Abstract
The present research extensively investigates the significance of the sac centerline length of the anterior cerebral artery (ACA) on aneurysm rupture risk. Hemodynamic factors influencing aneurysm rupture are assessed to identify critical regions prone to rupture. Wall shear stress is analyzed by modeling blood flow in three real saccular ACA cases with varying sac centerline lengths. A one-way fluid-solid interaction (FSI) model is employed to examine the interaction between blood flow and vessel deformation. The computational study reveals that wall shear stress on the ACA saccular aneurysm wall is significantly higher when the sac centerline length is shorter. Conversely, the mean oscillatory index changes with different sac centerline lengths, showing that an increase in sac centerline length leads to higher blood oscillation within the sac area in ACA cases.
Similar content being viewed by others
Introduction
Aneurysms, particularly those located in the cerebral circulation, pose significant clinical challenges due to their potential for rupture, leading to severe neurological consequences. Among the various factors influencing the stability of these vascular structures, geometrical characteristics play a crucial role. One such parameter is the sac centreline length, which refers to the distance along the centerline of the aneurysm sac from its origin to the apex1,2, and3.
Recent studies have highlighted the importance of hemodynamic evaluations in understanding how these geometrical factors correlate with the risk of rupture. The sac centreline length affects blood flow dynamics within the aneurysm, influencing parameters such as flow velocity, wall shear stress, and pressure distribution. Abnormalities in these hemodynamic factors can lead to increased stress on the aneurysm walls, potentially culminating in rupture4,5, and6.
This study aims to explore the relationship between the sac centreline length and the hemodynamic environment of cerebral aneurysms7,8, and9. By analyzing real anatomical models and employing advanced computational fluid dynamics (CFD) techniques, we seek to elucidate how variations in sac geometry can impact the hemodynamic forces at play10,11, and12. Understanding these relationships is critical for improving risk assessment, guiding clinical decision-making, and developing more effective treatment strategies for patients with cerebral aneurysms. Ultimately, this research endeavors to contribute to a more nuanced understanding of aneurysm stability, paving the way for personalized interventions that enhance patient outcomes13,14, and15.
. Aneurysms of the anterior cerebral artery (ACA) represent a significant clinical concern due to their potential for rupture, which can lead to devastating neurological outcomes16,17. Understanding the factors that contribute to the rupture risk of ACA aneurysms is crucial for effective patient management and treatment strategies. Several key hemodynamic and geometric factors play pivotal roles in determining the stability of these vascular structures18,19, and20.
Hemodynamic parameters such as wall shear stress, pressure distribution, and flow dynamics are critical in assessing the rupture risk of ACA aneurysms. Wall shear stress, in particular, reflects the frictional forces exerted by blood flow on the aneurysm wall21,22, and23. Elevated or irregular shear stress can weaken the vessel wall, increasing the likelihood of rupture. Additionally, oscillatory shear index (OSI) provides insights into the flow patterns within the aneurysm, with higher OSI values often associated with unstable hemodynamics that elevate rupture risk.
The geometric characteristics of ACA aneurysms, including sac size, shape, and sac centerline length, are also influential in rupture risk assessment. Larger aneurysms or those with complex shapes may experience more pronounced hemodynamic stresses, leading to increased vulnerability. The sac centerline length, in particular, is a critical parameter that correlates with flow dynamics and stress distribution along the aneurysm wall24.
Understanding these factors is essential for developing tailored treatment approaches and improving risk stratification in patients with ACA aneurysms25. By integrating hemodynamic and geometric analyses, clinicians can enhance their predictive capabilities regarding aneurysm behavior, potentially guiding interventions such as endovascular therapy or surgical options.
This article investigates how the geometric characteristics of a cerebral aneurysm affect blood hemodynamics within a sac located in the Anterior Cerebral Artery (ACA) in relation to rupture risk analysis. Utilizing the finite volume method of computational fluid dynamics, the study models blood flow dynamics within the ACA aneurysm to calculate key hemodynamic parameters associated with aneurysm rupture. The primary aim of this research is to explore the relationship between sac centerline length and wall shear stress, which is a critical hemodynamic factor for predicting aneurysm rupture in selected cases.
Computational methods and aneurysm geometries
It is confirming that all methods were carried out in accordance with relevant guidelines and regulations. Besides, all experimental protocols were approved by of the Emory University and it is confirmed that informed consent was obtained from all subjects and/or their legal guardian(s)26.
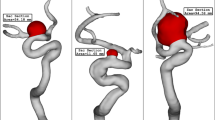
The ICA artery branches into the MCA and ACA, as shown in Fig. 1, which also illustrates the definition of the sac centerline length. In this study, specific ACA cases from the Aneurisk website26 have been selected for hemodynamic analysis. Focusing on the sac centerline length of ACA aneurysms, aneurysm with length of 3.10, 7.7 and 10.40 mm are chosen, while other geometrical parameters of the selected ACA aneurysms remain constant.
Sac centerline length definition.
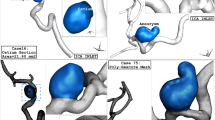
Figure 2 displays the geometry and shape of the chosen ACA saccular aneurysms. These cases are chosen from Aneurisk Web project website26. The real shape of these models with full details would result in precise results.
ACA aneurysm geometry of 3 different cases.
The modeling of transient blood flow in the ACA cases is conducted by solving the Navier-Stokes equations, treating pulsatile blood as a non-Newtonian fluid. The Casson model is employed to define the viscosity in the governing Eqs27,28. The Casson model is a widely used non-Newtonian fluid model specifically designed to describe the flow behavior of blood. Blood is a complex fluid composed of plasma, red blood cells, white blood cells, and platelets, and its viscosity does not behave like a simple Newtonian fluid, where viscosity remains constant irrespective of the shear rate. The Casson model is particularly advantageous for modeling blood viscosity due to several key features:
Despite its ability to capture the complex rheological behavior of blood, the Casson model remains relatively simple mathematically. This simplicity allows for easier implementation in computational simulations, providing a balance between accuracy and computational efficiency. The Casson model is derived empirically, meaning it is based on observed data of blood flow. This makes it particularly well-suited for practical applications where an empirical fit to real-world data is needed, such as in medical device design or in predicting blood flow under various pathological conditions.
The Casson model is versatile and can be applied in various scenarios, including large arteries, small capillaries, and even in pathological conditions such as in the presence of blood clots or altered blood rheology due to diseases like diabetes or sickle cell anemia. The Casson model’s consideration of yield stress and shear-thinning properties leads to improved accuracy in predicting how blood interacts with vessel walls, particularly in conditions where flow separation or recirculation occurs, such as in aneurysms or stenosed arteries.
A mass flow profile representative of normal bodily activity is applied at the inlet, along with an equivalent outlet pressure at the outlet. The results of the computational study are presented after the completion of the third cardiac cycle. A straightforward algorithm corrects pressure and velocity in the model. OpenFOAM software is used for the modeling of the non-Newtonian flow of blood29.
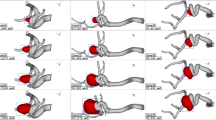
Grid generation for accurately modeling real aneurysm shapes is crucial, as grid size significantly impacts the data obtained, particularly near the vessel. Figure 3 illustrates the grid created for the three ACA cases. The grid size on the vessel wall is nearly uniform, while the cross-sectional grid shows that the density is greater near the vessel wall compared to the center30,31. This is important because the primary hemodynamic features related to aneurysm rupture are assessed on the sac wall2,32. The figure also includes a close-up view of the grid on the sac surface. The total number of grid cells for these three models ranges from 1,450,000 to 1,950,000.
Grid production for the chosen models.
Results and discussion
Figure 4 shows the variations in the oscillatory shear index (OSI) in the sac region at early diastole (end of the cardiac cycle). The highest OSI index is not found on the dome or ostium areas. Instead, a critical OSI value is observed near the dome, where flow changes are more pronounced than in other segments. Although this critical OSI region may appear small, the risk of rupture is elevated in these areas.
OSI contour of 3 selected cases.
The oscillatory shear index (OSI) is a hemodynamic parameter that is used to quantify the change in the direction of shear stress over a cardiac cycle. In the context of evaluating the risk of hemorrhage in saccular aneurysms, OSI provides important insights into the flow patterns within the aneurysm sac and its impact on the aneurysm wall. Saccular aneurysms are bulging, balloon-like dilations of a blood vessel wall, usually occurring at arterial bifurcations. The flow within these aneurysms is often complex, with regions of flow recirculation and stagnation. OSI is particularly important in this context because it reflects the degree of oscillatory or disturbed flow within the aneurysm. High OSI values indicate that the blood flow is highly disturbed, which can lead to abnormal endothelial cell behavior, inflammation, and potential degradation of the vessel wall. The comparison of the mean OSI value (Fig. 5) of these models indicates that the extension of the sac centerline length results in higher OSI values. Indeed, the risk of aneurysm rupture is increased by the increase of the sac centerline length in ACA aneurysms.
Mean OSI of 3 selected cases.
To analyze the hemodynamic of the blood flow, the iso-velocity of the bloodstream in the selected ACA cases is displayed in (Fig. 6). The core of the flow inside the sac area indicates that the angle of the main parent vessel is crucial to the structure of the bloodstream inside the sac section of ACA aneurysms. In the case with a sac centerline of 3.1 mm, most portion of the ACA aneurysm is covered by the high-velocity blood flow. The stream of the blood inside the sac region is also illustrated in (Fig. 7). The streamline indicates that the velocity of the incoming blood flow in the sac section is considerably reduced after passing the neck region. Accumulation of the blood inside the aneurysms reduces the velocity in the dome section. Consequently, a higher wall shear stream could occur in the neck area of the aneurysms.
Velocity Iso-surface of 3 selected cases.
Streamlines of 3 selected cases.
Figure 8 also disclosed the flow recirculation and contact of the bloodstream with the wall of ACA aneurysms. When the length of the sac centerline is increased, the velocity of the blood flow is reduced in the sac region as demonstrated in (Fig. 8). The alteration of the sac centerline directly changes the mean sac velocity and this confirms that the velocity of the blood inside the sac region is connected to the geometry of the aneurysms. Table 1 presents the results of the hemodynamic factors for the complete evaluation of the achieved data of three ACA cases.
Mean sac velocity of 3 selected cases.
The comparison of the velocity on the plane located at the sac ostium region is done and velocity profile are displayed as surface in (Fig. 9). The shape of velocity surface at this section indicates the high-velocity of blood flow enters to the sac domain. It is important since this make the wall shear stress on the sac surface. The figure demonstrates that the creeping flow in case 3 has high velocity near sac surface.
velocity changes on the ostium section of 3 selected cases.
Conclusion
This article provides a thorough hemodynamic analysis to highlight the significance of sac centerline length in ACA saccular aneurysms concerning hemodynamic factors associated with rupture risk. Three saccular ACA aneurysms with varying sac centerline lengths are selected to examine how this geometric factor influences rupture risk. Blood flow within the ACA cases is simulated using a computational fluid dynamics approach, assuming the flow is non-Newtonian, transient, and incompressible. Velocity surface at ostium section and oscillatory shear index (OSI) are evaluated at both peak systolic and early diastolic phases to assess the rupture risk across different sac sizes. The results indicate that wall shear stress is elevated near the neck region of the ACA cases, while OSI calculations during early diastole show that the oscillatory index is low in both the dome and neck areas. Additionally, the findings reveal that wall shear stress on the sac surface significantly decreases in ACA cases with longer sac centerline lengths.
Data availability
All data generated or analysed during this study are included in this published article.
References
Shiryanpoor, I., Valipour, P., Barzegar Gerdroodbary, M., Abazari, A. M. & Moradi, R. Using computational fluid dynamic for evaluation of rupture risk of micro cerebral aneurysms in the growth process: hemodynamic analysis. Int. J. Mod. Phys. C (2024).
Rostamian, A., Fallah, K. & Rostamiyan, Y. Reduction of rupture risk in ICA aneurysms by endovascular techniques of coiling and stent: numerical study. Sci. Rep. 13, 7216. https://doi.org/10.1038/s41598-023-34228-2 (2023).
Hu, R. & Feng, H. Lenticulostriate artery and lenticulostriate-artery neural complex: New concept for intracerebral hemorrhage. Curr. Pharm. Des. 23 (15), 2206–2211. https://doi.org/10.2174/1381612823666170220163750 (2017).
Rostamian, A., Fallah, K., Rostamiyan, Y. & Alinejad, J. Application of computational fluid dynamics for detection of high risk region in middle cerebral artery (MCA) aneurysm. Int. J. Mod. Phys. C 2350019. (2022).
Zhang, C. et al. Hematoma evacuation via image-guided para-corticospinal tract approach in patients with spontaneous intracerebral hemorrhage. Neurol. Ther. 10(2), 1001–1013. https://doi.org/10.1007/s40120-021-00279-8 (2021).
Jiang, H., Lu, Z., Barzegar Gerdroodbary, M., Sabernaeemi, A. & Sajad Salavatidezfouli. The influence of sac centreline on saccular aneurysm rupture: computational study. Sci. Rep. 13(1), 11288 (2023).
Shen, B. et al. Cerebral hemodynamics underlying ankle force sense modulated by high-definition transcranial direct current stimulation. Cereb. Cortex 34(6), bhae226. https://doi.org/10.1093/cercor/bhae226 (2024).
Huang, Y. et al. Lumican promotes calcific aortic valve disease through H3 histone lactylation. Eur. Heart J. ehae407. https://doi.org/10.1093/eurheartj/ehae407 (2024).
Yu, Y. et al. Targeting loop3 of sclerostin preserves its cardiovascular protective action and promotes bone formation. Nat. Commun. 13(1), 4241. https://doi.org/10.1038/s41467-022-31997-8 (2022).
Ge, H. et al. Targeting ASIC1a promotes neural progenitor cell migration and neurogenesis in ischemic stroke. Research 6, 105. https://doi.org/10.34133/research.0105 (2023).
Valipour, P. Effects of coiling embolism on blood hemodynamic of the MCA aneurysm: a numerical study. Sci. Rep. 12(1), 22029 (2022).
Liu, R., Ren, C., Fu, M., Chu, Z. & Guo, J. Platelet detection based on improved YOLO_v3. Cyborg Bionic Syst. 2022https://doi.org/10.34133/2022/9780569 (2022).
Fathollahi, R. et al. Examination of bio convection with nanoparticles containing microorganisms under the influence of magnetism fields on vertical sheets by five-order Runge-Kutta method. Heliyon (2023).
Mousavi, S.V., Barzegar Gerdroodbary, M., Sabernaeemi, A., Salavatidezfouli, S., Valipour, P. Impacts of the aneurysm deformation induced by stent on hemodynamic of blood flow in saccular internal carotid artery aneurysms. AIP Adv. 14, 095035 (2024).
Zafaripour, G. et al. Fabrication and evaluation of 3D bio-scaffold wound dressings for monitoring of chronic pH wounds using fuzzy logic analysis. Mater. Sci. Eng. B 294, 116542 (2023).
Shen, Xiao-Yong, M., Barzegar Gerdroodbary, A. M., Abazari & Moradi, R. Computational study of blood flow characteristics on formation of the aneurysm in internal carotid artery. Eur. Phys. J. Plus 136 (5), 541 (2021).
Shen, X. Y., Xu, H. Q., Barzegar Gerdroodbary, M., Valiallah Mousavi, S. & Abazari, A. M. Misagh Imani. Numerical simulation of blood flow effects on rupture of aneurysm in middle cerebral artery. Int. J. Mod. Phys. C 33 (03), 2250030 (2022).
Wu, W., Alizadeh, A. A. & Hekmatifar, M. The effect of initial temperature on the mechanical interaction of 3DN5/5OTF protein-based structures using molecular dynamics simulation. Int. J. Biol. Macromol., 123235. (2023).
Karimipour, A. & Toghraie, D. Lokman Aziz Abdulkareem, As’ ad Alizadeh, Majid Zarringhalam, and Arash Karimipour. Roll of stenosis severity, artery radius and blood fluid behavior on the flow velocity in the arteries: application in biomedical engineering. Med. Hypotheses 144 : 109864 (2020).
Sadeh, A. et al. Computational study of blood flow inside MCA aneurysm with/without endovascular coiling. Sci. Rep. 13, 4560. https://doi.org/10.1038/s41598-023-31522-x (2023).
Poueinak, M. M. Seyyed Amirreza Abdollahi, as’ad Alizadeh, Mahya Ahmadpour Youshanlui,Hussein Zekri, M. Barzegar Gerdroodbary. Computational study of blood hemodynamic in ICA aneurysm with coiling embolism. Int. J. Mod. Phys. C 34 (6), 2350138. https://doi.org/10.1142/S0129183123501383 (2023).
Sabernaeemi, A. et al. Influence of stent-induced vessel deformation on hemodynamic feature of bloodstream inside ICA aneurysms. Biomech. Model. Mechanobiol.https://doi.org/10.1007/s10237-023-01710-9 (2023).
Hariri, S., Poueinak, M. M., Hassanvand, A., Barzegar Gerdroodbary, M. & Faraji, M. Effects of blood hematocrit on performance of endovascular coiling for treatment of middle cerebral artery (MCA) aneurysms: computational study. Interdiscip. Neurosurg. 32, 101729 (2023).
Zan-Hui Jin, M., Barzegar Gerdroodbary, P., Valipour, M., Faraji, Nidal, H. & Abu-Hamdeh CFD investigations of the blood hemodynamic inside internal cerebral aneurysm (ICA) in the existence of coiling embolism. Alex. Eng. J.https://doi.org/10.1016/j.aej.2022.10.070 (2023).
Armin Sheidani, M., Barzegar Gerdroodbary, A., Poozesh, A., Sabernaeemi, S. & Salavatidezfouli Arash Hajisharifi, influence of the coiling porosity on the risk reduction of the cerebral aneurysm rupture: computational study. Sci. Rep. 12, 19082 (2022).
AneuriskWeb project website, Emory University, Department of Math&CS. http://ecm2.mathcs.emory.edu/aneuriskweb (2012).
Sajad Salavatidezfouli, A. et al. Investigation of the stent induced deformation on hemodynamic of internal carotid aneurysms by computational fluid dynamics. Sci. Rep. 13 (1), 7155 (2023).
Voss, S., Beuing, O., Janiga, G. & Philipp Berg. Stent-induced vessel deformation after intracranial aneurysm treatment–A hemodynamic pilot study. Comput. Biol. Med. 111, 103338 (2019).
OpenFOAM Foundation. OpenFOAM: The Open Source CFD Toolbox. User Guide. (2023). https://www.openfoam.com/documentation/user-guide
Sadeh, A., Kazemi, A., Bahramkhoo, M. & Barzegar Gerdroodbary, M. Computational analysis of the blood hemodynamic inside internal cerebral aneurysm in the existence of endovascular coiling. Int. J. Mod. Phys. C 34 (05), 2350059 (2023).
Sadeghi, A., Amini, Y. & Saidi, M. H. Shear-rate‐dependent rheology effects on mass transport and surface reactions in biomicrofluidic devices. AIChE J. 61(6), 1912–1924 (2015).
Mitsos, A. P., Nikolaos, M. P., Kakalis, Y. P. & Ventikos and James V. Byrne. Haemodynamic simulation of aneurysm coiling in an anatomically accurate computational fluid dynamics model. Neuroradiology 50, 4 : 341–347. (2008).
Author information
Authors and Affiliations
Contributions
Z.S. and W.T. wrote the main manuscript text and Y.L. and H.W. prepared figures and Z.G. supervised project. All authors reviewed the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Su, Z., Teng, W., Liu, Y. et al. Hemodynamics study of anterior cerebral artery with different sac centerline lengths for rupture risk assessment. Sci Rep 14, 22500 (2024). https://doi.org/10.1038/s41598-024-73231-z
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-024-73231-z