Abstract
Nonalcoholic fatty liver disease (NAFLD) has been linked with a number of extra hepatic diseases and could be a potential risk factor of decreasing bone mineral density. To determine whether Upper Egyptian patients with NAFLD are at risk of developing osteoporosis. Cross sectional study was done on a total 100 individuals; 50 patients diagnosed with NAFLD (based on ultrasound imaging) crossed-matched with 50 individuals without NAFLD based on age, sex and body mass index. Bone mineral density, serum calcium and phosphorus levels, serum parathyroid hormone, serum vitamin D and fasting insulin level were assessed. Osteoporosis was prevalent in NAFLD patients versus to controls (19/50 vs. 0/50; P < 0.001). There was significant decrease in bone mineral density in NAFLD patients than controls (− 2.29 ± 0.4 vs. − 1.53 ± 0.1; P < 0.001). There was a statistical significance decrease in serum vitamin D and calcium levels in NAFLD patients than controls. Furthermore, vitamin D levels in the NAFLD group was a predictor for osteoporosis (OR 0.614; 95% CI 0.348–0.825). Patients with NAFLD tend to have a significant decrease in bone density, vitamin D, and serum calcium levels than controls.
Similar content being viewed by others
Introduction
Nonalcoholic fatty liver disease (NAFLD) has emerged as noninfectious liver disease affecting between 17 and 33% in the general population and 75% in obese and/or diabetic individuals worldwide1. NAFLD is not confined to adult but it occurs in children and adolescents. Prevalence report from Egypt estimates NAFLD among school children was 15.8% in cross-sectional study2.
The pathological spectrum of NAFLD ranges from simple steatosis to nonalcoholic steatohepatitis (NASH) that ultimately may progress to fibrosis and cirrhosis which may be complicated by hepatocellular carcinoma3,4.
NAFLD has been linked with extra hepatic diseases as cardiomyopathy, cardiac arrhythmias, chronic kidney disease, type 2 diabetes mellitus (T2DM) and obstructive sleep apnea3,5,6. Moreover, NAFLD has been associated with increased risk of extra hepatic cancer including gastrointestinal, urinary, lung, breast, and gynecological cancers7.
Osteoporosis (OP) is a systemic skeletal disease characterized by low bone mineral density (BMD) and may be associated with pathological fracture8. In developed countries, OP affecting 9–38% of women and 1–8% of men aged > 50 years9.
Diagnosis of OP is based on dual-energy X-ray absorptiometry (DEXA)10. There are multiple sites that could be used to assess bone mineral density including hip, spine and wrist. In spite of these multiple sites, DEXA scanning of the hip or spine is validated by world health organization (WHO)11.
About 12–55% of patients with liver cirrhosis have imminent risk of vertebral fractures so patients with chronic liver disease need to be screened by DEXA scan for early detection of OP as vertebral fractures are usually asymptomatic12,13.
Several cross sectional studies have evaluated association between NAFLD and lower BMD. Unfortunately, the results of these studies were conflicting as some studies have identified a significant association between NAFLD and low BMD14,15 and other studies showed no significant associations between NAFLD and low BMD17,18.
Some Studies showed that NAFLD may contribute in the pathophysiology of bone demineralization and OP via production of multiple pro-inflammatory cytokines, tumor necrosis factor α (TNF-α), pro-oxidant mediators, bone-influencing molecules and/or via the direct effect on hepatic and insulin resistance12,19.
As association between NAFLD and low BMD is still a matter of controversy, so the study aimed to evaluate bone mineral density, serum calcium and vitamin D in patients with NAFLD to determine if patients who have NAFLD are at risk of developing OP.
Patients and methods
This cross section study was done in Hepato-Gastroentrology and rheumatology departments, Al-Azhar Assiut University hospital, from February 2019 to December 2020 to determine if patients who have NAFLD are at risk of OP.
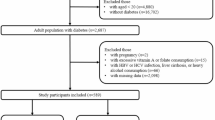
A total 100 individuals were enrolled in the study; 50 patients diagnosed by ultrasound to have NAFLD and 50 crossed matched individuals without NAFLD based on age, sex and BMI.
The study was approved by ethical committee of Al-Azhar Assiut faculty of medicine and an informed written consent was signed by every individual before being enrolled in the study. The study was conducted in accordance with ethical principles of the World Medical Association Declaration of Helsinki.
Inclusion criteria
Any individual diagnosed by ultrasound to have (NAFLD) and aged ≥ 18 years.
Exclusion criteria
Individual with any one of the following criteria were excluded from the study: (1) aged ≤ 18 years, (2) any liver disease that could lead to NAFLD such as viral hepatitis, autoimmune liver diseases, alcohol consumption, Wilson’s disease, hemochromatosis, (3) recent exposure to hepatotoxic drugs within 6 months or drugs containing or affecting vitamin D level, (4) diabetes mellitus, (5) renal disease (6) Pregnant or lactating women.
Investigatory work-up
Eligible individuals (cases and controls) were admitted to Hepato-Gastroentrology and rheumatology departments, et al.-Azhar Assiut University Hospital and full history taking, clinical examination and BMI were assessed for every individual.
After midnight fasting, eligible individuals were assessed by the following laboratory tests and imaging studies: complete blood count (CBC), liver function tests (serum bilirubin, AST, ALT, albumin and INR), renal function test (urea, creatinine and serum uric acid), fasting blood sugar (FBS), cholesterol, triglycerides and erythrocyte sedimentation rate (ESR).
Serum calcium and phosphorus levels were assessed by colorimetric method using (spinreact, S.A.U., Spain) and (5010 chemistry photometer, Germany) and reference range for serum calcium is 8.0–10.5 mg/dl and 2.5–5 mg/dl for phosphorus.
Serum parathyroid hormone level was assessed by ELISA using (Human PTH ELISA Kit, Bioassay technology laboratory) produced by (Shanghai Korian Biotech Co., Ltd, China) and (Robonik ELISA plate reader, India).
Serum 25-OH Vitamin D3/D2 level was assessed by ELISA using (ORGENTEC Diagnostika GmbH, Germany) and (Robonik ELISA plate reader, India).
Fasting insulin level was assessed by ELISA using (human insulin enzyme immunoassay test Kit , prechek Bio Inc , USA) and (Robonik ELISA plate reader , India) and Homeostasis Model Assessment of Insulin Resistance (HOMA-IR) was calculated by using the following equation: HOMA-IR = fasting insulin (micro unite/ml) × fasting blood glucose (mmol/ml)/22.520,21.
Pelvi-abdominal ultrasound examination for eligible individuals was done after a midnight fasting using a B-mode convex probe US equipment (Esaote ID, CE0051; Technos, Genoa, Italy) with a 4.5–7 MHz to assess severity of fatty liver and NAFLD was graded according to echogenicty of the liver22 into Grade I: minimal diffuse increase in hepatic echogenicty in which the liver appears bright compared with the cortex of the kidney with normal visualization of diaphragm and borders of intrahepatic vessel, Grade II: moderate diffuse increase in hepatic echogenicty with slightly impaired visualization of diaphragm and intrahepatic vessels, Grade III: marked increase in hepatic echogenicty which obscures visualization of intrahepatic vessels and diaphragm.
Bone mineral density (BMD) at lumbar spine was measured by dual energy X-ray absorptiometry (DEXA) (DEXA scan lunar DPX-NT 2013 made in USA by General Electric) and according to the World Health Organization (WHO) criteria for BMD, OP in adult is defined by a T score less than -2.5 and osteopenia is defined by a T score between − 1 and − 2.511,23,24,25.
Statistical analysis
The statistical analysis was performed using Windows 10 SPSS version 22 (IBM SPSS Inc., Chicago, Illinois, USA)) program. Normally distributed data were presented as mean ± standard deviation (SD) and categorical data were expressed as number and percentage. The Student’s t test was performed for continuous variables and categorical variables were compared by the chi-square (χ2) and Fisher's exact tests. Regression analysis was done to predict the independent associated factors that may affect BMD in patients with NAFLD. P value of < 0.05 was considered as statistical significance.
Results
A total of 100 individuals were included in the study. The baseline and laboratory characteristics of individuals with NAFLD (cases) and individuals without NAFLD (controls) is shown in (Table 1).
Our study showed no significant difference between cases and controls regarding to age, sex, smoking, BMI and the baseline and laboratory characteristic of studied groups. Moreover, cases had elevated ALT more than controls with significant P-value 0.003 (Table 1).
Among 50 patients enrolled in the study, 41 (82%) patients had grade I fatty liver and 9 (18%) patients had grade ≥ II (Table 2).
As regarded to factors affecting bone density, the study showed a significant difference in PTH, vitamin D, and serum calcium between cases and controls group (Table 3).
DEXA scan results showed a significant difference between cases (− 2.29 ± 0.4) and controls (− 1.53 ± 0.1) with p value < 0.001 (Table 4).
Our results showed a significant difference in bone density between cases and controls. Moreover, OP was more prevalent in cases than controls group with p value < 0.001. Among the NAFLD group, 19 patients had OP (16 of them had grade I fatty liver and 3 of them had grade ≥ II fatty liver), 28 patients had osteopenia (22 of them had grade I fatty liver and 6 had grade ≥ II fatty liver) 3 patients had normal bone density (Table 5).
In our study, we performed logistic regression model analysis to determine predictors of OP among the NAFLD group. We found among patients with NAFLD, serum vitamin D level was statistically significant predicts OP (OR 0.614; 95% CI 0.348–0.825) (P < 0.001). Also female gender, BMI, bilirubin, serum calcium, serum phosphorus PTH, fasting insulin, and insulin resistance were statistically significant predictors of OP among the NAFLD group (Table 6).
Discussion
Large numbers of cross sectional studies have identified a significant association between NAFLD and low BMD, but this association remains a matter of controversy14,18. One meta-analysis showed no significant difference in BMD between patients with NAFLD and controls26. Another meta-analysis showed NAFLD was associated with osteoporotic fracture, but not associated with low BMD27. However, recent meta-analysis done by Mantovani et al. found that NAFLD was significantly associated with low BMD in children and adolescents28.
Our study showed a significant difference in bone density between patients with NAFLD and controls. Our results agree with Xia et al. who found that patients with NAFLD were significantly associated with low BMD29. Lee et al. reported a positive association between NAFLD and lumbar spine BMD in postmenopausal females17. Moreover, in Shen et al. cohort study showed that males and females patients with NAFLD were associated with increased risk of low BMD30.
Our results showed that osteoporosis was more prevalent in patients with NAFLD compared to controls with p value < 0.001. Moreover among NAFLD group, 19 patients had osteoporosis and 28 patients had osteopenia. Our result is supported by meta-analysis study done by Pan et al.31 who reported that prevalence and risk of OP or osteoporotic fracture was significantly associated with NAFLD group than in controls. Also, Loosen et al.32 reported that incidence of OP was significantly higher in the NAFLD patients (6.4%) compared to controls (5.1%) with p value < 0.001). Moreover, our study agrees with Pirgon et al.33 who indicated that NAFLD has a noxious effect on BMD in adolescents and was correlated with increased insulin resistance. Another cohort study performed by Chen et al.34 showed increase risk of OP 1.35 times in patients with NAFLD than individuals without NAFLD. Also Li et al.16 showed that NAFLD was significantly associated with history of osteoporotic fractures in middle-aged and elderly Chinese men.
Our results can be interpreted in light that factors affecting bone mineral density including serum vitamin D and serum calcium were significantly lower in our studied patients with NAFLD than controls. Our results agree with Targher et al. who reported a potential link between decreased serum vitamin D and low BMD in patients with NAFLD19. Moreover, many studies proved that patients with NAFLD have lower levels of serum vitamin D than controls which lead to decrease BMD and increase risk of fractures35,36,37,38. Our study showed that patients with NAFLD had increased level of PTH compared to controls which indicates that patients with NAFLD had low BMD and OP.
The association between hypovitaminosis D and NAFLD have been found in many diseases such as metabolic syndrome and obesity39,40. But in observational studies like our study, it is difficult to judge if NAFLD is a cause or result to hypovitaminosis D. Moreover, association between hypovitaminosis D and NAFLD could be accidental without actual relations between them. Consequently, the relationship between hypovitaminosis D and NAFLD needs prospective randomized controlled trials to compare development of NAFLD in patients with hypovitaminosis D versus healthy subjects and to assess effect of vitamin D supplementation on regression of NAFLD.
In our study, we have performed logistic regression model analysis to determine predictors of osteoporosis among patients with NAFLD. We found that serum vitamin D levels was a statistically significant predictor for OP among NAFLD patients (OR 0.614; 95% CI 0.348–0.825) (P < 0.001).
Insulin resistance is considerable risk factor for NAFLD4,41 and insulin resistance and high fasting serum insulin may be associated with increased risk of low BMD42.
Although most people with NAFLD have metabolic dysfunction such as diabetes mellitus, but we excluded diabetic patients as diabetes mellitus deteriorates bone strength and increase susceptibility to bone fracture and this could affect results of our study43,44.
In our study, 10 patients had HOMA-IR more than 2, nine of them had decreased BMD (osteoporosis and osteopenia) and only one patient had significant insulin resistant (HOMA-IR more than 2.7). The patient who had HOMA-IR more than 2.7 also had osteopenia.
In our study logistic regression model analysis showed that fasting insulin and insulin resistance were statistical significant predictors of OP among the NAFLD group. Our result agrees Filip et al.8 who found that patients with NAFLD are associated with insulin resistance which is risk factors for low BMD.
Conclusion
Our study showed significant decreased in bone density and osteoporosis in patients with NAFLD compared to controls. Also serum vitamin D and serum calcium were significantly deceased in patients with NAFLD while level of PTH was increased in patients with NAFLD than controls which indicate that patients with NAFLD have potential risk of developing low BMD and OP. Additional further prospective studies are needed to determine the relationship between NAFLD and risk of low BMD and OP.
Study limitations and future recommendations
This study had some limitations. The first limitation was the small sample size so we recommend further studies with large sample size with different risk factors to determine the relationship between NAFLD and the risk of low BMD and osteoporosis. Second, ultrasound had been used to determine whether individuals had NAFLD or not and its degrees, instead of liver biopsy which is the gold standard in diagnosis of fatty liver. Third, minimal steatosis (NAFLD grade 1) might be missed by using ultrasound. Fourth, further studies are recommended to screen BMD in patients with NAFLD who have diabetes mellitus as most patients with NAFLD have metabolic dysfunction as diabetes mellitus or insulin resistance. Moreover, in this study we screened only lumbar spine to detect BMD in patients with NAFLD and individuals without NAFLD and we recommend further studies for screening BMD of other vulnerable sites of fracture such as neck of the femur.
Data availability
Regarding to the datasets used and/or analyzed during the current study, the datasets used and/or analyzed will be available from the corresponding author on reasonable request.
References
Federico, A., Dallio, M., Masarone, M., Persico, M. & Loguercio, C. The epidemiology of non-alcoholic fatty liver disease and its connection with cardiovascular disease: Role of endothelial dysfunction. Eur. Rev. Med. Pharmacol. Sci. 20, 4731–4741 (2016).
Alkassabany, Y. M., Farghaly, A. G. & El-Ghitany, E. M. Prevalence, risk factors, and predictors of nonalcoholic fatty liver disease among schoolchildren: A hospital-based study in Alexandria, Egypt. Arab. J. Gastroenterol. 15, 76–81 (2014).
VanWagner, L. B. & Rinella, M. E. Extrahepatic manifestations of nonalcoholic fatty liver disease. Curr. Hepatol. Rep. 15, 75–85 (2016).
Pan, B., Yan, J., Zhao, P., Liu, J., Fu, S. Relationship between nonalcoholic fatty liver disease and bone mineral density in type 2 diabetic patients. Int. J. Diabetes Dev. Ctries (2021).
Li, A. A., Ahmed, A. & Kim, D. Extrahepatic manifestations of nonalcoholic fatty liver disease. Gut Liver 14(2), 168–178 (2020).
Chung, G. E. et al. Nonalcoholic fatty liver disease is associated with the development of obstructive sleep apnea. Sci. Rep. 11, 13473 (2021).
Mantovani, A. et al. Non-alcoholic fatty liver disease and increased risk of incident extrahepatic cancers: a meta-analysis of observational cohort studies. Gut 71(4), 778–788 (2022).
Cosman, F. et al. Clinician’s guide to prevention and treatment of osteoporosis. Osteoporos. Int. 25(10), 2359–2381 (2014).
Wade, S. W., Strader, C., Fitzpatrick, L. A., Anthony, M. S. & O’Malley, C. D. Estimating prevalence of osteoporosis: Examples from industrialized countries. Arch Osteoporos. 9(1), 182 (2014).
Filip, R., Radzki, R. P. & Bieńko, M. Novel insights into the relationship between nonalcoholic fatty liver disease and osteoporosis. Clin. Interv. Aging 13, 1879–1891 (2018).
Premkumar, K. et al. Bone mineral density assessment in chronic liver disease. Arch Can. Res. 5, 1 (2017).
Guanabens, N. & Pares, A. Osteoporosis in chronic liver disease. Liver Int. 38, 776–785 (2017).
Collier, J. Bone disorders in chronic liver disease. Hepatology 46, 1271–1278 (2007).
Zhai, T., Chen, Q., Xu, J., Jia, X. & Xia, P. Prevalence and trends in low bone density, osteopenia and osteoporosis in U.S. adults with non-alcoholic fatty liver disease, 2005–2014. Front. Endocrinol. 12, 825448 (2022).
Cui, R., Sheng, H., Rui, X. F., Cheng, X. Y., Sheng, C. J., Wang, J. Y., et al. Low bone mineral density in Chinese adults with nonalcoholic fatty liver disease. Int. J. Endocrinol. 396545 (2013).
Li, M. et al. Association between nonalcoholic fatty liver disease (NAFLD) and osteoporotic fracture in middle-aged and elderly Chinese. J. Clin. Endocrinol. Metab. 97, 2033–2038 (2012).
Lee, S. H. et al. Association between bone mineral density and nonalcoholic fatty liver disease in Korean adults. J. Endocrinol. Invest. 39(11), 1329–1336 (2012).
Kaya, M. et al. Increased bone mineral density in patients with non-alcoholic steatohepatitis. World J. Hepatol. 5(11), 627–634 (2013).
Targher, G., Lonardo, A. & Rossini, M. nonalcoholic fatty liver disease and decreased bone mineral density: Is there a link?. J. Endocrinol. Invest. 38, 817–825 (2015).
Muniyappa, R., Lee, S., Chen, H. & Quon, M. J. Current approaches for assessing insulin sensitivity and resistance in vivo: advantages, limitations, and appropriate usage. Am. J. Physiol.-Endocrinol. Metab. 294(1), E15–E26 (2008).
Meyer, C. et al. Different mechanisms for impaired fasting glucose and impaired postprandial glucose tolerance in humans. Diabetes Care 29(8), 190914 (2006).
Saadeh, S. et al. The utility of radiological imaging in nonalcoholic fatty liver disease. Gastroenterology 123, 745–750 (2002).
Zhu, X. et al. Association between non-alcoholic fatty liver disease-associated hepatic fibrosis and bone mineral density in postmenopausal women with type 2 diabetes or impaired glucose regulation. BMJ Open Diabetes Res. Care 8, e000999 (2020).
Uziel, Y., Zifman, E. & Hashkes, P. J. Osteoporosis in children: pediatric and pediatric rheumatology perspective: A review. Pediatr. Rheumatol. 7(16), 1–8 (2020).
The International Society for Clinical Densitometry. ISCD official positions: Adult [Internet]. The International Society for Clinical Densitometry (2013).
Upala, S., Jaruvongvanich, V., Wijarnpreecha, K. & Sanguankeo, A. Nonalcoholic fatty liver disease and osteoporosis: A systematic review and metaanalysis. J. Bone Miner. Metab. 35, 685–693 (2017).
Mantovani, A. et al. Systematic review with meta-analysis: non-alcoholic fatty liver disease is associated with a history of osteoporotic fractures but not with low bone mineral density. Aliment. Pharmacol. Ther. 49, 375–388 (2019).
Mantovani, A. et al. Association between nonalcoholic fatty liver disease and reduced bone mineral density in children: a meta-analysis. Hepatology 70, 812–823 (2019).
Xia, M. F. et al. The association of liver fat content and serum alanine aminotransferase with bone mineral density in middle-aged and elderly Chinese men and postmenopausal women. J. Transl. Med. 14, 11. https://doi.org/10.1186/s12967-016-0766-3 (2016).
Shen, Z. et al. Increased risk of low bone mineral density in patients with non-alcoholic fatty liver disease: a cohort study. Eur. J. Endocrinol. 182, 157–164 (2020).
Pan, B. et al. Relationship between prevalence and risk of osteoporosis or osteoporotic fracture with non-alcoholic fatty liver disease: A systematic review and meta-analysis. Osteoporos. Int. 33, 2275–2286 (2022).
Loosen, S. H. et al. Non-alcoholic fatty liver disease is associated with increased incidence of osteoporosis and bone fracture. Z. Gastroenterol. 60(08), 1221–1227 (2022).
Pirgon, O., Bilgin, H., Tolu, I. & Odabas, D. Correlation of insulin sensitivity with bone mineral status in obese adolescents with nonalcoholic fatty liver disease. Clin. Endocrinol. 75, 189–195 (2020).
Chen, H. et al. Increased risk of osteoporosis in patients with nonalcoholic fatty liver disease: A population-based retrospective cohort study. Medicine 97, e12835 (2018).
Lips, P. Vitamin D deficiency and secondary hyperparathyroidism in the elderly: Consequences for bone loss and fractures and therapeutic implications. Endocr. Rev. 22(4), 477–501 (2001).
Shawky, M. A., Hassan, A. M., Mohammed, A. Q. & Oraby, M. I. Vitamin D levels in egyptian patients with non-alcoholic fatty liver disease. J. Gastroenterol. Hepatol. Res. 7(1), 2530–2534 (2018).
Wang, X., Li, W., Zhang, Y., Yang, Y. & Qin, G. Association between vitamin D and non-alcoholic fatty liver disease/non-alcoholic steatohepatitis: Results from a meta-analysis. Int. J. Clin. Exp. Med. 8, 17221–17234 (2015).
Eliades, M. et al. Meta-analysis: Vitamin D and non-alcoholic fatty liver disease. Aliment. Pharmacol. Ther. 38(3), 246–254 (2013).
Black, L. J. et al. Low serum 25-hydroxyvitamin D concentrations associate with nonalcoholic fatty liver disease in adolescents independent of adiposity. J. Gastroenterol. Hepatol. 29, 1215–1222 (2014).
Yang, K. C. et al. Association of non-alcoholic fatty liver disease with metabolic syndrome independently of central obesity and insulin resistance. Sci. Rep. 6, 27034. https://doi.org/10.1038/srep27034 (2016).
Gaggini, M. et al. Non-alcoholic fatty liver disease (NAFLD) and its connection with insulin resistance, dyslipidemia, atherosclerosis and coronary heart disease. Nutrients 5, 1544–1560 (2013).
Shin, D., Kim, S., Kim, K. H., Lee, K. & Park, S. M. Association between insulin resistance and bone mass in men. J. Clin. Endocrinol. Metab. 99, 988–995 (2014).
Farr, J. N. & Khosla, S. Determinants of bone strength and quality in diabetes mellitus in humans. Bone 82, 28–34 (2016).
Wongdee, K. & Charoenphandhu, N. Osteoporosis in diabetes mellitus: Possible cellular and molecular mechanisms. World J. Diabetes 2(3), 41–48 (2011).
Funding
Open access funding provided by The Science, Technology & Innovation Funding Authority (STDF) in cooperation with The Egyptian Knowledge Bank (EKB).
Author information
Authors and Affiliations
Contributions
H.A.M. create the research idea. H.A.M. and A.-G.M. designed the study. H.A.M., A.-G.M., S.M.Z., and H.M.A., A.T.M., perform the clinical examination. A.M.M., E.S.S., A.W.E. performed the laboratory analysis. M.T.M. performed the ultrasound examination. S.M.Z. and A.-A.T.M. performed the DEXA scan analysis. H.A.M., A.-G.M., S.M.Z., and H.M.A., A.T.M., A.M.M., E.S.S., A.W.E., M.T.M., Q.M.K., and K.E.M. shared in analyzing and interpreting the patient data and in writing the manuscript drafting. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Hassan, A.M., Haridy, M.A., Shoaeir, M.Z. et al. Non-alcoholic fatty liver disease is associated with decreased bone mineral density in upper Egyptian patients. Sci Rep 13, 4353 (2023). https://doi.org/10.1038/s41598-023-31256-w
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-023-31256-w