Abstract
Obesity has negative effects on comorbidities, health-related quality of life and survival. Telomere length (TL) changes after bariatric surgery have been reported, but the studies are contradictory, and analyses using state-of-the art techniques for TL measurement, such as flow-FISH, are sparse. We measured TL dynamics via flow-FISH in patients undergoing bariatric surgery and compared their TL with 105 healthy individuals. Patients with obesity who underwent bariatric surgery were included. Lymphocyte and granulocyte absolute and age-adjusted (aa) TL were analyzed by flow-FISH before (preoperative cohort, n = 45) and after surgery (follow-up cohort, n = 35) at month 5.5 ± 3.9 (mean ± standard deviation [SD]). The initial lymphocyte aaTL was significantly shorter (-0.37 kb ± 0.18 kb, P = 0.045) in patients with obesity, while the granulocyte aaTL was not different from that in the healthy comparison population (0.28 kb ± 0.17 kb, P = 0.11). The telomere dynamics after surgery showed an increase in mean TL in both lymphocytes and granulocytes of patients with a pronounced BMI loss of ≥ 10 kg/m2. We did not find any association between TL increase after surgery and age, sex or the type of procedure selected for bariatric surgery. We confirmed that patients suffering from obesity have significantly shorter lymphocyte TL using flow-FISH. Along with and dependent on the degree of weight reduction after bariatric surgery, TL significantly increased in both lymphocytes and granulocytes after a mean of 5.5 months. Our results show that bariatric surgery affects not only body weight but also biomarkers of aging, such as TL.
Similar content being viewed by others
Introduction
Telomeres are the end structures of chromosomes and they have a variety of functions, including protection and stabilization of the chromosomal architecture1 as well as preserving the integrity and organization of the DNA1,2. In vertebrates, telomeres consist of the repeating hexanucleotides 5′-TTAGGG-3′ that extend the end of chromosomes for several kilobases (kb). During cell division, telomeres shorten consecutively2 and thereby mirror the degree of replicative aging the individual somatic tissue has undergone3. Critically short telomeres have been associated with cellular senescence4, the appearance of a senescence-associated secretory phenotype5, increased genetic instability6 and eventually apoptosis7.
Prematurely shortened telomeres can be found in hereditary primary telomere biology disorders (TBDs), such as dyskeratosis congenita (DKC)8, as well as in various acquired hematological diseases, such as chronic myeloid leukemia (CML), acute myeloid leukemia (AML) and aplastic anemia (AA)2,9,10,11,12,13 due to a substantially increased replicative demand in the disease-affected cellular compartments. Furthermore, many diseases linked to inflammation, cancer and autoimmunity, such as AA and psoriasis, have been reported to be associated with shorter telomeres13,14,15.
In many countries, the number of people with obesity has been increasing for decades16, which is associated with the risk of comorbidities such as coronary heart disease, high blood pressure, stroke, diabetes type II, the risk of a severe course of COVID-19 disease and negative impacts on the quality of life17,18,19. Obesity is defined by a body mass index (BMI) of 30 kg/m2 or higher20 and is categorized into class I (BMI 30.0–34.9 kg/m2), class II (BMI 35.0–39.9 kg/m2) and class III (BMI ≥ 40.0 kg/m2)21. Bariatric surgery is indicated for obesity class III and class II with secondary diseases22. According to the therapy guidelines on obesity, initial preoperative management consists of interventions regarding nutrition, exercise and behavior23. In the absence of a response to conservative therapy, surgical intervention may be the only currently available effective therapy for patients with severe obesity24.
Obesity leads to a reduced life expectancy, as confirmed by large meta-analyses25. One underlying mechanism for the fatal consequences of severe overweight is oxidative stress and chronic inflammation in adipose tissue26. Both mechanisms are known to contribute to premature shortening of telomeres, which in turn leads to a preaged cellular status27. Concordantly, patients with obesity have an increased risk of shortened TL, and BMI is negatively associated with TL28. However, despite growing evidence that telomere dynamics are affected by obesity, there are conflicting data regarding the state of TL in patients with obesity and the TL dynamics of those who undergo bariatric surgery27,28,29,30,31,32,33,34,35,36,37,38,39,40,41. In this study, we aimed to elucidate the link between TL and obesity by using the gold standard for the measurement of TL, flow-FISH, and to determine whether bariatric surgery has an impact on telomeres as an established biomarker of aging42.
Materials and methods
Patient cohort
The cohort consisted of 45 patients with obesity undergoing bariatric surgery at the Department of Surgery and Transplantation (University Hospital RWTH Aachen). The inclusion criteria, according to the recommended guidelines for surgical interventions43, were BMI ≥ 40 kg/m2 or BMI ≥ 35 kg/m2 with secondary diseases, minimum age of 18 years and insufficient response during a structured conservative program for weight reduction. Following approval by the ethics committee of the Medical Faculty of RWTH Aachen (EK 206/09) and individual written informed consent, demographic and clinical data were collected. Baseline TL in lymphocytes and granulocytes was measured 11 ± 3.3 days (mean ± SEM) before surgery. In two patients, no granulocyte TL could be measured due to insufficient sample quality. In 35 patients, a follow-up TL measurement was performed 5.5 ± 3.9 months (mean ± SD) after surgery, depending on the individual plans for clinical follow-up visits (Fig. 1). For one patient, no granulocyte TL could be measured after surgery due to insufficient sample quality (Supplementary Fig. 1).
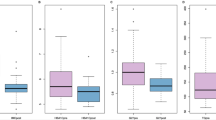
Absolute and age-adjusted (aa) telomere length (TL) in peripheral blood before bariatric surgery. Absolute TL in (A) lymphocytes (blue dots; n = 45) and (B) granulocytes (yellow dots; n = 43) in patients with obesity before bariatric surgery given in kilobases (kb). The TL of healthy subjects (n = 105) was used to show the 1%, 10%, 50%, 90% and 99% percentile of the TL, respectively (black lines). (C) Lymphocytes (blue dots; n = 45) show a mean aaTL of -0.37 kb (P = 0.0446) compared to the healthy control population. Granulocytes (yellow dots; n = 43) show a mean aaTL of 0.28 kb (P = 0.1108) compard to the healthy control population. Data are displayed as mean (red bar). *Significance of P < 0.05, **P < 0.01 (Student’s t-test).
TL measurement via flow-FISH
At the respective time points, mononuclear cells were isolated from whole blood and frozen at − 80 °C until further processing. TL analysis was performed by flow-FISH as previously described9,10,11,12,13,44.
In summary, we prepared the samples after thawing for cell denaturation and stained them with a telomere-specific (CCCTAA)3-peptide nucleic acid (PNA) FITC-labeled FISH probe (Panagene) for DNA hybridization, combined with DNA counterstaining using LDS 751 (Sigma). Fluorescence intensity was measured via an FC-500 (Becton Dickinson) using forward scatter (cell volume) and LDS 751 for identification of the cell subsets (thymocytes, lymphocytes and granulocytes). Autofluorescence values of the respective unstained lymphocytic, granulocytic and thymocytic subpopulations were subtracted from the stained samples, and the mean TL was calculated relative to control cells with a known TL (bovine thymocytes). The whole analysis was carried out in a single-blinded manner in triplicate. Samples from 105 healthy subjects were used as a comparison population for age-adjustment of the TL (aaTL) using a linear regression model. TL is given in kilobases (kb)2,10,11,12,44.
Statistical analysis
Statistical analysis was performed with Student´s t test. Age-adjusted (aa) TL measurements of the lymphocytes and granulocytes showed a normal distribution with different statistical tests (Shapiro‒Wilk, Kolmogorov‒Smirnov, D’Agostino & Pearson and Anderson‒Darling tests). The age adaptation for TL was carried out using a healthy control cohort of 105 subjects as described previously45. Preoperative samples were analyzed with unpaired Student’s t tests to compare the healthy control group with the patient cohort. A paired Student’s t test was used for the analysis of follow-up measurements to reduce interindividual differences. For each patient, a delta TL was calculated and compared pre- and post-surgery. We considered significance at a P value * < 0.05, ** < 0.01, and **** < 0.0001. The results are expressed as the mean ± standard deviation (SD). Graphics were created using GraphPad Prism Version 9.1.0, La Jolla, CA, USA.
Informed consent statement
Informed consent was obtained from all subjects involved in the study.
Institutional review board statement
The study was conducted according to the guidelines of the Declaration of Helsinki and approved by the Ethics Committee of the Medical Faculty of RWTH Aachen (EK 206/09).
Results
Patient characteristics
The mean age of the patient cohort was 46.5 ± 1.5 years (Table 1); 69% were female (n = 31), and 31% were male (n = 14). Different bariatric operations and endoscopic methods were used: sleeve gastrectomy (n = 18, 40%), gastric bypass (n = 24, 53%) and other procedures (intragastric balloon n = 2, 4%; one anastomosis gastric bypass n = 1, 2%, Table 1).
Initial TL in patients with obesity was significantly shortened compared to healthy controls
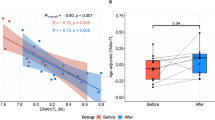
The initial absolute lymphocyte and granulocyte TL (n = 45 and n = 43, respectively) are shown in Fig. 1A (lymphocytes; blue dots) and Fig. 1B (granulocytes; yellow dots), compared to healthy individuals shown as percentile curves as used in previous publications42,44. No patient showed TL below the 1st percentile as a potential indicator of an inherited TBD. Compared to healthy controls, the lymphocyte aaTL of all patients before surgery was significantly shortened (-0.37 kb ± 0.18 kb, P = 0.045; Fig. 1C), while the granulocyte aaTL was not different (0.28 kb ± 0.17 kb, P = 0.11; Fig. 1C). Moreover, the lymphocyte TL was significantly shorter than that of the granulocytes (mean difference 0.65 kb ± 0.23 kb, P = 0.0041, Fig. 1C). Healthy individuals also had shorter TLs in lymphocytes than in granulocytes (data not shown). No correlation between preoperative BMI and the difference (Δ) in aaTL in lymphocytes (R2 = 0.04, P = 0.28) or granulocytes (R2 = 0.04, P = 0.26) could be detected (Fig. 2).
Difference (∆) of the age-adjusted (aa) telomere length (TL) before bariatric surgery. Depicted is the ΔaaTL correlation with the patients’ body mass index (BMI) before surgery. Positive values of the ΔaaTL represent an increase of the TL after surgery in lymphocytes (A) (blue dots; n = 35) and granulocytes (B) (yellow dots; n = 32). The regression line for lymphocytes (A) (P = 0.28) and granulocytes (B) (P = 0.26) shows no significance.
TL increase in patients with obesity after bariatric surgery
As expected, BMI was found to be significantly reduced from 51.7 ± 1.8 kg/m2 to 39.5 ± 1.5 kg/m2 after bariatric surgery (n = 35, P < 0.0001; Fig. 3). To estimate the effect of weight loss on telomere dynamics, we defined an absolute BMI loss of ≥ 10 kg/m2, representing a change of up to two clinical obesity classes, as a clinically relevant cutoff for weight loss. Moreover, in accordance with the guidelines for the management of overweight and obesity in adults, in which weight loss of at least 20% of the total body weight through bariatric surgery is considered effective46, the BMI cutoff value of 10 kg/m2 corresponds to a 20% reduction of mean preoperative BMI in our cohort. We observed a significant increase in the absolute lymphocyte TL in patients with a BMI loss ≥ 10 kg/m2 (n = 20; P = 0.003; Fig. 4A) compared to patients with a BMI loss < 10 kg/m2 (n = 15; P = 0.54; Fig. 4A). Of note, similar findings were observed for the granulocyte TL. Here, we observed a significant increase in the absolute granulocyte TL in patients with a BMI loss ≥ 10 kg/m2 (n = 18; P = 0.031; Fig. 4B) compared to patients with a less pronounced weight loss (n = 14; P = 0.64; Fig. 4B). Patients with a BMI loss ≥ 10 kg/m2 had a mean TL increase of 0.77 kb ± 0.23 kb in lymphocytes and 0.61 kb ± 0.26 kb in granulocytes, corresponding to an improvement in TL calculated as 17.9 life years of TL attrition. In comparison, in patients with a BMI loss < 10 kg/m2, no significant changes in lymphocyte TL or granulocyte TL were observed (0.14 kb ± 0.23 kb and 0.12 kb ± 0.25 kb, respectively).
Body mass index (BMI) reduction of the follow-up cohort. BMI before and after surgery of 35 patients is shown. BMI was significantly reduced from 51.7 ± 1.8 kg/m2 to 39.5 ± 1.5 kg/m2 (P < 0.0001; Student’s t-test).
Difference (∆) of the absolute telomere length (TL) of the follow-up population after bariatric surgery (n = 35): (A) Lymphocytes (n = 35) and (B) granulocytes (n = 32) separated in patients with a body mass index (BMI) loss ≥ 10 kg/m2 (shown in squares) or < 10 kg/m2 (shown in circles). Patients with BMI loss ≥ 10 kg/m2 had a significant increase of TL in lymphocytes and granulocytes. Data are displayed as mean (red bar). *Significance of P < 0.05, **P < 0.01 (Student’s t-test).
Discussion
Obesity is associated with chronic inflammation and cardiovascular diseases17,47,48,49, both of which are associated with shortened telomeres and reduced survival27,28,50. In line with this, recent studies have linked obesity with shortened telomeres28,42,51. In addition, a recent study also showed that pregnancy-related changes in weight gain during pregnancy had an impact on TL52. However, data on the association between TL and obesity, particularly telomere dynamics after bariatric surgery, are contradictory. Some studies found shortened telomeres in patients with obesity28,29, while others could not confirm these results30,31. Similar results on telomere lengthening after successful weight reduction were found, reviewed by Peña et al.37. Some data indicate a lengthening of telomeres after weight loss, while some do not19,32,33,34,42. Different timings of TL measurement and different periods of follow-up, ranging from a few months to several years, could explain these contradictory results. However, a major influencing factor is the technique used for TL measurement.
In the vast majority of the studies, PCR techniques were used29,34,36,39, and these have several disadvantages, such as reduced accuracy and noncell-specific TL measurement compared to telomere measurement using flow-FISH45. In addition, there is no way to differentiate among leukocyte subpopulations when using PCR. This is a significant limitation since lymphocytes (including their naïve and memory subpopulations)53 and granulocytes show different absolute values and kinetics of TL during normal physiological aging2. Various publications have demonstrated that PCR techniques are limited in the diagnostic potential of telomere biology-associated disorders, particularly when the phenotype is restricted to individual hematopoietic lineages, such as in leukemias8,45. Based on individual and prospective evaluations of telomere-associated disease states (e.g., linked to increased replicative aging) as well as in TBD, flow-FISH represents the gold standard of telomere measurement: leukocyte subpopulations such as lymphocytes and granulocytes (as well as their distributions) can be analyzed separately and with high accuracy45.
To our knowledge, only one previous study used flow-FISH as a standardized method for TL assessment in the context of bariatric surgery31, and our study is the first to specifically investigate the granulocyte compartment. We found a correlation between obesity and TL shortening in lymphocytes, which was not detectable in granulocytes. Following bariatric surgery, the weight reduction was correlated with a significant increase in both lymphocyte and granulocyte TL in those patients who had a BMI change of ≥ 10 kg/m2. This cutoff represents a relevant weight reduction, corresponding to at least a 20% weight reduction in baseline body weight46.
In the context of obesity, chronic inflammation is probably one of the main mechanisms leading to the consecutive shortening of TLs in immune cells. We hypothesize that the myeloid compartment is also affected by the systemic effects of obesity but to a lesser extent than the lymphatic compartment. The influence of obesity-induced inflammation on the hematopoietic stem cell niche48, as well as on myeloid immune cells54, has been described previously. Here, we confirmed that weight reduction had beneficial effects on TL in both lymphocytes and granulocytes of obese individuals. As previously shown, adipose tissue, as an important organ of the immune system, is instrumental in impairing immune cells and maintaining chronic inflammation in patients with obesity49,55. Lymphocytes in particular play an important role in chronic inflammation and insulin resistance in this patient population56. Because of this, we assumed that TL attrition caused by obesity-associated (chronic) inflammation mainly occurs in the lymphocyte compartment. This is also emphasized by the fact that we observed significant differences in lymphocyte and granulocyte TL (Fig. 1C), although this is a phenomenon that can also be observed in healthy individuals. Furthermore, we could not detect any significant change in the TL of the granulocytes in the preoperative cohort, possibly due to the small sample size.
Since we observed a postoperative increase in TL, particularly in patients with significant weight loss, we mechanistically assumed that a significant reduction in adipose tissue is required to reduce systemic inflammation before seeing beneficial effects on telomere biology. We hypothesize that proinflammatory cytokines, such as IL-1, IL-6, and IL-12, which are increased in patients with obesity57,58,59, lead to telomere shortening at the stem cell and progenitor levels. Moreover, an increased activation and proliferation of immune cells, indicated by more white blood cells, especially granulocytes and lymphocytes, in patients with obesity60, could be a mechanism of TL shortening. Another explanation for the decrease in TL could be telomere attrition by reactive oxygen species (ROS)61 and impairment of telomerase activity in immune cells and their progenitors62.
Our study has several limitations that need to be addressed. In addition to a small sample size and being a single-site study, we had a short follow-up time. One could speculate that the positive metabolic changes brought about by weight loss will not be completed in a short time. Further investigation of lymphocyte subpopulations, such as CD4+ cells, CD8+ cells and Tregs, as well as myeloid subpopulations, could reveal the impairment of the immune system more fully.
In conclusion, we showed that patients with obesity have significantly shortened telomeres in only lymphocytes. After bariatric surgery, we observed an increase in both lymphocyte and granulocyte TL in patients with significant weight reduction, arguing for a systemic effect of bariatric surgery and not only a shift between subcompartments. This TL increase following surgery was dependent on the overall degree of BMI reduction. Therefore, we assume that patients with a significant postoperative TL increase will benefit from bariatric surgery to the greatest extent. However, larger prospective studies with longer follow-up periods are needed to validate this assumption.
Data availability
The datasets generated during and/or analyzed during the current study are available from the corresponding authors on reasonable request.
References
O’Sullivan, R. J. & Karlseder, J. Telomeres: Protecting chromosomes against genome instability. Nat. Rev. Mol. Cell Biol. 11(3), 171–181. https://doi.org/10.1038/nrm2848 (2010).
Brümmendorf, T. H. & Balabanov, S. Telomere length dynamics in normal hematopoiesis and in disease states characterized by increased stem cell turnover. Leukemia 20(10), 1706–1716. https://doi.org/10.1038/sj.leu.2404339 (2006).
Jiang, H. et al. Proteins induced by telomere dysfunction and DNA damage represent biomarkers of human aging and disease. Proc. Natl. Acad. Sci. 105(32), 11299–11304. https://doi.org/10.1073/pnas.0801457105 (2008).
Fyhrquist, F., Saijonmaa, O. & Strandberg, T. The roles of senescence and telomere shortening in cardiovascular disease. Nat. Rev. Cardiol. 10(5), 274–283. https://doi.org/10.1038/nrcardio.2013.30 (2013).
Braig, M. et al. A ‘telomere-associated secretory phenotype’ cooperates with BCR-ABL to drive malignant proliferation of Leukemic cells. Leukemia 28(10), 2028–2039. https://doi.org/10.1038/leu.2014.95 (2014).
Hackett, J. A., Feldser, D. M. & Greider, C. W. Telomere dysfunction increases mutation rate and genomic instability. Cell 106(3), 275–286. https://doi.org/10.1016/S0092-8674(01)00457-3 (2001).
Mondello, C. & Scovassi, A. I. Telomeres, telomerase, and apoptosis. Biochem. Cell Biol. 82(4), 498–507. https://doi.org/10.1139/o04-048 (2004).
Vieri, M., Brümmendorf, T. H. & Beier, F. Treatment of telomeropathies. Best Pract. Res. Clin. Haematol. 34(2), 101282. https://doi.org/10.1016/j.beha.2021.101282 (2021).
Beier, F., Foronda, M., Martinez, P. & Blasco, M. A. Conditional TRF1 knockout in the hematopoietic compartment leads to bone marrow failure and recapitulates clinical features of dyskeratosis congenita. Blood 120(15), 2990–3000. https://doi.org/10.1182/blood-2012-03-418038 (2012).
Beier, F. et al. Accelerated telomere shortening in glycosylphosphatidylinositol (GPI)-negative compared with GPI-positive granulocytes from patients with paroxysmal nocturnal hemoglobinuria (PNH) detected by proaerolysin flow-FISH. Blood 106(2), 531–533. https://doi.org/10.1182/blood-2004-10-3996 (2005).
Beier, F. et al. Telomere dynamics in patients with del (5q) MDS before and under treatment with lenalidomide. Leuk. Res. https://doi.org/10.1016/j.leukres.2015.09.003 (2015).
Brümmendorf, T. H. et al. Prognostic implications of differences in telomere length between normal and malignant cells from patients with chronic myeloid leukemia measured by flow cytometry. Blood 95(6), 1883–1890 (2000).
Brümmendorf, T. H., Maciejewski, J. P., Mak, J., Young, N. S. & Lansdorp, P. M. Telomere length in leukocyte subpopulations of patients with aplastic anemia. Blood 97(4), 895–900. https://doi.org/10.1182/blood.V97.4.895 (2001).
Rolles, B. et al. telomere shortening in peripheral leukocytes is associated with poor survival in cancer patients treated with immune checkpoint inhibitor therapy. Front. Oncol. https://doi.org/10.3389/fonc.2021.729207 (2021).
Wenn, K. et al. Telomere length at diagnosis of chronic phase chronic myeloid leukemia (CML-CP) identifies a subgroup with favourable prognostic parameters and molecular response according to the ELN criteria after 12 months of treatment with nilotinib. Leukemia 29(12), 2402–2404. https://doi.org/10.1038/leu.2015.245 (2015).
Jaacks, L. M. et al. The obesity transition: Stages of the global epidemic. Lancet Diabetes Endocrinol. 7(3), 231–40. https://doi.org/10.1016/S2213-8587(19)30026-9 (2019).
Blüher, M. Obesity: Global epidemiology and pathogenesis. Nat. Rev. Endocrinol. 15(5), 288–298. https://doi.org/10.1038/s41574-019-0176-8 (2019).
Stefan, N., Birkenfeld, A. L. & Schulze, M. B. Global pandemics interconnected-obesity, impaired metabolic health and COVID-19. Nat. Rev. Endocrinol. 17(3), 135–149. https://doi.org/10.1038/s41574-020-00462-1 (2021).
Kolotkin, R. L. & Andersen, J. R. A systematic review of reviews: Exploring the relationship between obesity, weight loss and health-related quality of life. Clin. Obes. 7(5), 273–289. https://doi.org/10.1111/cob.12203 (2017).
Caballero, B. Humans against obesity: Who will win?. Adv. Nutr. 10(suppl_1), S4–S9. https://doi.org/10.1093/advances/nmy055 (2019).
Keating, C. et al. Prevalence of class-I, class-II and class-III obesity in Australian adults between 1995 and 2011–2012. Obes. Res. Clin. Pract. 9(6), 553–562. https://doi.org/10.1016/j.orcp.2015.02.004 (2015).
Dietrich, A. et al. Obesity surgery and the treatment of metabolic diseases. Dtsch Arztebl Int. 115(42), 705–711. https://doi.org/10.3238/arztebl.2018.0705 (2018).
Ruban, A., Stoenchev, K., Ashrafian, H. & Teare, J. Current treatments for obesity. Clin. Med. (Lond) 19(3), 205–212. https://doi.org/10.7861/clinmedicine.19-3-205 (2019).
Wirth, A., Wabitsch, M. & Hauner, H. The prevention and treatment of obesity. Dtsch Arztebl Int. 111(42), 705–713. https://doi.org/10.3238/arztebl.2014.0705 (2014).
Di Angelantonio, E. et al. Body-mass index and all-cause mortality: Individual-participant-data meta-analysis of 239 prospective studies in four continents. The Lancet 388(10046), 776–786. https://doi.org/10.1016/S0140-6736(16)30175-1 (2016).
Monteiro, R. & Azevedo, I. Chronic inflammation in obesity and the metabolic syndrome. Mediators Inflamm. https://doi.org/10.1155/2010/289645 (2010).
Gavia-García, G. et al. Telomere length and oxidative stress and its relation with metabolic syndrome components in the aging. Biology (Basel). https://doi.org/10.3390/biology10040253 (2021).
Gielen, M. et al. Body mass index is negatively associated with telomere length: A collaborative cross-sectional meta-analysis of 87 observational studies. Am. J. Clin. Nutr. 108(3), 453–475. https://doi.org/10.1093/ajcn/nqy107 (2018).
Morton, J. M., Garg, T. & Leva, N. Association of laparoscopic gastric bypass surgery with telomere length in patients with obesity. JAMA Surg. 154(3), 266–268. https://doi.org/10.1001/jamasurg.2018.4830 (2019).
Dershem, R. et al. Changes in telomere length 3–5 years after gastric bypass surgery. Int. J. Obes. 41(11), 1718–1720. https://doi.org/10.1038/ijo.2017.156 (2017).
Jongbloed, F. et al. Effects of bariatric surgery on telomere length and T-cell aging. Int. J. Obes. 43(11), 2189–99. https://doi.org/10.1038/s41366-019-0351-y (2019).
Carulli, L. et al. Telomere length elongation after weight loss intervention in obese adults. Mol. Genet. Metab. 118(2), 138–142. https://doi.org/10.1016/j.ymgme.2016.04.003 (2016).
Hohensinner, P. J. et al. Reduction of premature aging markers after gastric bypass surgery in morbidly obese patients. Obes. Surg. 28(9), 2804–2810. https://doi.org/10.1007/s11695-018-3247-3 (2018).
Kim, S. et al. Obesity and weight gain in adulthood and telomere length. Cancer Epidemiol. Biomarkers Prev. 18(3), 816–820. https://doi.org/10.1158/1055-9965.EPI-08-0935 (2009).
Laimer, M. et al. Telomere length increase after weight loss induced by bariatric surgery: Results from a 10 year prospective study. Int. J. Obes. 40(5), 773–778. https://doi.org/10.1038/ijo.2015.238 (2016).
Peña, E. et al. Telomere length in patients with obesity submitted to bariatric surgery: A systematic review. Eur. Eat Disorders Rev. https://doi.org/10.1002/erv.2865 (2021).
Peña, E. et al. Longitudinal changes in telomere length in a cohort of obese patients submitted to bariatric surgery: A 2-year follow-up. Surg. Obes. Relat. Dis. 16(11), 1794–1801. https://doi.org/10.1016/j.soard.2020.06.027 (2020).
Sonia, G.-C. et al. Telomere length as a biomarker for adiposity changes after a multidisciplinary intervention in overweight/obese adolescents: The EVASYON Study. PLOS ONE 9(2), e89828. https://doi.org/10.1371/journal.pone.0089828 (2014).
Welendorf, C. et al. Obesity, weight loss, and influence on telomere length: New insights for personalized nutrition. Nutrition (Burbank, Los Angeles County, Calif.) 66, 115–21. https://doi.org/10.1016/j.nut.2019.05.002 (2019).
Welendorf, C. R. et al. The impact of gastric bypass on telomere length and shelterin complex gene expression: 6 months prospective study. Obes. Surg. 31(6), 2599–606. https://doi.org/10.1007/s11695-021-05299-9 (2021).
Zhang, Y. et al. Association between weight change and leukocyte telomere length in U.S.. Adults. Front. Endocrinol. 12, 650988. https://doi.org/10.3389/fendo.2021.650988 (2021).
Himbert, C., Thompson, H. & Ulrich, C. M. Effects of intentional weight loss on markers of oxidative stress, DNA repair and telomere length-a systematic review. Obes. Facts 10(6), 648–665. https://doi.org/10.1159/000479972 (2017).
Runkel, N. et al. Evidence-based German guidelines for surgery for obesity. Int. J. Colorectal Dis. 26(4), 397–404. https://doi.org/10.1007/s00384-011-1136-5 (2011).
Werner, B. et al. Reconstructing the in vivo dynamics of hematopoietic stem cells from telomere length distributions. eLife https://doi.org/10.7554/eLife.08687 (2015).
Ferreira, M. S. V. et al. Comparison of flow-FISH and MM-qPCR telomere length assessment techniques for the screening of telomeropathies. Ann. N.Y. Acad. Sci. 1466(1), 93–103. https://doi.org/10.1111/nyas.14248 (2020).
Jensen, M. D. et al. 2013 AHA/ACC/TOS guideline for the management of overweight and obesity in adults. Circulation 129(25_suppl_2), S102-38. https://doi.org/10.1161/01.cir.0000437739.71477.ee (2014).
Aguilar, E. G. & Murphy, W. J. Obesity induced T cell dysfunction and implications for cancer immunotherapy. Curr. Opin. Immunol. 51, 181–186. https://doi.org/10.1016/j.coi.2018.03.012 (2018).
Bowers, E. & Singer, K. Obesity-induced inflammation: The impact of the hematopoietic stem cell niche. JCI Insight https://doi.org/10.1172/jci.insight.145295 (2021).
Reilly, S. M. & Saltiel, A. R. Adapting to obesity with adipose tissue inflammation. Nat. Rev. Endocrinol. 13(11), 633–643. https://doi.org/10.1038/nrendo.2017.90 (2017).
Yeh, J.-K. & Wang, C.-Y. Telomeres and telomerase in cardiovascular diseases. Genes Basel 7(9), 54. https://doi.org/10.3390/genes7090058 (2016).
Mundstock, E. et al. Effect of obesity on telomere length: Systematic review and meta-analysis. Obesity (Silver Spring) 23(11), 2165–2174. https://doi.org/10.1002/oby.21183 (2015).
Maugeri, A., Magnano San Lio, R., La Rosa, M. C., Giunta, G., Panella, M., & Cianci, A. et al. The relationship between telomere length and gestational weight gain: Findings from the mamma & bambino cohort. Biomedicines 2021. https://doi.org/10.3390/biomedicines10010067.
Rufer, N. et al. Telomere fluorescence measurements in granulocytes and T lymphocyte subsets point to a high turnover of hematopoietic stem cells and memory T cells in early childhood. J. Exp. Med. 190(2), 157–167. https://doi.org/10.1084/jem.190.2.157 (1999).
Gummlich, L. Obesity-induced neutrophil reprogramming. Nat. Rev. Cancer 21(7), 412. https://doi.org/10.1038/s41568-021-00372-y (2021).
Koenen, M., Hill, M. A., Cohen, P. & Sowers, J. R. Obesity, adipose tissue and vascular dysfunction. Circ. Res. 128(7), 951–968. https://doi.org/10.1161/CIRCRESAHA.121.318093 (2021).
Xu, E. et al. Temporal and tissue-specific requirements for T-lymphocyte IL-6 signalling in obesity-associated inflammation and insulin resistance. Nat. Commun. 8(1), 14803. https://doi.org/10.1038/ncomms14803 (2017).
Charles, B. A. et al. The roles of IL-6, IL-10, and IL-1RA in obesity and insulin resistance in African-Americans. J. Clin. Endocrinol. Metab. 96(12), E2018–E2022. https://doi.org/10.1210/jc.2011-1497 (2011).
Ballak, D. B., Stienstra, R., Tack, C. J., Dinarello, C. A. & van Diepen, J. A. IL-1 family members in the pathogenesis and treatment of metabolic disease: Focus on adipose tissue inflammation and insulin resistance. Cytokine 75(2), 280–290. https://doi.org/10.1016/j.cyto.2015.05.005 (2015).
Suárez-Álvarez, K. et al. Serum IL-12 is increased in Mexican obese subjects and associated with low-grade inflammation and obesity-related parameters. Mediators Inflamm. 2013, 967067. https://doi.org/10.1155/2013/967067 (2013).
Dixon, J. B. & O’Brien, P. E. Obesity and the white blood cell count: Changes with sustained weight loss. Obes. Surg. 16(3), 251–257. https://doi.org/10.1381/096089206776116453 (2006).
Wang, L. et al. Selective oxidative stress induces dual damage to telomeres and mitochondria in human T cells. Aging Cell 20(12), e13513. https://doi.org/10.1111/acel.13513 (2021).
Kordinas, V., Ioannidis, A. & Chatzipanagiotou, S. The telomere/telomerase system in chronic inflammatory diseases. Cause or Effect?. Genes (Basel) https://doi.org/10.3390/genes7090060 (2016).
Acknowledgements
We thank Anne Abels and Lucia Vankann for their excellent technical expertise and help concering sample measurements. We owe a great debt of gratitude to all persons involved at the University Hospital Aachen and due to the excellent cooperation between the surgical center and the oncology department (CIO Aachen).
Funding
Open Access funding enabled and organized by Projekt DEAL.
Author information
Authors and Affiliations
Contributions
Conceptualization: F.B., M.T., T.F.U., B.R. Methodology: B.R., F.B., M.S.V.F., M.V. Validation: B.R., M.T., F.B., M.S.V.F., M.V. Formal analysis: B.R., M.T. Investigation: T.F.U., M.T. Resources: M.T., T.F.U., B.R. Data curation: M.T., F.B. Writing—original draft preparation: B.R., M.T., T.F.U. Writing—review and editing: F.B., M.S.V.F., M.V., P.H.A., U.N., T.H.B., M.T., T.F.U., B.R., K.P.R., S.M.S. Visualization: B.R., M.T., T.F.U., F.B. Supervision: M.T., T.F.U. Project administration: M.T., T.F.U. All authors have read and agreed to the published version of the manuscript.
Corresponding author
Ethics declarations
Competing interests
THB and FB receive scientific support from Repeat Dx, Vancouver, Canada. The other authors declare that they have no conflict of interest.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Rolles, B., Ferreira, M.S.V., Vieri, M. et al. Telomere length dynamics measured by flow-FISH in patients with obesity undergoing bariatric surgery. Sci Rep 13, 304 (2023). https://doi.org/10.1038/s41598-022-27196-6
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-022-27196-6