Abstract
In the fast-evolving realm of communication technology, microstrip patch antennas (MPAs) are in high demand owing to their compact size, lightweight, inexpensive, ease of integration, and compatibility with modern electronic devices. This research focuses on the synthesis of ZnAl2O4Ca (ZAC) ceramic nanoparticles using an economical sol–gel method suitable for microstrip patch antenna applications. The structural analysis study of ZAC nanoparticles confirmed the polycrystalline nature with 8.1 nm of crystallite size whereas an investigation of functional groups showed the corresponding vibration modes. Morphological investigation revealed the spherical grains having their mean diameter of 12.32 nm. The dielectric property’s examination, revealed the dielectric permittivity of 13, loss tangent of 0.02, and conductivity of 67 μΩ−1 cm−1. Furthermore, a prototype patch antenna fabricated using ZAC ceramics demonstrated a dual-band performance at frequencies 2.8 GHz and 4.8 GHz, with return losses of − 25.78 dB and − 28.5 dB, respectively. This work suggests the suitability of ZAC ceramic nanoparticles for use in WLAN applications.
Similar content being viewed by others
Introduction
Due to increasing demand and rapid advancements in real-time communication systems, multiband antennas with wide bandwidth are becoming increasingly favorable due to their ability to provide high radiation patterns. The primary objective of multiband antennas is to ensure minimal interference with other frequency bands. In the ever-expanding landscape of wireless communication technology, certain key attributes are highly required. These include low-profile, lightweight construction, and the improvement of band designations such as the S-band from 2 to 4 GHz, C-band from 4 to 8 GHz for, Wireless LAN (WLAN) from 2.4 to 5 GHz, WiFi operating at 2.4 GHz, and X-band covering the 8–12 GHz range1. The crucial parameters for describing the microwave characteristics of a communication system include a high-quality factor (Qxf) helpful to wideband designations, an appropriate permittivity (εr) to facilitate miniaturization, and a temperature coefficient of resonant frequency (τf) to ensure the stability and reliability of microwave devices. Microwave communication technology has impacted cellular mobile communication, television broadcasting, radar technology, WiFi modules, signal tracking systems, cavity resonators, filters, isolators, circulators, and signal jamming systems. The primary factor behind this progress can be attributed to the utilization of microwave dielectric ceramics2.
Microwave-absorbing materials are gaining significant attention for their dual role in mitigating electromagnetic pollution in civilian settings and enhancing stealth capabilities in military applications. Researchers explored synthesis and applications of various nanomaterials such as CuCr2O4, La-substituted CuFe2O4, CuFe2O4, and ZnAl2O4, to enhance microwave absorption and reflection loss3,4,5,6. Additionally, studies on capped BaFe2O4 nanoparticles and expanded-carbon microspheres blended with ZnAl2O4 nanoparticles demonstrated an excellent absorption property, suggesting their potential for practical applications7,8. Presently, magnetic ferrites are highly valued for their properties such as high resistivity, moderate permeability, and low electric and magnetic losses, making them crucial across various applications. In this context, researchers highlighted the potential of Cu-substituted Ni–Zn nanocrystalline ferrites for industrial and domestic applications, while another work on CoSm ferrite-metamaterial absorbers evidenced promising electromagnetic features with potential applications in satellite communication9,10. Furthermore, a study on Gd–Ce-substituted garnet ferrites, Al-doped spinel and garnet nanoferrites showed potential for industrial and technological applications in different frequency bands11,12.
ZnAl2O4 nanoparticles possess exceptional electrical, mechanical, and optical properties, making them ideal for microwave applications. Researchers explored the applications of doped ZnAl2O4 nanoparticles for the enhanced thermal stability, mechanical strength, band gap, electrical properties and so on. Furthermore, synthesis approach such as the sol–gel method is a low-temperature and straightforward one. This technique yields nanoparticles with their uniform size distribution and allows ease incorporation of small-sized dopants13,14. Numerous researchers have undertaken the synthesis, characterization, and investigation of ceramic nanoparticles. For instance, Yin et al. employed the solid-state route method to synthesize Ba2CuGe2O7 ceramic nanoparticles doped with Mg. They investigated the microwave dielectric properties of Ba2CuMgGe2O7 sintered at 900 °C, and reported, εr = 9.43 and τf = -76 ppm/°C along with Qxf = 20,000 GHz15. Wu et al. synthesized and characterized, Li2WO4 and composite Ba3Ti4Nb4O2 microwave dielectric ceramic nanoparticles using the solid-state reaction method. The ceramic material exhibited, εr = 21.85 and τf = − 7.78 ppm/°C along with Qxf = 10,534 GHz16. Jiamao et al. adopted the stearic acid method to synthesize NdAlO3 ceramic nanoparticles at low temperature. They obtained εr = 23.02, Qxf = 65,320 GHz, and τf = − 32.4 ppm/°C17. Similarly, Mengjuan et al. prepared (1 − x)CoTiNb2O8-xZnNb2O6 ceramic material via the conventional solid-state reaction route to mitigate τf value. This material revealed, εr = 39.2, Qxf = 40,013 GHz, τf = 3.57 ppm/°C18. In another study, Jiamao et al. employed the stearic acid route method to synthesize SmAlO3 and examined its dielectric properties. By sintering at 1500 °C for 4 h, they achieved a consistent microstructure with an average grain size of 1.8 μm. The measured values of dielectric parameters were εr = 20.22, Qxf = 74,110 GHz, and τf = − 74.6 ppm/°C19. Likewise, Hsiang et al. explored the preparation of low-temperature co-fired Ca0.7Nd0.2TiO3 ceramic material incorporating CaO-B2O3-SiO2. Their investigation revealed εr and Qxf values of 22.8 and 2380 GHz, respectively20. Further, Rahman et al. employed the sol–gel method to synthesize CoxZn(0.90−x)Al0.10Fe2O4 ceramics for MPA applications. They observed the variation in crystallite size, dielectric permittivity, and tangential loss from 24.45 to 20.84 nm, 3.5 to 4.5, and 0.002 to 0.006, respectively with an increased Co concentration from 20 to 60%. The antenna of this composite exhibited a return loss (RL) varying from − 35 to − 26 dB, with a bandwidth of 2.20–2.5 GHz, at a resonant frequency (Fr) between 4.6 and 4.0 GHz21. Borah et al. employed magnetodielectric composites doped with cobalt ferrite for MPA applications whose average grain size was 10 nm. The real part of the dielectric permittivity and permeability were found to vary from 1 to 2.905 and 1.01 to 1.05, respectively, with an increased concentration from 1% volume fraction to 5% volume fraction. Additionally, they fabricated MPA, which exhibited an RL of − 19.45 dB at the resonance frequency of 9.5 GHz22. In a similar fashin, Ashish et al. prepared a miniaturized MPA using barium hexaferrite (BaFe12O19) through the co-precipitation method. They reported εr = 6.2 and εo = 1.9, with an average crystallite size of 60 nm. They subsequently constructed MPA with BaFe12O19, which showed its RL of − 25 dB at Fr of 4.25 GHz with a bandwidth of 166 MHz23. In other study, the same group reported the synthesis of Ni0.5Zn0.3Co0.2In0.1Fe1.9O4 nanoparticles for miniaturized microstrip antenna application and also investigated dielectric permittivity and permeability values of 5.3 and 5.5, respectively. The fabricated antenna demonstrated a remarkable RL of − 42 dB at the resonance frequency of 450 MHz24. Bhongale at el. fabricated Mg–Nd–Cd ferrite-based MPA prepared by the co-precipitation method having a grain size of 1.37 µm. The dielectric permittivity, permeability, and tangential loss were measured to be 3.18, 1.29, and 0.2, respectively. The fabricated MPA exhibited a return loss of − 19.16 dB at a frequency of 9.35 GHz, with a voltage standing wave ratio (VSWR) of 1.2625.
Among above-discussed materials, doped ZnAl2O4 is promising for increasing its dielectric permittivity, which facilitates the miniaturization of patch antennas, resulting in compact antenna designs suitable for integration into various electronic devices, such as smartphones, tablets, and wearable technology. Additionally, it reduces the dielectric loss indicating less signal energy lost in the form of heat within the substrate. This mechanism is fruitful in enhancing the antenna’s efficiency and minimizing signal distortion. Furthermore, Ca doping can be beneficial for tuning the thermal expansion coefficient of ZnAl2O4 so that thermal stresses which causes performance degradation can also be minimized. In this manner, doped zinc aluminate, as substrate material, exhibits excellent thermal, electrical, and chemical stability. This makes antennas fabricated with it suitable for harsh operating environments, including outdoor and industrial settings.
In this study, we synthesize Zn0.988Al2O4Ca0.012 ceramic nanoparticles using the sol–gel method and present a comprehensive study of various characteristics, including structural, morphological, and dielectric properties. Additionally, we fabricated a prototype MPA, which exhibited remarkable return loss values in both the S-band and C-band frequencies, measured to be − 25.78 dB and − 28.5 dB, respectively. Consequently, our research highlights the suitability of the prepared ceramic nanoparticles for dual-band MPA applications, unambiguously for the S-band and C-band. Notably, there is no prior similar work reported in the literature, particularly in the context of the same material and its application for dual-band communication, like WLAN. “Materials and Methods” Section presents the materials and methods employed in this study. A comprehensive study of results is discussed in “Results and Discussion” Section. Lastly, “Conclusions” Section reviews the findings.
Materials and methods
For the synthesis of ZnAl2O4Ca ceramic nanoparticles, various chemicals including C2H5OH (Ethanol, Sigma-Aldrich), Al(NO3)3.9H2O (Aluminum Nitrate Nonahydrate, purity of 99.99%, Sigma-Aldrich, C2H6O2 (Ethylene Glycol, SdFine), Zn(O2CCH3)2.2H2O (Zinc Acetate Dihydrate, purity of 99.99%, Lobychem), Ca(NO3)2H8O4 (Calcium Nitrate Tetrahydrate, purity of 99.99%, Sigma-Aldrich, and HNO3 (Nitric Acid, Lobychem) were used as purchased.
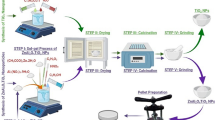
To prepare Ca doped zinc aluminate (Zn(1−x)Al2O4-Cax) ceramic nanoparticles with molar concentration x = 0.012, the following synthesis procedure was employed, and the prepared sample named as ‘ZAC’ throughout the manuscript. The synthesis procedure is illustrated in Fig. 1. Initially, 60 ml ethanol combined with 22.5 gm aluminum nitrate nonahydrate was stirred to get dissolved solution. Subsequently, 0.49 ml ethylene glycol and 10.87 gm zinc acetate were introduced into the solution. After stirring for 5 min, we added 0.17 gm calcium nitrate tetrahydrate under stirring at 75 °C for 1 h. Following this, we introduced 0.36 ml nitric acid into the solution and later kept it for drying at 180 °C for 1 h. The dried powder was further processed by calcining it in a furnace for 2 h at 900 °C and then the powder was finely ground to get fine ZAC nanoparticles.
The process of preparation of ZAC ceramic nanoparticles.
The prepared nanoparticles were examined using various techniques: X-ray Diffractometer (XRD, Bruker D8, Venture, Germany) for probing the crystallographic structure, Fourier-Transform Infrared Spectroscopy (FTIR, Perkin Elmer Spectrum Two, US) for analyzing the chemical composition, Transmission Electron Microscopy (TEM, Talos F200S G2, US) coupled with Energy-dispersive X-ray spectroscopy (EDS) for studying the morphology and compositional elements in the sample, LCR Meter (PSM 1735 N4L, Newtons 4th Limited, UK) for evaluating the electrical properties of the ZAC pellet (having diameter of 10 mm and thickness of 1.5 mm), and Vector Network Analyzer (VNA, R&S ZVL, Germany) for measuring the scattering parameters of the microstrip patch antenna.
Results and discussion
Figure 2a depicts the Rietveld refinement plot of the XRD pattern of ZAC ceramic nanoparticles, which revealed its polycrystalline characteristics. The intensity peaks, indexed at 2θ values of 31.49°, 36.8°, 44.9°, 47.7°, 49.0°, 55.68°, 56.8°, and 59.28° were attributed to the crystal planes of (220), (311), (400), (331), (331), (422), (422), and (511), respectively, which correspond to the characteristics of ZnAl2O4. Additionally, another peak observed at 2θ = 34.65° was attributed to the ZnO plane of (101).
Rietveld refinement plot of the XRD pattern of ZAC ceramic nanoparticles (a), representation of crystal structure of undoped (b) and Ca-doped ZnAl2O4 (c).
These characteristic peaks in the XRD pattern closely matched those found in #JCPDS Card Nos. 005-0669 and 89-0510. The crystallite size was calculated to be 8.1 nm using Scherer’s equation. Figure 2b,c illustrate the visualization of crystal structures of undoped and Ca-doped ZnAl2O4 obtained using the Vesta package.
The crystal structure of undoped ZnAl2O4 shown in Fig. 2b shows the presence of individual elements such as Zn, Al and O, while an additional element, Ca (dopant) is evident in the crystal structure of Ca–ZnAl2O4 as depicted in Fig. 2c. Table 1 summarizes the crystal structure parameters obtained from the Rietveld refinement analysis. It revealed the unit cell parameters for gahnite as a = b = c = 8.07098 Å, and for zincite, a = b = 3.24170 ≠ c = 5.18760 Å. The cell volume for gahnite was noticed to be 525.7494 (Å)3, and 47.2110 (Å)3 for zincite.
Figure 3 depicts the FTIR spectrum of Ca-doped ZnAl2O4 ceramic nanoparticles in the wave number range of 400–4000 cm−1. Several vibrational peaks were evident at various wavenumbers, specifically at 492, 560, 667, 1209, 1386, 1639, 2328, 2813, 3043, and 3424 cm−1, corresponding to different functional groups. The peaks at 3424, 3043, 1639, and 1386 cm−1 were found associated with the stretching vibration modes of O–H, contributed by the water molecules. Another stretching peaks, resulting from the O–O bond observed at 2813 and 2328 cm−1 were found associated with the FCC crystal lattice of oxygen atoms. Another peak noticed at 1209 cm−1 was assigned to Al–O–H indicating stretching vibration mode. In the low-frequency region below 1000 cm−1, peaks originating at 8667, 560, and 492 cm−1 were found associated with the mineral network. This complex region can be attributed to the stretching frequencies of aluminum-oxygen (Al-O) and metal–oxygen-aluminum (M–O–Al) bonds. Specifically, the band 492 cm−1 corresponds to the spinel characteristics of ZnAl2O4 and originated due to the Zn–O–Al, Al–O, and Zn–O modes27,28,29,30,31.
FTIR spectrum of ZAC ceramic nanoparticles.
The morphology of ZAC nanoparticles was examined using TEM. Figure 4 illustrates TEM and HRTEM micrographs along with an inverse FFT lattice and line-profile analyses, SAED pattern, and EDS spectrum. In Fig. 4a, a TEM micrograph of the prepared sample revealed spherical grains arranged with their clear boundaries. An inset histogram shows the distribution of grains. The estimated mean diameter of the grains was 12.32 nm. In Fig. 4b, a high-resolution TEM image provides a detailed view of the d-spacing planes with marked rectangular boxes. We performed the inverse FFT process of the lattices and conducted line-profiling, as depicted in Fig. 4c–f. The obtained d-spacing values were 0.24 nm and 0.28 nm, corresponding to ZnAl2O4 planes (311) and (220) along with 0.26 nm of ZnO plane (002). Figure 4g shows the selected area electron diffraction (SAED) pattern, confirming the co-centric solid rings that represent the polycrystalline nature of the ZAC nanoparticles. This SAED pattern aligns well with the XRD pattern depicted in Fig. 2a. The elemental composition of ZAC analyzed by EDS is depicted in Fig. 4h. This spectrum reveals elemental peaks corresponding to Zn, Al, O, and Ca, identified at energy levels of 8.6 keV, 1.49 keV, 0.5 keV, and 4.1 keV, respectively.
TEM micrograph with a histogram (a), HRTEM image (b) line-profile with an inset inverse FFT (c–f), SAED pattern (g) and EDS spectrum (h).
The comparison of obtained structural and morphological parameters with previously reported literature is presented in Table 2.
The study of real and imaginary values of dielectric permittivity is significant in analyzing the behavior of ZAC compound. The real part of it signifies energy storage capability, whereas the imaginary part indicates energy dissipation by the material. We have performed both analyses as plotted in Fig. 5 which illustrates the real and imaginary dielectric permittivity of ZAC over a frequency range from 3 to 10 MHz.
Dielectric permittivity (real and imaginary) of ZAC ceramic nanoparticles.
The dielectric permittivity was estimated using εr’ = Ct/(εoS), where εr’ represents the relative dielectric permittivity, C is the capacitance, t is its thickness, εo is the free space dielectric permittivity, and S is the surface area of the sample. From this figure, it is evident that the dielectric permittivity decreases as the frequency rises. This behavior aligns with the principles of the Maxwell–Wagner model and Koop's phenomenological theory. Specifically, the real dielectric permittivity was observed to be 13.75 at 3 MHz and 13 at 10 MHz. The imaginary dielectric permittivity decreases as the frequency increases. This phenomenon is attributed to polarization resonance. At lower frequencies, polarization resonance requires more energy, leading to higher energy absorption. Conversely, at higher frequencies, resonance occurs more easily with lower polarization, resulting in less energy absorption. This behavior aligns with the principle underlying the observed changes in the imaginary dielectric permittivity.
The imaginary dielectric permittivity of the ZAC ceramic nanoparticles was determined to be 0.62 and 0.32 at frequency 3 and 10 MHz, respectively using the formula εr’’ = εr’ tanδ, where εr’ represents the real dielectric permittivity and tanδ represents the dielectric loss. For the study of dielectric properties, the selection of this frequency range was based on the frequency limitation of the LCR meter. However, the goal of this investigation was to observe the typical dielectric behavior of ZAC nanoparticles.
Dielectric loss (tanδ), arises when the electric field remains constant with the polarization of dielectric materials. Figure 6 illustrates the dielectric loss of the ZAC sample. Due to domain wall resonance, the dielectric loss decreases at higher frequencies. The tangent loss values were 0.045 and 0.024, corresponding to the frequency values 3 and 10 MHz.
The dielectric loss of ZAC ceramic nanoparticles.
Figure 7 illustrates the conductivity of the ceramic nanoparticles over a frequency range from 3 to 10 MHz. Conductivity was determined using σ = εrεoωtanδ, where ω is the angular frequency, εr is the relative dielectric constant, εo is the dielectric constant for free space, and tanδ is the dielectric loss. An observed trend in Fig. 7 shows that conductivity increases with higher frequencies. This behavior can be attributed to the migration of electrons. Grain boundaries act as potential barriers opposing the movement of charged particles, in line with the Maxwell–Wagner interface model. At higher frequencies, electrons have sufficient energy to overcome the barrier potential, leading to higher conductivity37,38. The calculated conductivity values were 21.84 and 67.33 µm at frequencies 3 and 10 MHz, respectively.
Conductivity of ZAC ceramic nanoparticles.
Figure 8 provides an overview of the preparation process for the prototype antenna using ZAC ceramic nanoparticles. Initially, ceramic nanoparticles paste was applied onto the FTO (Fluorine-doped Tin Oxide) plate using the doctor blade method. This paste was then thermally treated to ensure adhesiveness and solidification processes. A layer of silver was applied over the previously prepared ZAC ceramic patch and the bottom of the substrate. This step is crucial to create a closed electrical circuit. In the next step, an SMA connector was connected and bonded to the antenna to enhance electrical conductivity, which is essential for antenna performance. Finally, a prototype MPA underwent performance testing.
Process illustrating the preparation of ZAC ceramic nanoparticles.
The return loss of an antenna quantifies the extent to which a signal is reflected back to the employed source, resulting from an impedance mismatch between the antenna and the transmission line. In other words, it tells us how much of the signal fed to the antenna is lost or reflected back instead of being transmitted. Figure 9 shows RL and VSWR characteristics of a prototype MPA prepared using ZAC ceramic nanoparticles in the frequency range from 2 to 6 GHz. This antenna exhibits two distinct resonant peaks, corresponding to the S-band and C-band frequencies. In specific, the return loss for the S-band was measured at − 25.78 dB, occurring at a resonant frequency of 2.8 GHz with a bandwidth of 1.2 GHz. For the C-band, the return loss was − 28.5 dB, resonating at 4.8 GHz with a bandwidth of 800 MHz.
RL and VSWR of ZAC ceramic nanoparticles based microstrip patch antenna.
These results demonstrate that the prepared ZAC material is well-suited for the fabrication of a dual-band suitable for both S- and C-band applications. A comparative study of dielectric properties of ceramics and microstrip patch antenna is shown in Table 3. Our antenna showed better performance as compared to others, which suggests the suitability of the prepared ZAC nanoparticles.
VSWR is a measure of how effectively an antenna transfers power to the load. It is calculated using the equation \(S=((|1+\rho |))/((|1-\rho |))\), where S is the standing wave ratio and ρ is the reflection coefficient. A low VSWR value signifies efficient power transfer from the antenna to the load, while a high VSWR value indicates poor power transfer and increased signal reflections. Ideally, a VSWR value close to 1 indicates a nearly perfect match between the antenna and the transmission line, resulting in minimal signal reflection. Figure 9 depicts the VSWR curve of the MPA plotted by varying frequency from 2–6 GHz. The VSWR values obtained were 1.1 for the S-band and 1.0 for the C-band resonant frequencies. These VSWR values suggest that the MPA is operating efficiently at both the S-band and C-band frequencies, indicating a well-matched antenna system with minimal power reflection.
MPAs possess numerous advantages, making them an excellent choice for designing and fabricating dual-band antennas. WLAN technology seamlessly integrates wireless communication into computer networks, utilizing the 2.4 GHz and 5 GHz spectrum within the Industrial Scientific Medical (ISM) band. The ability to use these frequency bands without requiring authorization has spurred the widespread adoption and application of WLAN, particularly due to advancements in wireless communication technology. It serves as a prevalent communication band in devices such as cell phones, smart televisions, computers, and laptops. This technology's user-friendly nature and its compatibility with printed MPA designs make it even more appealing43. Moreover, a dual-band MPA can be tailored to operate across two distinct frequency bands. This allows both communication systems to utilize the same antenna, leading to space savings and cost reduction. Nevertheless, fabricating a dual-band MPA that meets the requirements of both frequency bands poses a significant challenge, as the antenna must perform effectively across both frequency ranges2. In our case, the resonant frequencies closely coincide with those of WLAN antennas. This emphasizes the potentiality for further optimization of the antenna fabrication to meet the frequencies employed in WLAN applications.
Conclusions
In this study, Ca-doped ZnAl2O4 dielectric ceramic nanoparticles were synthesized and analyzed. Materials characterization revealed the polycrystalline characteristics of the nanoparticles with a crystallite size of 8.1 nm, the presence of functional groups associated with the prepared ceramic compound and the spherical grains of ZAC with a mean diameter of 12.32 nm. Further, investigation of dielectric properties showed a decrease in dielectric permittivity and tangent loss along with enhanced conductivity with the rise in frequency. Finally, the fabricated prototype microstrip patch antenna demonstrated dual-band performance, with return losses of − 25.78 dB at 2.8 GHz (S-band) and − 28.5 dB at 4.8 GHz (C-band). VSWR values were measured to be 1.1 and 1.0 for the resonant frequencies of 2.8 GHz and 4.8 GHz, respectively. Our findings suggest the potential of Ca-doped ZnAl2O4 ceramic nanoparticles as alternate substrate material for fabricating dual-band microstrip patch antenna in WLAN applications.
Data availability
All data generated or analyzed during this study are presented in this manuscript and can be obtained upon reasonable request from the corresponding author.
Change history
11 July 2024
A Correction to this paper has been published: https://doi.org/10.1038/s41598-024-66239-y
References
Hasan, M. M., Rahman, M., Faruque, M. R. I. & Islam, M. T. Bandwidth enhanced metamaterial embedded inverse L-slotted antenna for WiFi/WLAN/WiMAX wireless communication. Mater. Res. Expr. https://doi.org/10.1088/2053-1591/ab261a (2019).
Hasan, M. M., Faruque, M. R. I. & Islam, M. T. Dual band metamaterial antenna for LTE/bluetooth/WiMAX system. Sci. Rep. 8(1), 1240. https://doi.org/10.1038/s41598-018-19705-3 (2018).
Peymanfar, R. & Khodamoradipoor, N. Preparation and characterization of copper chromium oxide nanoparticles using modified sol–gel route and evaluation of their microwave absorption properties. Phys. Status Solidi https://doi.org/10.1002/pssa.201900057 (2019).
Peymanfar, R. & Azadi, F. La-substituted into the CuFe2O4 nanostructure: A study on its magnetic, crystal, morphological, optical, and microwave features. J. Mater. Sci.: Mater. Electron. https://doi.org/10.1007/s10854-020-03501-9 (2020).
Keykavous-Amand, S. & Peymanfar, R. Fabrication of clay soil/CuFe2O4 nanocomposite toward improving energy and shielding efficiency of buildings. Sci. Rep. https://doi.org/10.1038/s41598-021-00347-x (2021).
Peymanfar, R. & Rahmanisaghieh, M. Preparation of neat and capped BaFe2O4 nanoparticles and investigation of morphology, magnetic, and polarization effects on its microwave and optical performance. Mater. Res. Expr. 5(10), 105012. https://doi.org/10.1088/2053-1591/aadaac (2018).
Peymanfar, R. & Fazlalizadeh, F. Evaluating morphological and interfacial influence on microwave characteristics using ZnAl2O4. Chem. Eng. J. https://doi.org/10.1016/j.cej.2020.126089 (2020).
Peymanfar, R. & Fazlalizadeh, F. Fabrication of expanded carbon microspheres/ZnAl2O4 nanocomposite and investigation of its microwave, magnetic, and optical performance. J. Alloys Compd. https://doi.org/10.1016/j.jallcom.2020.157273 (2021).
Akhtar, M. N. et al. Evaluation of structural, morphological and magnetic properties of CuZnNi (CuxZn0.5−xNi0.5Fe2O4) nanocrystalline ferrites for core, switching and MLCI’s applications. J Magn. Magn. Mater. https://doi.org/10.1016/j.jmmm.2016.08.035 (2017).
Akhtar, M. N. et al. Enhanced structural, electromagnetic and absorption features of CoSm ferrite-metamaterial absorbers through synergistic effects. Ceram. Int. 48(20), 29561–29571. https://doi.org/10.1016/j.ceramint.2022.06.208 (2022).
Akhtar, M. N. et al. Impacts of Gd–Ce on the structural, morphological and magnetic properties of garnet nanocrystalline ferrites synthesized via sol–gel route. J. Alloys Compd. https://doi.org/10.1016/j.jallcom.2015.11.146 (2016).
Akhtar, M. N., Saleem, M. & Khan, M. A. Al doped spinel and garnet nanostructured ferrites for microwave frequency C and X- band applications. J. Phys. Chem. Solids 123, 260–265. https://doi.org/10.1016/j.jpcs.2018.08.007 (2018).
Ismail, S. N., Ali, E. M., Alwan, B. J. & Abd, A. N. Potassium chloride nanoparticles: Synthesis, characterization, and study the antimicrobial applications. Macromol. Symp. 401, 2100312. https://doi.org/10.1002/masy.202100312 (2022).
Mohanty, P., Mohapatro, S., Mahapatra, R. & Mishra, D. K. Low cost synthesis route of spinel ZnAl2O4. Mater. Today: Proc. https://doi.org/10.1016/j.matpr.2020.03.508 (2020).
Changzhi, Y. I. N. et al. A low-firing melilite ceramic Ba2CuGe2O7 and compositional modulation on microwave dielectric properties through Mg substitution. J. Adv. Ceram. 10(1), 108–119. https://doi.org/10.1007/s40145-020-0424-3 (2021).
Xiaohui, Wu. & Hua, Su. Li2WO4 composite Ba3Ti4Nb4O21 microwave dielectric ceramics with near-zero temperature coefficient and low-temperature sintering for 5G communication devices. J. Phys.: Conf. Ser. 2519, 012050. https://doi.org/10.1088/1742-6596/2519/1/012050 (2023).
Li, J., Zhang, C., Li, L. & Fan, C. Synthesis of NdAlO3 with good microwave dielectric properties by stearic acid method. Ceram. Int. 46(11), 18940–18947. https://doi.org/10.1016/j.ceramint.2020.04.217 (2020).
Wu, M., Zhang, Y. & Xiang, M. Synthesis, characterization and dielectric properties of a novel temperature stable (1–x)CoTiNb2O8−xZnNb2O6 ceramic. J. Adv. Ceram. 8(2), 228–237. https://doi.org/10.1007/s40145-018-0308-y (2019).
Li, J., Zhang, C., Liu, H., Qiu, T. & Fan, C. Structure, morphology, and microwave dielectric properties of SmAlO3 synthesized by stearic acid route. J. Adv. Ceram https://doi.org/10.1007/s40145-020-0394-5 (2020).
Hsianga, H. I., Chen, C.-C. & Yang, S.-Y. Microwave dielectric properties of Ca0.7Nd0.2TiO3 ceramic-filled CaO–B2O3–SiO2 glass for LTCC applications. J. Adv. Ceram. 8(3), 345–351. https://doi.org/10.1007/s40145-019-0316-6 (2019).
Rahman, M. A. et al. Magnetic, dielectric and structural properties of CoxZn(0.90–x)Al0.10Fe2O4 synthesized by sole–gel method with application as flexible microwave substrates for microstrip patch antenna. J. Mater. Res. Tech. 16, 934–943 (2021).
Borah, K. & Bhattacharyya, N. S. Magnetodielectric composite with ferrite inclusions as substrates for microstrip patch antennas at microwave frequencies. Compos. Part B: Eng. 43(3), 1309–1314. https://doi.org/10.1016/j.compositesb.2011.11.067 (2012).
Saini, A., Thakur, A. & Thakur, P. Miniaturization and bandwidth enhancement of a microstrip patch antenna using magneto-dielectric materials for proximity fuze application. J. Electron. Mater. 46(3), 1902–1907. https://doi.org/10.1007/s11664-016-5256-0 (2017).
Saini, A., Thakur, A. & Thakur, P. Matching permeability and permittivity of Ni0.5Zn0.3Co0.2In0.1Fe1.9O4 ferrite for substrate of large bandwidth miniaturized antenna. J. Mater. Sci.: Mater. Electron. 27(3), 2816–2823. https://doi.org/10.1007/s10854-015-4095-8 (2016).
Bhongale, S. R. Mg–Nd–Cd ferrite as substrate for X-band microstrip patch antenna. J. Magn. Magn. Mater. https://doi.org/10.1016/j.jmmm.2019.165918 (2019).
Didde, S., Dubey, R. S. & Panda, S. K. et al. Experimental study of doped zinc aluminate nanoparticles by bottom-up approach for microstrip patch antenna applications. J. Mater. Sci. 57, 21069–21079. https://doi.org/10.1007/s10853-022-07929-8 (2022).
Zhang, D. et al. The improvement of structure and photoluminescence properties of ZnAl2O4:Cr3+ ceramics synthesized by using solvothermal method. Mater. Des. 115, 37–45. https://doi.org/10.1016/j.matdes.2016.11.034 (2017).
Miron, I., Enache, C., Dabici, A. & Grozescu, I. Characterization and luminescence properties of ZnAl2O4:Tb3+ (2.5%) phosphor. J. Optoelectron. Adv. Mater. 14(9–10), 820–825 (2012).
Jamal, E. M. A., Kumar, D. S. & Anantharaman, M. R. On structural, optical and dielectric properties of zinc aluminate nanoparticles. Bull. Mater. Sci. 34(2), 251–259. https://doi.org/10.1007/s12034-011-0071-y (2011).
Lu, Q. et al. Electronic structure and optical properties of spinel structure Zn1−xNixAl2O4 nanopowders synthesized by sol–gel method. Chem. Phys. Lett. https://doi.org/10.1016/j.cplett.2021.138582 (2021).
Abd-Allah, A. A., Amin, A. M. M., Youssef, A. O. & Ahmed, Y. M. Z. Fabrication of zinc aluminate (ZnAl2O4) nanoparticles from solid industrial wastes. Egypt. J. Pure Appl. Sci. 60(2), 14–26 (2022).
Bhongale, S. R., Ingavale, H. R., Shinde, T. J. & Vasambekar, P. N. Effect of microwave sintering on synthesis and structural properties of nanocrystalline Mg–Cd ferrites. J. Mater. Sci.: Mater. Electron. 28(15), 11070–11077. https://doi.org/10.1007/s10854-017-6891-9 (2017).
Huang, S., Wei, Z., Wu, X. & Shi, J. Optical properties and theoretical study of Mn doped ZnAl2O4 nanoparticles with spinel structure. J. Alloys Compd. https://doi.org/10.1016/j.jallcom.2020.154004 (2020).
Rahman, M. A., Faruque, M. R. I. & Islam, M. T. Preparation of flexible substrate for patch antenna based on nickel aluminate (NiAl2O4) synthesized by sol–gel method. J. Electron. Mater. https://doi.org/10.1007/s11664-019-07041-9 (2019).
Meijie, D. I. N. G. et al. Influence of Cr and Mn co-doping on the microstructure and optical properties of spinel structured Zn0.95–xCr0.05MnxAl2O4 nanoparticles. J. Ceram. Soc. Jpn. 128(11), 927–935. https://doi.org/10.2109/jcersj2.20029 (2020).
Huang, S.-P., Wei, Z.-Q., Wu, X.-J. & Shi, J.-W. Optical properties of Cr doped ZnAl2O4 nanoparticles with Spinel structure synthesized by hydrothermal method. Mater. Res. Expr. 7(1), 015025. https://doi.org/10.1088/2053-1591/ab6125 (2020).
Hussain, T. et al. Tunable dielectric behaviour and energy band gap range of ZnAl2O4 ceramics mediated by Mg substitution. J. Alloys Compd. 724, 940–950. https://doi.org/10.1016/j.jallcom.2017.07.104 (2017).
Moussa, N. M., Ebrahim, F. M., Adly, K. & Hassaan, M. Y. Chromium doped ZnO nanoparticles for energy storage, gas and humidity sensing and spin based electronic devices applications. Opt. Quant. Electron. 54, 683. https://doi.org/10.1007/s11082-022-04075-y (2022).
Ansari, S. A., Nisar, A., Fatma, B., Khan, W. & Naqvi, A. H. Investigation on structural, optical and dielectric properties of Co doped ZnO nanoparticles synthesized by gel-combustion route. Mater. Sci. Eng. 177(5), 428–435. https://doi.org/10.1016/j.mseb.2012.01.022 (2012).
Wang, D. et al. Cold-sintered temperature stable Na0.5Bi0.5MoO4–Li2MoO4 microwave composite ceramics. ACS Sustain. Chem. Eng. https://doi.org/10.1021/acssuschemeng.7b03889 (2017).
Thiruramanathan, P., Marikani, A., Madhavan, D., Suyambulingam, T. & Raja, G. Development of performance-improved global positioning system (GPS) patch antenna based on sol–gel-synthesized dual-phase (Bi4Ti3O12)X -(CaCu3Ti4O12)1-X composites. Cryst. Res. Technol. 51(6), 366–379. https://doi.org/10.1002/crat.201500213 (2016).
Bhongale, S. R., Ingavale, H. R., Shinde, T. J. & Vasambekar, P. N. Performance of a wideband cadmium ferrite microstrip patch antenna in the X-band region. J. Electron. Mater. https://doi.org/10.1007/s11664-017-5807-z (2017).
Yang, J., Wang, H., Lv, Z. & Wang, H. Design of miniaturized dual-band microstrip antenna for WLAN application. Sensors 16(983), 5. https://doi.org/10.3390/s16070983 (2016).
Author information
Authors and Affiliations
Contributions
R.S.D. conceptualized the synthesis of ceramic nanoparticles for microstrip patch antenna applications and S. D. performed the experiments under his supervision. R.S.D. involved in the data analysis and presentation. R.S.D. and S.D. drafted and revised the manuscript. All authors discussed the results and approved the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
The original online version of this Article was revised: The original version of this Article contained an error in Affiliation 1, which was incorrectly given as ‘Department of ECE, Koneru Lakshmaiah Education Foundation, Greenfields, Vaddeswaram, Guntur, A.P., India.’ The correct Affiliation is listed in the correction notice. Additionally, a Reference, which is now listed as Reference 26, and the reprint permission for Figure 1, were omitted. Full information regarding the corrections made can be found in the correction for this Article.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Didde, S., Dubey, R.S. Sol–gel derived ceramic nanoparticles as an alternative material for microstrip patch antenna in WLAN applications. Sci Rep 14, 13684 (2024). https://doi.org/10.1038/s41598-024-63454-5
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-024-63454-5
Keywords
This article is cited by
-
Preparation of low-temperature sintered (Mg0.4Zn0.6)2SiO4 ceramics with CuO addition and simulation for application in microwave antennas
Journal of Materials Science: Materials in Electronics (2025)