Abstract
Hyponatremia can worsen the outcomes of patients with liver cirrhosis. However, it remains unclear about how to predict the risk of death in cirrhotic patients with hyponatremia. Patients with liver cirrhosis and hyponatremia were screened. Eligible patients were randomly divided into the training (n = 472) and validation (n = 471) cohorts. In the training cohort, the independent predictors for in-hospital death were identified by logistic regression analyses. Odds ratios (ORs) were calculated. An artificial neural network (ANN) model was established in the training cohort. Areas under curve (AUCs) of ANN model, Child-Pugh, model for end-stage liver disease (MELD), and MELD-Na scores were calculated by receiver operating characteristic curve analyses. In multivariate logistic regression analyses, ascites (OR = 2.705, P = 0.042), total bilirubin (OR = 1.004, P = 0.003), serum creatinine (OR = 1.004, P = 0.017), and international normalized ratio (OR = 1.457, P = 0.005) were independently associated with in-hospital death. Based on the four variables, an ANN model was established. Its AUC was 0.865 and 0.810 in the training and validation cohorts, respectively, which was significantly larger than those of Child-Pugh (AUC = 0.757), MELD (AUC = 0.765), and MELD-Na (AUC = 0.769) scores. An ANN model has been developed and validated for the prediction of in-hospital death in patients with liver cirrhosis and hyponatremia.
Similar content being viewed by others
Introduction
Hyponatremia, the most common electrolyte disorder in liver cirrhosis, is significantly associated with worse outcome1,2. Hyponatremia is defined as serum sodium (Na) level < 135mmol/L3, and its prevalence is approximately 50% in patients with liver cirrhosis and ascites4. In a registry of 6796 patients awaiting liver transplantation, an increase of 5% in the risk of death was observed per unit decrease in serum Na level5. Thus, serum Na level has been included in the MELD-Na score to determine the priority of liver transplantation6. However, at present, effective treatment of hypervolemic hyponatremia, especially moderate and severe hyponatremia, is still lacking in patients with liver cirrhosis7. Therefore, it is crucial to clarify the patients with poor prognosis in the early stage of disease.
Artificial intelligence (AI), especially machine learning, has been widely used to develop the diagnosis and prognosis model of various diseases8. Nowadays, several specific AI approaches, such as support vector machine, random forest, decision tree, and artificial neural network (ANN), outperform conventional approaches in screening and diagnosis of various diseases and drug design9. Among them, ANN is one of the most commonly used to construct a predictive model in the nonlinear complex biological systems10,11. It has been used to predict post-hepatectomy survival of early hepatocellular carcinoma12, evaluate liver transplantation prioritization13, manage colorectal cancer liver metastases14, differentiate between prostate cancer from benign hyperplasia of prostate15, and diagnose chronic atrophic gastritis16. Generally, ANN models are more accurate than multiple logistic regression and multiple linear discriminant analysis models17,18.
This study aimed to develop a predictive model for the in-hospital death of patients with liver cirrhosis and hyponatremia by using an ANN approach.
Method
Study design
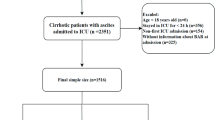
This retrospective study conformed to the ethical guidelines of the 1975 Declaration of Helsinki and was approved by the Medical Ethical Committee of the General Hospital of Northern Theater Command with an approval number of Y(2022)171. The requirement of informed written consent was waived, because we only extracted the data from the inpatients’ medical records. As mentioned in our previous studies19,20, patients with liver cirrhosis who were consecutively admitted to the General Hospital of Northern Theater Command from January 2010 to June 2014 were selected. Patients who lacked baseline data were excluded. Hyponatremia was defined as a serum Na level of less than 135mmol/L. The Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) checklist for research reporting of observational studies was followed.
Dataset acquisition
Baseline data included age, sex, etiology of liver cirrhosis, ascites, acute upper gastrointestinal bleeding (AUGIB), hepatocellular carcinoma (HCC), hemoglobin (Hb), white blood cell (WBC), platelet (PLT), total bilirubin (TBIL), alanine aminotransferase (ALT), albumin (ALB), alkaline phosphatase (AKP), serum creatinine (Scr), potassium (K), Na, and international normalized ratio (INR). Child-Pugh and model for end-stage liver disease (MELD) scores were calculated. The outcome data was the death during hospitalization.
Statistical analyses
Continuous variables were expressed as mean ± standard deviation and median (range), and categorical variables were expressed as frequency (percentage). Random sampling was used to divide patients into training and validation cohorts with an approximate percentage of 50%. Difference between training and validation cohorts was compared by the nonparametric Mann-Whitney U test and the Chi-square test. Logistic regression analyses were performed to identify the independent predictors associated with in-hospital death in the training cohort. Odds ratios (ORs) with 95% confidence intervals (CIs) were calculated. In the training cohort, an ANN model was established by the independent predictors by the nnet package of the R software. Then, receiver operating characteristic curve (ROC) analysis was performed to evaluate the predictive performance of the ANN model. Its area under curve (AUC) was calculated and compared with those of Child-Pugh, MELD, and MELD-Na scores. The predictive performance of the ANN model was also validated in the validation cohort. Additionally, the calibration ability of the ANN model was measured using the calibration plot and Hosmer-Lemeshow test. All statistical analyses were performed by the SPSS software (version 25.0, IBM Corp, Armonk, NY, USA), R software (version 4.0.3, R Core Team, R Foundation for Statistical Computing, Vienna, Austria), and MedCalc software (version 11.4.2.0, MedCalc Software, Mariakerke, Belgium). A two-tailed P < 0.05 was considered statistically significant.
Results
Characteristics of included patients
Overall, 943 patients with cirrhosis and hyponatremia were included. Their mean age was 59.35 years. Among them, 669 patients were male, and 396, 325, 247, 219, and 626 patients had HBV infection, alcohol-related liver disease, HCC, AUGIB, and ascites at their admissions. One hundred patients died during hospitalization (Table 1).
Characteristics of training and validation cohorts
Among them, 472 and 471 patients were assigned to the training and validation cohorts, respectively. The training cohort has significantly higher proportion of AUGIB (26.30% vs. 20.20%, P = 0.027) and lower Hb level (92.06 ± 30.06 vs. 96.49 ± 28.12, P = 0.010) than the validation cohort. Other baseline data was not significantly different between training and validation cohorts (Table 2).
Development of ANN model
Univariate logistic regression analyses showed that ascites, WBC, TBIL, ALB, ALT, Scr, K, INR, Child-Pugh score, and MELD score were significantly associated with the in-hospital death of cirrhotic patients with hyponatremia in the training cohort (Table 3). Multivariate logistic regression analyses showed that ascites (OR = 2.705, 95%CI: 1.036–7.060, P = 0.042), TBIL (OR = 1.004, 95%CI: 1.001–1.006, P = 0.003), Scr (OR = 1.004, 95%CI: 1.001–1.007, P = 0.017), and INR (OR = 1.457, 95%CI: 1.123–1.891, P = 0.005) were independently associated with in-hospital death of cirrhotic patients with hyponatremia (Table 3). Accordingly, the ANN model for predicting the in-hospital death in cirrhotic patients with hyponatremia was developed (Supplementary Fig. 1). The ANN model had a significantly higher AUC than the Child-Pugh (0.865 vs. 0.757, P = 0.0018), MELD (0.865 vs. 0.765, P = 0.004), and MELD-Na (0.865 vs. 0.769, P = 0.0016) scores (Fig. 1). Calibration plots showed a satisfactory accordance between the predicted probabilities based on the ANN model and actual values (Supplementary Fig. 2A). The Hosmer-Lemeshow test value was 10.369 (P = 0.240).
ROC curves of ANN model, Child-Pugh score, MELD score, and MELD-Na score for predicting the risk of in-hospital death in patients with cirrhosis and hyponatremia in the training cohort.
Validation of ANN model
The AUC of this ANN model remained significantly higher than Child-Pugh (0.810 vs. 0.502, P < 0.001), MELD (0.810 vs. 0.520, P < 0.001), and MELD-Na (0.810 vs. 0.510, P < 0.001) scores in the validation cohort (Fig. 2). Calibration plots showed the satisfactory accordance between the predicted probabilities based on the ANN model and actual values (Supplementary Fig. 2B). The Hosmer-Lemeshow test value was 5.045 (P = 0.538).
ROC curves of ANN model, Child-Pugh score, MELD score, and MELD-Na score for predicting the risk of in-hospital death in patients with cirrhosis and hyponatremia in the validation cohort.
Discussion
The current study developed and validated an ANN model for the prediction of in-hospital death in patients with liver cirrhosis and hyponatremia. It includes only four clinical variables, including ascites, TBIL, Scr, and INR, which can be easily obtained in clinical practice.
Hyponatremia is a common electrolyte dysfunction in liver cirrhosis. Its major pathogenesis is portal hypertension, which is induced by increased hepatic resistance to portal blood flow through the liver in cirrhosis21. Portal hypertension can induce the development of hyperdynamic circulation and splanchnic vasodilation22. Additionally, the impairment of the intestinal mucosal barrier caused by portal hypertension will lead to bacterial translocation to the submucosa, and then induce the development of inflammation and release of vasoactive mediators23,24,25, which can further worsen hyperdynamic circulation and splanchnic vasodilation in liver cirrhosis26. Splanchnic vasodilation can activate the renin angiotensin aldosterone system (RAAS) and cause abnormal secretion of antidiuretic hormone (ADH)27. Aldosterone can reserve sodium and water28, and ADH can reserve a large amount of water29, thereby inducing the development of hypervolemic hyponatremia30. Thus, hepatic and renal dysfunction as well as water-sodium retention are important for the development and progression of hyponatremia in liver cirrhosis. Accordingly, in our ANN model, TBIL and INR indicate the severity of liver dysfunction in liver cirrhosis, Scr is a conventional parameter for the severity of renal dysfunction, and ascites reflects the status of water-sodium retention.
Some existing models include Child-Pugh31,32, ALBI33,34, NLR35,36, and MELD37,38,39 scores for predicting the outcomes of cirrhotic patients. However, all of them focus on general patients with liver cirrhosis, but not cirrhotic patients with hyponatremia. In 2021, based on the National Inpatient Sample database, Thuluvath et al. developed a model to predict the risk of in-hospital death in patients with liver cirrhosis and hyponatremia40. The characteristics of the study population are different between the Thuluvath’s and our studies. Our study included all cirrhotic patients with hyponatremia, and the Thuluvath’s study included cirrhotic patients with ascites and hyponatremia but without HCC. Additionally, the Thuluvath’s model included age ≥ 65, variceal bleeding, ACLF, sepsis, and coagulopathy. But it should be noted that the diagnostic criteria of ACLF are greatly different among regions41,42,43,44,45. In contrast, four variables from our ANN model were objective. Finally, the Thuluvath’s model was based on the traditional logistic regression analysis, but our model was based on the ANN, as a mathematical method governed by the biological nervous system, which is similar to the information processing in the central nervous system and can complete statistical analyses of numerous complex relationships46.
Our study has several limitations. First, it was retrospective, so a potential patient selection bias was inevitable. Second, only internal validation was performed in our study, but not external validation. Third, the treatment selection during hospitalization might influence the patients’ outcomes, and we just included the baseline data to predict the risk of in-hospital death in patients with liver cirrhosis and hyponatremia. Fourth, we did not follow these patients or analyze their long-term outcomes in the current study. Fifth, the potential influence of previous decompensation and drugs cannot be analyzed in the current study. Sixth, we did not distinguish patients into acute decompensated cirrhosis, chronic decompensated cirrhosis, and acute-on-chronic liver failure groups.
In conclusion, an ANN model was constructed to predict the in-hospital death of patients with liver cirrhosis and hyponatremia with a good performance in both training and validation cohorts.
Data availability
The data used to support the findings of this study are available from the corresponding author upon request.
References
Borroni, G., Maggi, A., Sangiovanni, A., Cazzaniga, M. & Salerno, F. Clinical relevance of hyponatraemia for the hospital outcome of cirrhotic patients. Dig. Liver Dis. 32, 605–610 (2000).
Jenq, C. C. et al. Serum sodium predicts prognosis in critically ill cirrhotic patients. J. Clin. Gastroenterol. 44, 220–226 (2010).
Aithal, G. P. et al. Guidelines on the management of ascites in cirrhosis. Gut. 70, 9–29 (2021).
Angeli, P., Wong, F., Watson, H., Ginès, P. Hyponatremia in cirrhosis: results of a patient population survey. Hepatology. 44, 1535–1542 (2006).
Kim, W. R. et al. Hyponatremia and mortality among patients on the liver-transplant waiting list. N Engl. J. Med.359, 1018–1026 (2008).
Kalra, A., Wedd, J. P. & Biggins, S. W. Changing prioritization for transplantation: MELD-Na, hepatocellular carcinoma exceptions, and more. Curr. Opin. Organ. Transpl. 21, 120–126 (2016).
Alukal, J. J., John, S. & Thuluvath, P. J. Hyponatremia in cirrhosis: an update. Am. J. Gastroenterol. 115, 1775–1785 (2020).
Haug, C. J. & Drazen, J. M. Artificial Intelligence and Machine Learning in Clinical Medicine, 2023. N Engl. J. Med. 388, 1201–1208 (2023).
Nayarisseri, A. et al. Artificial Intelligence, Big Data and Machine Learning approaches in Precision Medicine & Drug Discovery. Curr. Drug Targets. 22, 631–655 (2021).
Shao, F. & Shen, Z. How can artificial neural networks approximate the brain? Front. Psychol. 13, 970214 (2022).
Topol, E. J. High-performance medicine: the convergence of human and artificial intelligence. Nat. Med. 25, 44–56 (2019).
Qiao, G. et al. Artificial neural networking model for the prediction of post-hepatectomy survival of patients with early hepatocellular carcinoma. J. Gastroenterol. Hepatol. 29, 2014–2020 (2014).
Bagheri Lankarani, K. et al. Predictors of death in the liver transplantation adult candidates: an Artificial neural networks and support Vector Machine Hybrid-based Cohort Study. J. Biomed. Phys. Eng. 12, 591–598 (2022).
Rompianesi, G., Pegoraro, F., Ceresa, C. D., Montalti, R. & Troisi, R. I. Artificial intelligence in the diagnosis and management of colorectal cancer liver metastases. World J. Gastroenterol. 28, 108–122 (2022).
Sadoughi, F. & Ghaderzadeh, M. A hybrid particle swarm and neural network approach for detection of prostate cancer from benign hyperplasia of prostate. Stud. Health Technol. Inf. 205, 481–485 (2014).
Zhang, Y. et al. Diagnosing chronic atrophic gastritis by gastroscopy using artificial intelligence. Dig. Liver Dis. 52, 566–572 (2020).
Montie, J. E. & Wei, J. T. Artificial neural networks for prostate carcinoma risk assessment. An overview. Cancer. 91, 1647–1652 (2001).
Grossi, E., Mancini, A. & Buscema, M. International experience on the use of artificial neural networks in gastroenterology. Dig. Liver Dis. 39, 278–285 (2007).
Bai, Z. et al. Effects of Short-Term Human Albumin Infusion for the Prevention and Treatment of Hyponatremia in patients with liver cirrhosis. J. Clin. Med. 12, 107 (2022).
Bai, Z. et al. Albumin infusion may decrease the incidence and severity of overt hepatic encephalopathy in liver cirrhosis. Aging (Albany NY). 11, 8502–8525 (2019).
Bosch, J., Groszmann, R. J. & Shah, V. H. Evolution in the understanding of the pathophysiological basis of portal hypertension: how changes in paradigm are leading to successful new treatments. J. Hepatol. 62, S121–S130 (2015).
Martell, M., Coll, M., Ezkurdia, N., Raurell, I. & Genescà, J. Physiopathology of splanchnic vasodilation in portal hypertension. World J. Hepatol. 2, 208–220 (2010).
Byl, B., Roucloux, I., Crusiaux, A., Dupont, E. & Devière, J. Tumor necrosis factor alpha and interleukin 6 plasma levels in infected cirrhotic patients. Gastroenterology. 104, 1492–1497 (1993).
Albillos, A. et al. Increased lipopolysaccharide binding protein in cirrhotic patients with marked immune and hemodynamic derangement. Hepatology. 37, 208–217 (2003).
Wiest, R., Lawson, M. & Geuking, M. Pathological bacterial translocation in liver cirrhosis. J. Hepatol. 60, 197–209 (2014).
Arroyo, V., García-Martinez, R. & Salvatella, X. Human serum albumin, systemic inflammation, and cirrhosis. J. Hepatol. 61, 396–407 (2014).
John, S. & Thuluvath, P. J. Hyponatremia in cirrhosis: pathophysiology and management. World J. Gastroenterol. 21, 3197–3205 (2015).
Funder, J. W. Aldosterone and mineralocorticoid receptors-physiology and pathophysiology. Int. J. Mol. Sci. 18, 1032 (2017).
Nielsen, S., Marples, D., Frøkiaer, J., Knepper, M. & Agre, P. The aquaporin family of water channels in kidney: an update on physiology and pathophysiology of aquaporin-2. Kidney Int. 49, 1718–1723 (1996).
Ginès, P. & Guevara, M. Hyponatremia in cirrhosis: pathogenesis, clinical significance, and management. Hepatology. 48, 1002–1010 (2008).
Child, C. G. & Turcotte, J. G. Surgery and portal hypertension. Major Probl. Clin. Surg. 1, 1–85 (1964).
Pugh, R. N., Murray-Lyon, I. M., Dawson, J. L., Pietroni, M. C. & Williams, R. Transection of the oesophagus for bleeding oesophageal varices. Br. J. Surg. 60, 646–649 (1973).
Knox, J. J. Addressing the interplay of liver disease and hepatocellular carcinoma on patient survival: the ALBI scoring model. J. Clin. Oncol. 33, 529–531 (2015).
Shao, L. et al. Albumin-to-bilirubin score for assessing the in-hospital death in cirrhosis. Transl Gastroenterol. Hepatol. 2, 88 (2017).
Duffy, B. K. et al. Usefulness of an elevated neutrophil to lymphocyte ratio in predicting long-term mortality after percutaneous coronary intervention. Am. J. Cardiol. 97, 993–996 (2006).
Qi, X., Wang, C. & Shan, X. Peripheral blood cell ratios as prognostic predictors of mortality in patients with Hepatitis B Virus-related decompensated cirrhosis. Clin. Lab. 67, 12 (2021).
Malinchoc, M. et al. A model to predict poor survival in patients undergoing transjugular intrahepatic portosystemic shunts. Hepatology. 31, 864–871 (2000).
Wiesner, R. et al. Model for end-stage liver disease (MELD) and allocation of donor livers. Gastroenterology. 124, 91–96 (2003).
Kim, W. R. et al. MELD 3.0: The Model for End-Stage Liver Disease Updated for the Modern Era. Gastroenterology ; 161: 1887-95 e4. (2021).
Thuluvath, P. J., Alukal, J. J. & Zhang, T. A model to predict inhospital mortality in patients with cirrhosis, ascites and hyponatremia. Eur. J. Gastroenterol. Hepatol. 34, 591–597 (2022).
Sarin, S. K. et al. Acute-on-chronic liver failure: consensus recommendations of the Asian Pacific association for the study of the liver (APASL): an update. Hepatol. Int. 13, 353–390 (2019).
Moreau, R. et al. Acute-on-chronic liver failure is a distinct syndrome that develops in patients with acute decompensation of cirrhosis. Gastroenterology. 144, 1426–1437 (2013).
Bajaj, J. S. et al. Survival in infection-related acute-on-chronic liver failure is defined by extrahepatic organ failures. Hepatology. 60, 250–256 (2014).
Mochida, S. et al. Proposed diagnostic criteria for acute-on-chronic liver failure in Japan. Hepatol. Res. 48, 219–224 (2018).
Liver Failure and Artificial Liver Group, Chinese Society of Infectious Diseases, Chinese Medical Association. Severe liver Disease and Artificial Liver Group, Chinese Society of Hepatology, Chinese Medical Association. Guideline for diagnosis and treatment of liver failure(2018). J. Clin. Hepatol. 35, 38–44 (2019).
Amato, F. et al. Artificial neural networks in medical diagnosis. J. Appl. Biomed. 11, 47–58 (2013).
Acknowledgements
We are indebted to our study team for establishing and updating our retrospective database, including Junna Dai, Cuihong Zhu, Yun Li, Ying Peng, Zheng Ning, Feifei Hou, Jiancheng Zhao, Han Deng, Ran Wang, Jing Li, Xintong Zhang, Dan Han, Tingxue Song, Zhong Peng, Wenchun Bao, Yingying Li, and Yang An, of whom all had worked for our study group.
Funding
This paper was partially funded by the Outstanding Youth Foundation of Liaoning Province [2022-YQ-07].
Author information
Authors and Affiliations
Contributions
Conceptualization: XQ and GC Methodology: ZB and XQ. Formal analysis: ZB, YY, and XQ. Data curation: ZB, YY, WX, and XQ. Writing-original draft: ZB and XQ. Writing-review and editing: ZB, YY, WX, GC, and XQ. Supervision: XQ and GC. All the listed authors meaningfully participated in the study and they have seen and approved the final manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Bai, Z., Yin, Y., Xu, W. et al. Predictive model of in-hospital mortality in liver cirrhosis patients with hyponatremia: an artificial neural network approach. Sci Rep 14, 28719 (2024). https://doi.org/10.1038/s41598-024-73256-4
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-024-73256-4
Keywords
This article is cited by
-
The EZ-ALBI and PALBI scores contribute to the clinical application of ALBI in predicting postoperative recurrence of HCC
Scientific Reports (2025)
-
28-day all-cause mortality in patients with alcoholic cirrhosis: a machine learning prediction model based on the MIMIC-IV
Clinical and Experimental Medicine (2025)