Abstract
Silver nanoparticles (AgNPs) have been successfully synthesized using leaf extract of Neem (Azadirachta Indica), Mint (Mentha Piperita), Tulsi (Ocimum Tenuiflorum), Bermuda grass (Cynodon Dactylon) and silver salt. As plant extracts produce best capping material for the stabilization of nanoparticles. AgNPs were characterized by UV–Vis spectroscopy in range of 200–800 nm and transmission electron microscopy TEM, XRD and FTIR. The nanoparticles synthesized were mainly in sizes between 25 and 100 nm. They appeared to be spherical, nanotriangles and irregular in shape. Catalytic application was observed for all the aqueous solution of leaves, quantity taken was 1 ml, 2 ml, 3 ml, 4 ml and 5 ml. Furthermore, prepared Ag nanoparticles are also used for seed germination.
Similar content being viewed by others

Introduction
Nanotechnology is the study of minute Particles. Particles having dimension in the range of 1–100 nm. Which involves altering materials within that size range. Nano is a Greek word meaning dwarf. One nanometer (nm) is one billionth of a meter. A human hair is 10,000 times bigger than a nm1. Nanotechnology is based on the fact that nanoscale particles hold properties of their own which are different from that of the bulk materials altogether2. The particles have physiochemical properties which get influenced with alteration in the particle size. Nanotechnology produce materials of different types which can be used for different purposes according to their properties and their sizes3. Traditional methods used for the synthesis of nanoparticles are very time taking, costly, and they require toxic chemicals for the process. Also the by- products formed in the reaction are very hazardous for the environment. Thus an alternate method is required which can overcome the shortcomings of the chemical methods such as it has to be non- toxic, less time consuming and should be economically cheap. Herein, the green synthesis plays a convenient role as it is non- toxic, requires less time and is economically cheap4. A lot of work has been done on green synthesis of nano-particles using living organisms including bacteria, fungi and plants because of their antioxidant properties capable of reducing metal compounds in their respective nanoparticle. Plant extracts produce best capping material for the stabilization of nanoparticles3. Extracts of leaves, fruits, stems are being utilized for the production of nanoparticles as they contain phytochemicals like polyphenols, alkaloids, flavonoids that act as reducing as well as stabilizing agents all together5.
Considering the advantages of green synthesis over other methods, this study aims to study the synthesis of AgNPs using aqueous Neem (Azadirachta Indica)6,7,8,9,10, Mint (Mentha Piperita)11,12,13, Tulsi (Ocimum Tenuiflorum)14,15,16,17, Bermuda grass (Cynodon Dactylon)4,18,19 leaves extract. Furthermore, silver based composites like Ag–CuO20, Ag/NiO21, Ag/ZnO22 and AgSnO2/Nb2O5-SiO223 also reported for various photocatalytic applications such as degradation of methylene blue with their antibacterial activity. Similarly, other composites like Pd/TiO224 and Co/ZnO25 also prepared by green methods and used for anti-bacterial activities.
It focuses on the study of the of catalytic reduction of Ag nanoparticles with 4- Nitroaniline. We also attempt to investigate the effect of NPs in seed germination with Cyamopsis Tetragonoloba (Cluster beans) seed.
Materials and methods
Preparation of leaf extracts
Silver nitrate was obtained from Thermo Fisher Scientific. All the glassware were washed with distilled water and dried in oven. The petri plates were also washed and were autoclaved before using. Leaves were collected from their natural habitats, which are available widely in farms and thoroughly washed with deionized water (D.W.) numerous times to remove any impurities and debris (Fig. 1). Then the leaves were shredded and allowed to dry in the oven overnight drying at 60 °C. Afterwards, dry leaves were grounded in a stain- less steel mixer to have a fine powder (Fig. 2). 20 gm from the resultant powder of each plant material were mixed with 250 ml of distilled water. The mixture then was heated at 50 °C for approximately 45 min for the extraction of leaves. Eventually, the hot solution was filtered and the filtrate was stored in a glass beaker at 4.0 °C for further use.
Pictures of used plants in nanomaterials.
Powder of all the plant material.
Biosynthesis of silver nanoparticles
1 mM (170 mg) silver nitrate (AgNO3) is mixed with 90 ml distilled water. To this 90 ml silver salt solution 10 ml of leaves extract of each sample was added separately in different conical flask (Fig. 3) and allow to stir. Change in color was observed from light yellow to dark brown indicating the formation of silver nanoparticles.
Nanoparticles made from all the plant material.
Characterization
UV–Visible analysis
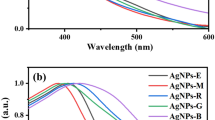
UV–Visible spectrophotometer model Multiskan GO 1.01.12 was used for absorbance measurements. 20 µL each of the samples was diluted with DI water. It is the most important technique and the simplest way to confirm the formation of nanoparticles. The spectrum of the colloidal sample was obtained in the range of 200–800 nm. The UV–Vis spectrum shows the important role of AgNO3 and the presence of ingredients in the leaves for the formation of silver nanoparticles.
The absorption spectra exhibit a gradual decrease of the absorbance, accompanied by a shift in the wavelength from 381 to 307 nm. It is also observed that the surface Plasmon peak is slowly shifted toward lower wavelength at high concentrations. The appearance of Ag peak at shorter wavelength indicates small size of Ag-NPs that were formed, while a longer wavelength reveals bigger Ag-NPs.
The UV–visible absorption spectra of the Ag nanoparticles, concentration of Neem (Azadirachta Indica), Mint (Mentha Piperita), Tulsi (Ocimum Tenuiflorum), Bermuda grass (Cynodon Dactylon) leaf extract of 4 ml are recorded and shown in Figs. 4, 5, 6, and 7 respectively.
The UV–visible absorption spectra of the Ag nanoparticles, concentration of Neem (Azadirachta Indica).
The UV–visible absorption spectra of the Ag nanoparticles, concentration of Mint (Mentha Piperita).
The UV–visible absorption spectra of the Ag nanoparticles, concentration of Tulsi (Ocimum Tenuiflorum).
The UV–visible absorption spectra of the Ag nanoparticles, concentration of Bermuda grass (Cynodon Dactylon).
TEM (Transmission electron microscopy)
Transmission electron microscopy images of AgNP’s are shown in below. According to TEM micrographs the obtained silver nanoparticles had different morphology. The nanoparticles synthesized with 4 ml of extract were mainly in sizes between 25 and 100 nm (Fig. 8). In TEM micrographs spherical, nanotriangles and irregular nanoparticles were observed. The fact, that in the present work at higher concentrations the AgNP’s presented more diversity in morphology could be attributed to the high amount and variety of chemical constituents present in the extract of mint, in which each one presents different reducing, chelating and binding properties (Fig. 9).
The UV–visible absorption spectra of the 4-Nitro Aniline and reduced 4-Nitro Aniline.
TEM of the synthesized AgNPs.
XRD of synthesised Ag NPs
Crystalline size and structure of the silver nanoparticle is carried out by XRD (Fig. 10). The three distinct diffraction peaks of the 2 h values of 28°, 38.1°, 46.1°, and 27.8° can be assigned the plane of (100), (1 1 1), and (2 00) respectively indicating the silver nanoparticles are fcc and crystalline (JCPDS file no. 84-0713 and 04-0783).
XRD analysis of the synthesized AgNPs.
FTIR study of synthesised Ag NPs
FTIR analysis of leaf and synthesised silver nanoparticles were carried out to identify the possible functional groups which are responsible for capping and efficient stabilization of the metal nanoparticles synthesized by leaf extract. Figure 11 shows the peaks near 3334.20 cm−1, 2927 cm−1, and 2846 cm−1 assigned to OH stretching and aldehydic C–H stretching, respectively. The N–H bend of primary amines and C–H bend of alkanes occurred at 1609.90 cm−1 and 1365.77 cm−1, respectively. As reported in the previous studies, this N–H bend is responsible for stability of AgNPs26. FTIR spectra of silver nanoparticles is showing prominent peaks at 2911 cm−1, 1618 cm−1 and 1317 cm−1.Peak at 1618 cm−1 denoted to the stretching vibration of (NH) C=O group, while peak at 1317 cm−1 and 2911 cm−1 observed for C–H and C–H (methoxy compounds) stretching vibration, respectively27.
Comparative FTIR study of the synthesized AgNPs.
Catalytic activity
To check the catalytic activity of synthesized Ag NPs, the reduction of 4-nitroaniline (4-NA) was carried out in the test tube in the presence of the sodium borohydride. The catalytic reduction was performed with all the leaf samples and were optimized to find out which quantity of synthesized AgNPs were fast reducing 4-nitroaniline. The quantity of AgNPs solution taken for the optimization was 1 ml, 2 ml, 3 ml, 4 ml and 5 ml. The quantity taken for 4-NA was 14 mg and for the NaBH4 the quantity taken was 18 mg.
Experimental procedure
14 mg of 4-NA is added to the test tube along with distilled water (DI) and is heated till it dissolves allowing to be stirred for 5 min on magnetic stirrer. After this, AgNPs solution was added to the test tube letting all this gets mixed altogether (Tables 1, 2, 3, and 4). Post of which 18 mg NaBH4 is mixed with 1 ml of DI and then is added to the test tube solution.
Seed germination experiment
Silver nanoparticles (AgNPs) have been implicated nowadays to enhance seed germination, plant growth, improvement of photosynthetic quantum efficiency and as antimicrobial agents to manage plant diseases. The interactions of nanomaterials with plants have not been fully elucidated. There are different and often conflicting reports on the absorption, translocation, accumulation, biotransformation, and toxicity of nanoparticles on various plant species. Silver nanoparticles (AgNPs) is one of the nanomaterials that its effects are under investigation. The impact of AgNPs on higher plants appears to depend on the species and age of plants, the size and concentration of the nanoparticles, the experimental conditions such as temperature, and the duration and method of exposure. This study showed that exposure to AgNPs caused both positive and negative effects on seed germination. The seeds taken here for the experiment are Cyamopsis Tetragonoloba (cluster beans).
Seeds were washed with a 0.5% mercury chloride solution for 3 min to ensure surface sterility then, they were washed with distilled water for 5 min, and then soaked in serial of different prepared leaf AgNPs concentration suspension with 10 ml of a test solution for respective timings of 24 h (Fig. 12), 3 h (Fig. 13), 6 h (Fig. 14) and 9 h (Fig. 15). One piece of filter paper was put into each 100 mm X 15 mm Petri dish. Seeds were transferred onto the filter paper, with 4 seeds per dish at 2 cm or larger distance between each seed. Petri dishes were covered and sealed with tape, and were incubated at room temperature. Figure 9 shows, the after 96 h with the sample solution, the seeds showed signs of damage, becoming burnt during the subsequent 24-h soaking period with the same solution. Figure 10 shows, the Following the initial 96-h, prior soaking of seeds for 3 h. Observation revealed that seedlings had emerged in nearly all Petri dishes, with only one left with no seedlings. Figure 11 shows, the seeds underwent a 6-h soaking period. Upon observation after 96 h, it was revealed that out of 5 batches of Petri dishes, only 3 showed signs of seedling emergence, while the remaining two did not. Furthermore, the number of observed seedlings was notably low, suggesting varied responses to the solution and indicating potential limitations in promoting germination. Figure 12 shows, The seeds underwent a 9-h soaking period, which yielded the most favourable results in terms of seedling emergence. Upon observation after 96 h, it was revealed that out of 5 batches of Petri dishes all but one didn’t showed signs of seedling emergence. Furthermore, the seedlings exhibited substantial growth to the extent that their length could be measured.
After 96 h with sample solutions prior 24 h of soaking seeds with sample solution.
After 96 h prior 3 h soaking of seeds with sample solution.
After 96 h prior 6 h soaking of seeds.
After 96 h prior 9 h soaking of seeds.
Results and discussions
Reduction of the solution was observed in different time intervals with UV spectrometry and the time for different samples were noted down. As shown in Fig. 8., 4-nitroaniline observed at 380 nm, after reduction with Ag NPs, it disappeared. The best results were shown with the 5 ml quantity of AgNPs solution of different leaf extract and as well as the quantity of Ag NPs solution decreases, the reduction time of 4-Nitro aniline increases due to availability of low concentration of Ag ions. NPs act as an electron transfer medium between the adsorbed molecules High concentrations of Ag ions are enhancing the transmission of an electron from a donor-reducing agent (BH4−1) to acceptor dye molecules (4-NA)28 (Scheme 1).
Reduction of 4–Nitro Aniline.
All the seed germination results were taken after 96 h. All results revealed after 3 h (Table 5), 6 h (Table 6) and 9 h (Table 7) soaking in aqueous solution of Ag NPs. After 3 h soaking, results reveal consistent seedling development with Neem, minimal germination with Mint, and mixed responses with Grass and Tulsi as shown in Table 5. The Control treatment showed steady seedling emergence. After 6 h soaking (Table 6), across different Petri plates, varying degrees of seedling emergence were observed. Notably, Neem and Control treatments consistently promoted seedling growth, while Mint and Grass treatments exhibited limited effectiveness. Tulsi treatment yielded mixed results.
After 9 h of soaking (Table 7), the Neem treatment showed no significant impact on germination, while the Control treatment consistently led to the highest seedling emergence. Mint and Grass treatments displayed effectiveness. Tulsi treatment did not result in any seedling. These findings underscore the diverse effects of treatments on seed germination, necessitating further investigation for optimization.
AgNPs have been implicated nowadays in agriculture to improve crops. The study showed that the cluster beans revealed different dosage response to AgNPs on germination percentage and the measured growth characters. Result indicated that exposure to AgNPs had significant effects on seed germination and seedling growth of plant. The best dose of AgNPs for plant detected by our results is in 9 h soaking of 10 ml solution of each leaf sample separately, which enhanced germination percentage and germination rate for highest values.
Conclusion
Silver nanoparticles (AgNPs) were manufactured using aqueous extract of Neem (Azadirachta Indica), Mint (Mentha Piperita), Tulsi (Ocimum Tenuiflorum), Bermuda grass (Cynodon Dactylon) leaves. Employment of plant extracts in the synthesis of metallic NPs is fast, environment friendly, non-pathogenic, and economical. Metallic ions are reduced and stabilized by the phytochemicals present in the leaves. The formed nanoparticles were showing surface Plasmon peak from 307 to 381 nm. The synthesized NPs were efficient in terms of stability. The rate formation of NPs using green method is easier, single step, eco-friendly, cheap, and requires absolute no chemical energy process. Furthermore, the synthesized NPs were utilized for the catalytic application and the best results obtained was with the 4 ml leaf extract for all the samples. Germination is important for determining the final plant density if planted seeds germinate completely and vigorously. The obtained results from germination were best found in 9 soaking of seeds.
Data availability
The datasets used and/or analysed during the current study available from the corresponding author on reasonable request.
References
Bayda, S., Adeel, M., Tuccinardi, T., Cordani, M. & Rizzolio, F. The history of nanoscience and nanotechnology: From chemical-physical applications to nanomedicine. Molecules 25, 1–15. https://doi.org/10.3390/molecules25010112 (2020).
Khan, I., Saeed, K. & Khan, I. Nanoparticles: Properties, applications and toxicities. Arab. J. Chem. 12, 908–931. https://doi.org/10.1016/j.arabjc.2017.05.011 (2019).
Sahu, N. et al. Synthesis and characterization of silver nanoparticles using Cynodon dactylon leaves and assessment of their antibacterial activity. Bioprocess Biosyst. Eng. 36, 999–1004. https://doi.org/10.1007/s00449-012-0841-y (2013).
Eltaweil, A. S. et al. Green synthesis of platinum nanoparticles using Atriplex halimus leaves for potential antimicrobial, antioxidant, and catalytic applications. Arab. J. Chem. 15, 103517. https://doi.org/10.1016/J.ARABJC.2021.103517 (2022).
Ulaeto, S. B. et al. Biogenic Ag nanoparticles from neem extract: Their structural evaluation and antimicrobial effects against Pseudomonas nitroreducens and Aspergillus unguis (NII 08123). ACS Biomater. Sci. Eng. 6(1), 235–245. https://doi.org/10.1021/acsbiomaterials.9b01257 (2020).
Bhat, R. S., Almusallam, J., Al Daihan, S. & Al-Dbass, A. Biosynthesis of silver nanoparticles using Azadirachta indica leaves: Characterisation and impact on Staphylococcus aureus growth and glutathione-S-transferase activity. IET Nanobiotechnol. 13, 42–46. https://doi.org/10.1049/iet-nbt.2018.5133 (2019).
Sajesh Kumar, N. K., Vazhacharickal, J., Mathew, J. J. & Joy, J. Synthesis of silver nano particles from neem leaf (Azadirachta Indica) extract and its antibacterial activity. Int. J. Appl. Pharm. Biol. Res. 2, 38–49 (2017).
Verma, A. & Mehata, M. S. Controllable synthesis of silver nanoparticles using Neem leaves and their antimicrobial activity. J. Radiat. Res. Appl. Sci. 9, 109–115. https://doi.org/10.1016/j.jrras.2015.11.001 (2016).
Roy, P., Das, B., Mohanty, A. & Mohapatra, S. Green synthesis of silver nanoparticles using azadirachta indica leaf extract and its antimicrobial study. Appl. Nanosci. 7, 843–850. https://doi.org/10.1007/s13204-017-0621-8 (2017).
Gabriela, Á. -M. et al. Biosynthesis of silver nanoparticles using mint leaf extract (Mentha piperita) and their antibacterial activity. Adv. Sci. Eng. Med. 9, 914–923. https://doi.org/10.1166/asem.2017.2076 (2017).
Sarkar, D. & Paul, G. Green synthesis of silver nanoparticles using Mentha asiatica (Mint) extract biosciences and plant biology green synthesis of silver nanoparticles using Mentha asiatica (Mint) extract and evaluation of their antimicrobial potential. Int. J. Curr. Res. Biosci. Plant Biol. 4, 77. https://doi.org/10.20546/ijcrbp.2017.401.009 (2017).
Alex, K. V. et al. Green synthesized Ag nanoparticles for bio-sensing and photocatalytic applications. ACS Omega https://doi.org/10.1021/acsomega.0c01136 (2020).
Singhal, G. & Bhavesh, R. Biosynthesis of silver nanoparticles using Ocimum sanctum (Tulsi) leaf extract and screening its antimicrobial activity. J. Nanoparticle Res. https://doi.org/10.1007/s11051-010-0193-y (2011).
J. Singh, L. Singh, R. Bajaj, A.S. Batth, M. Rawat Green Synthesis of Silver nanoparticles using Ocimum sanctum (Tulsi) leaf extract and evaluation of their antimicrobial activities Antimicrobial Chart, (2016) 16–17.
Ramteke, C., Chakrabarti, T., Sarangi, B. K. & Pandey, R. Synthesis of silver nanoparticles from the aqueous extract of leaves of Ocimum sanctum for enhanced antibacterial activity. J. Chem. 2013, 278625 (2013).
Tailor, G., Yadav, B. L., Chaudhary, J., Joshi, M. & Suvalka, C. Green synthesis of silver nanoparticles using Ocimum canum and their anti-bacterial activity. Biochem. Biophys. Rep. 24, 100848. https://doi.org/10.1016/j.bbrep.2020.100848 (2020).
Supraja, S. & Arumugam, P. Antibacterial and anticancer activity of silver nanoparticles synthesized from Cynodon dactylon leaf extract. J. Acad. Ind. Res. 3, 629–631 (2015).
Sharma, R. K. et al. Cynodon dactylon leaf extract assisted green synthesis of silver nanoparticles and their anti-microbial activity. Adv. Sci. Eng. Med. 5, 858–863. https://doi.org/10.1166/asem.2013.1352 (2013).
Supraja, S. et al. Green synthesis of silver nanoparticles from Cynodon dactylon leaf extract. Int. J. Chem. Tech. 5, 271–277 (2013).
Farooq, M. et al. Phytoassisted synthesis of CuO and Ag–CuO nanocomposite, characterization, chemical sensing of ammonia, degradation of methylene blue. Sci. Rep. 14, 1618 (2024).
Khan, A. U. et al. Uncaria rhynchophylla mediated Ag/NiO nanocomposites: A new insight for the evaluation of cytotoxicity, antibacterial and photocatalytic applications. Photodiagn. Photodyn. Ther. 37, 102681 (2022).
Khan, M. J. et al. Facile synthesis of silver modified zinc oxide nanocomposite: An efficient visible light active nanomaterial for bacterial inhibition and dye degradation. Photodiagn. Photodyn. Ther. 36, 102619 (2021).
Ullah, M. A. et al. Photocatalytic removal of alizarin red and photoinhibition of microbes in the presence of surfactant and Bio-template mediated Ag/SnO2/Nb2O5-SiO2 nanocomposite. J. Mol. Liq. 370, 121042 (2023).
Subhan, A. et al. A new study of biomediated Pd/TiO2: A competitive system for Escherichia coli inhibition and radical stabilization. Mater. Res. Express 6(12), 125430 (2020).
Alam, N. et al. Effect of light-dark conditions on inhibition of Gram positive and gram negative bacteria and dye decomposition in the presence of photocatalyst Co/ZnO nanocomposite synthesized by ammonia evaporation method. Photodiagn. Photodyn. Ther. 38, 102853 (2022).
Niraimathi, K. L., Sudha, V., Lavanya, R. & Brindha, P. Biosynthesis of silver nanoparticles using Alternanthera sessilis (Linn.) extract and their antimicrobial, antioxidant activities. Colloids Surf. B Biointerfaces 102, 288–291 (2013).
Marimuthu, S. et al. Evaluation of green synthesized silver nanoparticles against parasites. Parasitol. Res. 108, 1541–1549 (2011).
Din, M. I. et al. Critical review on the chemical reduction of nitroaniline. RSC Adv. 10(32), 19041–19058 (2020).
Author information
Authors and Affiliations
Contributions
AS and RV has conducted the experiments, RT and AJ has edited and formatted the manuscript. AD has helped technically in manuscript. SC and MSC has designed and conceptualized the experiment work.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Saini, A., Verma, R., Tiwari, R. et al. Green synthesis of silver nanoparticle for catalytic applications and priming study by seed germination. Sci Rep 14, 20744 (2024). https://doi.org/10.1038/s41598-024-69120-0
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-024-69120-0
Keywords
This article is cited by
-
Ficus religiosa Bark Extract-Mediated Synthesis of Calcium Oxide Nanoparticles: Physicochemical Characterization and Evaluation of their Therapeutic Properties
Applied Biochemistry and Biotechnology (2026)