Abstract
Recent theoretical advances in the One Health approach have suggested that cancer pathologies should be given greater consideration, as cancers often render their hosts more vulnerable to infectious agents, which could turn them into super spreaders within ecosystems. Although biologically plausible, this hypothesis has not yet been validated experimentally. Using a community of cnidarians of the Hydra genus (Hydra oligactis, Hydra viridissima, Hydra vulgaris) and a commensal ciliate species (Kerona pediculus) that colonizes them, we tested whether tumoral polyps of H. oligactis, compared to healthy ones, played an amplifying role in the number of ciliates, potentially resulting in a higher likelihood of infection for other community members through spillovers. Our results indicate that K. pediculus has a higher proliferation rate on tumoral polyps of H. oligactis than on healthy ones, which results in the infestation of other hydras. However, the magnitude of the spillover differed between recipient species. This study provides to our knowledge the first elements of proof of concept that tumoral individuals in communities could act as super spreaders of symbionts within and between species, and thus affect biotic interactions and dynamics in ecosystems.
Similar content being viewed by others
Introduction
In the face of the numerous consequences of human activities on the planet (destruction of ecosystems, pollution, climate change…) together with the emergence or re-emergence of diseases, it is now established that a holistic approach, such as in the One Health framework, is essential1,2. This integrated approach aims to understand and address the complex links between human, animal, and environmental health3. By adopting a global perspective, One Health provides an effective response to contemporary challenges, fostering collaboration between disciplines that do not routinely interact (e.g. medical, veterinary, ecological, and social disciplines). Although there has been a plethora of work in the One Health perspective in recent years (see4,5,6 for recent reviews), this area of research is still very active, and major implications are yet to be revealed.
Given the diversity of the links between infectious agents and malignant pathologies7,8,9, Dujon et al.10 recently advocated for a greater reflection on the integration of cancer into the One Health perspective. For example, it is increasingly accepted that human activities are responsible for an increase in cancers in wildlife11,12,13,14, and this could affect the dynamics of parasite communities, given that cancers often lead to a greater vulnerability to infections through, for instance, immune suppression8,15,16,17. More specifically, Dujon et al.10 predicted that cancer pathologies could increase the circulation rate of infectious agents in ecosystems, when hosts harboring tumors become super spreaders which could exacerbate zoonotic risks. Although conceptually appealing, the importance of tumor bearing individuals as super spreaders has, to our knowledge, never been tested.
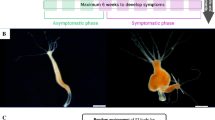
Hydras, notably Hydra oligactis, Hydra vulgaris, and Hydra viridissima, are common freshwater cnidarians regularly found in ponds and rivers in the northern hemisphere18,19. Remarkably, individuals (polyps) in the species H. oligactis frequently develop tumors under laboratory conditions20, with a specific strain even harboring a tumor line that is vertically transmitted during asexual reproduction (i.e. budding)21 (see Fig. 1A,B). These hydras, as well as other hydra species, are sometimes colonized by the ciliate Kerona pediculus, described as a commensal species22,23 (see however24) (see Fig. 1C). These unicellular organisms live on the ectoderm of hydra and feed on cellular waste and leftover food. Boutry et al.25 have demonstrated that ciliates not only exhibit a strong colonization preference for tumoral hydras over healthy hydras but also display an increased population growth rate on the former. Hydra is therefore a promising experimental model for exploring the potential role of tumoral individuals as super spreader of symbionts in communities.
Healthy (A) and tumoral (B) H. oligactis of the St Petersburg strain, the ciliate K. pediculus (C), and the three other introduced hydra strains (C,D). (A) Healthy H. oligactis of the St Petersburg strain (the body is long and thin). (B) Tumoral H. oligactis of the St Petersburg strain presenting numerous masses thickening the body column. (C) Three ciliates on the bottom of the plate. (D) H. vulgaris AEP with testis on his body column. (E) H. oligactis CR. (F) H. viridissima. A trinocular magnifier was used to take the pictures, scale bars: 1 mm for pictures A and B, 200 µm for the C, and 2 mm for D, E, and F.
In this study, using several species of hydras and the ciliate K. pediculus, we aim to (i) first confirm the higher proliferation rate of ciliates on tumoral hydras compared to healthy ones, observed by Boutry et al.25; and then (ii) evaluate the spillover role of tumoral individuals within and between hydra species.
Material and methods
The protocol used in this study is summarized in Fig. 2.
Illustration of the protocol (see text for details).
Origin of hydras and ciliates
We used hydras from different species and health status: (i) healthy and tumoral polyps from Hydra oligactis originating from the St Petersburg clonal strain maintained in culture for over 15 years, differ not only in phenotype (body shape and number of tentacles) but also in microbiota (see21,26); (ii) healthy polyps from H. oligactis strain CR, H. viridissima strain and H. vulgaris strain AEP, all obtained from Professor Galliot’s lab, in Geneva (Switzerland) (see Fig. 1). The latter three strains have been chosen because they present similar body size (although within-strain variability in the body size of polyps is present [personal observation]). All hydras were cultivated in ciliate-free mass cultures filled with Volvic© at 18 °C under a 12-h photoperiod and fed ad libitum three times a week with artemia nauplii (as described in27). The ciliates originated from a mass culture of wild H. oligactis samples collected from the field (Montaud lake in France; 43°44′52″ N; 3°59′23″ E). This study was conducted in accordance with relevant institutional, national, and international guidelines and legislation.
Protocol
Ciliate amplification
To confirm the results of Boutry et al.25, we isolated 18 healthy and 18 tumoral hydras from the St. Petersburg strain and placed them individually in standard cell culture plates (12-well plates, Thermo Scientific, 1.5 ml/well). These hydras are referred to as resident hydras. Each individual was infected with two ciliates sampled from wild hydras, using a P100 pipette (Thermo Finnpipette™) under a binocular magnifier, by gently aspirating the water around the hydra. Polyps were not fed during the experiment so as not to interfere with the amplification of ciliates, which may feed on hydra food leftovers. Over the duration of the experiments, the inoculated resident hydras produced buds which were removed from the wells to preserve the resident hydras as the only possible host. The buds were removed taking care to leave any ciliates they might have hosted in the well, to avoid altering their amplification. After 7 days, the ciliate load on healthy and tumoral hydras was counted. For one tumoral hydra, infection failed, reducing the planned sample size to 5 (from 6) in the infection experiment (see below).
Spillover role of tumoral hydras within and between species
To estimate the spillover potential of healthy and tumoral hydras within and between hydra species, we introduced with the resident hydras, one week after they have been infected by ciliates, one healthy polyp of the other H. oligactis strain (i.e. CR) or of the other hydra species. We therefore paired the 17 tumoral and infected St Petersburg hydras with 5 H. oligactis CR, 6 H. viridissima or 6 H. vulgaris (i.e., a total of 17 pairs). As a control, we paired the 18 non-tumoral and infected St. Petersburg hydras with 6 H. oligactis CR, 6 H. viridissima or 6 H. vulgaris for a total of 18 pairs (see Fig. 2). As before, the polyps were no longer fed from the start of the experiment, and if buds were produced, they were removed, leaving any ciliates they may have hosted in the well. The number of ciliates on each polyp was counted on the next four consecutive days on each resident and introduced hydra. All the ciliates were attached to the hydras and none were found moving freely within the well. When conducting preliminary trials, we observed that within our experimental settings, hydras started to decompose only during the third week of fasting (e.g., loss of tentacles, loss of body shape, and shedding of ectoderm cells), producing organic material that ciliates could feed on. Therefore, we used two weeks as the maximum duration to conduct our experiments.
Data analysis
We used the R software (version 4.2.2)28 to analyze the data; and "GGplot2" package29 to create graphical representations.
Generalized linear mixed-effects models (GLMMs) from the “glmmTMB” package30 were utilized to analyze ciliate load, as this variable represents repeated count data that are non-normally distributed. The ciliate load on resident hydras was analyzed based on their status (healthy or tumoral) and time, using a Poisson distribution. The analysis of ciliate load on introduced hydras according to the status of the resident hydra in their well and time used a negative binomial distribution, to account for underdispersion of residuals when using a Poisson distribution. For the two analyses, a random intercept effect for each individual was added to account for the repeated measurements on the same individuals. Following the three-step protocol of Zuur et al.31, model selection initially focused on the random effect, followed by the fixed effect, guided by the corrected Akaike information criterion (AICc) weights obtained using the "MuMIn" package32. Subsequently, the adequacy of the obtained model fit (e.g. distribution and variance of residuals) was verified using the "DHARMa" package33, which conducts Kolmogorov–Smirnov, outlier, and overdispersion tests on model residuals. The detail of the variable types (i.e. in this case, continuous or categorical variables) as well as all constructed models and their associated AICc weights, are presented in Table S1 in the supplementary materials.
Results and discussion
Ciliate amplification rate is higher on tumoral hydras
One week after the inoculation of two ciliates in the wells containing resident hydras, tumoral hydras harbor an estimated average 2.4 times more ciliates than healthy ones throughout the week (Fig. 3A, Days 0 to 4; GLMM, effect of resident hydra status, IRR = 2.38, SE = 0.49, p-value < 0.001) with an observed weekly average ciliate load of 2.85 ± 0.19 for healthy polyps, and 9.25 ± 073 for tumoral. Furthermore, while the ciliate load was stable on healthy hydras, it increased in average by close to 15% per day in tumoral ones, after the introduction of the healthy polyps (Fig. 3A; GLMM, interaction effect of resident hydra status and time, IRR = 1.15, SE = 0.06, p-value = 0.008). Thus, tumoral hydras harbor a higher ciliate load than healthy hydras even after new hydras were introduced in the system (see Fig. 3A).
Ciliate load on resident and introduced hydras at the introduction of the latest, seven days after the infection of the resident hydras with two ciliates. Measurements begin at Day 0 when the introduced hydras have been added to the well, which is seven days after infecting the resident hydras with two ciliates. The trend line represents the average value predicted by a generalized linear model and the bands the associated confidence interval at 95%. (A) Ciliate load of resident hydras according to their status and time, seven days after their infection with two ciliates. Individual data are connected with a thin colored line. (B) Ciliate load of introduced hydras according to the status of the resident hydra (next to which they were placed at day 0), and time, for each species.
These data confirm the higher amplification rate of ciliates on tumoral hydras compared to healthy ones, as reported by Boutry et al.25. Precise reasons behind this phenomenon are unclear, but could simply be due to the fact that tumoral polyps are bigger (see20), offering more space and/or resources for proliferating ciliates or due to immunodepression caused by the tumor and/or associated microbiome (see25 for a more exhaustive list). Also, significant variability in ciliate load is observed among hydras within each status, and at least two non-exclusive explanations could be advanced to explain this observation. As above, the body size, which is variable between polyps, could provide different surface area and cellular waste for ciliates to develop and feed upon (see for instance34). The general health condition could be another explanation for the variability in both healthy and tumoral hydras' ciliate load. For instance, it has been observed that ciliate load increases drastically when a hydra's life comes to an end (personal observation, S.T, J.B, and J.M), independently of the presence of a tumor. The presence of tumors could decrease the general physical condition of the hydras and accentuate the amplification of ciliates. Consequently, the general physical condition of some hydras may have been poorer or deteriorated faster than others, especially in the context of fasting, thereby introducing variability in ciliate load. Further studies are therefore needed to evaluate the impact of body size and general physical condition of healthy and tumoral hydras on their ciliate load.
Tumoral hydras can act as super spreaders but the magnitude of the effect is species dependent
The analysis of ciliate load on introduced hydras shows a global increased colonization rate over time regardless of the status of the resident hydra (Fig. 3B; GLMM, effect of time, IRR = 1.64, SE = 0.09, p-value < 0.001). However, a hydra (all species combined) placed next to a tumoral hydra harbors on average around 2.4 times more ciliates than one placed next to a healthy hydra (Fig. 3B; GLMM, effect of resident hydra status, IRR = 2.38, SE = 0.85, p-value = 0.015) with an observed weekly average ciliate load: 1.00 ± 0.14 next to a healthy polyp, 2.60 ± 0.39 next to a tumoral polyp). Furthermore, this ciliate load varies according to the species of the introduced hydras; H. vulgaris exhibits a reduced ciliate load about 76% compared to H. oligactis CR and H. viridissima (Fig. 3B; GLMM, effect of introduced hydra species, IRR = 0.24, SE = 0.11, p-value = 0.001), which display comparable ciliate loads (observed weekly average ciliate load: 2.62 ± 0.51 for H. oligactis, 2.18 ± 0.34 for H. viridissima, and 0.60 ± 0.13 for H. vulgaris).
These data support the hypothesis that tumoral individuals could act as super spreaders of symbionts, resulting in a higher ciliate load on the introduced hydra when the resident hydra was tumoral rather than healthy. This observation could be caused by two non-exclusive mechanisms. First, it could result from a higher spillover rate from the tumoral hydras compared to healthy ones, due to their enhanced ciliate amplification on the former, followed by a dispersal process. Second, it could be that the ciliate amplification rate on the introduced hydra is increased in the presence of tumoral hydra in the well. Because the first hypothesis seems more parsimonious, we favor it. Not only tumoral hydras transfer a higher number of ciliates to other hydras, it is also likely that the subsequent local amplification of ciliates on each tumoral hydra accentuates the final difference observed. Interestingly, this spillover effect, for reasons that remain to be determined, varies in intensity depending on the species introduced. Further studies, both in the field and/or in the laboratory, would be necessary to determine whether this differential colonization pattern is naturally observed between the Hydra species used here. For field specimens, this exploration will however need to correct for body size heterogeneity since, for instance, H. viridissima individuals are usually the smallest in natura. Similarly, it would be relevant to explore if the mean ciliate load of each recipient species is, in the long term, enhanced in the presence of super spreader tumoral polyps, as well as the fitness consequence for the colonized hydra. In addition, it would be necessary to explore the general health status (and immunity) of hydra placed near a tumoral one. Indeed, we cannot exclude the hypothesis that tumoral hydras transmit infectious agents to other hydras, leading to an immunosuppressive effect that increases ciliate load. However, this alternative hypothesis would still support the idea that tumoral hydras can act as super spreaders of pathogenic species. Since it is plausible that the larger size of the hydra body, rather than the presence of the tumors itself, contributes to the increased ciliate load, future studies should assess whether oversized healthy individuals can reproduce the spillover effects observed in tumoral individuals. This could also be tested experimentally by transplanting healthy tissues (see for instance35,36) to enhance the body size of hydras with reason other than tumors. Such research is crucial to distinguish the contributions of tumor-related factors from those related to polyp size. Furthermore, it is important to note that K. pediculus is defined as an epibiotic commensal and, according to the literature, does not harm its host (see however24). Although our results represent a first step towards such a proof of concept, a more definitive demonstration would require the study of pathogenic organisms and their impacts on both tumor and healthy hosts. It is essential to consider that pathogens, unlike commensals, might shorten the lifespan of tumoral hosts, potentially preventing them from becoming effective super spreaders. This hypothetical difference warrants further discussion and investigation to understand how pathogens might alter the dynamics observed in our study. In any case, this suggests that the spillover role of tumoral individuals, and its consequences, may not be an easy phenomenon to understand and predict in ecosystems, and further studies are necessary before generalizations can be made.
Conclusion
One of the central goals in research within the One Health theme is to understand the complexity of direct and/or indirect factors that drive the dynamics of pathogens in ecosystems and facilitate spillovers between species37,38,39. Although there are multiple connections between cancerous pathologies, infections and pathogen transmission9, to our knowledge, no study has attempted to link the two within a One Health perspective. Such concerns are nevertheless important because several human activities influence the prevalence of cancers in nature11,12,14. In this work, we explored a key prediction proposed by Dujon et al.10—namely, that tumoral individuals within communities could play the role of symbiont amplifiers, increasing the likelihood of spillover to other species.
Our study provides the first preliminary evidence that tumoral individuals, at least in Hydra, could act as a super spreader of symbionts within and between species. Our findings confirm that tumoral hydras exhibit higher ciliate loads and demonstrate that they have a more pronounced spillover effect compared to healthy ones, although with different consequences according to the species introduced. While the reasons for the higher ciliate colonization of tumoral hydras remain unclear (see also25), these results represent a first step towards full proof of concept, assessing the potential implications of tumor-induced vulnerability to infections for ecosystem functioning8. Further research using more complex experimental approaches is needed to understand the proximate mechanisms underlying the spillover effect of tumoral individuals and to generalize the results obtained to other models, thus exploring their ecological realism. By improving our understanding of these processes through the One Health approach, we can enhance strategies for managing disease transmission and preserving ecosystem stability in natural populations.
Data availability
Scripts and data associated are provided in supplementary information.
Change history
10 February 2025
A Correction to this paper has been published: https://doi.org/10.1038/s41598-025-88642-9
References
Peyre, M. et al. PREZODE: Preventing zoonotic disease emergence. The Lancet 397, 792–793 (2021).
Leal Filho, W., Ternova, L., Parasnis, S. A., Kovaleva, M. & Nagy, G. J. Climate change and zoonoses: A review of concepts, definitions, and bibliometrics. Int. J. Environ. Res. Public. Health 19, 893 (2022).
Atlas, R. M. One health: Its origins and future. In One Health: The Human-Animal-Environment Interfaces in Emerging Infectious Diseases: The Concept and Examples of a One Health Approach (eds Mackenzie, J. S. et al.) 1–13 (Springer, 2013).
Djordjevic, S. P. et al. Genomic surveillance for antimicrobial resistance: A One Health perspective. Nat. Rev. Genet. 25, 142–157 (2024).
Xiao, K. & Zhang, L. Wastewater pathogen surveillance based on One Health approach. Lancet Microbe 4, e297 (2023).
Banerjee, S. & van der Heijden, M. G. A. Soil microbiomes and one health. Nat. Rev. Microbiol. 21, 6–20 (2023).
Ewald, P. W. & Swain Ewald, H. A. Infection and cancer in nature. In Ecology and Evolution of Cancer (eds Ujvari, B. et al.) 47–56 (Academic Press, 2017).
Vittecoq, M. et al. Cancer: A missing link in ecosystem functioning?. Trends Ecol. Evol. 28, 628–635 (2013).
Thomas, F. et al. The importance of cancer cells for animal evolutionary ecology. Nat. Ecol. Evol. 1, 1592–1595 (2017).
Dujon, A. M. et al. On the need for integrating cancer into the One Health perspective. Evol. Appl. 14, 2571–2575 (2021).
Giraudeau, M., Sepp, T., Ujvari, B., Ewald, P. W. & Thomas, F. Human activities might influence oncogenic processes in wild animal populations. Nat. Ecol. Evol. 2, 1065–1070 (2018).
Sepp, T., Ujvari, B., Ewald, P. W., Thomas, F. & Giraudeau, M. Urban environment and cancer in wildlife: Available evidence and future research avenues. Proc. R. Soc. B Biol. Sci. 286, 20182434 (2019).
Klaassen, H. et al. Behavioural ecology meets oncology: Quantifying the recovery of animal behaviour to a transient exposure to a cancer risk factor. Proc. R. Soc. B 291, 20232666 (2024).
Baines, C. et al. Linking pollution and cancer in aquatic environments: A review. Environ. Int. 149, 106391 (2021).
Kareva, I. Immune suppression in pregnancy and cancer: Parallels and insights. Transl. Oncol. 13, 100759 (2020).
Yürekli, A. & Erbaş, O. Cancer and immunosuppression. J. Exp. Basic Med. Sci. 2, 116–121 (2021).
Dujon, A. M. et al. The complex effects of modern oncogenic environments on the fitness, evolution and conservation of wildlife species. Evol. Appl. https://doi.org/10.1111/eva.13763 (2024).
Martínez, D. E. et al. Phylogeny and biogeography of Hydra (Cnidaria: Hydridae) using mitochondrial and nuclear DNA sequences. Mol. Phylogenet. Evol. 57, 403–410 (2010).
Campbell, R. D. Taxonomy of the European Hydra (Cnidaria: Hydrozoa): A re-examination of its history with emphasis on the species H. vulgaris Pallas, H. attenuata Pallas and H. circumcincta Schulze. Zool. J. Linn. Soc. 95, 219–244 (1989).
Boutry, J. et al. Spontaneously occurring tumors in different wild-derived strains of hydra. Sci. Rep. 13, 7449 (2023).
Domazet-Lošo, T. et al. Naturally occurring tumours in the basal metazoan Hydra. Nat. Commun. 5, 4222 (2014).
Coleman, D. C. The laboratory population ecology of Kerona Pediculus (O.F.M.) epizoic on hydra SPP. Ecology 47, 705–711 (1966).
Warren, A. & Robson, E. A. The identity and occurrence of Kerona pediculus (Ciliophora: Hypotrichida), a well-known epizoite of Hydra vulgaris (Cnidaria: Hydrozoa). Zool. Verh. 323, 235–245 (1998).
Stiven, A. E. Experimental Methods for Epizootilogical Research with Hydras. In Hydra: Research Methods (ed. Lenhoff, H. M.) 417–425 (Springer, 1983).
Boutry, J. et al. Tumors (re)shape biotic interactions within ecosystems: Experimental evidence from the freshwater cnidarian Hydra. Sci. Total Environ. 803, 149923 (2022).
Rathje, K. et al. Dynamic interactions within the host-associated microbiota cause tumor formation in the basal metazoan Hydra. PLoS Pathog. 16, e1008375 (2020).
Boutry, J. et al. Tumors alter life history traits in the freshwater cnidarian, Hydra oligactis. iScience 25, 105034 (2022).
R Development Core Team. R: A Language and Environment for Statistical Computing (R Foundation for Statistical Computing, 2020).
Wickham, H. et al. ggplot2: Create Elegant Data Visualisations Using the Grammar of Graphics (2023).
Brooks, M. et al. glmmTMB: Generalized Linear Mixed Models using Template Model Builder (2022).
Zuur, A. F., Ieno, E. N., Walker, N., Saveliev, A. A. & Smith, G. M. Mixed Effects Models and Extensions in Ecology with R (Springer, 2009).
Bartoń, K. MuMIn: Multi-Model Inference (2022).
Hartig, F. & Lohse, L. DHARMa: Residual Diagnostics for Hierarchical (Multi-Level/Mixed) Regression Models (2022).
Poulin, R. Body size vs abundance among parasite species: Positive relationships?. Ecography 22, 246–250 (1999).
Boutry, J. et al. First evidence for the evolution of host manipulation by tumors during the long-term vertical transmission of tumor cells in Hydra oligactis. eLife 13, 891 (2024).
Dujon, A. M. et al. A review of the methods used to induce cancer in invertebrates to study its effects on the evolution of species and ecosystem functioning. Methods Ecol. Evol. 13, 1885–1898 (2022).
Rogalski, M. A., Gowler, C. D., Shaw, C. L., Hufbauer, R. A. & Duffy, M. A. Human drivers of ecological and evolutionary dynamics in emerging and disappearing infectious disease systems. Philos. Trans. R. Soc. B 372, 20160043 (2017).
Sánchez, C. A., Venkatachalam-Vaz, J. & Drake, J. M. Spillover of zoonotic pathogens: A review of reviews. Zoonoses Public Health 68, 563–577 (2021).
Thompson, R. C. A. Parasite zoonoses and wildlife: One health, spillover and human activity. Int. J. Parasitol. 43, 1079–1088 (2013).
Acknowledgements
We are grateful to Chrystelle Perruchoud and Professor Galliot’s lab for providing us with H. oligactis CR, H. vulgaris AEP, and H. viridissima strains. We want to thank the two anonymous referees for their constructive comments that contributed to improving this article. This work was carried out within the framework of the Camargue Health-Environment "Zone Atelier" (ZACAM) of the "Long-Term Socio-Ecological Research network" (LTSER-RZA), funded by the Ecology & Environment of the French National Center for Scientific Research (EE-CNRS).
Funding
This work was funded by the CNRS, the HOFFMANN Family and by the following grant: EVOSEXCAN project (ANR-23-CE13-0007).
Author information
Authors and Affiliations
Contributions
The original idea for the article and the design of the study came from F.T, A.M.N, R.H, S.T, and A.M.D. M.C and J.B carried out the initial design tests and J.M bred the hydras used in this study. S.T realized the experiment and collected the data. S.T performed the statistical analysis with the support of A.M.D. S.T wrote the manuscript with the support of F.T, A.M.N, J.T, J.B, B.U, R.H, A.M.D, J.M, and B.R.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
The original online version of this Article was revised: The original version of this Article contained an error in Figure 3, where the results for Hydra viridissima and Hydra vulgaris were interchanged.
Supplementary Information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Tissot, S., Meliani, J., Chee, M. et al. Cancer and One Health: tumor-bearing individuals can act as super spreaders of symbionts in communities. Sci Rep 14, 21283 (2024). https://doi.org/10.1038/s41598-024-72171-y
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-024-72171-y