Abstract
To develop a simple scoring system based on baseline inflammatory and nutritional markers to predict the long-term prognosis of patients with nasopharyngeal carcinoma (NPC). Conducted a retrospective analysis of clinical data from 1024 newly diagnosed non-metastatic NPC patients. A total of 15 pre-treatment inflammatory and nutritional markers were collected as candidate variables. Receiver operating characteristic (ROC) curves were used to determine the optimal cutoff points for each parameter. Survival analysis was performed using Kaplan–Meier method and Cox regression analysis. Besides, the Inflammation Nutrition Risk Score (INRS) was calculated for each patient by assigning each independent prognostic factor a score of 1. Multivariate Cox regression analysis showed that serum albumin (ALB), systemic immune-inflammation index, and monocyte count (M) were independent prognostic factors for OS (P < 0.05). Survival analysis showed that higher INRS was associated with a worsened prognosis. Patients in the high-risk group had shorter OS than in the low-risk group. In the training group, the 3-, 5-, and 8-years OS rates for the low-risk group versus high-risk group were 92.5% versus 87.8%, 87.4% versus 75.1%, and 84.6% versus 62.2%, respectively (P < 0.05). In the validation group, the 3-, 5-, and 8-years OS rates for the low-risk group vs. high-risk group were 95.0% versus 86.4%, 92.1% versus 82.2%, and 89.5% versus 74.3%, respectively (P < 0.05). Further subgroup analysis showed a significant difference in the OS between the high-risk group and low-risk group in patients with locally advanced disease (P < 0.05). The ROC curve demonstrated that INRS had a similar predictive value for long-term survival in NPC patients compared to TNM staging and serum EBV-DNA levels. Pretreatment ALB, M, and SIRI are independent prognostic factors for long-term survival in patients with NPC. INRS constructed based on these three factors can serve as a long-term prognostic indicator for NPC.
Similar content being viewed by others
Introduction
Nasopharyngeal carcinoma (NPC) originates from the epithelial cells of the nasopharynx and most commonly occurs in the pharyngeal recess and roof of the nasopharynx. Previous data indicate that in recent years, the incidence and mortality rates of nasopharyngeal carcinoma (NPC) in global have been significant. According to the latest statistics, an estimated 120,416 people were diagnosed with NPC, and 73,476 died from the disease1. Currently, comprehensive radiotherapy-based treatment is the main therapy for patients with NPC2.
The American Joint Committee on Cancer (AJCC) TNM staging system and serum EBV-DNA levels are important indicators for predicting NPC3,4,5. However, the TNM staging primarily reflects the anatomical extent of tumor invasion and does not consider variations in the overall systemic function among different patients6. Approximately 40% of patients with NPC have negative serum EBV-DNA levels before treatment, and the normal reference values for serum EBV-DNA levels vary among different medical centers7. Therefore, clinicians require reliable biomarkers or evaluation indicators to assist in clinical diagnosis and treatment decision-making.
In recent years, researchers have focused on the effects of nutrition and systemic inflammation on the prognosis of patients with cancer8. Previous studies have confirmed that systemic inflammation and nutritional status have good predictive value for short-term disease control in NPC9. Li et al.10 found that inflammatory and nutritional indicators can be used to evaluate short-term disease control rates in patients with newly diagnosed metastatic NPC. Systemic inflammatory response has also been shown to be closely associated with weight loss and malnutrition in patients with NPC. However, the effects of systemic inflammation and nutritional status on long-term survival outcomes remain unclear. More proactive or tailored treatment approaches may be necessary for patients with poor levels of inflammation and nutritional indicators.
Therefore, this study aimed to establish a prognostic scoring system based on inflammatory and nutritional indicators in patients newly diagnosed with non-metastatic NPC. Our study introduced a novel Inflammation Nutrition Risk Score (INRS) based on baseline inflammatory and nutritional markers to predict the long-term prognosis of NPC patients. This scoring system was unique in its simplicity and accessibility, offering a new method for risk stratification that could complement existing prognostic indicators. The significance of our research lied in its potential to enhance personalized treatment strategies. By identifying serum albumin, systemic immune-inflammation index (SII), and monocyte count as independent prognostic factors, our study provided a comprehensive assessment that could lead to improved patient outcomes. The INRS has shown predictive value comparable to established tools, underscoring its potential clinical utility.
Methods and materials
Patients
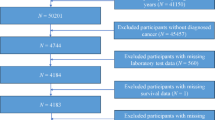
Among 1068 patients with NPC who sought treatment at our research center between January 2012 and December 2013, 1024 eligible subjects were included in the analysis. The main inclusion criteria were as follows: (1) treatment received at our research center between January 2012 and December 2013; (2) newly diagnosed primary NPC confirmed by pathology; (3) absence of distant metastasis at the time of diagnosis, stage I to IVA; (4) availability of complete clinical, laboratory, and follow-up data, with all treatment plans completed at our research center; (5) age between 11 and 82 years. The exclusion criteria were as follows: (1) patients who refused or did not complete the treatment, (2) patients lost to follow-up, and (3) incomplete relevant clinical or laboratory data. In our hospital, EBV-DNA levels were measured using the polymerase chain reaction (PCR) technique, and the results were expressed in copies/ml. All patients were reclassified according to the eighth edition of the AJCC on Cancer Staging System. This retrospective study was conducted in accordance with the ethical principles outlined in the Declaration of Helsinki and approved by the ethics committee of our center. The committee waived the requirement for individual informed consent because the retrospective nature of the study and the use of anonymized data.
Inflammatory and nutritional indicators
There were 15 variables included in the analysis: white blood cell count (WBC), neutrophil count (N), lymphocyte count (L), monocyte count (M), platelet count (PLT), serum hemoglobin level (HB), serum albumin level (ALB), platelet-to-lymphocyte ratio (PLR), neutrophil-to-lymphocyte ratio (NLR), systemic immune-inflammation index (SII), systemic inflammation response index (SIRI), platelet-to-albumin ratio (PAR), body mass index (BMI), nutritional risk index (NRI), and prognostic nutritional index (PNI). All laboratory data used in this study were collected at the time of the patients' first medical visit. This baseline data was crucial for assessing the initial inflammatory and nutritional status of the patients before the commencement of any treatment.
PLR was calculated by dividing the platelet count by the lymphocyte count. NLR was calculated by dividing the neutrophil count by the lymphocyte count. The SII was calculated as PLT (*109/L) * N (*109/L)/L (*109/L). The SIRI was calculated as N (*109/L) * M (*109/L)/L (*109/L). PAR is calculated by dividing the platelet count by the serum albumin level. BMI was calculated by dividing the patient's pretreatment weight (kg) by the square of height (m). The NRI was calculated as 1.489 × ALB (g/L) + 41.7 × (pretreatment weight/ideal weight) (ideal weight calculated as the square of the height (m) multiplied by 22.5). The PNI was calculated as ALB (g/L) + 5 × L (× 10^9/L). Besides, the Inflammation Nutrition Risk Score (INRS) was calculated for each patient by assigning each independent prognostic factor a score of 1.
Determination of optimal cutoff points
To determine the optimal cutoff points for the pre-treatment inflammatory and nutritional markers, we utilized the Receiver Operating Characteristic (ROC) curve analysis. We generated ROC curves for each of the 15 pre-treatment inflammatory and nutritional markers. The ROC curve plots the true positive rate (sensitivity) against the false positive rate (1-specificity) at various threshold settings. We employed Youden’s index to determine the optimal cutoff point for each marker. Youden’s index is defined as: Youden’s Index = Sensitivity + Specificity − 1. The optimal cutoff point is the value that maximizes Youden’s index, which represents the point on the ROC curve that balances sensitivity and specificity, providing the best overall diagnostic effectiveness. For each marker, we calculated Youden’s index at different threshold values. The threshold value with the highest Youden’s index was selected as the optimal cutoff point for that marker. The ROC curves aimed to predict long-term overall survival (OS) at specified time points. By identifying the optimal cutoff values for each marker, we were able to classify patients into high-risk and low-risk groups, thereby providing a robust prognostic tool for long-term outcomes in NPC.
Treatment methods
Among the patients in this study, those in stage I received radiotherapy alone, whereas those in stages II–IVA underwent comprehensive treatment combining radiotherapy and chemotherapy.
All chemotherapy regimens were administered in accordance with the previously described protocol. T1-4N1-3M0 patients received two cycles of platinum-based concurrent chemotherapy during radiotherapy, and patients with stage III–IVA cancer received 2–4 cycles of neoadjuvant chemotherapy before radiotherapy. Neoadjuvant chemotherapy regimens included TP (paclitaxel plus cisplatin, paclitaxel: 135–175 mg/m2 on day 1, Cisplatin: Total dose of 80 mg/m2, administered on days 1–3 or day 2) or GP (gemcitabine plus cisplatin, Gemcitabine: Total dose of 1000 mg/m2, administered on days 1 and 8, Cisplatin: 80 mg/m2 on days 1–3 or day 2).
All patients underwent intensity-modulated radiotherapy (IMRT) according to our center's predefined treatment plan11. Clinical target volume 1 (CTV-1) was defined as the high-risk area, including the gross tumor volume (GTV) and 5 mm margin around the nasopharyngeal mucosa. Clinical target volume 2 (CTV-2) was designed for potentially involved regions, such as the nasopharynx, maxillary sinus, pterygopalatine fossa, skull base, posterior ethmoid sinus, and sphenoid sinus. The prescribed dose was 70 Gy, delivered in daily fractions of 2.0–2.25 Gy over 31–35 treatment sessions. Radiotherapy was administered five times a week, once daily, from Monday to Friday.
Follow-up and clinical endpoints
The follow-up time was defined as the interval from the diagnosis of NPC to the last follow-up or death. Outpatient re-examinations were conducted every 3 months within the first 2 years after treatment. From the 3rd to the 5th year after treatment, outpatient re-examinations were conducted every 6 months. After 5 years post-treatment, outpatient re-examinations were conducted annually. The primary endpoint of this study was OS, defined as the time interval from diagnosis of NPC to death from any cause.
Statistical analysis
A total of 1024 patients with newly diagnosed non-metastatic NPC were included in the study and randomly allocated in a 2:1 ratio to the training (n = 675) or validation (n = 349) groups using a computer-generated randomization schedule. The randomization process was conducted by an independent statistician to prevent any potential bias. To ensure balanced distribution of clinicopathological factors, stratified randomization was employed, with stratification factors12.
ROC curves were used to determine the optimal cutoff points for the 15 inflammatory and nutritional variable indicators. The area under the curve (AUC) was calculated for each ROC curve. We compared the AUCs of the different predictive models using the DeLong test, which is a non-parametric approach for comparing the areas under two or more correlated ROC curves. The DeLong test evaluates whether the differences between the AUCs are statistically significant. The accuracy of the INRS, TNM staging, and serum EBV-DNA level in predicting long-term survival in patients with newly diagnosed non-metastatic NPC was assessed using the area under the ROC curve (AUC value). Survival outcomes were evaluated using the log-rank test and Cox regression model. All statistical analyses were performed using the SPSS (IBM version 26.0) and R software (version 4.2.1). We have obtained a copyright license of SPSS statistical software. In the statistical analysis, a p value < 0.05 was considered statistically significant.
Result
Patient characteristics and outcomes
A total of 1024 eligible patients with a median age of 49 years (range: 11–82 years) were included in the analysis. The enrolled patients were randomly allocated in a 2:1 ratio to the training (n = 675) and validation (n = 349) groups. No significant differences were observed between the training and validation groups regarding baseline characteristics, including sex, age, histological type, and TNM staging (Table 1).
The median follow-up time of the included patients was 99 months (range: 14–119 months), and the median survival time was 98 months (range: 14–119 months). No significant difference was observed in the median survival time between the training and validation groups (99 vs. 98 months, p = 0.34). The 3-, 5-, and 8-years OS rates were 92.8%, 87.2%, and 82.9%, respectively (Fig. 1A). The 3-, 5-, and 8-years OS rates in the training group were 92.5%, 85.6%, and 81.6%, respectively (Fig. 1B), whereas those in the validation group were 92.8%, 89.7%, and 86.1%, respectively (Fig. 1C).
OS curves of enrolled patients: (A) Total (n = 1024); (B) training group (n = 675); (C) validation group (n = 349). (The images were created using R version 4.2.1 (https://www.r-project.org)).
Development of risk-scoring system
We employed the method of drawing ROC curves and selecting the optimal threshold point on the curves to stratify the risk based on the selected 15 variables. The calculated cutoff values for WBC, N, L, M, PLT, HB, ALB, NLR, PLR, SII, SIRI, BMI, PNI, NRI, and PAR were as follows: 6.3 (× 109/L), 2.1 (× 109/L), 2.3 (× 109/L), 0.49 (× 109/L), 136 (× 109/L), 111 (g/L), 39.7 (g/L), 2.21, 226.15, 1040, 1.2, 21.7, 56.9, 105.3, and 3.58. Univariate analysis of the training group revealed significant correlations between WBC, M, ALB, NLR, SII, SIRI, and PLR with OS (Table 2, all P values < 0.05). Further multivariate analysis revealed that ALB, M, and SII were independent prognostic factors for OS (Table 3, all P values < 0.05).
The Inflammation Nutrition Risk Score (INRS) was calculated for each patient by assigning each independent prognostic factor a score of 1, resulting in a score sum ranging from 0 to 3. The specific cutoff values were as follows: ALB: < 39.7 g/L, M: > 0.49 × 109/L, and SII: > 1040. Subsequently, based on the total risk score, the patients were classified into low group (0–1 points) and high-risk group (2–3 points) groups (Supplementary Tables 1 and 2).
The following six factors, considered significant predictors of prognosis, were included in further multivariate analyses along with INRS: sex, age, T stage, N stage, TNM stage, and pretreatment serum EBV-DNA levels. Multivariate Cox regression analysis demonstrated that INRS, TNM stage, and age were independent prognostic factors for OS in the training group (Table 4). In the validation group, the INRS, TNM stage, age, and sex were independent prognostic factors for OS (Table 5).
Predictive performance of INRS
The survival curves of patients with different INRS scores in the training and validation groups are shown in Fig. 2A,B. In both the validation and training groups, higher INRS scores were associated with lower OS rates.
OS curves of patients with inflammatory nutritional risk score 0–3 points: (A) training group (n = 675); (B) validation group(n = 349). (The images were created using R version 4.2.1 (https://www.r-project.org)).
Patients in the high-risk group had shorter OS than those in the low-risk group. In the training group, the 3-, 5-, and 8-years OS rates were 92.5% versus 87.8%, 87.4% versus 75.1%, and 84.6% versus 62.2% for the low-risk and high-risk groups, respectively (all P < 0.005; Supplementary Fig. 1A). In the validation group, the 3-, 5-, and 8-years OS rates were 95.0% versus 86.4%, 92.1% versus 82.2%, and 89.5% versus 74.3% for the low-risk and high-risk groups, respectively (all P < 0.005; Supplementary Fig. 1B).
Similarly, significant differences in OS were observed between the high-risk and low-risk groups of patients with advanced-stage NPC. In the training group, the 3-, 5-, and 8-years OS rates for the low-risk group were 92.9%, 87.0%, and 84.5%, respectively, compared with 86.3%, 74.0%, and 61.5% for the high-risk group (P < 0.05) (Fig. 3A). In the validation group, the 3-, 5-, and 8-years OS rates for the low-risk group were 94.0%, 91.3%, and 88.7%, respectively, compared to 83.9%, 80.8%, and 72.4% in the high-risk group (P < 0.05) (Fig. 3B).
OS curves of advanced-stage patients in high-risk group and low-risk group: (A) training group (n = 609); (B) validation group (n = 318). (The images were created using R version 4.2.1 (https://www.r-project.org)).
The comparison of the prognostic value
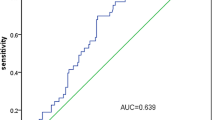
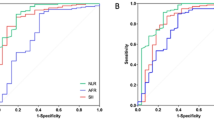
The predictive abilities of the INRS, TNM staging, and serum EBV-DNA levels in determining the long-term survival rates of patients with NPC were assessed using ROC curves. In the training group, the AUC values for predicting 5- and 8-years survival rates using the INRS, TNM staging, and serum EBV-DNA levels were 0.60 versus 0.63 versus 0.57, respectively (all pairwise comparisons, P = 0.270 for INRS vs TNM staging, P = 0.088 for INRS vs serum EBV-DNA levels, P = 0.003 for TNM staging vs serum EBV-DNA levels) (Fig. 4A) and 0.65 vs. 0.62 versus 0.56, respectively (all pairwise comparisons, P = 0.659 for INRS versus TNM staging, P = 0.027 for INRS vs serum EBV-DNA levels, P = 0.065 for TNM staging vs serum EBV-DNA levels) (Fig. 4B). In the validation group, the AUC values for predicting 5- and 8-years survival rates using the INRS, TNM staging, and serum EBV-DNA levels were 0.67 versus 0.61 versus 0.54, respectively (all pairwise comparisons, P = 0.185 for INRS vs TNM staging, P = 0.446 for INRS versus serum EBV-DNA levels, P = 0.033 for TNM staging vs serum EBV-DNA levels) (Fig. 4C) and 0.66 versus 0.60 versus 0.54, respectively (all pairwise comparisons, P = 0.066 for INRS versus TNM staging, P = 0.279 for INRS versus serum EBV-DNA levels, P = 0.407 for TNM staging versus serum EBV-DNA levels) (Fig. 4D). These results demonstrate that the ability of INRS to predict long-term survival rates in patients with NPC is comparable to that of TNM staging and serum EBV-DNA levels.
ROC curve, long-term prognostic value of inflammatory nutrition score, TNM stage, and serum EBV-DNA level before treatment: (A) 5-years OS in the training group; (B) 8-years OS in the training group; (C) 5-years OS in the validation group; (D) 8-years OS in the validation group. (The images were created using R version 4.2.1 (https://www.r-project.org)).
Discussion
The present study identified ALB, SII, and M as independent prognostic factors for OS in patients with NPC. Our findings suggested that the INRS, which incorporates these factors, can serve as a valuable prognostic tool for long-term survival. The INRS demonstrated comparable predictive value to traditional indicators such as TNM staging and serum EBV-DNA levels, thereby offering a complementary approach to existing prognostic assessments. Disease-related malnutrition is a common syndrome among hospitalized patients with a prevalence of up to 30%. Among patients with cancer, the prevalence of disease-related malnutrition can reach 40%13. Li et al.14 found that in a population of patients with NPC prior to treatment, the proportions of malnutrition assessed by BMI and ALB were 36.5% and 34.6%, respectively. Similarly, compared with the general population or the non-tumor disease population, patients with NPC are more prone to abnormal inflammatory markers. A study by Zeng et al. followed up and compared 255 patients with NPC with 500 patients with sinusitis and found significant differences in most pretreatment inflammatory markers between the two groups. PLT, L, and WBC counts are independent prognostic factors of OS in NPC15. Our results were consistent with these findings. After conducting an INRS for NPC, we found that most patients in both the training and validation groups had abnormal nutritional and inflammatory status before treatment (risk score of 1 or above) (68.5% and 72.2%, respectively). It is worth mentioning that the correlation analysis suggested a weak correlation between the inflammation-nutrition status and TNM staging before treatment (correlation coefficients of 0.24 and 0.22, respectively, P < 0.05). Patients with more advanced TNM stages tended to have poorer inflammation-nutritional status, which aligns with our clinical expectations.
An increasing number of studies have focused on the relationships among inflammation, nutritional status, treatment efficacy, and prognosis in patients with cancer. Multiple studies have shown that assessing the nutritional and inflammatory statuses of patients with cancer may reflect their true tumor status and predict treatment efficacy. Patients with systemic inflammation and malnutrition represent an underestimated subgroup that may require more personalized comprehensive treatment8,9,16. Our results were consistent with these findings. In the training group, we found significant correlations (P < 0.05) between baseline inflammatory and nutrition-related variables, such as WBC, M, PNI, ALB, NLR, SII, SIRI, PLR, and prognosis. Further multivariate analysis revealed that baseline ALB, SII, and M levels were independent prognostic factors for long-term OS in patients with newly diagnosed non-metastatic NPC.
It is well known that ALB is widely used for nutritional assessment in patients with cancer. Recently, Evans et al.17 proposed that hypoalbuminemia should be considered a chronic disease characterized by inflammation and closely associated with adverse outcomes. Low ALB levels disrupt metabolic function, impair immunity, increase susceptibility to infection, compromise the response to tumor treatment, and exacerbate the toxicity of radiotherapy and chemotherapy, severely affecting survival and outcomes18,19. Our study validated these conclusions in a population of patients with newly diagnosed non-metastatic NPC. In the validation group, patients with high inflammation-nutrition risk scores based on serum ALB, SII, and M levels had a 1.64-fold, 1.72-fold, and 1.60-fold higher risk of death, respectively, than patients with low scores. Therefore, controlling systemic inflammation is particularly important for malnourished patients with cancer, and early nutritional intervention should be emphasized for patients with cancer with high levels of inflammation to improve their prognosis. Based on the risk stratification using the INRS, specific treatment strategies can be applied to high-risk patients to improve their prognostic outcomes. High-risk patients, characterized by poor levels of inflammation and nutritional indicators, may benefit from intensified chemotherapy regimens, integration of immunotherapy, proactive nutritional support, close monitoring, and a multidisciplinary care approach. These tailored treatment strategies aim to address the unique challenges faced by high-risk patients, potentially enhancing their survival and quality of life.
To accurately predict the long-term prognosis of patients with NPC, we constructed an INRS based on the independent prognostic factors for OS (ALB, SIRI, and M). Kaplan–Meier curve analysis demonstrated that INRS could effectively stratify patients into different risk groups. By comparing the area under the ROC curve, the INRS showed no significant difference in predicting long-term survival capability compared to the TNM staging system and pretreatment serum EBV-DNA levels. Therefore, it can be considered a long-term prognostic indicator of NPC. These findings were consistent with those of previous studies. A prospective cohort study by EMC et al. involving 2470 patients with stage I to III colorectal cancer demonstrated that pre-diagnosis inflammation was associated with treatment-induced sarcopenia and that the sarcopenia and high NLR groups had worse OS16. Muscle atrophy can lead to metabolic and endocrine abnormalities and impaired muscle contraction, often accompanied by hypoalbuminemia, which affects systemic metabolism and immune or inflammatory responses. We acknowledge the growing evidence indicating that sarcopenia, particularly secondary sarcopenia associated with cachexia, significantly impacts the prognosis of cancer patients. However, our study did not include specific data on sarcopenia, such as muscle mass measurements or functional assessments. Therefore, we could not directly evaluate the relationship between sarcopenia and long-term prognosis. In this study, the inflammation markers (WBC count, PLR, and NLR) mentioned in the two studies mentioned above were also included in the analysis, and all showed correlations with prognosis (OS) in the Cox univariate analysis. However, further stratification analysis revealed that the SII and M demonstrated better predictive ability. Specifically, the PLR was calculated from the peripheral blood platelet count and lymphocyte ratio, the NLR was calculated from the peripheral blood neutrophil count and lymphocyte ratio, and the SII was calculated based on three indicators: peripheral blood platelet count, lymphocyte count, and neutrophil count. Based on the results of this study, it can be inferred that the SII may better reflect the inflammatory status of patients with cancer. On the other hand, Li et al. developed a scoring system based on inflammation and nutritional markers in patients with newly diagnosed metastatic NPC. The system comprises an NRI and three acute inflammation-related biomarkers (C-reactive protein, alkaline phosphatase, and lactate dehydrogenase). It performed well in evaluating short-term progression-free survival (PFS): the low, moderate, and high-risk groups based on this scoring system had 3-years PFS rates of 55.8%, 29.1%, and 11.9%, respectively (P < 0.05)9. This study also validated the potential association between inflammation, nutritional status, and short-term prognosis in patients with cancer. The inflammation-nutritional status of patients may affect the occurrence of short-term disease recurrence and metastasis, thereby influencing their long-term survival.
Our results indicate that the baseline nutritional and inflammatory status before treatment in patients with newly diagnosed non-metastatic NPC may reflect the true tumor condition. Patients with systemic inflammation and malnutrition represent a subgroup that may require individualized treatment. By assessing the baseline inflammatory-nutrition status of patients before treatment, in conjunction with TNM staging, clinicians can achieve more accurate risk stratification. Moreover, based on existing research findings, early interventions targeting inflammation and nutrition in patients with higher inflammation-nutrition scores may improve treatment efficacy and prognosis. However, further prospective studies are required to validate these findings.
Nevertheless, our study has certain limitations. First, patients with cancer in clinical practice often have comorbidities such as metabolic syndrome, liver disease, and skeletal disorders, which can influence the studied markers and thereby affect the inflammation-nutrition scoring results. Second, this study was retrospective, and the patient cohorts used were from a single institution, which cannot prevent potential selection bias. However, the study had comprehensive clinical and pathological characteristics, a relatively long follow-up period, and a large sample size, which partially compensated for these limitations. Third, despite our efforts to mitigate the effects of multicollinearity, we recognized that correlations among variables can still pose limitations. Therefore, we acknowledged that our model's estimates may be influenced by these correlations to some extent. In our study, OS was defined as death from any cause, capturing a comprehensive measure of patient survival over an extended follow-up period. While this approach provides a broad overview of patient outcomes, we acknowledge that cancer-specific mortality could offer more precise insights into the direct impact of NPC on survival. Future studies should consider evaluating cancer-specific mortality alongside OS to differentiate between cancer-related and non-cancer-related deaths, thereby providing a clearer association with oncologic outcomes. Despite this, OS remains a widely accepted and valuable endpoint in oncology research, facilitating comparisons with other studies and clinical practice guidelines. In addition, while OS was the primary endpoint in our study, we acknowledge the importance of examining additional survival outcomes, such as disease-free survival, to provide a more comprehensive assessment of oncologic outcomes. Unfortunately, detailed data on DFS were not collected in this study. Future research should aim to incorporate disease-free survival and other relevant survival outcomes to enhance the understanding of treatment efficacy and disease control in NPC. Our study did not include data on smoking or alcohol consumption, we were unable to analyze their effects on survival outcomes in our study. Finally, some inflammatory or nutritional markers that have been shown to be associated with tumor prognosis in other cancers, such as C-reactive protein, IL-6, and alkaline phosphatase, were not included in this study because of the limitations of retrospective data collection. Therefore, future large-scale, multicenter prospective studies are required to validate our findings.
Conclusion
In conclusion, baseline ALB, M, and SII were independent prognostic factors for long-term survival in patients with newly diagnosed non-metastatic NPC. Based on these three factors, we developed an inflammation-nutrition risk scoring system that facilitates effective risk stratification for patients with NPC, particularly those with advanced-stage disease. INRS, along with TNM staging and serum EBV-DNA levels, can serve as long-term prognostic indicators, enabling the development of personalized treatment strategies for patients.
Data availability
The data that support the findings of this study are available from the corresponding author upon reasonable request.
References
Bray, F. et al. Global cancer statistics 2022: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J. Clin. 74(3), 229–263 (2024).
He, Q. et al. A novel prognostic marker based on risk stratification with prognostic nutritional index and age for nasopharyngeal carcinoma patients who received neoadjuvant chemotherapy. Biomark. Med. 13(12), 1013–1023 (2019).
Chen, Y. P. et al. Nasopharyngeal carcinoma. Lancet 394(10192), 64–80 (2019).
Wei, W. et al. Pretreatment Epstein-Barr virus DNA load and cumulative cisplatin dose intensity affect long5-term outcome of nasopharyngeal carcinoma treated with concurrent chemotherapy: Experience of an institute in an endemic area. Oncol. Res. Treat. 37, 88 (2014).
Wang, W. Y. et al. Long-term survival analysis of nasopharyngeal carcinoma by plasma Epstein-Barr virus DNA levels. Cancer 119(5), 963–970 (2013).
Lee, A. et al. The strength/weakness of the AJCC/UICC staging system (7th edition) for nasopharyngeal cancer and suggestions for future improvement]. Oral Oncol. 48(10), 1007–1013 (2012).
Huang, C. et al. Plasma Epstein-Barr virus DNA load after induction chemotherapy predicts outcome in locoregionally advanced nasopharyngeal carcinoma. Int. J. Radiat. Oncol. Biol. Phys. 104(2), 355–361 (2019).
Zhang, Q. et al. Association of systemic inflammation with survival in patients with cancer cachexia: Results from a multicentre cohort study. J. Cachexia Sarcopenia Muscle 12(6), 1466–1476 (2021).
Li, W. Z. et al. A scoring system based on nutritional and inflammatory parameters to predict the efficacy of first-line chemotherapy and survival outcomes for de novo metastatic nasopharyngeal carcinoma. J. Inflamm. Res. 14, 817–828 (2021).
Hong, J.-S. et al. Effect of chemoradiotherapy on nutrition status of patients with nasopharyngeal cancer. Nutr. Cancer 68(1), 1–7 (2015).
Lin, S. et al. Nasopharyngeal carcinoma treated with reduced-volume intensity-modulated radiation therapy: Report on the 3-year outcome of a prospective series. Int. J. Radiat. Oncol. Biol. Phys. 75(4), 1071–1078 (2009).
Schulz, K. F., Altman, D. G. & Moher, D. CONSORT 2010 statement: Updated guidelines for reporting parallel group randomized trials. Ann. Intern. Med. 152(11), 726–732 (2010).
Schuetz, P. et al. Management of disease-related malnutrition for patients being treated in hospital. Lancet (London, England) 398(10314), 1927–1938 (2021).
Li, G. et al. Influence of pretreatment ideal body weight percentile and albumin on prognosis of nasopharyngeal carcinoma: Long-term outcomes of 512 patients from a single institution. Head Neck 36(5), 660–666 (2014).
Zeng, X. et al. Development and validation of immune inflammation-based index for predicting the clinical outcome in patients with nasopharyngeal carcinoma. J. Cell. Mol. Med. 24(15), 8326–8349 (2020).
Cespedes Feliciano, E. M. et al. Association of systemic inflammation and sarcopenia with survival in nonmetastatic colorectal cancer: Results from the C SCANS study. JAMA Oncol. 3, e172319 (2017).
Evans, D. The use of visceral proteins as nutrition markers: An ASPEN position paper (vol 36, pg 22, 2021). Nutr. Clin. Pract. Offic. Public. Am. Soc. Parenter. Enter. Nutr. 4, 36 (2021).
Miao, J. et al. The value of the prognostic nutritional index (PNI) in predicting outcomes and guiding the treatment strategy of nasopharyngeal carcinoma (NPC) patients receiving intensity-modulated radiotherapy (IMRT) with or without chemotherapy. J. Cancer Res. Clin. Oncol. 143(7), 1263–1273 (2017).
Peng, H. et al. Prognostic value of nutritional risk screening 2002 scale in nasopharyngeal carcinoma: A large-scale cohort study. Cancer Sci. 109(6), 1909–1919 (2018).
Funding
This work was supported by the National Clinical Key Specialty Construction Program (Grant No. 2021), the National Clinical Key Specialty Construction Program, Fujian Provincial Clinical Research Center for Cancer Radiotherapy and Immunotherapy (2020Y2012), and Fujian Clinical Research Center for Radiation and Therapy of Digestive, Respiratory and Genitourinary Malignancies (2021Y2014).
Author information
Authors and Affiliations
Contributions
W.N.F., H.X.W., and L.Q.M. designed this study. Z.P.W., Z.D.F., D.Z. and F.C. contributed to the data collection. C.L., H.X.W., and Z.P.W. analyzed the data. C.L., L.Q.M., and W.N.F. supervised the study. W.N.F., H.X.W. and Z.P.W. wrote the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethics approval and consent to participate
This study was approved by the ethics committee of Fujian Medical University Cancer Hospital, Fuzhou, China (K2023-307-01). The committee waived the requirement for individual informed consent because the patient medical data and follow-up data were extracted retrospectively. All the information was anonymized prior to analysis.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Fang, Wn., Wu, Hx., Wu, Zp. et al. A scoring system based on inflammatory and nutritional indicators to predict the long-term survival of patients with non-metastatic nasopharyngeal carcinoma. Sci Rep 14, 20229 (2024). https://doi.org/10.1038/s41598-024-71360-z
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-024-71360-z