplementary File, supplementary Table S2). Two strains were MRSA, both of them mecA-positive and SCCmec type IV, with only three different pattern bands in the pulsed field gel electrophoresis, sharing the same spa type (t002). Both strains were isolated in two patients hospitalised in the same ward at the same time, suggesting possible nosocomial transmission.
MR-GNB colonisation and associated risk factors
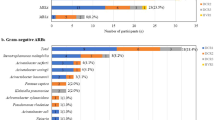
MR-GNB rectal colonisation was present in 24 patients (27%). Twenty-seven resistant strains were isolated in these 24 patients: 16 of which corresponded to ESBL-producing strains, 10 to carbapenemase producers, and 1 producing both ESBL and carbapenemase. The Verona integron-encoded metallo-β-lactamase was the most frequent carbapenemase (6/10). Klebsiella spp. was the most frequently isolated MR-GNB (Supplementary File, supplementary Table S3).
The factors associated with MR-GNB colonisation are shown in Table 2. In the univariate analysis, several conditions were identified as potential risk factors for MR-GNB colonisation: younger age, previous antibiotic exposure, history of previous skin infections, immunosuppressive therapies at inclusion, surgery in the previous year, and duration of hospital stays in the previous 12 months. Children who attended school or nurseries presented lower MR-GNB colonisation rates than those who did not attend school or nurseries (p = 0.022). No association was found with a history of S. aureus or MR-GNB colonisation or infection (p > 0.05).
In the age-adjusted multivariate analysis, factors significantly associated with MR-GNB colonisation were previous skin infections were (aOR 2.9; 1.07–8.14; p = 0.03); immunosuppressive therapies, (aOR 31; 2.02–47; p = 0.01); antibiotic prophylaxis (aOR 4.56; 1.4–14.86; p = 0.012); surgery in the previous year (aOR 1.4; 1.06–1.8; p = 0.014); and hospital admission in the previous year (aOR 1.79; [1.26–2.56];p = 0.001). Antibiotic exposure in the previous month or previous 6 months was not associated with MR-GNB colonisation (aOR 2.6; 0.95–7.3; p = 0.06 and aOR 3.2; 0.6–16; p = 0.16). After adjusting for age, school attendance was not a protective factor. The OR and aOR values for these factors are shown in Table 3.
Discussion
In this study that analysed the colonisation by resistant pathogens in a referral unit for children with CCC, the prevalence of S. aureus colonisation was lower than expected, taking into account the features of included patients, with practically no cases of MRSA. However, 1 of every 4 participants had MR-GNB colonisation, and several risk factors associated with the healthcare system were identified, including previous admissions, surgeries, immunosuppressive therapies, and antibiotic prophylaxis. This high rate of MR-GNB could be explained due to the peculiarities of the large referral hospital were the study was performed. Identifying the risk factors can help in implementing strategies for preventing colonisation and optimising treatment in this unique population. The novelty of our study is to study colonization rates in a unique population who is especially vulnerable to infections, analysing both MRSA and MDR-GNB colonization.
The prevalence of S. aureus colonisation in this series (16%) is below the 33% reported in community-dwelling children in Spain 9, although our included patients were young, and the prevalence of colonization is usually increased in older children7,9. However, as we have analyzed children with CCCs, a higher rate of carriage status were expected to be found, at least for MRSA strains, compared to children from the community, as other authors have reported higher colonization rates of S. aureus and MRSA in chronically institutionalized children7. The use of broad-spectrum antibiotics in children with CCC might have eradicated methicillin-sensitive S. aureus and CA-MRSA in this population, as these strains lack other multi-resistance genes29. In fact, exposure to antibiotics was also identified as a protective factor for MRSA colonisation in the study by Gesualdo et al.12. Half of our non-colonized children had taken antibiotics during the previous month, and up to 70% during the previous 6 months. In addition, none of the children from our cohort who had undergone previous decolonisation were found to be colonised. Antibiotic exposure might have contributed not only to the low prevalence of S. aureus and MRSA observed in our cohort but also to the lack of association with previous hospitalisation or surgeries observed in this study. In our study, S. aureus colonisation was associated with technology dependency. Other authors have reported higher rates of S. aureus colonization in tracheostomized children30, or in patients with percutaneous gastrostomy31 In the same way, tracheostomy, nasoenteric or gastrostomy tube, or the use of intravenous devices have been reported as risk factors for MRSA acquisition32,33,34,35. Therefore, invasive devices might favor colonization via bacterial biofilm formation.
The MRSA colonisation rates observed (2%) are similar to those reported in our previous community studies (1.4%)9, highlighting the fact that MRSA is not currently a relevant issue among children in Spain. A number of authors have also reported that, in children with CCCs, infections are mostly caused by Enterobacteriaceae and Pseudomonas, while S. aureus has a less relevant role36,37. However, other authors have reported a worrisome rise in MRSA cases in other European countries, with an increase specifically among children and youths4,38. Epidemiological surveillance is therefore essential.
The risk of S. aureus transmission appears to be greater after exposure to other carriers within the household or school/nursery9,39,40 than after antibiotic consumption41, while resistant Enterobacteriaceae colonisation is more frequent when individuals have been exposed to antibiotics41. In our cohort, the S. aureus colonisation rate did not increase in the children with prolonged hospitalisations, while colonisation appears to be more frequently observed in patients attending schools/nurseries, although our small sample size and low rate of S. aureus colonisation did not allow us to observe statistical significances.
Interestingly, the only two MRSA strains that appeared to be genetically related were isolated in patients admitted to the same ward during the same week, being the first admitted case of Latin American origin. Both strains shared the SCCmec IV cassette chromosome and were t002 spy type, which is usually found in CA-MRSA29. We hypothesize that nosocomial transmission of CA-MRSA strains might have occurred4. Higher CA-MRSA rates have been observed in patients of foreign origin in Spain and other European countries, including Latin American populations4,42.
There has been a worrying increase in MR-GNB infections in hospital settings in the last decade, with associated high rates of resistance to multiple antimicrobials and high morbidity and mortality37,43,44. Although several studies have analysed the prevalence of MR-GNB infections in adult populations, there is paucity of data addressing paediatric populations for whom screening of rectal colonisation is not routinely advised45. In addition, a significant variability among different regions and countries has been observed2,3,15,21. Our sample consisted of children with CCCs requiring frequent contact with healthcare facilities, use of medical devices, multiple surgeries, intensive care admissions, and recent and prolonged hospitalisations, all of which have been reported as risk factors for MR-GNB infection2,14,18,21.
Exposure to broad-spectrum antibiotics, especially carbapenems, fluoroquinolones, and cephalosporin, has been associated with infection by resistant Enterobacteriaceae20,23. Our data are in line with these findings, given that antibiotic prophylaxis decreased the risk of S. aureus colonisation but increased the prevalence of MR-GNB colonisation. In children, the relationship between exposure to non-carbapenem antibiotics and resistant infections is less clear43. Unfortunately, the prescribed regimens and administration characteristics were not collected among the study variables and could not be analysed, which, in addition to the sample size, might explain the fact that the association between MR-GNB colonisation and previous antibiotic exposure was non-significant (p = 0.06). In our series, immunosuppressive therapy was also associated with MR-GNB colonisation. Other authors have also reported a high rate of infections due to these microorganisms in immunocompromised children37,43,44.
Our results highlight the importance of MR-GNB surveillance in this population, to guide empiric antibiotic therapy and implement strict infection control measures to avoid the spread of this worrisome healthcare problem. In addition, there is a need for specific guidelines that stablish the recommendations for routine screening among hospitalised children and among those who have several CCCs. Some authors have reported that patients previously colonized by MRSA, carbapenemase-producing or ESBL-producing Enterobacteriaceae might be more susceptible to severe infections once colonized5,46,47,48. This colonization was associated, not only with significantly higher frequency of subsequent infections, but also with increased mortality48. Therefore, antibiogram-guided antimicrobial stewardship might be interesting in selected high-risk colonized patients in order to improve prognosis in the case of infections. Knowing colonization state of our patients might help to implement antibiotic stewardship policies in terms of antimicrobial prophylaxis or empirical treatments.
The small sample size and low prevalence of S. aureus colonisation in our cohort are the main limitations of this study and impaired our ability to stratify the risk and analyse the potential confounders. In addition, the variability in the ages of included patients probably implies diversity also at the level of the microbiota, with potential impact of age differences on bacterial colonization. The study’s retrospective design precluded us from analysing certain risk factors in detail, such as antibiotic exposure (length, dosing, posology, and type of antimicrobial drug). Lastly, the clinical relevance of MR-GNB colonisation in children with CCCs needs to be clarified. Prospective studies are needed to establish the actual risk of future infections by resistant bacteria in this paediatric group.
In summary, the rate of S. aureus colonisation was low in this series of children with CCCs and was associated with technology dependency. Only 2% of the patients were colonised by MRSA. However, we found a high prevalence of MR-GNB colonisation, which was associated with medical complexity and related factors (admissions, surgery, immunosuppressive therapy, antibiotic prophylaxis, and previous infections). Surveillance programs are needed to deepen the knowledge of colonisation and infection risk factors, to optimise management, and to guide empirical therapy in this vulnerable population.
Data availability
Relevant data from our study are provided in the manuscript and the supplementary file.
Abbreviations
- aOR:
-
Adjusted odds ratio
- CA-MRSA:
-
Community-associated methicillin-resistant Staphylococcus aureus
- CCC:
-
Complex chronic conditions
- CI:
-
Confidence interval
- ESBL:
-
Extended-spectrum beta-lactamases
- GNB:
-
Gram-negative bacilli
- MRSA:
-
Methicillin-resistant Staphylococcus aureus
- MR-GNB:
-
Multidrug-resistant Gram-negative bacilli
- OR:
-
Odds ratio
- SCCmec:
-
Staphylococcal cassette chromosome mec
References
Meropol, S. B., Haupt, A. A. & Debanne, S. M. Incidence and outcomes of infections caused by multidrug-resistant Enterobacteriaceae in Children, 2007–2015. J. Pediatr. Infect. Dis. Soc. 7, 36–45 (2018).
Moxon, C. A. & Paulus, S. Beta-lactamases in Enterobacteriaceae infections in children. J. Infect. 72, S41–S49 (2016).
Morrissey, I. et al. A review of ten years of the study for monitoring antimicrobial resistance trends (SMART) from 2002 to 2011. Pharmaceuticals 6, 1335–1346 (2013).
Junnila, J. et al. Changing epidemiology of methicillin-resistant Staphylococcus aureus in a low endemicity area—new challenges for MRSA control. Eur. J. Clin. Microbiol. Infect. Dis. 39, 2299–2307 (2020).
Milstone, A. M. et al. Methicillin-resistant Staphylococcus aureus colonization and risk of subsequent infection in critically ill children: Importance of preventing nosocomial methicillin-resistant Staphylococcus aureus transmission. Clin. Infect. Dis. 53, 853–859 (2011).
Lakhundi, S. & Zhang, K. Methicillin-resistant Staphylococcus aureus: Molecular characterization, evolution, and epidemiology. Clin. Microbiol. Rev. 31, e00020-18 (2018).
Schlesinger, Y. et al. Methicillin-resistant nasal colonization in children in Jerusalem: Community vs. chronic care institutions. Isr. Med. Assoc. J. 5, 847–851 (2003).
Liang, B. et al. Active surveillance, drug resistance, and genotypic profiling of Staphylococcus aureus among school-age children in China. Front. Med. 8, 701494 (2021).
Del Rosal, T. et al. Staphylococcus aureus nasal colonization in Spanish children. The COSACO Nationwide Surveillance Study. Infect. Drug Resist. 13, 4643–4651 (2020).
Feudtner, C., Feinstein, J. A., Zhong, W., Hall, M. & Dai, D. Pediatric complex chronic conditions classification system version 2: Updated for ICD-10 and complex medical technology dependence and transplantation. BMC Pediatr. 14, 199 (2014).
Climent Alcalá, F. J., García Fernández de Villalta, M., Escosa García, L., Rodríguez Alonso, A. & Albajara Velasco, L. A. Unidad de niños con patología crónica compleja. Un modelo necesario en nuestros hospitales. Anales de Pediatría 88, 12–18 (2018).
Gesualdo, F. et al. Methicillin-resistant Staphylococcus aureus nasal colonization in a department of pediatrics: A cross-sectional study. Ital. J. Pediatr. 40, 3 (2014).
Yamamoto, M. et al. Effective surveillance to identify the surgical patients carrying methicillin-resistant Staphylococcus aureus on admission in a pediatric ward. Osaka City Med. J. 62, 1–9 (2016).
Lukac, P. J., Bonomo, R. A. & Logan, L. K. Extended-spectrum-lactamase-producing Enterobacteriaceae in children: Old foe, emerging threat. Clin. Infect. Dis. https://doi.org/10.1093/cid/civ020 (2015).
Fedler, K. A., Biedenbach, D. J. & Jones, R. N. Assessment of pathogen frequency and resistance patterns among pediatric patient isolates: Report from the 2004 SENTRY Antimicrobial Surveillance Program on 3 continents. Diagn. Microbiol. Infect. Dis. 56, 427–436 (2006).
Caselli, D. et al. Incidence of colonization and bloodstream infection with carbapenem-resistant Enterobacteriaceae in children receiving antineoplastic chemotherapy in Italy. Infect. Dis. 48, 152–155 (2016).
Logan, L. K. et al. Multidrug- and Carbapenem-Resistant Pseudomonas aeruginosa in Children, United States, 1999–2012. JPIDSJ piw064 (2016) https://doi.org/10.1093/jpids/piw064.
Flokas, M. E., Alevizakos, M., Shehadeh, F., Andreatos, N. & Mylonakis, E. Extended-spectrum β-lactamase-producing Enterobacteriaceae colonisation in long-term care facilities: A systematic review and meta-analysis. Int. J. Antimicrob. Agents 50, 649–656 (2017).
Bharadwaj, R. et al. Drug-resistant Enterobacteriaceae colonization is associated with healthcare utilization and antimicrobial use among inpatients in Pune, India. BMC Infect. Dis. 18, 504 (2018).
Logan, L. K. Carbapenem-resistant Enterobacteriaceae: An emerging problem in children. Clin. Infect. Dis. 55, 852–859 (2012).
Badal, R. E. et al. Etiology, extended-spectrum β-lactamase rates and antimicrobial susceptibility of gram-negative bacilli causing intra-abdominal infections in patients in general pediatric and pediatric intensive care units—global data from the Study for Monitoring Antimicrobial Resistance Trends 2008 to 2010. Pediatr. Infect. Dis. J. 32, 636–640 (2013).
Wang, Q. et al. Risk factors and clinical outcomes for carbapenem-resistant Enterobacteriaceae nosocomial infections. Eur. J. Clin. Microbiol. Infect. Dis. 35, 1679–1689 (2016).
Sahbudak Bal, Z. et al. The prospective evaluation of risk factors and clinical influence of carbapenem resistance in children with gram-negative bacteria infection. Am. J. Infect. Control 46, 147–153 (2018).
Simon, T. D. et al. Pediatric medical complexity algorithm: A new method to stratify children by medical complexity. Pediatrics 133, e1647–e1654 (2014).
Román, F. et al. Characterization of methicillin-resistant Staphylococcus aureus strains colonizing the nostrils of Spanish children. MicrobiologyOpen 10, e1235 (2021).
EUCAST. European committee on antimicrobial susceptibility testing breakpoint tables for interpretation of MICs and zone diameters. The European Committee on Antimicrobial Susceptibility Testing. (2018).
Oteo, J. et al. Prospective multicenter study of carbapenemase-producing Enterobacteriaceae from 83 hospitals in Spain reveals high in vitro susceptibility to colistin and meropenem. Antimicrob. Agents Chemother. 59, 3406–3412 (2015).
Maseda, E. et al. Risk factors for colonization by carbapenemase-producing enterobacteria at admission to a Surgical ICU: A retrospective study. Enferm. Infecc. Microbiol. Clin. 35, 333–337 (2017).
Bassetti, M., Nicco, E. & Mikulska, M. Why is community-associated MRSA spreading across the world and how will it change clinical practice?. Int. J. Antimicrob. Agents 34, S15–S19 (2009).
El Cheikh, M. R., Barbosa, J. M., Caixêta, J. A. S. & Avelino, M. A. G. Microbiology of tracheal secretions: What to expect with children and adolescents with tracheostomies. Int. Arch. Otorhinolaryngol. 22, 50–54 (2018).
González-Del Castillo, J. et al. BAHNG score: Predictive model for detection of subjects with the oropharynx colonized by uncommon microorganisms. Rev. Esp Quimioter. 30, 422–428 (2017).
Hu, X. et al. Risk factors for methicillin-resistant Staphylococcus aureus colonization and infection in patients with human immunodeficiency virus infection: A systematic review and meta-analysis. J. Int. Med. Res. 50, 3000605211063019 (2022).
Gleeson, A., Larkin, P., Walsh, C. & O’Sullivan, N. Methicillin-resistant Staphylococcus aureus: Prevalence, incidence, risk factors, and effects on survival of patients in a specialist palliative care unit: A prospective observational study. Palliat. Med. 30, 374–381 (2016).
Hogardt, M. et al. Current prevalence of multidrug-resistant organisms in long-term care facilities in the Rhine-Main district, Germany, 2013. Euro Surveill. 20, 21171 (2015).
Warren, D. K. et al. Epidemiology of methicillin-resistant Staphylococcus aureus colonization in a surgical intensive care unit. Infect. Control Hosp. Epidemiol. 27, 1032–1040 (2006).
Folgori, L. et al. Healthcare-associated infections in pediatric and neonatal intensive care units: Impact of underlying risk factors and antimicrobial resistance on 30-day case-fatality in Italy and Brazil. Infect. Control Hosp. Epidemiol. 37, 1302–1309 (2016).
Béranger, A. et al. Early bacterial infections after pediatric liver transplantation in the era of multidrug-resistant bacteria: Nine-year single-center retrospective experience. Pediatr. Infect. Dis. J. 39, e169–e175 (2020).
Bouras, D. et al. Staphylococcus aureus osteoarticular infections in children: An 8-year review of molecular microbiology, antibiotic resistance and clinical characteristics. J. Med. Microbiol. 67, 1753–1760 (2018).
Rodriguez, M., Hogan, P. G., Krauss, M., Warren, D. K. & Fritz, S. A. Measurement and impact of Staphylococcus aureus colonization pressure in households. J. Pediatr. Infect. Dis. Soc. 2, 147–154 (2013).
Messina, N. L., Williamson, D. A., Robins-Browne, R., Bryant, P. A. & Curtis, N. Risk factors for carriage of antibiotic-resistant bacteria in healthy children in the community: A systematic review. Pediatr. Infect. Dis. J. 39, 397–405 (2020).
Dualleh, N. et al. Colonization with multiresistant bacteria in acute hospital care: The association of prior antibiotic consumption as a risk factor. J. Antimicrob. Chemother. 75, 3675–3681 (2020).
Daskalaki, M. et al. Panton-Valentine leukocidin-positive Staphylococcus aureus skin and soft tissue infections among children in an emergency department in Madrid, Spain. Clin. Microbiol. Infect. 16, 74–77 (2010).
Aguilera-Alonso, D., Escosa-García, L., Saavedra-Lozano, J., Cercenado, E. & Baquero-Artigao, F. Carbapenem-resistant gram-negative bacterial infections in children. Antimicrob. Agents Chemother. 64, e02183-e2219 (2020).
Phichaphop, C. et al. High prevalence of multidrug-resistant gram-negative bacterial infection following pediatric liver transplantation. Medicine 99, e23169 (2020).
Tacconelli, E. et al. ESCMID guidelines for the management of the infection control measures to reduce transmission of multidrug-resistant Gram-negative bacteria in hospitalized patients. Clin. Microbiol. Infect. 20, 1–55 (2014).
McConville, T. H., Sullivan, S. B., Gomez-Simmonds, A., Whittier, S. & Uhlemann, A.-C. Carbapenem-resistant Enterobacteriaceae colonization (CRE) and subsequent risk of infection and 90-day mortality in critically ill patients, an observational study. PLoS ONE 12, e0186195 (2017).
Tamma, P. D. et al. The likelihood of developing a carbapenem-resistant Enterobacteriaceae Infection during a hospital stay. Antimicrob. Agents Chemother. 63, e00757-e819 (2019).
Detsis, M., Karanika, S. & Mylonakis, E. ICU acquisition rate, risk factors, and clinical significance of digestive tract colonization with extended-spectrum beta-lactamase-producing Enterobacteriaceae: A systematic review and meta-analysis. Crit. Care Med. 45, 705–714 (2017).
Funding
This study was supported by The Spanish Ministry of Science and Innovation –Carlos III Health Institute, and European Regional Development Funds; Grant No. PI18CIII/00372 [Fondo de Investigaciones Sanitarias-Spanish Health Research Fund (ISCIII)]; Grant Award “Jose María Corretger” from the Spanish Society for Paediatric Infectious Diseases; Grant Research Award from the Spanish Association of Paediatric Primary Care; and a small grant award from the European Society for Paediatric Infectious Diseases.
Author information
Authors and Affiliations
Contributions
M.A.: Participated in research design. Participated in the performance of the research and data collection. Participated in the writing of the initial draft. Participated in reviewing the definitive manuscript. I.d.M.: Participated in research design. Participated in the performance of the research and data collection. Participated in data analysis. Participated in the writing of the initial draft. A.M.E.: Participated in research design. Participated in the performance of the research and data collection. Participated in data analysis. Participated in the writing of the initial draft. Participated in reviewing the definitive manuscript. T.S.: Participated in data analysis. Participated in the writing of the initial draft. Participated in reviewing the definitive manuscript. F.R., G.R.C., L.E.G., C.M.A., F.J.C., A.R., M.G.F.d.V.: Participated in the performance of the research. Participated in reviewing the definitive manuscript. C.C.: Participated in research design. Participated in the writing of the initial draft. Participated in reviewing the definitive manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Agud, M., de Medrano, I., Mendez-Echevarria, A. et al. Risk factors for antibiotic-resistant bacteria colonisation in children with chronic complex conditions. Sci Rep 12, 7223 (2022). https://doi.org/10.1038/s41598-022-11295-5
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-022-11295-5