Abstract
Studies have shown that the co-occurrence of diabetes mellitus (DM) and thyroid dysfunction (TD) exacerbates diabetes complications and imposes a financial burden on the healthcare system. Therefore, this study aimed to investigate the prevalence of TD-DM comorbidity and its associated risk factors. This cross-sectional study was conducted on enrollment phase data of the TABARI cohort population which consisted of 10,255 adults aged between 35 to 70 years old residing in Sari, Mazandaran, Iran from 2015 to 2017. A total of 9939 out of 10,255 individuals (96.92%) entered the study. The prevalence of TD among T2DM patients was 13.2%. The prevalence of T2DM among patients with TD was 9.2%. Furthermore, the prevalence of TD-DM comorbidity in the overall population was 2.2%. Logistic regression analysis revealed that the odds of TD-DM comorbidity was significantly higher in women (OR 2.85; 95% CI 1.58–5.11), in the age group of 60–70 years (OR 9.62; 95% CI 3.69–25.10), in smokers (OR 2.32; 95% CI 1.19–4.52), in individuals with high waist circumference (WC) (OR 2.22; 95% CI 1.32–3.75), in individuals with low high-density lipoprotein (HDL) (OR 1.60; 95% CI 1.20–2.14), in individuals with high total cholesterol (TC) (OR 1.71; 95% CI 1.21–2.41), in individuals with high triglycerides (TG) (OR 1.79; 95% CI 1.27–2.51), and significantly lower in individuals with higher physical activity (PA) (OR 0.67; 95% CI 0.49–0.93). The present study demonstrated a prevalence of 2.2% in patients with both TD and T2DM. Additionally, female gender, older age, smoking, high WC, low HDL, high TC, high TG, and low PA were predictors of TD-DM comorbidity.
Similar content being viewed by others
Introduction
Diabetes mellitus (DM) and thyroid dysfunction (TD) are among the most prevalent endocrine disorders, with overlapping pathologies1. DM is a chronic metabolic disorder characterized by elevated levels of glucose in the blood mostly due to insulin resistance2. Similarly, thyroid hormones play a significant role in the regulation of glucose homeostasis3. Thyroid hormones could alter blood glucose levels through various mechanisms, including impairing insulin secretion4,5. Thyroid hormones regulate carbohydrate metabolism and pancreatic function, while on the other hand, DM impacts the function and performance of the thyroid gland3,6. Hyperthyroidism affects insulin secretion, function, and clearance, potentially leading to hyperglycemia, while hypothyroidism may mask clinical symptoms and signs of DM7,8. Additionally, diabetic patients with hypothyroidism may require different levels of insulin compared to diabetic patients without TD8,9,10. Moreover, as demonstrated by Raval et al.11, T2DM patients who also suffer from TD have greater healthcare expenditures mostly due to more frequent diabetic complications. Thus, recognizing and treating TD is an essential part of DM management for patients and society.
Studies on the prevalence of TD in diabetic patients have shown variable results from 10.8% up to 24.2% which highlight the need for large population-based studies12,13. Additionally, researches identifying potential risk factors for TD-DM comorbidity have shown differing results14,15,16. Thus, such inconsistent results further necessitate population-based studies to be performed.
Population assessments regarding TD and DM comorbidity can yield valuable insights. It has been reported that recognizing and treating TD in diabetic patients contributes to better blood sugar (BS) control, reduces the risk of cardiovascular events, and improves overall health17. However, not only do investigations indicate conflicting results among primary studies, but also there is also limited evidence that examines the mutual relationship between TD and diabetes, and sometimes a limited number of samples in existing studies restricts researchers and clinicians from drawing strong conclusions. Additionally, available evidence is restricted to certain nations. The results of such studies may not be generalizable due to the considerable confounding effects of ethnicity and genetics, which correlate with geographical patterns.
Therefore, considering the aforementioned reasons which underscore the importance of further studies, the present study aimed to assess the prevalence of TD in T2DM patients and the general population, and identify its potential risk factors in a large cohort of Northern Persian adults.
Materials and methods
Population and study design
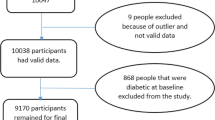
This cross-sectional study was conducted on enrollment phase data of the TABARI cohort population which is a part of the Iranian national cohort entitled “Prospective Epidemiological Research Studies in Iran (PERSIAN)”. The enrollment phase of the TABARI consisted of 10,255 individuals between 35 and 70 years old in the urban and mountainous regions of Sari (the capital city of Mazandaran situated in the foothills of the Alborz mountain range and along the shores of the Caspian Sea, north of Iran) from 2015 to 2017. In the present study, a census-based sampling method was used and required data were acquired through the TABARI cohort registration system. The questionnaire used in this study was a standardized questionnaire, details and characteristics of which are described in the methodology articles and cohort profile18,19,20,21. Data were recorded by trained interviewers, who had participated in national and provincial workshops, and standardized web-based (online) questionnaires According to the PERSIAN cohort protocol. Further details regarding the methodology of the PERSIAN and TABARI cohorts are provided in the cohort profile and methodology articles18,19,20,21. The entire population of the TABARI cohort was enrolled in this study. However, patients were excluded if they had cancer or kidney failure.
TD and DM criteria
Individuals with a fasting blood sugar (FBS) level of ≥ 126 mg/dL, individuals with a previous history of DM, and individuals taking DM medications were considered as our DM population. Additionally, participants were marked as functional TD (hypo or hyperthyroidism) based on self-reporting and health documents. It should be noted that in epidemiological studies where large sample sizes are utilized, assessment with specialized tests is not common practice, and self-reporting does not introduce significant limitations. Furthermore, if a participant was simultaneously affected by DM and TD, the participant was considered as TD-DM comorbidity.
Measurements
In the present study, data regarding demographic variables such as age, gender, residential area (urban/mountainous), socioeconomic status (rated from one to five, representing the lowest to highest socioeconomic status), marital status, education level, occupation, smoking status, physical activity (PA) level, diabetes status, and thyroid disorders were extracted from the TABARI cohort data repository. Also, data regarding biochemical blood measurements and anthropometric assessments were obtained.
Blood samples were collected from all study participants after a 12-h fasting period. FBS, total cholesterol (TC), triglycerides (TG), low-density lipoprotein (LDL), and high-density lipoprotein (HDL) were measured using an AutoAnalyzer (BT 1500, Biotechnica, Italy). Additionally, anthropometric indices including height, weight, waist circumference (WC), hip circumference (HC), waist-to-hip ratio (WHR), and body mass index (BMI) were measured using standardized tools. Height was measured by SECA 226 (SECA, Hamburg, Germany) stadiometer and weight measurement was done by SECA 755 (SECA, Hamburg, Germany) analogue standing scale. Also, WC and HC were measured based on National Institute of Health (NIH) definitions22.
Statistics
The data were received in Excel format after approval and confirmation by the ethics committee. Subsequently, the variables were transferred to SPSS software version 26. Variables were described using frequencies and percentages. Comparison between the frequency of DM, TD, and TD-DM comorbidity with demographic and epidemiological variables was conducted using chi-square tests. Variables with significance levels less than 0.1 in univariate analysis were entered into the multivariable model. Based on the results of the univariate analysis, three variables (including marital status, socioeconomic status, and LDL) were excluded, and 13 variables (including gender, age, WC, WHR, BMI, smoking status, PA, HDL, TG, TC, residential area, education level, and occupation) were included in the multivariable model. Considering the binary outcome (TD-DM comorbidity: yes/no), multiple logistic regression was used to adjust for confounding variables.
Ethics
This study was conducted without commercial input or involvement in the design, implementation, analysis, or reporting. This study was approved by the Research Ethics Committees of Mazandaran University of Medical Sciences (Ethics Approval Code: IR.MAZUMS.IMAMHOSPITAL.REC.1401.010, 11-05-2022). Written informed consent was obtained from all participants before entering the study. All procedures performed in this study were in accordance with the ethical standards of the Institutional Research Ethics Committee of Mazandaran University of Medical Sciences and with the 1964 Helsinki Declaration and its later amendments or comparable ethical standards.
Results
Baseline characteristics
After considering inclusion and exclusion criteria, a total of 9939 individuals out of 10,255 (92.96%) participants from the TABARI cohort population were enrolled in the analysis, of which 5,896 individuals (59.59%) were female. Further baseline characteristics of the participants are presented in Table 1.
TD in T2DM participants
Out of 9939 participants, 1686 individuals (16.96%) had T2DM. The prevalence of TD among all diabetic patients, diabetic males, and diabetic females was 223 (13.23%), 31 (5%), and 192 (18%), respectively (P-value < 0.001). Also, the prevalence of TD among non-diabetics was 797 (9.7%) which was significantly lower than the diabetic population (P < 0.001). Furthermore, the prevalence of TD among T2DM patients differed significantly based on BMI categories. Among individuals with a BMI less than 25, 29.9–25, and 30 and above, the prevalence rates were 6.8%, 11.4%, and 16.8%, respectively (P-value < 0.001). The prevalence of TD among patients with T2DM had no statistically significant difference within various age groups, residential areas, socioeconomic status, and marital status (P-value < 0.05) (Table 1).
T2DM in participants with TD
A total of 1020 individuals (10.26%) had TD in the population. The prevalence of T2DM among all TD patients, male, and female TD patients, was 223 (21.9%), 31 (23.7%), and 192 (21.6%), respectively (P = 0.573). Also, out of 8919 participants without TD, 1463 participants had T2DM (16.4%) which was significantly lower compared to participants with TD (P < 0.001). Table 1 demonstrates a trend in the prevalence of T2DM among patients with TD which increases with age. The prevalence of T2DM ranges from 4.2% in the 35–39 years-old group to 36.1% in the age group of 60–70 years-old (P < 0.001). Additionally, the prevalence of T2DM among patients with TD varies significantly based on BMI, ranging from 10.7% in the BMI < 25 group, 18.2% in the 25–29.9 group, and 29.2% among those with a BMI ≥ 30 (P < 0.001).
TD-DM comorbidity
Table 2 presents the results of univariate analysis of TD-DM comorbidity. The prevalence of TD-DM comorbidity in the TABARI cohort population, as well as among men and women, was 223 (2.2%), 31 (0.8%), and 192 (3.3%), respectively (P < 0.001). The prevalence of TD-DM comorbidity increased with age, ranging from 0.3% in the 35–39-year-old age group to 3.5% in the age group of 60–70 years (P < 0.001). Moreover, the TD-DM comorbidity was higher among individuals with a metabolic equivalent of task (MET) score below the median compared to those with a MET score equal to or above the median (2.9% vs. 1.6%, P < 0.001). Additionally, the prevalence of TD-DM comorbidity varied significantly based on WC, WHR, and BMI categories, and HDL, TG, and TC levels (P < 0.001) (Table 3).
Table 4 demonstrates the results of multiple logistic regression analysis. According to the results, females were more likely to have TD-DM comorbidity compared to men (OR = 2.85, 95% CI 1.58–5.11). Participants aged between 40 and 49 years old, 50–59 years old, and 60–70 years old had higher chances of getting TD-DM comorbidity compared to participants aged 35–39 years old as the reference group (OR = 3.76, 95% CI 1.48–9.57; OR = 8.52, 95% CI 3.38–21.48; and OR = 9.62, 95% CI 3.69–25.10, respectively). Also, smokers (OR = 2.32, 95% CI 1.19–4.52), participants with higher WC (OR = 2.22, 95% CI 1.32–3.75), abnormal HDL levels (OR = 1.60, 95% CI 1.20–2.14), high TC levels (OR = 1.71, 95% CI 1.21–2.41), and high TG levels (OR = 1.79, 95% CI 1.27–2.51) had higher chance of having TD-DM comorbidity, and those with higher PA levels (OR = 0.67, 95% CI 0.49–0.93) had lower chance of having TD-DM comorbidity.
Discussion
In the present study, we aimed to assess the prevalence of TD in T2DM patients and the general population and identify its potential risk factors in a large cohort of Northern Persian adults. Our results indicated that the prevalence of T2DM among patients with TD was 21.9%, the prevalence of TD among T2DM patients was 13.2%, and the prevalence of patients with TD-DM comorbidity was 2.2%. After adjusting for the confounding variables, the TD-DM comorbidity was significantly higher in females, smokers, participants of older age, higher WC, and individuals with elevated TG, higher TC, lower HDL, and lower PA.
Concerning these two important comorbidities, various studies have assessed the prevalence of TD in diabetic patients, with significantly differing results. Yet, evidence has shown that TD is more prevalent in individuals with type 2 DM (T2DM) compared to the general population16. In a study by Sreelatha et al.23, the prevalence of TD in diabetic patients was reported to be 13%. However, in a meta-analysis conducted by Sharma et al.24, this prevalence was found to be 24.2%. In another study, the prevalence of thyroid disease among T2DM patients was estimated to be 10.8%13. Such dissimilar results necessitate large population-based studies to be conducted.
In the present study, the prevalence of TD in T2DM patients was lower compared to the studies by Hussain et al. (21.7%)25, Zhu et al. (23.7%)26, Sharma et al. (24.2%)12, Talwalkar et a. (24.8%)27, and Khassawneh et al. (14%)16; but consistent with the studies by Vamshidhar et al.13 and Hassan-Kadle et al.28. In these studies, the most common TD among diabetic patients was hypothyroidism, which was not differentiated in the present study. Additionally, in the study by Sadatamini et al.29, contrary to the present study, the incidence of TD among T2DM diabetic patients was lower than among non-diabetic individuals. Also, in the present study, the prevalence of T2DM among individuals with TD (21.9%) was higher than the prevalence of T2DM in the entire population (16.96%) which may indicate that patients with TD may be prone to T2DM. Consistent with our results, Elgazar et al.30 found that patients with TD have significantly higher HbA1c levels and lower insulin levels compared to those with normal thyroid function. Similarly, Rong et al.1 demonstrated that high baseline TSH levels, low FT3, and low FT4 are significantly associated with increased risk of T2DM.
Regarding the role of TD in diabetes complications, Reddy et al.31 found that the prevalence of diabetic retinopathy was significantly higher in T2DM patients with subclinical hypothyroidism than in euthyroid T2DM patients. Also, Raval et al.11 have reported higher health expenditures in patients with TD-DM comorbidity compared to T2DM patients without TD. This significant increase in health expenditures was reported mostly due to more frequent diabetic complications among this population. On the other hand, Khassawneh et al.16 and Rong et al.1 found no significant relationship between TD and diabetes complications or duration. Considering that some patients may not be aware of their TD, the importance of this issue increases, and special attention is warranted in public health policies. In the studies by Khassawneh et al.16 and Fregenal et al.32, 12.6% and 40% of cases, respectively, were unaware of their TD. Given the role of TD in BS levels, TD may exacerbate or intensify diabetes complications in the long term as demonstrated by previous studies. In the study by Elgazar et al.30, the prevalence of TD was associated with an increase in HbA1c levels and a decrease in insulin levels, and in the study by Barmpari et al.33, T2DM patients with hypothyroidism had higher HbA1c levels compared to individuals without hypothyroidism.
The results regarding the higher risk and prevalence of TD-DM comorbidity in women and its direct correlation with age in the present study are consistent with the findings of existing studies such as those by Khassawneh et al.16, Zhu et al.26, Demitrost et al.34, Ershad et al.35, and Barmpari et al.33. Overall, to the best of our knowledge, no evidence contradicting these results has been observed. These results can aid public health systems in screening the target group in diabetic patients for TD; thus, indirectly leading to better control and prevention of diabetes complications by early detection and treatment.
Additionally, studies have been conducted to identify potential risk factors for DM and TD comorbidity. Al-Geffari et al.14 found female gender and family history of TD as potential risk factors for TD among Saudi T2DM patients. In another study, Khassawneh et al.16 demonstrated old age, female gender, and poor control DM as risk factors for TD in T2DM patients. Meanwhile, Díez et al.15 only found thyroid autoimmunity to be a risk factor in diabetic patients TD. Thus, such inconsistent results further necessitate population-based studies to be performed.
In addition to gender and age, other variables contribute as TD-DM comorbidity risk factors. Regarding anthropometric indices and PA, the likelihood of TD-DM comorbidity was higher in individuals with higher BMI, higher WHR, higher WC, and lower PA. However, statistically significant associations were observed only for WC and PA in the multiple logistic regression analysis. These results confirm the findings of previous studies. In the studies by Khatiwada et al.36, Khassawneh et al.16, Umpierrez et al.37, Al-Geffari et al.14, and Diez et al.15, there was no significant difference in BMI among individuals with TD-DM comorbidity compared to others.
Furthermore, in the current study, smoking showed a statistically significant association with TD-DM comorbidity. Similarly, in the study by Khatiwada et al.36, a significant difference in the prevalence of smoking was observed between diabetic individuals with hypothyroidism and diabetic individuals without TD (12.17% versus 6.44%). This may be explained by the cyanide as one of the components of tobacco which, when smoked, is converted to the chemical thiocyanate38. Thiocyanate is known to interfere with thyroid function through inhibition of iodine uptake which leads to reduced production of thyroid hormones39. Additionally, smoking causes oxidative stress. Overproduction of mitochondrial reactive oxygen species (ROS) leads to the activation of pathways involved in the pathogenesis of insulin resistance and diabetic complications40. Thus, the increased chance of TD-DM comorbidity in smokers can be attributed to the combined effects of smoking on both thyroid function and glucose metabolism. For instance, the inhibition of iodine uptake into the thyroid gland can lead to hypothyroidism, which in turn can cause insulin resistance and exacerbate diabetes mellitus41,42. In contrast, Al-Geffari et al.14 did not observe any significant difference in the prevalence of smoking between the two groups of TD-DM comorbidity and others.
Regarding the relationship between lipid profile and TD-DM comorbidity, the variables of low HDL, high TG, and high TC significantly increased the likelihood of TD-DM comorbidity. This finding could be explained in a molecular level by the synergistic effects of thyroid hormones and insulin levels on the lipid profile. Insulin induces the synthesis of HMG-CoA reductase, a key enzyme in cholesterol biosynthesis43. Thyroid hormones also induce the synthesis of HMG-CoA reductase and promote the conversion of cholesterol into bile acid43. Furthermore, thyroid hormones can influence HDL metabolism by increasing cholesteryl ester transfer protein (CETP) activity44. There is molecular evidence that the synergistic effects of thyroid hormones and insulin determine the pathways of glucose and lipid metabolism. For instance, FT4 has been found to increase the insulin resistance index (HOMA-IR) by positively correlating with TG levels. In patients with T2DM with hyperthyroidism, decreasing HDL levels showed an association with HOMA-IR45. Also, recent literature shows that on a molecular level, some newly identified regulatory factors, such as proprotein convertase subtilisin/kexin type 9, angiogenin-like proteins, and fibroblast growth factors are the underlying causes of dyslipidemia in hypothyroidism46. In contrast to the findings of the current study, Khatiwada et al.36 and Al-Geffari et al.14 did not observe any significant difference in any of the lipid profile parameters (including HDL, LDL, TC, and TG) among individuals with TD-DM comorbidity compared to others.
Limitations
The lack of differentiation between various TD disorders (including hyperthyroidism and hypothyroidism) is a limitation in the present study. Another limitation is the assessment of TD based on self-reporting by participants and their previous documentation (if any), which may lead to underreporting. However, it should be noted that the sample size of the current study was high, close to ten thousand participants, and in epidemiological studies of this nature, conducting laboratory evaluations may not be necessary. Another point is that for assessing TD, available documents such as medical records, date of diagnosis, and treatment status were used to confirm self-reported cases.
Conclusion
The present study showed that prevalence of TD-DM comorbidity, especially in women and older adults, was considerable. Additionally, female gender, older age, smoking, high WC, low HDL, high TC, high TG, and low PA were predictors of TD-DM comorbidity.
Data availability
The data are available upon reasonable request from the corresponding author.
References
Rong, F. et al. Association between thyroid dysfunction and type 2 diabetes: A meta-analysis of prospective observational studies. BMC Med. 19, 1–13 (2021).
Kumar, R. et al. A review on diabetes mellitus: Type1 & Type2. World J. Pharm. Pharmaceut. Sci. 9, 838–850 (2020).
Eom, Y. S., Wilson, J. R. & Bernet, V. J. Links between thyroid disorders and glucose homeostasis. Diabetes Metab. J. 46, 239 (2022).
Jang, J. et al. Association between thyroid hormones and the components of metabolic syndrome. BMC Endocr. Disord. 18, 29. https://doi.org/10.1186/s12902-018-0256-0 (2018).
Chen, C., Xie, Z., Shen, Y. & Xia, S. F. The roles of thyroid and thyroid hormone in pancreas: Physiology and pathology. Int. J. Endocrinol. 2018 (2018).
Potenza, M., Via, M. A. & Yanagisawa, R. T. Excess thyroid hormone and carbohydrate metabolism. Endocr. Pract. 15, 254–262 (2009).
Mullur, R., Liu, Y.-Y. & Brent, G. A. Thyroid hormone regulation of metabolism. Physiol. Rev. (2014).
Ward, R. J., Heald, A. H., Ogunmekan, S., Fryer, A. A. & Duff, C. J. Should we be screening for thyroid dysfunction in patients with type 2 diabetes mellitus?. Br. J. Gen. Pract. 68, 94–95 (2018).
Ashwini, S., Bobby, Z. & Joseph, M. Mild hypothyroidism improves glucose tolerance in experimental type 2 diabetes. Chem. Biol. Interact. 235, 47–55 (2015).
Yang, W. et al. Subclinical hypothyroidism increases insulin resistance in normoglycemic people. Front. Endocrinol. 14, 1106968 (2023).
Raval, A. D. & Sambamoorthi, U. Incremental healthcare expenditures associated with thyroid disorders among individuals with diabetes. J. Thyroid Res. 2012 (2012).
Sharma, A. & Shivgotra, V. Prevalence of thyroid disease among the diabetic patients: A meta analysis. J. Med. Sci. Clin. Res. 5, 1–5 (2015).
Vamshidhar, I. & Rani, S. S. A study of association of thyroid dysfunctions in patients with type 2 diabetes mellitus. Maedica 15, 169 (2020).
Al-Geffari, M. et al. Risk factors for thyroid dysfunction among type 2 diabetic patients in a highly diabetes mellitus prevalent society. Int. J. Endocrinol. 2013 (2013).
Diez, J., Sánchez, P. & Iglesias, P. Prevalence of thyroid dysfunction in patients with type 2 diabetes. Exp. Clin. Endocrinol. Diabetes 119, 201–207 (2011).
Khassawneh, A. H. et al. Prevalence and predictors of thyroid dysfunction among type 2 diabetic patients: A case–control study. Int. J. Gen. Med. 803–816 (2020).
Kadiyala, R., Peter, R. & Okosieme, O. E. Thyroid dysfunction in patients with diabetes: Clinical implications and screening strategies. Int. J. Clin. Pract. 64, 1130–1139 (2010).
Eghtesad, S. et al. The PERSIAN cohort: Providing the evidence needed for healthcare reform. Arch. Iran. Med. 20, 691–695 (2017).
Poustchi, H. et al. Prospective epidemiological research studies in Iran (the PERSIAN Cohort Study): Rationale, objectives, and design. Am. J. Epidemiol. 187, 647–655 (2018).
Moosazadeh, M. et al. Enrolment phase results of the Tabari cohort study: Comparing family history, lipids and anthropometric profiles among diabetic patients. Osong Public Health Res. Perspect. 10, 289 (2019).
Kheradmand, M. et al. Tabari Cohort Profile and Preliminary Results in Urban Areas and Mountainous Regions of Mazandaran, Iran (2019).
Statistics, N. C. F. H. National Health and Nutrition Examination Survey (NHANES) Anthropometry Procedures Manual. 2015–2016 (US Department of Health and Human Services, 2016).
Sreelatha, M., Kumar, V. S., Shekar, G. C. & Shekar, V. C. Study of thyroid profile in patients with type 2 diabetes mellitus. Int. J. Sci. Study 5, 211–220 (2017).
Sharma, A. & Shivgotra, V. Prevalence of Thyroid Disease Among the Diabetic Patients: A Meta Analysis.
Hussain, T. et al. Prevalence and predictors of thyroid dysfunction among patients with type 2 diabetes mellitus attending a tertiary care hospital in an urban area of Bhubaneswar, Odisha. Thyroid Res. Pract. 16, 26–35 (2019).
Zhu, Y. et al. Prevalence of thyroid dysfunction in older Chinese patients with type 2 diabetes—A multicenter cross-sectional observational study across China. PLoS ONE 14, e0216151 (2019).
Talwalkar, P., Deshmukh, V. & Bhole, M. Prevalence of hypothyroidism in patients with type 2 diabetes mellitus and hypertension in India: A cross-sectional observational study. Diabetes Metab. Syndr. Obes. Targets Ther. 369–376 (2019).
Hassan-Kadle, M. A. et al. Spectrum and prevalence of thyroid diseases at a tertiary referral hospital In Mogadishu, Somalia: A retrospective study of 976 cases. Int. J. Endocrinol. 2021 (2021).
Sadatamini, M., Delshad, H., Amozghar, A., Tohidi, M. & Azizi, F. The Incidence of Thyroid Dysfunctions in Patients with Type 2 Diabetes: A Twelve-Year Follow-Up of the Tehran Lipid and Glucose Study (2015).
Elgazar, E. H., Esheba, N. E., Shalaby, S. A. & Mohamed, W. F. Thyroid dysfunction prevalence and relation to glycemic control in patients with type 2 diabetes mellitus. Diabetes Metab. Syndr. 13, 2513–2517 (2019).
Reddy, N., Pradeep, T., Tirupati, S., Sarathi, V. & Kumar, D. Thyroid dysfunction and its association with microvascular complications in patients with type 2 diabetes mellitus in south India. Diabetes Metab. Syndr. 14, 615–617 (2020).
Fregenal, M., D’Urso, M. & Luciardi, H. Prevalence of thyroid dysfunction in patients with type 2 diabetes mellitus. Medicina 76, 355–358 (2016).
Barmpari, M. E. et al. Thyroid dysfunction among Greek patients with type 1 and type 2 diabetes mellitus as a disregarded comorbidity. J. Diabetes Res. 2017 (2017).
Demitrost, L. & Ranabir, S. Thyroid dysfunction in type 2 diabetes mellitus: A retrospective study. Indian J. Endocrinol. Metab. 16, S334–S335 (2012).
Ershad, E., Ghaderian, S. B., Rashidi, H., Moradi, L. & Zaman, F. The Prevalence of Overt and Subclinical Hypothyroidism in Patients with Type 2 Diabetes Mellitus Admitted to Imam and Golestan Hospitals, Ahvaz, in 2020–2021 (2021).
Khatiwada, S. et al. Thyroid dysfunction and associated risk factors among Nepalese diabetes mellitus patients. Int. J. Endocrinol. 2015 (2015).
Umpierrez, G. E. et al. Thyroid dysfunction in patients with type 1 diabetes: A longitudinal study. Diabetes Care 26, 1181–1185 (2003).
Mervish, N. A. et al. Thyroid antagonists (perchlorate, thiocyanate, and nitrate) and childhood growth in a longitudinal study of US girls. Environ. Health Perspect. 124, 542–549 (2016).
Serrano-Nascimento, C. & Nunes, M. T. Perchlorate, nitrate, and thiocyanate: Environmental relevant NIS-inhibitors pollutants and their impact on thyroid function and human health. Front. Endocrinol. 13, 995503 (2022).
Alnima, T., Meijer, R., Spronk, H., Warlé, M. & Cate, H. T. Diabetes-versus smoking-related thrombo-inflammation in peripheral artery disease. Cardiovasc. Diabetol. 22, 257 (2023).
Hussein, S. M. M. & Abd Elmageed, R. M. The relationship between type 2 diabetes mellitus and related thyroid diseases. Cureus 13 (2021).
Biondi, B., Kahaly, G. J. & Robertson, R. P. Thyroid dysfunction and diabetes mellitus: Two closely associated disorders. Endocr. Rev. 40, 789–824 (2019).
Zhang, D. et al. Important hormones regulating lipid metabolism. Molecules 27, 7052 (2022).
Rizos, C., Elisaf, M. & Liberopoulos, E. Effects of thyroid dysfunction on lipid profile. Open Cardiovasc. Med. J. 5, 76 (2011).
Lee, Y.-W., Yang, T.-T., Lin, Y.-Y. & Hsieh, Y.-S. Elevated free thyroxine levels might alter the effect of the lipid profile on insulin resistance in type 2 diabetes mellitus. Diagnostics 13, 2656 (2023).
Liu, H. & Peng, D. Update on dyslipidemia in hypothyroidism: the mechanism of dyslipidemia in hypothyroidism. Endocr. Connect. 11 (2022).
Acknowledgements
We would like to thank all the members of PERSIAN and TABARI cohort study (Ministry of Health and Medical Education and Mazandaran University of Medical Sciences) for all they did for this program.
Author information
Authors and Affiliations
Contributions
MM, SK, and EG acquired data, performed the statistical analyses, interpreted data, and drafted and revised the manuscript for important intellectual content and approved the final version. RA and AH reviewed the analyses and the final version of the manuscript. AB and MK interpreted data and revised the manuscript for important intellectual content and approved the final version. All authors have read and approved the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Moosazadeh, M., Khakhki, S., Bahar, A. et al. The prevalence and determinants of diabetes mellitus and thyroid disorder comorbidity in Tabari cohort population. Sci Rep 14, 17577 (2024). https://doi.org/10.1038/s41598-024-68569-3
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-024-68569-3