Abstract
The prognosis of extensive-stage small cell lung cancer is usually poor. In this study, a combined model based on pre-treatment CT radiomics and clinical features was constructed to predict the OS of extensive-stage small cell lung cancer after chemotherapy with immunotherapy.Clinical data of 111 patients with extensive stage small-cell lung cancer who received first-line immunotherapy combined with chemotherapy in our hospital from December 2019 to December 2021 were retrospectively collected. Finally, 93 patients were selected for inclusion in the study, and CT images were obtained through PACS system before treatment. All patients were randomly divided into a training set (n = 66) and a validation set (n = 27). Images were imported into ITK-SNAP to outline areas of interest, and Python software was used to extract radiomics features. A total of 1781 radiomics features were extracted from each patient’s images. The feature dimensions were reduced by MRMR and LASSO methods, and the radiomics features with the greatest predictive value were screened. The weight coefficient of radiomics features was calculated, and the linear combination of the feature parameters and the weight coefficient was used to calculate Radscore. Univariate cox regression analysis was used to screen out the factors significantly associated with prognosis from the radiomics and clinical features, and multivariate cox regression analysis was performed to establish the prognosis prediction model of extensive stage small cell lung cancer. The degree of metastases was selected as a significant clinical prognostic factor by univariate cox regression analysis. Seven radiomics features with significance were selected by LASSO-COX regression analysis, and the Radscore was calculated according to the coefficient of the radiomics features. An alignment diagram survival prediction model was constructed by combining Radscore with the number of metastatic lesions. The study population was stratified into those who survived less than 11 months, and those with a greater than 11 month survival. The C-index was 0.722 (se = 0.044) and 0.68(se = 0.074) in the training and the validation sets, respectively. The Log_rank test results of the combination model were as follows: training set: p < 0.0001, validation set: p = 0.00042. In this study, a combined model based on radiomics and clinical features could predict OS in patients with extensive stage small cell lung cancer after chemotherapy with immunotherapy, which could help guide clinical treatment strategies.
Similar content being viewed by others
Introduction
Small cell lung cancer (SCLC) is an aggressive neuroendocrine malignancy, accounting for 10-15% of all lung cancers, and is commonly associated with smoking1. Most patients present in an extensive stage with multiple metastases at the time of diagnosis, and the prognosis is often extremely poor, with a 5-year survival rate of only 6.3%2. The TNM staging according to the AJCC guidelines was used to guide clinical decision-making and prognosis evaluation. However, a previous study3 had shown that the applicability of TNM staging to assess extensive-stage small-cell lung cancer (ES-SCLC) prognosis in clinical practice remains uncertain. Therefore, it is necessary to search for more reliable biomarkers to predict the prognosis of ES-SCLC, to accurately guide clinical treatment.
In recent years, there have been many applied studies on efficacy prediction and the prognosis assessment of radiomics. Many studies4,5,6,7,8,9,10 have shown that radiomics has a good application value in predicting immunotherapy efficacy with different types of cancer. Yet, the number of studies describing using radiomics to predict the outcomes of SCLC are few. Herein, a machine learning algorithm was performed on pre-treatment CT images to extract radiomics features. These results were then combined with clinical features to construct a combined model to predict the prognosis of patients with ES-SCLC after first-line chemotherapy with immunotherapy.
Materials and methods
Patients
A total of 111 patients with initial ES-SCLC in our hospital from December 2019 to December 2021 were retrospectively collected. The experiment was approved by the Ethics Committee of Zhejiang Cancer Hospital (Approval Number: IRB-2022-445, Approval Date: August 9, 2022) and was conducted in accordance with the Declaration of Helsinki and the national legislation and the institutional requirements, all the participants provided written informed consent before participating in the study. The clinical characteristics of the patients collected in this study included: gender, age, clinical TNM stage, smoking history, basic disease, location of the lesion, metastatic organs, count of metastatic lesions, and so on. The count of metastatic lesions(transfer-ct.) was referred to the total number of single-organ metastases or multi-organ metastases lesions, using the concept of oligo-metastasis in the guidelines of the American Society of Radiation Oncology (ASTRO) and the European Society of Radiation Oncology (ESTRO)11 for reference, so groups were divided by whether they had more than, or less than/equal to 5 metastatic lesions.
Inclusion criteria: (1) patients were pathologically or cytologically diagnosed with ES-SCLC; (2) received at least 2 doses of chemoimmunotherapy as first line treatment therapy; (3) CT images before treatment; (4) With follow-up. Exclusion criteria: (1) received any other anti-tumor therapy before immunotherapy; (2) No CT images before treatment; (3) patients without follow up data. Finally, a total of 93 cases were included in this study. The immunotherapy regimen was durvalumab or atezolizumab. And the chemotherapy regimen was platinum etoposide.
CT scanning protocol
The CT scanning equipment used in our hospital included GE, Siemens and Philips models; Tube voltage was120KV, tube current: GE、Siemens、Philips: automatic exposure control (AEC); Collimation: GE and Philips: 64 × 0.625MM; Siemens: 64 × 0.6MM; Matrix: GE and Siemens: 512 × 512; Philips: 1024 × 1024; Pitch: GE:1.375P; Siemens: 1.2P; Philips: 1.0P; Tube speed: GE:0.4s; Siemens and Philips: 0.5s. During the examination, the patient was supine, and the scan was conducted during the end of deep inspiration. The visual range ranged from the thoracic entrance to the level below the costophrenic angle. The image was reconstructed with 5 mm layer thickness and 5 mm layer interval. Prior to scanning, 85 ml-95 ml of CT contrast agent was injected into the cubital vein with a high-pressure syringe at a rate of 3.0 ml/s. The scans were performed 60 s after the injection, and the scans were performed in one phase.
Segmentation of the tumor
The chest CT images were imported into ITK-SNAP (version 3.8.0, USA)12. The window position used was the enhanced mediastinal window, which could show a good boundary between the lesion and the surrounding large blood vessels, obstructive atelectasis or obstructive pneumonia. An outline was made along the inner boundary of the tumor focus, and the entire tumor focus was outlined to obtain the 3D-ROI. The delineation of ROI was carried out by two cardiothoracic diagnostic physicians alone with more than 5 years of experience blinded to the outcome of study patients. In the delineation process, the trachea, vasculature, distal obstructive atelectasis and regions of pneumonia were avoided.
Feature extraction and screening
python (3.7) software was used to extract features based on Pyradiomics conforming to IBSI standard13. The steps of feature screening were as follows: (1) Outliers in data are replaced by median values; (2) Conducted Z-Score standardization for each characteristic data value. For feature dimension reduction, MRMR and least absolute shrinkage and selection operator (LASSO) method were used to select the most predictive radiomics features. The LASSO-cox regression analysis model was used to construct the radiomics survival analysis model, and the Radscore was calculated.
Data analysis and model construction
All patients were randomly divided into the training set and validation set according to the ratio 7:3. Univariate cox regression was used to select significant predictors of radiomics characteristics (P < 0.05). Correlation analysis was performed among features.
Univariate cox regression analysis model was used to screen out statistically significant predictors of clinical features (p < 0.05). Multiple Cox regression analyses were performed to the construct the combined model based on the features retained by Radscore and clinical features.
Statistical analysis
SPSS 26.0 was used for statistical analysis, and classified data were compared by Chi-square test or Fisher exact probability method. Quantitative data were first tested for normality. Quantitative data with a normal distribution were represented by mean ± standard deviation and compared by an independent sample t-test. Quantitative data without the normal distribution were compared by Mann-Whitney U test, p < 0.05 indicated statistically significant differences.
Model verification: C-index and KM curve (Log-Rank) were used for survival analysis. The research process was shown in Fig. 1.
Results
Clinical features
A total of 93 cases were included, which were divided into training set (n = 66) and validation set (n = 27). Two groups with an average age of 64.1 (±6.9) and 62.7 (±6.8) were obtained. There were 62 males (93.9%) and 4 females (6.1%) in the training set. There were 26 males (96.3%) and 1 female (3.7%) in the validation set. There were no statistically significant differences in clinical features between the training and validation sets (p > 0.05) (Table 1). In addition, univariate cox regression analysis showed that the risk ratio of the number of metastatic lesions in the training set was 2.43 (p = 0.007), which was statistically correlated with OS.
Feature selection and radscore calculation
Python software (3.7) was used to screen 1781 radiomics features extracted from CT enhanced mediastinal window sequences by LASSO-COX regression analysis, and 7 features were finally screened.
exponential_gldm_LargeDependenceHighGrayLevelEmphasis,
wavelet.LLH_firstorder_90Percentile,
exponential_gldm_SmallDependenceHighGrayLevelEmphasis,
wavelet.HHH_glrlm_LowGrayLevelRunEmphasis,
wavelet.HHH_firstorder_Median,
original_gldm_LargeDependenceHighGrayLevelEmphasis,
wavelet.HHH_glcm_Correlation.
The corresponding radscore was calculated according to the coefficients of the radiomics features.
Clinical predictors
Univariate cox regression analysis showed that the count of metastatic lesions(transfer-ct.) was an independent prognostic factor (p < 0.05), those with more than 5 metastatic lesions were considered a high-risk group, while those with less than 5 fit into the low-risk group. A multivariate cox regression analysis showed that the Radscore was an independent risk factor (Table 2).
Establishment of a combined model with radiomics and clinical features
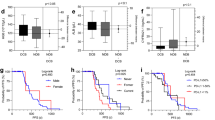
An alignment Diagram model was constructed based on Radscore and the number of metastatic lesions (Fig. 2). The KM curve (Fig. 3) and the Log-rank test showed that the combined model based on radiomics and clinical features could predict the OS of ES-SCLC after first-line chemotherapy and immunotherapy in both the training set (p < 0.0001) and the validation set (p = 0.00042). The C-index of the line graph model was 0.722 (se = 0.044) in the training set and 0.68 (se = 0.074) in the validation set, respectively.
Combined nomogram. A nomogram based on a survival combined model. The nomogram showed the clinical application of the survival combined model. Red highlights showed an example (N0.2 patient in validation set), the point of Met-num is 55, and point of Radscore is 89, so the total point of the patient was 144 and the probability of OS less than 11 months was 0.977, The actual OS of this patient is 4 months. Met-num: metastasis lesions number; Met-num: 0 = Met-num ≤ 5; Met-num:1 = Met-num> 5.
survival status The Figure showed the Kaplan-Meier curves of two risk groups in the training set (a) and validation set (b).
Discussion
In this study, a combined model based on the Radscore selected from radiomics features and the number of metastatic lesions selected from clinical features was constructed to predict the OS for ES-SCLC after chemotherapy and immunotherapy. The test results of the C-index and the KM curve were satisfactory, which indicated that the model established in this study can objectively and accurately stratify the survival of ES-SCLC patients.
Chen et al.14 reported on the value of radiomics for progression-free survival prediction for ES-SCLC after chemotherapy with etoposide and cisplatin. 5 and 6 radiomics features were extracted from the lung window and the enhanced mediastinal window, respectively. The C-index of the model constructed with 11 features was 0.7531 and the average C/D AUC was 0.8487 in the validation set, which was greater than that of the model constructed with lung window (C-index 0.6951, the mean C/D AUC was 0.7836) and enhanced mediastinal window (C-index was 0.7192, the mean C/D AUC was 0.7964). Another study15 used radiomics to predict the efficacy of platinum-based chemotherapy and found an OS of 153 SCLC patients with lung window feature modeling. The results showed that the radiomics risk score was correlated with the OS, with a C-index of 0.72 in training set and 0.69 in validation set. The results of the two studies showed that the effectiveness of the lung window feature model was essentially identical, and the C-index of the validation set was 0.69. In this study, the radiomics features of the enhanced mediastinal window were extracted, and the C-index of the combined model established with the clinical features was 0.722 in the training set and 0.68 in the validation set, respectively. These results were similar to what was reported above.
Chen et al. showed that the predictive value of the model constructed by the radiomics features of different window positions was higher than that of the radiomics model with a single window position. According to the correlation study between pathological and radiomics features16,17, plain CT images reflected the uneven tissue and cell density caused by necrosis, bleeding, and degeneration inside the tumor. The enriched and deficient blood supply areas in the enhanced scan images reflect the heterogeneity of blood supply vessels in the tumor. The heterogeneity within the tumor was translated into a quantitative expression of radiomics pixel density and distribution characteristics. Therefore, the radiomics features acquired from both the plain sequence and the enhanced sequence is greater than that of a single sequence, and the combination of multiple sequences was needed for disease diagnosis in practice. It was suggested that the multi-sequence radiomics features can be extracted for modeling in subsequent studies, and the differences with the single sequence model can be analyzed statistically.
This study was the first to use radiomics to predict the prognosis for SCLC after first-line chemotherapy plus immunotherapy, which is unique from previous radiomics studies which predicted prognosis after chemotherapy was used alone. The C-index of this model was noted to be slightly different from other models examining only conventional chemotherapy.
There was also a limited amount of literature that reports on the application of radiomics in predicting the efficacy of immunotherapy. The study of melanoma indicated that radiomics has a certain value in predicting the efficacy of first-line immunotherapy18. By combining radiomics and clinical characteristics, a more realistic prognostic prediction model was constructed to accurately predict the survival rate of patients with ES-SCLC after first-line immunotherapy plus chemotherapy.
The first staging criteria for small cell lung cancer (SCLC) was cited in randomized clinical trials by the Veterans Administration Lung Study Group (VALSG) in the 1950s. This approach divides SCLC into two categories, called limited disease (LD) and extensive disease (ED). There is no clear evidence of correlation between TNM stage and prognosis of small cell lung cancer。The patients included in this study were in an extensive stage, consistent with the overall TNM stage. However, only cT, cN and cM parameters could not reflect the difference in prognosis of the whole population, which was also verified by our results. A relevant clinical feature of this study is the number of distant metastases, which reflects the correlation between subgroup stages in the M stage and prognosis.
Regarding the correlation between diabetes and hypertension with the prognosis of small cell lung cancer, it has been reported19 that hypertension and type 2 diabetes may be related to the poor prognosis of patients with extensive small cell lung cancer, but it is not certain and there is not enough literature support. The results of this study show that hypertension and diabetes are not independent prognostic factors of patients with extensive small cell lung cancer. On the one hand, it may be that the proportion of patients with/without hypertension and diabetes is not equal, which cannot be accurately analyzed; on the other hand, it may be that hypertension and diabetes are not independent prognostic factors in patients with extensive small cell lung cancer. Therefore, this issue needs further study.
In this study, the patients were divided into two subgroups based on those which had more than, or less than 5 metastatic lesions. The greater the number of metastatic lesions, the worse the prognosis, which was consistent with clinical practice.
This study did not find any evidence to suggest that smoking is an independent risk factor for OS in SCLC. In addition, the imbalance of male to female participants in this study may add a confounding variable. However, it has been well-documented that brain metastasis is a poor prognostic indicator20. Prophylactic whole brain irradiation (PCI) has been shown to reduce the incidence of brain metastasis and improve prognosis. In this study, some patients received PCI independently. As such, brain metastasis had no overall correlation with OS. Several studies21,22 have reported that thoracic radiotherapy could improve the OS of ES-SCLC. Some patients in this study cohort received chest radiotherapy after first-line immunotherapy plus chemotherapy. Therefore, the value of the combined radiomics model in this study may be affected by the treatment strategies that were used.
There are several limitations to this study. Firstly, as a single-center retrospective study, the findings may be subject to selection bias, limiting the generalizability to broader populations. The relatively small sample size may reduce the robustness of the study and its applicability to diverse populations. Finally, the gender ratio of this study biases males due to the demographic of SCLC, a more balanced study may be required to draw definite conclusions.
Conclusion
Herein, this study determined that radiomics information combined with the clinical examination was able to predict the OS of patients objectively and accurately with ES-SCLC after first-line immunotherapy plus chemotherapy. The radiomics nomogram used in this study may help guide treatment strategy by identifying key prognostic indicators for the overall outcome.
Data availability
The original data could be provided on reasonable request from the authors. Requests to access these datasets should be directed to HT, jianght@zjcc.org.cn.
References
Riaz, S. P. et al. Lung Cancer - Small Cell: Statistics [Webpage on the Internet]. Alexandria, VA: American Society of Clinical Oncology (ASCO; Cancer.Net (2019). https://www.cancer.net/cancer-types/lung-cancer-small-cell/ statistics.
Zou, J. et al. Ageing as key factor for distant metastasis patterns and prognosis in patients with extensive-stage small cell Lung Cancer. J. Cancer 12 (6), 1575–1582 (2021).
Arriola, E. et al. Prognostic value of clinical staging according to TNM in patients with SCLC: a real-world surveillance epidemiology and end-results database analysis. JTO Clin. Res. Rep.3 (1), 100266. https://doi.org/10.1016/j.jtocrr.2021.100266 (2021). PMID: 35024640; PMCID: PMC8728577.
Xu, Q. et al. Advanced gastric cancer: CT radiomics prediction and early detection of downstaging with neoadjuvant chemotherapy. EurRadiol.31 (11), 8765–8774 (2021).
Xie, K. et al. Pretreatment contrast-enhanced computed Tomography Radiomics for Prediction of pathological regression following neoadjuvant chemotherapy in locally advanced gastric Cancer: a preliminary Multicenter Study. Front. Oncol.11, 770758 (2021).
Li, Z. Y. et al. Multi-modal radiomics model to predict treatment response to neoadjuvant chemotherapy for locally advanced rectal cancer. World J. Gastroenterol.26 (19), 2388–2402 (2020).
Chen, X. et al. Combining Dynamic contrast-enhanced magnetic resonance imaging and apparent diffusion coefficient maps for a Radiomics Nomogram to predict pathological complete response to neoadjuvant chemotherapy in breast Cancer patients. J. Comput. Assist. Tomogr.44 (2), 275–283 (2020).
Hu, C. et al. Application value of magnetic resonance Radiomics and Clinical nomograms in evaluating the sensitivity of Neoadjuvant Chemotherapy for nasopharyngeal carcinoma. Front. Oncol.11, 740776 (2021).
Trebeschi, S. et al. Predicting response to cancer immunotherapy using noninvasive radiomic biomarkers. Ann. Oncol.30 (6), 998–1004 (2019).
Qu, J. et al. DCE-MRI radiomics nomogram can predict response to neoadjuvant chemotherapy in esophageal cancer. Discov Oncol.13 (1), 3 (2022).
Iyengar, P. et al. Treatment of Oligometastatic Non-small Cell Lung Cancer: an ASTRO/ESTRO clinical practice Guideline. Pract. Radiat. Oncol.13 (5), 393–412 (2023 Sep-Oct). Epub 2023 Apr 25. PMID: 37294262.
Yushkevich, P. A. et al. User-guided 3D active contour segmentation of anatomical structures: significantly improved efficiency and reliability. Neuroimage31 (3), 1116–1128 (2006).
Zwanenburg, A. et al. The image Biomarker Standardization Initiative: standardized quantitative Radiomics for High-Throughput Image-based phenotyping. Radiology 295 (2), 328–338. https://doi.org/10.1148/radiol.2020191145 (2020). Epub 2020 Mar 10. PMID: 32154773; PMCID: PMC7193906.
Chen, N. et al. Progression-free survival prediction in small cell Lung Cancer based on Radiomics analysis of contrast-enhanced CT. Front. Med. (Lausanne) 9, 833283 (2022).
Jain, P. et al. Novel non-invasive Radiomic signature on CT scans predicts response to platinum-based chemotherapy and is Prognostic of overall survival in small cell Lung Cancer. Front. Oncol.11, 744724 (2021).
Ganeshan, B. et al. Non-small cell lung Cancer: histopathologic correlates for texture parameters at CT. Radiology 266 (1), 326–336 (2013).
Sun, J., Yu, X. R., Shi, B. B., Zheng, J. & Wu, J. T. CT features of retroperitoneal solitary fibrous tumor: report of three cases and review of the literature. World J. Surg. Oncol.12, 324 (2014).
Motzer, R. J. et al. Nivolumab versus Everolimus in Advanced Renal-Cell Carcinoma. N Engl. J. Med.373 (19), 1803–1813 (2015).
Xiu, W., Huang, Y., Li, Y., Yu, M. & Gong, Y. Comorbidities and mortality risk among extensive-stage small-cell lung cancer patients in mainland China: impacts of hypertension, type 2 diabetes mellitus, and chronic hepatitis B virus infection. Anticancer Drugs 33 (1), 80–90 (2022). PMID: 34183497; PMCID: PMC8670332.
Wei, L. J. et al. Construction of a nomogram model for predicting 2year survival rate of small cell lung cancer based on more comprehensive variables. Natl. Med. J. China 102 (17), 1283–1289. https://doi.org/10.3760/cma.j.cn112137-20211106-02467 (2022).
ZengH, H. L. et al. Risk factors for neurocognitive decline in lung cancer patients treated with prophylactic cranial irradiation: a systematic review. Cancer Treat. Rev.88, 102025. https://doi.org/10.1016/j.ctrv.2020.102025 (2020).
LiB, J. C. Prognostic value of a nomogram based on the dynamic albumin-to-alkaline phosphatase ratio for patients with extensive-stage small-cell lung cancer. Onco. Targets Ther.13, 9043–9057. https://doi.org/10.2147/OTT.S262084 (2020).
Author information
Authors and Affiliations
Contributions
HT, FW and FM designed the study; FW, WJ, JL, FM and YJ performed the experiments; FW and HT analyzed the data; and FW, HT and FM drafted the manuscript. All authors reviewed the manuscript, approved the final version submitted for publication.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Wang, F., Chen, W., Chen, F. et al. Risk stratification and overall survival prediction in extensive stage small cell lung cancer after chemotherapy with immunotherapy based on CT radiomics. Sci Rep 14, 22659 (2024). https://doi.org/10.1038/s41598-024-73331-w
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-024-73331-w