Abstract
Lazertinib is a recently developed third-generation epidermal growth factor receptor (EGFR)-tyrosine kinase inhibitors used for patients with advanced EGFR T790M-positive non-small-cell lung cancer. We evaluated the effectiveness of lazertinib compared with osimertinib using an external control. We obtained individual patient data for the lazertinib arm from the LASER201 trial and the osimertinib arm from registry data at the Samsung Medical Center. In total, 75 and 110 patients were included in the lazertinib and osimertinib groups, respectively. After propensity score matching, each group had 60 patients and all baseline characteristics were balanced. The median follow-up duration was 22.0 and 29.6 months in the lazertinib and osimertinib group, respectively. The objective response rate (ORR) were 76.7% and 86.7% for lazertinib and osimertinib, respectively (p = 0.08). The median progression-free survival (PFS) was 12.3 months (95% confidence interval [CI] 9.5–19.1) and 14.4 months (95% CI 11.8–18.1) for the lazertinib and osimertinib group, respectively (hazard ratio [HR] 0.97; 95% CI 0.64–1.45, p = 0.86). The median overall survival with lazertinib was not reached and that with osimertinib was 29.8 months (HR 0.44; 95% CI 0.25–0.77, p = 0.005). Our study suggests that lazertinib has an ORR and PFS comparable to those of osimertinib and has the potential for superior survival benefits.
Similar content being viewed by others
Introduction
Currently, two treatment options are available for patients with epidermal growth factor receptor (EGFR) T790M-positive non-small-cell lung cancer (NSCLC) who experienced disease progression following first- and second-generation EGFR-tyrosine kinase inhibitor (TKI) therapies in South Korea. Lazertinib (Leclaza®) is a recently developed third-generation EGFR-TKI1,2. LAZER201, a phase 1/2 single-arm study of lazertinib, showed that the objective response rate (ORR) was 55.3% and the median progression-free survival (PFS) was 11.1 months, which were evaluated by a blinded independent review (ORR of 72.4% and median PFS of 12.4 months by investigators)3. Another option is osimertinib (Tagrisso®), which is the first-approved third-generation EGFR-TKI and showed the ORR and median PFS of 70.1% and 10.1 months, respectively, in the AURA3 study4. Since both treatments have demonstrated excellent safety profiles, evidence on comparative effectiveness is needed to help oncologists decide the ideal treatment; however, there was no direct head-to-head study comparing the two drugs. Furthermore, the absence of data on the efficacy of lazertinib compared with that of the control group, including osimertinib or platinum-based chemotherapy, makes the comparison of the two treatments more challenging.
An external control arm is a good alternative in this situation. An external control is the comparator arm with no treatment or standard of care drawn from another population in different settings5,6. Since the background rates of outcomes cannot be adjusted in uncontrolled single-arm studies, a single-arm study with external control is considered to have a higher quality of evidence than those without5,7. Possible sources of external control are patient-level data from previous clinical trials and real-world data8. Therefore, in this study, we conducted an external control study to compare the effectiveness of lazertinib with that of osimertinib using individual patient data from the LASER201 study and the patient registry of a tertiary hospital in South Korea.
Materials and methods
Study population and data collection
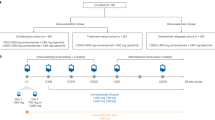
Individual patient data (IPD) for the lazertinib arm were obtained from the LASER201 study (ClinicalTrials.gov Identifier: NCT03046992). LASER201 is a multicenter, open-label, phase 1/2, single-arm study that enrolled patients with locally advanced or metastatic EGFR T790M-positive NSCLC who had disease progression after prior therapy with an EGFR-TKI from November 27, 2017 to May 10, 20191. Full details on the eligibility criteria and variables collected were described elsewhere1. Data cut-off for PFS and overall survival (OS) was January 8, 2021 and April 8, 2022, respectively.
As an external comparator, data from the osimertinib arm were extracted from real-world data of the Samsung Medical Center (SMC). Patients with locally advanced or metastatic EGFR T790M-positive NSCLC who had disease progression after prior therapy with an EGFR-TKI and started osimertinib between December 1, 2017, and May 31, 2019, were included. The SMC database includes sociodemographic information (e.g., age, sex, and smoking status) and various clinical information (e.g., Eastern Cooperative Oncology Group (ECOG) performance status, tumor histology, type of EGFR mutation, presence of brain metastasis, clinical stage at diagnosis, number of prior systemic therapies, type of prior EGFR-TKIs, number of previous EGFR-TKIs, results of laboratory tests, and history of other comorbidities). The data cutoffs for PFS and OS were the same as those for the lazertinib group (January 2021 for PFS and April 2022 for OS).
The same inclusion criteria in LASER201 were used for the osimertinib arm: (1) EGFR T790M-positive NSCLC and progression despite previous first- or second-generation EGFR-TKI therapies, (2) age ≥ 20 years, (3) ECOG 0–1, and (4) at least one measurable extracranial lesion. Among exclusion criteria of study subjects in LASER201, all applicable criteria in the SMC database were used. Patients who have the following history before the first use of osimertinib were excluded: (1) use of an investigational product within 30 days, (2) use of other EGFR-TKIs within 8 days, (3) cytotoxic chemotherapy or other anticancer drugs within 14 days, (4) major surgery within 4 weeks, (5) radiotherapy with a wide field within 4 weeks or a limited field within 1 week, (6) EGFR-TKIs targeting T790M mutation, (7) comorbidities within 8 weeks (spinal cord compression, brain metastases with steroid treatment, intracranial hemorrhage, central nervous system (CNS) complications with resection or shunt placement, leptomeningeal metastasis, interstitial lung disease, radiation pneumonitis with steroid treatment, symptomatic congestive heart failure, cardiac arrhythmia, or myocardial infarction/unstable angina within 6 months), and 8) clinical laboratory reports within 4 weeks (absolute neutrophil count < 1.5 × 10⁹ cells/L, platelet count < 100 × 10⁹ cells/L, hemoglobin < 90 g/L, alanine aminotransferase or aspartate aminotransferase > 2.5 × the upper limit of normal [ULN] if no demonstrable liver metastases or > 5.0 × ULN in presence of liver metastases, total bilirubin > 1.5 × ULN if no liver metastases or > 3.0 × ULN in the presence of documented Gilbert’s syndrome [unconjugated hyperbilirubinemia] or liver metastasis, or serum creatinine > 1.5 × ULN with creatinine clearance less than 50 mL/min). Additionally, we excluded patients with EGFR mutations other than exon 19 del and exon 21 Leu858Arg, because the number of patients was too small, making it difficult to include this variable in a matching model.
Outcome measures
The endpoints of interest were ORR, PFS, and OS. In the IPD of the lazertinib arm, we used the best overall response, progression, and death assessed by investigators, not an independent central review, as we believe that these were more comparable with those from real-world data. Responses were assessed using CT or MRI according to the Response Evaluation Criteria in Solid Tumors (version 1.1). ORR was defined as the proportion of patients with a complete response or partial response. PFS was defined as the time from the first drug administration to disease progression or death due to any cause, whichever occurred first. OS was defined as the time from the first drug administration to death from any cause. The durations of PFS and OS were censored at the last date when they were examined as event-free in patients without observed events. We also examined the distribution of subsequent anticancer therapy after progression and calculated the time to next treatment (TTNT) to investigate whether a significant discordance existed in sequential treatment after drug resistance.
Statistical analysis
To increase comparability between the two groups, we established a 1:1 propensity score-matched cohort. The propensity scores of each individual were estimated using a logistic regression model adjusted for variables that were considered clinically important or showed a significant difference between the groups before propensity score matching.
The ORR and median PFS and OS were calculated before and after PSM. Kaplan–Meier curves for PFS and OS were generated for the matched cohort and compared using a log-rank test. Hazard ratios (HRs) and their 95% confidence intervals (CIs) for progression and death with lazertinib versus osimertinib were estimated using Cox proportional hazard regression models. The risks of progression and death were examined according to age, sex, type of EGFR mutation, and the presence of brain metastasis, which can significantly affect the prognosis of patients with NSCLC. For sensitivity analysis, we applied the propensity score weighting approach [inverse probability of treatment weighting and standardized mortality ratio weighting instead of the matching method.
All analyses were performed using SAS (version 9.4, SAS Institute, Cary, NC, USA). A p-value of 5% or lower was considered statistically significant.
Ethical statement
This study was approved with a waiver of the requirement for informed consent from participants by the Institutional Review Board of Samsung Medical Center (SMC 2021-11-130-002) because we used anonymously collected data. All methods were performed following the relevant guidelines and regulations.
Results
Baseline characteristics
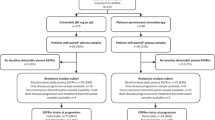
Between November 27, 2017, and May 10, 2019, 78 patients were enrolled and received lazertinib 240 mg. Two T790M-negative patients and one patient with EGFR mutations other than exon 19 del and exon 21 Leu858Arg were excluded; consequently, 75 patients remained (Fig. 1). In the SMC database, 203 patients started osimertinib treatment between December 1, 2017, and May 31, 2019. Among them, 93 patients were excluded as per protocol (14 with other EGFR-TKIs within 8 days, 4 with inappropriate laboratory results, 40 with a history of diseases of interest, 17 with major surgery or radiotherapy, 16 with American Joint Committee on Cancer stage I or II, and 2 with EGFR mutations other than exon 19 del or exon 21 Leu858Arg), and 110 patients were included in the final analysis.
Flowchart for the identification and selection of the study cohort.
The median follow-up duration for OS was 20.9 months [interquartile range (IQR) 8.8–35.8] in the lazertinib group and 29.3 months (IQR 17.9–36.8) in the osimertinib group. The median age of the lazertinib group was 62.0 years (range: 33–82), which was slightly but not significantly older than that of the osimertinib group (58.9 years, range: 29.5–88.1), and 52.0% and 40.0% of the patients in the lazertinib and osimertinib groups were male, respectively (Table 1). Regarding the type of EGFR mutation, 22 (29.3%) patients in the lazertinib group had an exon 21 Leu858Arg mutation, similar to that in the osimertinib group (30.9%). The proportion of patients with brain metastases was significantly higher in the lazertinib group (52.0%) than in the osimertinib group (27.3%) (p < 0.001). The mean number of prior EGFR-TKIs was 1.1 (standard deviation: 0.3) in both groups. The majority of patients received other EGFR-TKIs before lazertinib (92.0%) or osimertinib (83.7%). Additionally, 94.7% of patients in the lazertinib group and 94.6% in the osimertinib groups used prior EGFR-TKIs for over 6 months.
Propensity score matching
PSM was conducted by using clinical variables including age (< 65 years and ≥ 65 years), sex, smoking history (never-smoker and ever-smoker), presence of brain metastasis, the type of EGFR mutation (exon 19 del and exon 21 Leu858Arg), prior EGFR-TKI (gefitinib or erlotinib and afatinib), and duration of prior EGFR-TKI (< 6 months and ≥ 6 months). Sixty patients were included in each group after the PSM. The median follow-up duration for OS was still approximately 8 months longer in the osimertinib group (29.6 months, IQR 16.7–36.5) compared with the lazertinib group (22.0 months, IQR 12.0–37.2). The median age was 61.0 years in both groups (range: 33.0–82.0 in the lazertinib group and 38.3–80.5 in the osimertinib group). Although significant differences were observed in tumor histology (p = 0.045) and brain metastasis (p < 0.001) before PSM, all covariates were well balanced after PSM (Table 1).
Objective response rate and progression-free survival
Before PSM, the ORR of osimertinib (84.5%) was higher than that of lazertinib (72.0%) (Table 2). The median PFS was 12.4 months (95% CI 9.6–17.7) in the lazertinib group and 15.7 months (95% CI 12.8–18.0) in the osimertinib group (p = 0.594). After PSM, ORRs in both groups increased slightly but remained higher for osimertinib (76.7% for lazertinib and 86.7% for osimertinib) (p = 0.078). Among the 60 patients, 42 (70%) and 55 (91.7%) patients progressed were identified in the lazertinib and osimertinib groups, respectively. The median PFS was still numerically longer in the osimertinib group (14.4 months; 95% CI 11.8–18.1) than in the lazertinib group (12.3 months; 95% CI 9.5–19.1; p = 0.861) (Fig. 2A). HR for progression in the lazertinib group was 0.97 (95% CI 0.64–1.45) compared with the osimertinib group.
Kaplan–Meier curve for progression-free survival (A) and overall survival (B) of the lazertinib and osimertinib group after propensity score matching.
Overall survival
Before PSM, the median OS was 38.9 months (95% CI 30.2-not reached) and 29.6 months (95% CI 25.6–31.8) in the lazertinib and osimertinib group, respectively (p = 0.019). The 1-year OS rates in the lazertinib and osimertinib arms were 89.6% and 91.8%, respectively (p = 0.62). After PSM, the number of death events was 16 (26.7%) and 43 (71.7%) in the lazertinib and osimertinib groups, respectively. The median OS with lazertinib was not reached, and that with osimertinib was 29.8 months (95% CI 23.2–34.0; p = 0.004). The 1-year OS rates with lazertinib was 92.7%, while that with osimertinib was 86.6% (p = 0.27). The Kaplan–Meier curves for OS in the lazertinib and osimertinib groups are shown in Fig. 2B. HR for death in the lazertinib group versus osmertinib group was 0.44 (95% CI 0.25–0.77) after PSM, which was statistically significant (p = 0.005). The median TTNT and the distribution of patients who received subsequent anticancer therapy in the matched groups were shown in supplementary Table S1. The median TTNT was 26.6 months (range, 13.8- not reached) with lazertinib and 15.7 months (range, 12.4–20.3) with osimertinib. Out of 60 patients, 24 (40%) and 39 (65%) received subsequent treatment in the lazertinib and osimertinib groups, respectively. The proportion of patients who received treatments with 3rd-generation EGFR TKI was higher in the lazertinib group than in the osimertinib group (15% vs. 8.4%).
Subgroup and sensitivity analysis
In the subgroup analyses, the overall trend in the risk of death was favorable for lazertinib (Fig. 3). Among them, the risk of death in lazertinib was significantly lower than that in osimertinib regardless of age (< 65 years: HR 0.47; 95% CI 0.23–0.96; p = 0.037; and ≥ 65 years: HR, 0.33; 95% CI 0.11–0.99; p = 0.049) and in subgroups male (HR 0.31; 95% CI 0.11–0.83; p = 0.019), with exon 19 del (HR, 0.42; 95% CI 0.20–0.85; p = 0.017), and without brain metastasis (HR, 0.42; 95% CI 0.20–0.86, p = 0.017). However, the risk of progression in all subgroups was not statistically significant (Supplementary Fig. S1). The results of sensitivity analysis using propensity score weighting methods were consistent with those of the main analysis (Supplementary Tables S2–4).
Subgroup analysis for overall survival in patients who received lazertinib or osimertinib after propensity score matching.
Discussion
This study showed that lazertinib had ORR, PFS, and OS outcomes comparable to those of osimertinib before and after PSM. There was no significant difference in ORR and PFS between the two groups. The results of the subgroup analysis also revealed that the risk of death tended to be lower with lazertinib than with osimertinib, regardless of age group, sex, type of EGFR mutation, and brain metastasis status. We verified the robustness of these findings using sensitivity analyses.
The median PFS and OS in matched patients treated with osimertinib were estimated as 14.4 and 29.8 months, respectively. These results are similar to those of previous studies on osimertinib. In AURA3, a phase III trial investigating the efficacy and safety of osimertinib compared with platinum-pemetrexed among patients with EGFR T790M-positive locally advanced or metastatic NSCLC, the median PFS and OS were 10.1 and 26.8 months, respectively, in the osimertinib arm4,9. A study of real-world data of osimertinib from 19 medical centers reported the median PFS of 14.2 months and the median OS of 36.7 months10. Thus, it seems that the osimertinib group of this study demonstrated strong external validity.
Our study showed a 56% reduced risk of death with lazertinib compared to osimertinib, despite a similar risk of progression. Given the higher selectivity and potency of lazertinib compared to those of osimertinib in an in vitro study11, this finding may have resulted from the excellent efficacy of lazertinib. In particular, lazertinib showed superior intracranial activity compared to osimertinib in vivo and an intracranial ORR of 86% and a median intracranial PFS of 26.0 months in the LASER201 study3,11. The results of the subgroup analysis in the present study also support that lazertinib was likely to have a better survival benefit irrespective of brain metastasis. Although osimertinib showed good brain penetration and a good CNS objective response rate12,13,14, no significantly lower risk of death in patients with CNS metastasis in the osimertinib arm than in the platinum-pemetrexed arm was demonstrated in the subgroup analysis of the AURA 3 study (HR 1.19; 95% CI 0.79–1.83)9. Therefore, the potent CNS efficacy of lazertinib may contribute to the prolonged OS. Furthermore, the potentially better efficacy of lazertinib in the exon 21 Leu858Arg mutation could have led to a longer OS compared to osimertinib. In the subgroup analysis, lazertinib showed a remarkable point estimate of HR (0.49, 95% CI 0.19–1.29) in patients with exon 21 Leu858Arg mutation although CI was wide due to the small sample size. Previous studies reported that patients with exon 21 Leu858Arg have a worse prognosis than those with exon 19 del after anticancer therapies, including osimertinib15,16,17,18,19. Our results suggest that lazertinib may be more effective in patients with exon 21 Leu858Arg mutation and should be considered first for treatment.
However, a cautious interpretation of the longer OS is needed because of the considerable differences in the clinical settings of the groups. As shown in the Kaplan–Meier curves, many participants in the lazertinib trial dropped out within 2 years after treatment initiation due to withdrawal by the subject or loss to follow-up, whereas few patients did so in the registry data. In the same context, the median follow-up duration for OS was shorter in the lazertinib group than that in the osimertinib group. This could be attributed to the differential way death records were collected in the two databases; death could not be captured once a patient withdrew consent in the clinical trial, whereas death records in the SMC data was derived from the national cancer registry. This unequal early dropout rate and short follow-up duration would work in favor of lazertinib, especially in terms of long-term outcomes, as the investigators lost the chance to identify additional deaths in participants. Furthermore, differences in the distribution of subsequent treatments that patients underwent after progression between the two groups may have influenced the favorable results in OS for lazertinib. In the lazertinib group, 9 patients (15%) received osimertinib as a next-line treatment, while 4 patients (8.4%) received osimertinib plus savolitinib and only one patient (1.7%) received lazertinib plus amivantamab in the osimertinib group. However, 15 of 42 patients who experienced progression in the lazertinib group used lazertinib as a treatment beyond progressive disease, with the median duration of the treatment was 158 days (Supplementary Table S5). Considering the longer TTNT and the higher proportion of patients who maintained the treatment beyond progressive disease in the lazertinib group compared to the osimertinib group, the effect of subsequent treatment on the longer OS might not be considerable.
This study had several limitations. We attempted to make the osimertinib group as similar as possible to the lazertinib group by applying the same eligibility criteria; however, some criteria were unavailable in the retrospective real-world data. In addition, residual confounding factors may have originated from different clinical settings. Patients’ psychological status, measurement methods or techniques, standards of clinical judgement, and other prognostic factors can differ between the two settings. In our study, the different response assessment intervals (6 weeks for the lazertinib arm and 2–3 months for the osimertinib arm) may have affected the results. As mentioned above, different follow-up durations for OS due to different methods of collecting death information also impede a clear interpretation of OS differences. In addition, the small sample size and data from a single institute impede generalization of the results. Although we used all IPD of LASER201 and real-world data of SMC, which is a hospital with the largest data of NSCLC patients, further confirmative studies with a large population should be conducted. Lastly, we only evaluated the effectiveness of the drug, even though the safety of the drug is also an important factor when selecting a drug. Therefore, a study comparing the safety of the two drugs in a real-world setting needs to be conducted in the future.
Despite these limitations, this study has several important implications. We produced the first comparative evidence on the efficacy of lazertinib as a second-line or more EGFR-TKI at a time when there is no other study with direct, randomized head-to-head comparisons. Many researchers agree that studies with external comparators cannot substitute randomized studies with a direct comparison20,21,22. Nevertheless, our findings would provide helpful insights on the novel therapy to policymakers and practitioners since single-arm studies inherently have methodological limitations regarding interpreting the results23. We also present limitations of the study that should be considered and discussed for the appropriate regulatory use of this study design in the future. To generate valid comparative evidence using real-world data, regulatory agencies may need to develop guidelines for the uniform collection of real-world data, such as registries or electronic health records24.
In conclusion, our study strengthens the evidence on the comparable efficacy of lazertinib with osimertinib and the potential beneficial effect on the survival in patients with advanced EGFR T790M-positive NSCLC who experienced disease progression following EGFR-TKI, which was shown in a single-arm trial. However, the small sample size and different clinical settings between the groups hinder the generalizability of the results. A large-scale, controlled study is required to confirm these findings.
Data availability
The data analyzed in this study are available from Yuhan Corporation and Samsung Medical center. Restrictions apply to the availability of these data, which were used under license for this study. Data are available from the authors upon reasonable request with the permission of Yuhan Corporation and Samsung Medical center.
References
Ahn, M. J. et al. Lazertinib in patients with EGFR mutation-positive advanced non-small-cell lung cancer: Results from the dose escalation and dose expansion parts of a first-in-human, open-label, multicentre, phase 1–2 study. Lancet Oncol. 20, 1681–1690. https://doi.org/10.1016/S1470-2045(19)30504-2 (2019).
Dhillon, S. Lazertinib: First approval. Drugs 81, 1107–1113. https://doi.org/10.1007/s40265-021-01533-x (2021).
Cho, B. C. et al. A phase 1/2 study of lazertinib 240 mg in patients with advanced EGFR T790M-positive NSCLC after previous EGFR tyrosine kinase inhibitors. J. Thorac. Oncol. 17, 558–567. https://doi.org/10.1016/j.jtho.2021.11.025 (2022).
Mok, T. S. et al. Osimertinib or platinum-pemetrexed in EGFR T790M-positive lung cancer. N. Engl. J. Med. 376, 629–640. https://doi.org/10.1056/NEJMoa1612674 (2017).
Seeger, J. D. et al. Methods for external control groups for single arm trials or long-term uncontrolled extensions to randomized clinical trials. Pharmacoepidemiol. Drug Saf. 29, 1382–1392. https://doi.org/10.1002/pds.5141 (2020).
International Conference on Harmonization of Technical Requirements for Regulation of Pharmaceuticals for Human Use. ICH Harmonised Tripartite Guideline: Choice of Control Groups and Related Issues in Clinical Trials E10. https://database.ich.org/sites/default/files/E10_Guideline.pdf (2000).
Gray, C. M., Grimson, F., Layton, D., Pocock, S. & Kim, J. A Framework for methodological choice and evidence assessment for studies using external comparators from real-world data. Drug Saf. 43, 623–633. https://doi.org/10.1007/s40264-020-00944-1 (2020).
Schmidli, H. et al. Beyond randomized clinical trials: Use of external controls. Clin. Pharmacol. Ther. 107, 806–816. https://doi.org/10.1002/cpt.1723 (2020).
Papadimitrakopoulou, V. A. et al. Osimertinib versus platinum-pemetrexed for patients with EGFR T790M advanced NSCLC and progression on a prior EGFR-tyrosine kinase inhibitor: AURA3 overall survival analysis. Ann. Oncol. 31, 1536–1544. https://doi.org/10.1016/j.annonc.2020.08.2100 (2020).
Lee, J. H. et al. Real-world study of osimertinib in Korean patients with epidermal growth factor receptor T790M mutation-positive non-small cell lung cancer. Cancer Res. Treat. 55, 112–122. https://doi.org/10.4143/crt.2022.381 (2023).
Yun, J. et al. YH25448, an irreversible EGFR-TKI with potent intracranial activity in EGFR mutant non-small cell lung cancer. Clin. Cancer Res. 25, 2575–2587. https://doi.org/10.1158/1078-0432.CCR-18-2906 (2019).
Ballard, P. et al. Preclinical comparison of osimertinib with other EGFR-TKIs in EGFR-Mutant NSCLC brain metastases models, and early evidence of clinical brain metastases activity. Clin. Cancer Res. 22, 5130–5140. https://doi.org/10.1158/1078-0432.CCR-16-0399 (2016).
Goss, G. et al. CNS response to osimertinib in patients with T790M-positive advanced NSCLC: Pooled data from two phase II trials. Ann. Oncol. 29, 687–693. https://doi.org/10.1093/annonc/mdx820 (2018).
Wu, Y. L. et al. CNS efficacy of osimertinib in patients with T790M-positive advanced non-small-cell lung cancer: Data from a randomized phase III trial (AURA3). J. Clin. Oncol. 36, 2702–2709. https://doi.org/10.1200/JCO.2018.77.9363 (2018).
Ahn, M. J. et al. Osimertinib in patients with T790M mutation-positive, advanced non-small cell lung cancer: Long-term follow-up from a pooled analysis of 2 phase 2 studies. Cancer 125, 892–901. https://doi.org/10.1002/cncr.31891 (2019).
Auliac, J. B. et al. Real-life efficacy of osimertinib in pretreated patients with advanced non-small cell lung cancer harboring EGFR T790M mutation. Lung Cancer 127, 96–102. https://doi.org/10.1016/j.lungcan.2018.11.037 (2019).
Igawa, S. et al. Impact of EGFR genotype on the efficacy of osimertinib in EGFR tyrosine kinase inhibitor-resistant patients with non-small cell lung cancer: A prospective observational study. Cancer Manag. Res. 11, 4883–4892. https://doi.org/10.2147/CMAR.S207170 (2019).
Lee, C. K. et al. Impact of specific epidermal growth factor receptor (EGFR) mutations and clinical characteristics on outcomes after treatment with EGFR tyrosine kinase inhibitors versus chemotherapy in EGFR-mutant lung cancer: A meta-analysis. J. Clin. Oncol. 33, 1958–1965. https://doi.org/10.1200/JCO.2014.58.1736 (2015).
Zhang, Y. et al. Patients with exon 19 deletion were associated with longer progression-free survival compared to those with L858R mutation after first-line EGFR-TKIs for advanced non-small cell lung cancer: A meta-analysis. PLoS One 9, e107161. https://doi.org/10.1371/journal.pone.0107161 (2014).
Di Lorenzo, G. et al. An adjusted indirect comparison of everolimus and sorafenib therapy in sunitinib-refractory metastatic renal cell carcinoma patients using repeated matched samples. Expert Opin. Pharmacother. 12, 1491–1497. https://doi.org/10.1517/14656566.2011.587119 (2011).
Signorovitch, J. E. et al. Comparative efficacy of nilotinib and dasatinib in newly diagnosed chronic myeloid leukemia: A matching-adjusted indirect comparison of randomized trials. Curr. Med. Res. Opin. 27, 1263–1271. https://doi.org/10.1185/03007995.2011.576238 (2011).
Tan, D. S. et al. Comparative efficacy of ceritinib and crizotinib as initial ALK-targeted therapies in previously treated advanced NSCLC: An adjusted comparison with external controls. J. Thorac. Oncol. 11, 1550–1557. https://doi.org/10.1016/j.jtho.2016.05.029 (2016).
Cucherat, M. et al. From single-arm studies to externally controlled studies. Methodological considerations and guidelines. Therapie 75, 21–27. https://doi.org/10.1016/j.therap.2019.11.007 (2020).
Wu, J., Wang, C., Toh, S., Pisa, F. E. & Bauer, L. Use of real-world evidence in regulatory decisions for rare diseases in the United States-Current status and future directions. Pharmacoepidemiol. Drug Saf. 29, 1213–1218. https://doi.org/10.1002/pds.4962 (2020).
Acknowledgements
We would like to thank Yuhan Corporation for providing the clinical trial data, and IQVIA and Dr. JungAe Kim for mediating the acquisition of the SMC registry data. We also thank Editage (www.editage.co.kr) for English language editing.
Funding
This research was supported by a grant (21183MFDS542) from the Ministry of Food and Drug Safety, South Korea, from to 2021–2022. This work was supported by a grant (21153MFDS607) from the Ministry of Food and Drug Safety of South Korea in 2021–2025. This study was supported by grants from the Korean Society of Medical Oncology (KSMO) 2021 and the National Research Foundation of Korea (NRF) funded by the Ministry of Science and Information and Communication Technology (ICT) (No. NRF-2021R1F1A1054782). This paper was supported by research funds for newly appointed professors of Jeonbuk National University in 2022.
Author information
Authors and Affiliations
Contributions
Conceived and designed the analysis: J.-Y.S., H.A.J.; Collected the data: H.-Y.Y.; Contributed data or analysis tools: H.-L.J., M.K.; Performed the analysis: H.-L.J., M.K.; Wrote the paper: H.-L.J., S.K., J.-Y.S., H.A.J.
Corresponding authors
Ethics declarations
Competing interests
HLJ has received grants from the National Research Foundation of Korea. JYS received research grants from the Ministry of Food and Drug Safety, Ministry of Health and Welfare, the National Research Foundation of the Republic of Korea, and pharmaceutical companies, including Amgen, Pfizer, Hoffmann-La Roche, Dong-A ST, and Yungjin. No other relationships or activities that could appear to have influenced the submitted work. All other authors do not hold any competing interest.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Jeon, HL., Kwak, M., Kim, S. et al. Comparative effectiveness of lazertinib in patients with EGFR T790M-positive non-small-cell lung cancer using a real-world external control. Sci Rep 14, 14659 (2024). https://doi.org/10.1038/s41598-024-65220-z
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-024-65220-z