Abstract
Potatoes (Solanum tuberosum L.) are a significant food crop cultivated around the world. Caffeic acid (CA) can enhance plant growth by promoting antioxidant activity and stimulating root development, contributing to overall plant health and vigor. Cobalt sulfate (CoSO4) boosts plant growth by promoting nitrogen (N) fixation, healthier root development, and chlorophyll synthesis, enhancing photosynthesis and overall plant health. Nanoparticle-coated urea (NPCU) improves nutrient uptake, promoting plant growth efficiency and reducing environmental impact. This study investigates the effects of combining CA, CoSO4, and NPCU as amendments on potatoes with and without NPCU. Four treatments, control, 20 μM CA, 0.15 mg/L CoSO4, and 20 μM CA + 0.15 mg/L CoSO4 with and without NPCU, were applied in four replications using a completely randomized design. Results demonstrate that the combination of CA + CoSO4 with NPCU led to an increase in potato stem length (~ 6%), shoot dry weight (~ 15%), root dry weight (~ 9%), and leaf dry weight (~ 49%) compared to the control in nutrient stress. There was a significant rise in chlorophyll a (~ 27%), chlorophyll b (~ 37%), and total chlorophyll (~ 28%) over the control under nutrient stress also showed the potential of CA + CoSO4 with NPCU. In conclusion, the findings suggest that applying CA + CoSO4 with NPCU is a strategy for alleviating potato nutrient stress.
Similar content being viewed by others
Introduction
Nutrient deficiency can essentially affect plant growth and development1. Plants require essential nutrients, such as phosphorus (P), nitrogen (N), potassium (K), and various micronutrients, to perform key physiological processes. When these nutrients decrease in the soil, plants perform functions like photosynthesis, root development, and cell expansion2. Plants with nutrient deficiencies had smaller tubers, lower yields, and slowed growth. Certain signs indicate a lack of a nutrient, such as dark green leaves with purple discoloration for a P deficit and chlorosis of the leaves in an N shortage3. Low nutrient levels can prevent roots from growing, which limits a plant's capacity to absorb water and other necessary components. These deficiencies caused the risk of disease and pest attacks4. The growing global population and increasing food demands are putting pressure on agricultural farming and natural resources, resulting in a shortage of nutritious food and environmental effect. By 2050, a worldwide population of 9.7 billion will request 70% more food than at present consumed5. Recognizing and mitigating nutrient inadequacy is crucial for protecting soil and advancing manageable farming, meeting the nutritional requirements of a developing worldwide population6.
Caffeic acid (CA) has shown promising effects on plant growth and stress tolerance. At low concentrations (20–30 ppm), CA significantly improved growth, yield, and physiological processes in faba beans and lentils7,8. CA enhanced photosynthetic pigments, amino acids, and seed yield while reducing antinutritional factors like vicine8. In drought-stressed lentils, CA combined with ACC-deaminase-producing rhizobacteria improved plant height, pod number, and nutrient uptake while reducing stress indicators7. However, higher CA concentrations can inhibit root growth in soybeans by increasing lignification through the phenylpropanoid pathway9. Early research also demonstrated CA growth-promoting activity in pea epicotyl straight growth tests10.
Cobalt has been shown to enhance nitrogen uptake and utilization in plants, particularly legumes. Studies on pea11, cowpea12, and soybean13 demonstrated that cobalt application increased plant growth, yield, and nutrient content, especially nitrogen. Cobalt's role in improving nitrogen fixation efficiency is attributed to its essential function in vitamin B12 synthesis, required by enzymes involved in N2 fixation14. The optimal cobalt concentration for plant growth varies, with 8 ppm for pea11, 7.5 ppm for cowpea12 , and 12 mg/kg for soybean showing the best results13. However, excessive cobalt can be toxic to plants14. Adding cobalt can reduce the required nitrogen fertilizers while maintaining or improving plant growth and yield11,12,13.
Applying nanoparticle-coated urea enhances plant growth by facilitating efficient nutrient uptake and utilization15. The nanoparticles serve as carriers, gradually releasing urea, which promotes sustained nutrient availability to plants16. This controlled release mechanism minimizes N loss through leaching or volatilization, maximizing plant nutrient absorption. Incorporating silica nanoparticles into polyacrylate emulsions for urea coating has been shown to extend nutrient release duration and improve wear resistance17,18,19. Consequently, plants exhibit improved physiological processes, including enhanced photosynthesis, increased biomass accumulation, and heightened resistance to environmental stressors20.
Potato (Solanum tuberosum L.) ranks as one of the most widely cultivated vegetable crops globally, with a presence in 79% of countries worldwide21. It is fourth in production volume, followed by rice, wheat, and maize. Potatoes are a valuable source of affordable and readily available proteins, essential amino acids, carbohydrates, minerals, antioxidants, and vitamins globally22. Nutrient deficiency in potatoes leads to stunted growth, reduced yield, and poor-quality tubers. Potatoes need a balanced supply of N, P, K, and micronutrients. Addressing these deficiencies through appropriate fertilization strategies is crucial for optimal growth and yield.
That’s why the present study was planned to investigate the influence of CA and CoSO4 in combination with NPCU on potato growth cultivated in N-deficient soil. This study examines the individual and combined effects of CA and CoSO4 with and without NPCP on the growth of potato plants in N-deficient soil. The study covers the knowledge gap regarding the sole and combined use of CA and CoSO4 with and without NPCU for the improvement in the growth of potato plants. Compared to the control, potato growth might be improved in N-deficient soils by applying CA and CoSO4 with NPCU.
Material and methods
Experimental site
In 2022, a pot experiment was conducted in the experimental area of ResearchSolution (30°09′41.6″N 71°36′38.0″E). During the experiment conduction, the temperature was maintained at 25–30 °C. Humidity was maintained at 60–70%. The day sunlight exposure was 6–8 h. Random soil sampling was done from the research area to characterize soil physicochemical properties. The soil and irrigation water parameters before experimentation were given in Table 1.
Nanoparticles coated urea
A standard protocol23 was utilized to synthesize Urea-covered nanoparticles, integrating specific modifications. Polyvinyl alcohol (PVA) was chosen as the coating material due to its stable ecological compatibility, biocompatibility, and film-framing properties. The surface of a single superphosphate was treated with a solution of 10 mg ZnO nanoparticles and PVA (7%) 24 and coated urea dried at ambient temperature to create a durable PVA coating layer.
Experimental design and treatment plan
In this experiment, a fully randomized design (CRD) was used to deliver four treatments in four replicates: control, caffeic Acid (20 µM CA), cobalt sulfate (0.15 mg/L CoSO4), and 20 µM CA + 0.15 mg/L CoSO4. Nanoparticles-coated urea (NPCU) (5 mg ZnO NP/kg soil) was mixed in the soil during soil preparation for pot filling. However, CA and CoSO4 foliar application were done in 2 splits. The first foliar was done after three weeks of germination, while the second was done before tuber initiation (42 days after germination).
Seed collection, sowing, and thinning
For experimental purposes, the potato variety Sahiwal White (FD 73–110) was sown in 2nd fortnight of October. Each pot filled with 10 kg of soil was then sown with seventeen seeds. Two seedlings in each pot were maintained by thinning after germination.
Fertilizer
To meet the nutritional needs of potatoes, we implemented a N application at a rate of 247 kg per ha (equivalent to around 0.31 g/10 kg soil) obtained from urea. Simultaneously, P was introduced at a rate of 124 kg per ha (almost 0.15 g/10 kg soil), sourced from a single superphosphate, by recommended application protocols. Additionally, to enhance K levels, we augmented the soil with 124 kg per acre of K sulfate (about 0.15 g/10 kg soil). In the control plant, simple urea was added, but there was no nanoparticle coating. All fertilizers were applied in the soil during soil preparation for pot filling.
Irrigation
The irrigation control for individual pots was meticulously handled using a moisture indicator (ADVANCED™; 4 in 1 Soil Meter; China). Regular monitoring and daily supervision were conducted to maintain moisture levels within the specified range, ensuring 70% of the soil field capacity was maintained.
Harvesting and collection of data
After 65 days of sowing, harvesting was performed for data collection (tuber bulking stage). The fresh weight of samples was assessed using digital balance soon after harvesting. For dry weight assessment, samples were oven-dried for 72 h at 65 °C.
Chlorophyll contents and gas exchange attributes
To assess the chlorophyll contents in freshly harvested leaf samples, we ground the samples with 5 ml of 80% acetone24. After that filtration was done to remove the plant tissue residues. The absorbance of the aliquot was assessed at 645, 663 nm, and 480 nm wavelength using a UV-spectrophotometer (Multipurpose Shimadzu UV-1280)25. Using a CI-340 photosynthesis instrument to measure gas exchange attributes including intercellular CO2, leaf stomatal conductance, and net transpiration rate26. Fully expanded leaves were placed in the clip during the sunny morning from 10:45 to 11:35 AM, and readings of attributes were recorded.
Total soluble sugar, protein, and free proline
The method proposed by27 was used for assessing total sugar content using anthrone and 80% H2SO4, subjected to a 10-min water bath, heated and cooled, and measured optical density at 620 nm using a UV-spectrophotometer (Multipurpose Shimadzu UV-1280). Fresh leaf samples were standardized with 50 mM K phosphate buffer, centrifuged at 10,000×g for 15 min at 4 °C, and protein concentration in the extract was measured using the method of28. The glacial acetic acid, ninhydrin solutions, and sulfosalicylic acid were used to measure free proline content, following protocol29. The combined solution was heated at 100 °C, and 5 ml of toluene was added. Subsequently, the absorbance was recorded at 520 nm.
Antioxidants
Using the protocol outlined by30, the amount of superoxide dismutase (SOD) was measured by observing the decrease of nitro blue tetrazolium (NBT) at 560 nm. At 420 nm, peroxidase (POD) activity was measured31. The activity of catalase (CAT) was assessed by measuring the decrease in absorbance at 240 nm, which results in the breakdown of hydrogen peroxide (H2O2) Aebi (1984). Following the protocol, Ascorbate peroxidase (APX) activity was calculated by checking ascorbate oxidation in the presence of H2O2 at 290 nm. The study determined Malondialdehyde (MDA) levels by reacting the sample extract with thiobarbituric acid (TBA), forming a colored complex, and measuring its absorbance at 532 nm32.
Free proline determination
N, P, K, and Ca in leave
Initially, N sulfuric acid33 and P and K di-acid mixture34 were used for digestion. To assess N content using glass Kjeldahl’s distillation apparatus standard method was followed35. The P content was assessed at 420 nm using protocol35. A flame photometer (microprocessor-1385) was used to quantify K and Ca content36,37.
Statistical analysis
The data was subjected to standard statistical analysis38. A two-way ANOVA was applied using OriginPro 2024b (64-bit) software; 10.1.5.132. The paired comparison was done for making graphs and statistical lettering on bars at p ≤ 0.05 using the Tukey test. Convex hull and hierarchical cluster analysis examined the best treatment and most representable attribute using OriginPro 2024b (64-bit) software; 10.1.5.13239.
Results
Stem length, shoot, root, and leaf dry weight
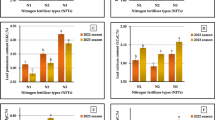
Applying 20 μM CA, 0.15 mg/L CoSO4 and 20 μM CA + 0.15 mg/L CoSO4 with no nanoparticle resulted increase in stem length (~ 8%, ~ 3%, and ~ 11%), shoot dry weight (~ 12%, ~ 4%, and ~ 19%), root dry weight (~ 4%, ~ 3%, and ~ 10%), and leaf dry weight (~ 47%, ~ 15%, and ~ 79%) than the control. Treatment 20 μM CA, 0.15 mg/L CoSO4, and 20 μM CA + 0.15 mg/L CoSO4 with NPCU caused a rise in stem length (~ 4%, ~ 3%, and ~ 6%), shoot dry weight (~ 11% ~ 8%, and ~ 15%), root dry weight (~ 5%, ~ 4%, and ~ 9), and leaf dry weight (~ 32%, ~ 11%, and ~ 49%) over the control (Fig. 1A–D).
Effect of caffeic acid (CA), cobalt sulfate (CoSO4), and nanoparticle coated urea (NPCU) on stem length (A), shoot dry weight (B), root dry weight (C) and leaf dry weight (D) concentration of leaves of potatoes. The Tukey test measured significant differences (p ≤ 0.05); distinct letters on the bars are the mean of four replicates ± SE.
Chlorophyll content and carotenoids
A significant increase in chlorophyll a (~ 46%, ~ 25%, and ~ 58%), chlorophyll b (~ 21%, ~ 70% ~ 46%), total chlorophyll (~ 46%, ~ 26%, and ~ 61%), and carotenoids (~ 42%, ~ 18%, and ~ 61%) were recorded than the control. Applying 20 μM CA, 0.15 mg/L CoSO4, and 20 μM CA + 0.15 mg/L CoSO4 treatments exhibit an increase in chlorophyll a (~ 18%, ~ 8%, and ~ 27%) chlorophyll b (~ 19%, ~ 80%, and ~ 45), total chlorophyll (~ 17%, ~ 7%, and ~ 28%), and carotenoids (~ 21%, ~ 10%, and ~ 30%) from the control under NPCU (Fig. 2A–D).
Effect of caffeic acid (CA), cobalt sulfate (CoSO4), and nanoparticle coated urea (NPCU) on chlorophyll a (A), chlorophyll b (B), total chlorophyll (C) and carotenoid (D) concentration of leaves of potatoes. The Tukey test measured significant differences (p ≤ 0.05); distinct letters on the bars are the mean of four replicates ± SE.
Gas exchange attributes
By adding 20 μM CA, 0.15 mg/L CoSO4, and 20 μM CA + 0.15 mg/L CoSO4 treatment showed an increase in photosynthetic rate (~ 13%, ~ 54%, and ~ 32%), stomatal conductance (~ 37%, ~ 22%, and ~ 52%), transpiration rate (~ 29%, ~ 13%, and 41%), and intercellular CO2 concentration (~ 49%, ~ 42%, and ~ 99%) under no NP than the control. Applying 20 μM CA, 0.15 mg/L CoSO4, and 20 μM CA + 0.15 mg/L CoSO4 treatment with NPCU showed a maximum increase in photosynthetic rate (~ 14%, ~ 59%, and ~ 45%), stomatal conductance (~ 20%, ~ 11%, and ~ 27%), transpiration rate (~ 35%, ~ 23%, and ~ 42%), and intercellular CO2 concentration (~ 82%, ~ 31%, and ~ 97%) compared to the control (Fig. 3A–D).
Effect of caffeic acid (CA), cobalt sulfate (CoSO4), and nanoparticle coated urea (NPCU) on photosynthetic rate (A), stomatal conductance (B), transpiration rate (C) and intercellular CO2 rate (D) concentration of leaves of potatoes. The Tukey test measured significant differences (p ≤ 0.05); distinct letters on the bars are the mean of four replicates ± SE.
Total soluble protein, soluble sugar, and free amino acid
Contrast to the control, treatments 20 μM CA, 0.15 mg/L CoSO4, and 20 μM CA + 0.15 mg/L CoSO4 with no nanoparticle showed a rise in total soluble protein (~ 41%, ~ 21%, and ~ 62%), soluble sugar (~ 32%, ~ 18%, and ~ 53%), and free amino acid (~ 44%, ~ 20%, and ~ 69%). Applying of NPCU with 20 μM CA, 0.15 mg/L CoSO4, and 20 μM CA + 0.15 mg/L CoSO4 resulted an increase in total soluble protein (~ 22%, ~ 10%, and ~ 30%), soluble sugar (~ 22%, ~ 14%, and ~ 30%), and free amino acid (~ 26%, ~ 14%, and ~ 38%) over the control (Fig. 4A–C).
Effect of caffeic acid (CA), cobalt sulfate (CoSO4), and nanoparticle coated urea (NPCU) on total soluble sugar (A), soluble protein (B) and free amino acid (C) concentration of leaves of potatoes. The Tukey test measured significant differences (p ≤ 0.05); distinct letters on the bars are the mean of four replicates ± SE.
H2O2, MDA, and POD
A significant decrease in H2O2 content (~ 8%, 5%, and 13%), MDA (~ 15%, ~ 7%, and ~ 29%), and POD (~ 12%, ~ 7%, and ~ 24%) activity were observed by adding 20 μM CA, 0.15 mg/L CoSO4, and 20 μM CA + 0.15 mg/L CoSO4 treatment under no NPCU than the control. NPCU with 20 μM CA, 0.15 mg/L CoSO4, and 20 μM CA + 0.15 mg/L CoSO4 resulted significant decrease in H2O2 content (~ 27%, ~ 28%, and ~ 99%), MDA (~ 25%, ~ 15%, and ~ 64%), and POD (~ 13% ~ 9, and ~ 51%) activity than the control (Fig. 5A–C).
Effect of caffeic acid (CA), cobalt sulfate (CoSO4), and nanoparticle coated urea (NPCU) on H2O2 (A), MDA (B) and POD (C) concentration of leaves of potatoes. The Tukey test measured significant differences (p ≤ 0.05); distinct letters on the bars are the mean of four replicates ± SE.
SOD, CAT, and proline
By adding 20 μM CA, 0.15 mg/L CoSO4, and 20 μM CA + 0.15 mg/L CoSO4 treatment showed a decrease in SOD (~ 12%, 7%, and ~ 28%), CAT (~ 13%, ~ 6%, and ~ 32%), and proline (~ 20%, ~ 16%, and ~ 38%) under no NP from the control. Applying 20 μM CA, 0.15 mg/L CoSO4, and 20 μM CA + 0.15 mg/L CoSO4 treatment with NPCU showed a maximum decrease in SOD (~ 32%, ~ 18%, and ~ 68%), CAT (~ 34%, ~ 27%, and ~ 77%), and proline (~ 17%, ~ 6%, and ~ 21%) in contrast to the control (Fig. 6A–C).
Effect of caffeic acid (CA), cobalt sulfate (CoSO4), and nanoparticle coated urea (NPCU) on CAT (A), proline content (B) and SOD (C) concentration of leaves of potatoes. The Tukey test measured significant differences (p ≤ 0.05); distinct letters on the bars are the mean of four replicates ± SE.
N, P, K, and Ca in leaves
Adding 20 μM CA, 0.15 mg/L CoSO4, and 20 μM CA + 0.15 mg/L CoSO4 with no NP caused an increase in leave N (~ 2%, ~ 1%, and ~ 3%), leave P (~ 56%, ~ 26%, and ~ 97%), leave K (~ 12%, ~ 5%, and ~ 17%), and leave Ca (~ 6%, ~ 3%, and ~ 10%) compared to control. Increase in leaves N (~ 3%, ~ 2%, and ~ 3%), leave P (~ 25%, ~ 13%, and ~ 33%), leave K (~ 7%, ~ 7%, and ~ 21%), and leave Ca (~ 6%, ~ 4%, and ~ 12%) by applying 20 μM CA, 0.15 mg/L CoSO4, and 20 μM CA + 0.15 mg/L CoSO4 treatment with NPCU contrast to the control (Table 2).
The Tukey test measured significant differences (p ≤ 0.05); distinct letters on the bars are the mean of four replicates ± SE.
Convex hull and hierarchical cluster analysis
The control group demonstrates scores clustered around negative values on PC 1, with varying scores on PC 2, ranging from − 7.88126 to 1.62146. In contrast, the treatment group administered with 20 μM CA shows scores distributed across PC 1 and PC 2, ranging from − 3.99695 to 6.10181. Similarly, the treatment group exposed to 0.15 mg/L CoSo4 displays a spread of scores from -5.77306 to 4.31424. The combination treatment of 20 μM CA and 0.15 mg/L CoSo4 exhibits scores extending from − 2.17416 to 8.21149, showing a distinct pattern compared to individual treatments (Fig. 7A).
Convex Hull cluster plots for treatments (A), NP levels (B), and Hierarchical Cluster analysis (C) for the studied attributes.
With Principal Component (PC) 1 explaining the vast majority of variance at 96.76%, it emerges as the dominant factor shaping the dataset's structure. Meanwhile, PC 2 contributes a minor 1.16% to the overall variance. Different treatment response patterns are presented by examining the treatment scores and distinct clusters. Most data points cluster tightly under the label no NP, indicated by negative values on PC 1 and positive values on PC 2, suggesting a consistent treatment response within this subgroup. In contrast, another cluster labeled NPCU exhibits positive values on PC 1 and PC 2, indicating a distinct treatment response pattern (Fig. 7B).
The analysis reveals clusters of variables based on their similarities. For instance, variables such as total soluble sugar and free amino acids exhibit a similarity of 0.0816, suggesting a close association in their properties. Similarly, chlorophyll a and total chlorophyll are similar to 0.1404, indicating a related nature. Soluble protein and leaf dry weight, photosynthetic rate, and leaf dry weight form clusters with similarities of 0.14656 and 0.23607, respectively, implying shared characteristics within each cluster. Furthermore, the analysis highlights significant associations among variables such as shoot dry weight and leaf N percentage, which exhibit a high similarity of 0.25467, suggesting a strong relationship between these parameters. Stomatal conductance and transpiration rate also exhibit a substantial resemblance of 0.27344, showing their close correlation in plant physiology.
Interestingly, variables such as stem length and leaf K percentage display a similarity of 0.6761 and 0.71185, respectively, suggesting a significant association between these parameters. Additionally, the analysis identifies variables such as H2O2 and intercellular CO2 rate, which exhibit higher similarities of 1.83681 and 2.21367, respectively, indicating potential physiological relationships between these parameters. The analysis concludes with variables such as SOD and chlorophyll b, which show similarities of 3.65837 and 7.91511, respectively, suggesting distinct characteristics within the dataset (Fig. 7C).
Pearson correlation analysis
Variables like stem length, shoot, root, and leaf dry weight exhibit strong positive correlations ranging from 0.97256 to 0.98804, revealing their close connection with plant development and biomass accumulation. Furthermore, chlorophyll-related parameters, including chlorophyll a, b, total, and carotenoid content, demonstrate high positive correlations, ranging from 0.83071 to 0.99724, highlighting the interconnectedness of these pigment-related processes in photosynthesis and plant metabolism. The photosynthetic rate, stomatal conductance, and transpiration rate also exhibit strong positive correlations with values ranging from 0.94456 to 0.99551, suggesting their synchronized roles in gas exchange and carbon assimilation within plant tissues. Conversely, oxidative stress-related parameters such as H2O2, MDA, POD, SOD, and CAT demonstrate adverse relationships with other variables, representing their inverse relationships. For instance, H2O2 and MDA exhibit negative correlations with most variables, suggesting their roles as indicators of oxidative stress levels in plants. Additionally, parameters related to leaf N, P, and K percentages demonstrate positive correlations, ranging from 0.81827 to 0.99337, reflecting their interconnected roles in plant nutrient uptake and metabolism (Fig. 8).
Pearson correlation analysis for the studied attributes.
Discussion
Caffeic acid
Caffeic acid has been shown to improve nitrogen uptake and utilization in plants, particularly under stress conditions. It can stimulate net nitrogen uptake at low concentrations40 and mitigate the adverse effects of abiotic stresses in plants by enhancing improving antioxidant activity41. Improving nitrogen use efficiency through altered amino acid transport processes can lead to increased biomass and seed yields, even in nitrogen-poor soils42. Plants can directly absorb amino acids, bypassing the mineralization step and potentially accelerating nitrogen turnover43. This ability varies among species, with deciduous shrubs and ectomycorrhizal species generally showing higher amino acid uptake rates43. These findings suggest that caffeic acid and related compounds play a crucial role in enhancing plant nitrogen uptake and overall stress tolerance. Hierarchical cluster analysis in current study also in agreement of above arguments. It clearly showed that free amino acids improvement in current study was the most representable attribute that played a crucial role in improvement of potato growth in N deficient soil.
Cobalt
Cobalt plays a crucial role in enhancing nitrogen uptake and improving plant growth across various crops. Studies on pea, cowpea, and groundnut have shown that cobalt application significantly increases nodule formation, nitrogenase activity, and nitrogen fixation efficiency11,44,45. Cobalt supplementation allows for a 25% reduction in nitrogen fertilizer use while maintaining or improving crop yields and quality11,44,45. The element enhances the uptake of macronutrients (N, P, K) and micronutrients (Mn, Zn), although it may reduce iron content due to antagonistic relationships46. Cobalt application results in increased plant growth parameters, improved pod and seed yields, and enhanced chemical composition of crops46,47. These findings suggest that cobalt supplementation can optimize nitrogen fertilization efficiency, reduce environmental pollution from excess nitrogen, and potentially lower agricultural costs for farmers.
Nanoparticle-coated urea
Nanoparticles can boost photosynthetic rate, biomass, chlorophyll content, and upregulate stress-related genes in plants48. Nanoparticle-coated urea has shown promising results in improving nitrogen uptake and enhancing crop performance. Fe3O4-urea nanocomposites significantly increased photosynthetic efficiency, nitrogen metabolism, and crop yield in rice49. Similarly, ZnO nanoparticle-coated urea improved wheat performance under drought stress, enhancing grain yield and zinc uptake50. Zinc oxide nanoparticle-coated urea also demonstrated superior results in terms of slow nutrient release, nitrogen and zinc uptake, and grain yield in wheat compared to uncoated urea51.
Conclusion
In conclusion, treatment 20 μM CA + 0.15 mg/L CoSO4 with NPCU can improve potato growth in N deficient soil. Application of 20 μM CA + 0.15 mg/L CoSO4 with NPCU can improve free amino acids, chlorophyll content, and uptake of macronutrients (i.e., N, P and K) that act as key factors for potato growth enhancement. Growers are recommended to apply 20 μM CA + 0.15 mg/L CoSO4 with NPCU for better growth and uptake of nutrients in potato in N deficient soils compared to simple urea application. More investigations are recommended at field levels to declare 20 μM CA + 0.15 mg/L CoSO4 with NPCU as most efficacious amendment for improvement of potato growth in different agro climates soils.
Data availability
All data generated or analysed during this study are included in this published article.
References
Kumar, S. Abiotic stresses and their effects on plant growth, yield and nutritional quality of agricultural produce. Int. J. Food Sci. Agric 4, 367–378 (2020).
Ameen, M. et al. Nutrient acquisition, transport and metabolism within the plant cells. In Sustainable plant nutrition 51–70 (Elsevier, 2023).
Lommen, W. J. M. Environmental conditions after planting affect the expression of differences in tuber formation of in vitro-derived potato plantlets from different ages and cultivars. Potato Res. 67(1), 117–137 (2023).
Bethke, P. C. & Jansky, S. H. Genetic and environmental factors contributing to reproductive success and failure in potato. Am. J. Potato Res. 98, 24–41 https://doi.org/10.1007/s12230-020-09810-3 (2021).
Zarzyńska, K., Trawczyński, C. & Pietraszko, M. Environmental and agronomical factors limiting differences in potato yielding between organic and conventional production system. Agriculture 13(4), 901 (2023).
Saeed, R. et al. Tea [Camellia sinensis (L.) kuntze] leaf compost ameliorates the adverse effects of salinity on growth of cluster beans (Cyamopsis tetragonoloba L.). Pak. J. Bot. 48, 495–501 (2016).
Zafar-ul-Hye, M. et al. Rhizobacteria inoculation and caffeic acid alleviated drought stress in lentil plants. Sustainability 13, 9603 (2021).
El-Awadi, M. E. Investigations of growth promoting activity of some phenolic acids. Agric. Eng. Int. CIGR J. 19, 53–60 (2017).
Bubna, G. A. et al. Exogenous caffeic acid inhibits the growth and enhances the lignification of the roots of soybean (Glycine max). J. Plant Physiol. 168, 1627–1633 (2011).
Vendrig, J. C. & Buffel, K. Growth-promoting activity of caffeic acid. Nature 193, 1204–1204 (1962).
Gad, N. Increasing the efficiency of nitrogen fertilization through cobalt application to pea plant. Res. J. Agric. Biol. Sci. 2, 433–442 (2007).
Siam, H. S., Mahmoud, A. & Abdel Hady, M. Increasing the utilization efficiency of nitrogenous fertilizers by cobalt addition and its effect on growth, yield parameters and nutrients contents of cowpea plants. J. Appl. Sci. Res. 8(2), 915-929 (2012).
Kandil, H. M., Farid, I. M. & el-Maghraby, A. M. Effect of cobalt level and nitrogen source on quantity and quality of soybean plant. J. Appl. Sci. Res. 8(2), 915–929 (2013).
Hu, X., Wei, X., Ling, J. & Chen, J. Cobalt: an essential micronutrient for plant growth? Front Plant. Sci. 12, 768523 (2021).
Xin, X., Judy, J. D., Sumerlin, B. B. & He, Z. Nano-enabled agriculture: from nanoparticles to smart nanodelivery systems. Environ. Chem. 17, 413 (2020).
Amin, S. et al. Zinc oxide nanoparticles coated urea enhances nitrogen efficiency and zinc bioavailability in wheat in alkaline calcareous soils. Environ. Sci. Pollut. Res. 30, 70121–70130 (2023).
Elhassani, C. E. et al. Lignin nanoparticles filled chitosan/polyvinyl alcohol polymer blend as a coating material of urea with a slow‐release property. J. Appl. Polym. Sci. 140, (2023).
Shen, Y., Zhou, J. & Du, C. Development of a polyacrylate/silica nanoparticle hybrid emulsion for delaying nutrient release in coated controlled-release urea. Coatings 9, 88 (2019).
Beig, B. et al. Development and testing of zinc sulfate and zinc oxide nanoparticle-coated urea fertilizer to improve N and Zn use efficiency. Front. Plant Sci. 13, (2023).
Pramanik, A. et al. Nanocatalyst-based biofuel generation: an update challenges and future possibilities. Sustainability 15, 6180 (2023).
De Vrieze, M., Germanier, F., Vuille, N. & Weisskopf, L. Combining different potato-associated pseudomonas strains for improved biocontrol of phytophthora infestans. Front. Microbiol. 9, 2573 (2018).
Chourasia, K. N. et al. Salinity stress in potato: understanding physiological, biochemical and molecular responses. Life 11, 545 (2021).
Klimczyk, M., Siczek, A. & Schimmelpfennig, L. Improving the efficiency of urea-based fertilization leading to reduction in ammonia emission. Sci. Total Environ. 771, 145483 (2021).
Arnon, D. I. Copper enzymes in isolated chloroplasts. Polyphenoloxidase in Beta vulgaris. Plant Physiol. 24, 1 (1949).
Kirk, J. T. O. & Allen, R. L. Dependence of chloroplast pigment synthesis on protein synthesis: Effect of actidione. Biochem. Biophys. Res. Commun. 21, 523–530 (1965).
Nazar, R., Khan, M. I. R., Iqbal, N., Masood, A. & Khan, N. A. Involvement of ethylene in reversal of salt-inhibited photosynthesis by sulfur in mustard. Physiol. Plant. 152, 331–344 (2014).
Yemm, E. W. & Willis, A J. The estimation of carbohydrates in plant extracts by anthrone. Biochem. J. 57, 508–514 (1954).
Bradford, M. M. A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal. Biochem. 72, 248–254 (1976).
Bates, L. S., Waldren, R. P. & Teare, I. D. Rapid determination of free proline for water-stress studies. Plant Soil 39, 205–207 (1973).
Hori, M. et al. Changes in the hepatic glutathione peroxidase redox system produced by coplanar polychlorinated biphenyls in Ah-responsive and-less-responsive strains of mice: Mechanism and implications for toxicity. Environ. Toxicol. Pharmacol. 3, 267–275 (1997).
Dhindsa, R. S., Plumb-Dhindsa, P. L. & Reid, D. M. Leaf senescence and lipid peroxidation: Effects of some phytohormones, and scavengers of free radicals and singlet oxygen. Physiol. Plant 56, 453–457 (1982).
Cakmak, I. & Horst, W. J. Effect of aluminium on lipid peroxidation, superoxide dismutase, catalase, and peroxidase activities in root tips of soybean (Glycine max). Physiol. Plant 83, 463–468 (1991).
Mills, H. A. & Jones, J. B. Jr. Plant Analysis Handbook II: A Practical Sampling, Preparation, Analysis, and Interpretation Guide. Plant analysis handbook. A practical sampling, preparation, analysis, and interpretation guide. (Micro-Macro Publishing, Inc., USA, 1991).
Miller, R. Nitric-perchloric acid wet digestion in an open vessel. In Handbook of Reference Methods for Plant Analysis (ed. Kalra, Y.) 57–62 (CRC Press, Washington, D.C., 1997). https://doi.org/10.1201/9781420049398.ch6.
Estefan, G., Sommer, R. & Ryan, J. Methods of Soil, Plant , and Water Analysis : A Manual for the West Asia and North Africa Region. (International Center for Agricultural Research in the Dry Areas (ICARDA), Beirut, Lebanon, 2013).
Donald, A. H. & Hanson, D. Determination of potassium and sodium by flame emmision spectrophotometery. In Handbook of Reference Methods for Plant Analysis (ed. Kalra, Y.) 153–155 (CRC Press, Washington, D.C., 1998).
Pratt, P. F. Potassium. in Methods of Soil Analysis, Part 2: Chemical and Microbiological Properties (ed. Norman, A. G.) 1022–1030 (John Wiley & Sons, Ltd, Madison, WI, USA, 2016). https://doi.org/10.2134/agronmonogr9.2.c20.
Steel, R. G., Torrie, J. H. & Dickey, D. A. Principles and Procedures of Statistics: A Biometrical Approach (McGraw Hill Book International Co., 1997).
OriginLab Corporation. OriginPro. (OriginLab, Northampton, MA, USA., 2021).
Abenavoli, M. R., Lupini, A., Oliva, S. & Sorgonà, A. Allelochemical effects on net nitrate uptake and plasma membrane H+-ATPase activity in maize seedlings. Biol. Plant 54, 149–153 (2010).
Zafar-ul-Hye, M., Nawaz, M. S., Asghar, H., Waqas, M. & Mahmood, F. Caffeic acid helps to mitigate adverse effects of soil salinity and other abiotic stresses in legume. J. Genet. Genomes 4, 1-6 (2020).
Perchlik, M. & Tegeder, M. Improving plant nitrogen use efficiency through alteration of amino acid transport processes. Plant Physiol. 175, 235–247 (2017).
Kielland, K. Amino acid absorption by arctic plants: implications for plant nutrition and nitrogen cycling. Ecology 75, 2373–2383 (1994).
Gad, N. Interactive effect of salinity and cobalt on tomato plants. II. Some physiological parameters as affected by cobalt and salinity. Res. J. Agric. Biol. Sci. 1, 270–276 (2005).
Gad, N., Moez, A. & Kandil, H. Response of Okra (Hibiscus esculentus) growth and productivity to cobalt and humic acid rates. Int. J. Chem. Tech. Res. 8, 1782–1791 (2015).
Gad, N. Role and importance of cobalt nutrition on groundnut (Arachis hypogaea) production. World Appl. Sci. J. 20(3), 359–367 (2012).
Gad, N. Role of cobalt on cowpea growth and yield under different levels of nitrogen. World Appl. Sci. J. 22(4), 470-478 (2013).
Singh, A., Tiwari, S., Pandey, J., Lata, C. & Singh, I. K. Role of nanoparticles in crop improvement and abiotic stress management. J. Biotechnol. 337, 57–70 (2021).
Guha, T., Gopal, G., Mukherjee, A. & Kundu, R. Fe3O4-urea nanocomposites as a novel nitrogen fertilizer for improving nutrient utilization efficiency and reducing environmental pollution. Environ. Pollut. 292, 118301 (2022).
Dimkpa, C. O. et al. Facile coating of urea with low-dose ZnO nanoparticles promotes wheat performance and enhances Zn uptake under drought stress. Front. Plant. Sci. 11, 168 (2020).
Beig, B. et al. Development and testing of zinc sulfate and zinc oxide nanoparticle-coated urea fertilizer to improve N and Zn use efficiency. Front. Plant Sci. 13, 1058219 (2023).
Acknowledgements
This project was supported by Researchers Supporting Project number (RSP2025R230) King Saud University, Riyadh, Saudi Arabia.
Funding
This project was supported by Researchers Supporting Project number (RSP2025R230) King Saud University, Riyadh, Saudi Arabia.
Author information
Authors and Affiliations
Contributions
Conceptualization; M.H.; S.D.; N.T.; F.G.; Conducted experiment; S.D.; M.H.; M.I.; Formal analysis; M.H.; M.I.; Methodology; N.A.; R.D.; N.T.; F.G.; Writing—original draft; M.J.A.; S.D.; S.A.A.; T.A.A.; Writing—review & editing; M.J.A.; R.D.; S.A.A.; T.A.A.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethics approval and consent to participate
We all declare that manuscript reporting studies do not involve any human participants, human data, or human tissue. So, it is not applicable. Experimental research and field studies on plants (either cultivated or wild), including the collection of plant material, must comply with relevant institutional, national, and international guidelines and legislation. We confirmed that all methods were performed in accordance with the relevant guidelines/regulations/legislation. The seeds were purchased from a local certified seed dealer of the Government of Punjab, Pakistan.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Danish, S., Hareem, M., Imran, M. et al. Effect of caffeic acid and cobalt sulfate on potato (Solanum tuberosum L.) plants in the presence and absence of nanoparticles-coated urea. Sci Rep 14, 20663 (2024). https://doi.org/10.1038/s41598-024-70998-z
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-024-70998-z