Abstract
Immune checkpoint inhibitors (ICIs) have become an important treatment modality for various malignancies. Due to excessive inflammatory and immune responses, immune-related adverse events (irAEs), such as rash, pruritis, pneumonitis, hepatitis, hypothyroidism, hyperthyroidism and fatigue, can occur. Acquired immune thrombocytopenia is a rare irAE that can lead to severe low platelet counts and hemorrhage. A retrospective observational analysis was conducted on data from the United States Food and Drug Administration Adverse Event Reporting System (FAERS). We searched for all reports recorded in the FAERS covering the period from 2011 Q1 to 2023 Q4 for target agents. Signal analysis was performed using the reporting odds ratio (ROR), proportional reporting ratio (PRR), Bayesian confidence propagation neural network (BCPNN) and multi-item gamma Poisson shrinker (MGPS) algorithm. Nonparametric tests were used to compare the time to onset of thrombocytopenia-associated fractures between different regimens. There were 404 reports of immune thrombocytopenia in the FAERS database. Immune thrombocytopenia was associated with all ICIs except tremelimumab, and signals were detected by all 4 methods. The median time to the onset of immune thrombocytopenia caused by PD-1, PD-L1 and CTLA-4 inhibitors was 47 days (range: 21-139.2), 21 days (range: 13–131) and 9 days (range: 7–27), respectively. The Weibull shape parameter test revealed that pembrolizumab-, durvalumab-, and ipilimumab-induced thrombocytopenia had a random failure-type profile, and nivolumab- and atezolizumab-induced thrombocytopenia were characterized by an early failure-type profile. There was a significant reporting signal of ICI-induced thrombocytopenia associated with most ICIs. Immune thrombocytopenia has a greater incidence in males, elderly individuals and Asian populations, and PD-1 inhibitors were the most common cause. Clinicians should be aware of the signs of potential serious adverse events.
Similar content being viewed by others
Introduction
Immune checkpoint inhibitors (ICIs) targeting programmed cell death 1/programmed death-ligand 1 (PD-1/PD-L1) and cytotoxic T-lymphocyte-associated protein 4 (CTLA-4) are important treatments for various malignancies1. ICIs inhibit immune checkpoint pathways and stimulate a T-cell-mediated antitumor response, which upregulates the immune system to kill tumor cells2. Because upregulation of the immune system can lead to excessive inflammatory and immune responses, immune-related adverse events (irAEs) are the primary cause of ICI-related toxicity. Common irAEs include rash, pruritus, pneumonitis, hepatitis, hypothyroidism, hyperthyroidism, and fatigue3.
However, hematologic irAEs, including pure red cell aplasia, acquired hemophilia, acquired thrombotic thrombocytopenic purpura, and autoimmune hemolytic anemia, are uncommon4. Acquired immune thrombocytopenia is another rare irAE5. Thrombocytopenia can lead to severe low platelet counts, which can cause hemorrhage, a life-threatening adverse event.
It is important to characterize the incidence of ICI-induced thrombocytopenia in a real-world setting. Spontaneous adverse drug reaction (ADR) reporting databases have been used to detect potential ADRs. The purpose of this study was to characterize reports of ICI-induced thrombocytopenia events and to use several models to analyze the onset profile to determine the reporting signal for ICI-induced thrombocytopenia.
Materials and methods
Data sources
Our study was designed as a retrospective, disproportionality analysis and did not involve human subjects. The data were obtained from the publicly available United States Food and Drug Administration (FDA) Adverse Event Reporting System (FAERS) database. This resource contains identified and anonymized spontaneous adverse event reports submitted to the FDA. The FAERS data were downloaded from https://fis.fda.gov/. According to the FDA approval time of our target drugs, all reports recorded in the FAERS covering the period from 2011 Q1 to 2023 Q4 were included in our analysis.
Adverse events and drug identification
The selected drugs, including nivolumab, pembrolizumab, cemiplimab, atezolizumab, durvalumab, avelumab, ipilimumab, and tremelimumab, were identified in the database using both the generic name and trade name, and the role_cod was set as primary suspected (PS). The incidence of thrombocytopenia associated with the use of ICIs was the primary outcome. The Medical Dictionary for Regulatory Activities (MedDRA) was used to define thrombocytopenia.
Data mining
The database contained seven files: patient demographics (DEMO), drug (DRUG), adverse events (REAC), patient outcomes (OUTC), report source (RPSR), drug therapy date (THER) and drug indication (INDI). Furthermore, the deleted files contained cases that were removed by the FDA or manufacturers for various reasons, including combined cases. All the data were imported into MySQL software (v8.0; Oracle, Sweden), and the deduplication process was performed before statistical analysis. The primary ID served as the primary linking field (primary key) between different files, and the case ID was chosen as the key filter to eliminate duplicate records.
The standardized MedDRA preferred term (PT) was used to classify adverse events (AEs). Notably, a PT can be linked to more than one system organ class (SOC) in MedDRA; therefore, we used MedDRA 24.0 to classify the AEs in each report into the appropriate SOC levels in MySQL 8.0.
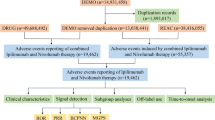
Subsequently, the clinical characteristics of the reported patients were described in detail, including sex, age, weight, indications, outcomes, reporters and country. The serious outcomes included hospitalization, death, life-threatening conditions, disability, other serious outcomes and missing data. In some cases, multiple outcomes were listed, so the total number of outcomes exceeded the total number of reports. A flow diagram of the multistep process is shown in Fig. 1.
Statistical analysis
This analysis was designed to investigate whether there is an association between ICI exposure and thrombocytopenia development in the FAERS database. We used the reporting odds ratio (ROR), proportional reporting ratio (PRR), Bayesian confidence propagation neural network (BCPNN) and multi-item gamma Poisson shrinker (MGPS) algorithm6 to compare reported cases of thrombocytopenia with ICI use with all other reported events in the FAERS to determine if a safety signal was present. Additionally, we used nonparametric tests (the Mann‒Whitney test for dichotomous variables and the Kruskal‒Wallis test when there were > 2 subgroups of respondents) to compare the time to onset of thrombocytopenia-associated fractures between different regimens7.
Selection process for reports of thrombocytopenia associated with ICIs from the Food and Drug Administration Adverse Event Reporting System database.
Results
Patient and reporter characteristics
A total of 379,490 adverse events and 404 reports of thrombocytopenia induced by ICIs were reported in the FAERS database after the exclusion of duplications. Patient and reporter characteristics are listed in Table 1. For thrombocytopenia reports, The reported sex was most commonly male (60.1%), and the age range was 65~ 5 years (51.0%). Most reports originated from Japan (45.0%) and the United States (20.1%). The most common reporters for thrombocytopenia were medical doctors (52.0%), followed by health professionals (16.3%) and other health professionals (12.9%). In our reports, some patients have multiple outcomes, and hospitalization was the most common outcome (28.6%), excluding the category of other serious outcomes. Although 9.3% of the patients died, and 10.7% of the patients have life-threatening condition, if delete duplicate outcomes, 31% of patients are deaths or life-threatening. In general, the number of thrombocytopenia reporters per year seemed to increase over the study period, and the number of reports has remained stable in recent years.
Signal detection for immune thrombocytopenia
There are 404 immune thrombocytopenia was associated with all ICIs except tremelimumab. Signal detection was positive for all 4 methods (Table 2).
Cumulative incidence and time to onset
Among 404 reports of immune thrombocytopenia associated with ICIs, 157 reports contained time-to-onset data. Of these, 147 reports included age, and 154 reports included sex. The Kaplan‒Meier curves are presented in Fig. 2. The cumulative incidence significantly differed between different ages (log-rank test, P < 0.001), whereas it did not significantly differ by sex (log-rank test, P = 0.3).
The results of the Weibull shape parameter (WSP) test for immune thrombocytopenia associated with ICIs are summarized in Table 3, and the time to event onset is presented in Fig. 3. A total of 157 reports described the time of onset. Most cases of immune thrombocytopenia (72 reports) occurred within the first month of drug use. The median time to the onset of immune thrombocytopenia caused by PD-1, PD-L1 and CTLA-4 was 47 days (range: 21-139.2), 21 days (range: 13–131) and 9 days (range: 7–27), respectively. Patients with immune thrombocytopenia caused by PD-1 showed a significantly longer time to onset than did those treated with other immune checkpoint inhibitors.
In the WSP test for immune thrombocytopenia associated with nivolumab and atezolizumab, the upper bound of the 95% CI for the shape parameter was less than 1, indicating an early failure-type profile. In contrast, in the WSP test for immune thrombocytopenia associated with pembrolizumab, durvalumab and ipilimumab, the lower limit of the 95% CI of the shape parameter exceeded 1, suggesting a random failure-type profile. There were no reports of immune thrombocytopenia caused by tremelimumab.
Onset time of immune thrombocytopenia. The times to onset of immune thrombocytopenia between different ages (log-rank test, P < 0.001) and sexes (log-rank test, P = 0.3) were compared using nonparametric tests (Kruskal‒Wallis test).
Time to onset of immune thrombocytopenia.
Discussion
To our knowledge, this is a study to utilize the FAERS pharmacovigilance database to assess the association between ICIs and an increased risk of immune thrombocytopenia, included the patient characteristics, occurrence time irregularity etc. With the widespread use of ICIs, an increasing number of patients have achieved excellent treatment efficacy, but various immune related adverse reactions (irAEs) have also been reported. Rash, pruritis, pneumonitis, hepatitis, hypothyroidism, hyperthyroidism and fatigue are common irAEs8, hematologic irAEs are uncommon9. In particular, immune thrombocytopenia associated with ICIs is a rare irAE but is one of the most serious hematological toxicities among hematological irAEs10, as we can see about one-third of the patients died or have life-threatening condition in our study. An observational study based on registries in three French drug monitoring databases revealed that the incidence of thrombocytopenia associated with ICIs was less than 1%11. Le Burel et al.12 retrospectively reviewed 908 patients with any cancer type treated with ICIs and reported that 0.2% of patients developed ICI-induced thrombocytopenia. Petrelli et al.13 performed a systematic review and meta-analysis of thirteen trials and reported that the incidence of all-grade thrombocytopenia was 2.84% and that of grade 3–5 thrombocytopenia was 1.83%. According to an observational study, immune thrombocytopenia seems to have a greater frequency, accounting for 25–29% of hematological irAEs14,15.
Pharmacovigilance analysis is important to describe events observed for medications after they are put on the market, especially for literature limited to case reports. But there is no universal method that can perfectly identify signals from datas or adverse events. The different statistical methods are all authentic qualitative signal detection tools, but they have own advantages and drawbacks. For example, The ROR can eliminate biases, that is the disadvantages of PRR16. BCPNN and MGPS are more stable for detecting rare safety signals17. Comprehensive use of different statistical methods can improve the specificity and positivity rate of the results.In the present analysis, we observed statistically significant RORs, PRRs, BCPNN, and MGPS results for most ICIs. The results indicate a safety signal and the potential association of ICI-induced thrombocytopenia with these agents. Thrombocytopenia caused by PD-1 inhibitors had a higher score than those of PD-L1 and CTLA-4 inhibitors according to each analysis method, and CTLA-4 inhibitors had the lowest score. Among 404 reports, PD-1 inhibitors made up the highest proportion, followed by PD-L1, which is consistent with the results of our literature search. We searched PubMed using the search terms “thrombocytopenia” and “immune checkpoint inhibitors”, most case reports also involved PD-1 inhibitors like nivolumab and pembrolizumab. The pathophysiologic mechanism of ICI-induced thrombocytopenia is currently unknown18. The possible mechanisms included elevated platelet-associated IgG titers19, CD4 + helper T cells and CD8 + cytotoxic T cells reactivated to damage to hematopoietic stem cells20, abnormal cytokine secretion leads to increased platelet death21 and platelet surface expresses PD-L1 receptor which is a direct target cell of ICIs etc. And the high proportion of thrombocytopenia induced by PD-1 inhibitors is considered to be due to their widespread use. In our study, although tremelimumab did not reportedly cause thrombocytopenia, it is explicitly mentioned in the instructions that monitoring platelet count is still necessary in clinical applications.
It is unclear when immune thrombocytopenia starts to develop. The risk of developing irAEs is usually highest during the first 4 weeks of immunotherapy22. Among the included reports, 157 reported the time to onset of immune thrombocytopenia, and in 72 reports, it occurred within 1 month after administration. In a majority of reported cases, thrombocytopenia occurred as early as the first 1–2 cycles of treatment23. To examine the onset profiles of thrombocytopenia and ICIs, time-to-onset analyses were conducted. According to the Weibull shape parameter test for immune checkpoint inhibitors associated with immune thrombocytopenia, the median time to onset and failure type of ICI-induced thrombocytopenia were significantly different depending on the ICI. Cemiplimab and avelumab do not have WSP test results because there were very few reports involving these ICIs. The WSP test results for atezolizumab, durvalumab, and ipilimumab may be unstable and inaccurate for the report less than 20 cases. The median time to onset of PD-1 inhibitor-induced thrombocytopenia was 47 days (range: 21-139.2), the median time to onset of PD-L1 inhibitor-induced thrombocytopenia was 21 days (range: 13–131), and the median time to onset of CTLA-4 inhibitor-induced thrombocytopenia was 9 days (range: 7–27). The median time to onset for PD-1 inhibitors was similar to that reported by Donald C. M. et al.24 (41–44 days). Therefore, patients receiving different ICIs may develop thrombocytopenia at different times. Ipilimumab exhibited the shortest time to immune thrombocytopenia onset after treatment, which indicates that ipilimumab-induced thrombocytopenia is rare, but if it develops, it can occur quickly. The WSP test also revealed that pembrolizumab-, durvalumab-, and ipilimumab-induced thrombocytopenia had a random failure-type profile, suggesting that thrombocytopenia can occur at any time when using these ICIs. In contrast, nivolumab- and atezolizumab-induced thrombocytopenia was characterized by an early failure-type profile, suggesting that thrombocytopenia can develop in the early stages of treatment, but the likelihood gradually decreases over time. This finding suggests that different ICIs may have different onset profiles. However, considering the long time span of the occurrence of irAEs, full monitoring throughout the entire process is still necessary, even after the end of treatment.
Previous reports have consistently shown that ICI-induced thrombocytopenia is strongly associated with male sex and advanced age5,24. However, in this study, the Kruskal‒Wallis test showed that the cumulative incidence of ICI-induced thrombocytopenia did not significantly differ between patients of different ages, whereas the incidence did significantly differ between males and females. In addition, ICI-induced thrombocytopenia is linked to patient weight and race, with weights of 50 ~ 100 kg and Asian ethnicity being the most commonly reported. Although there are currently no data indicating a correlation between irAEs and race, this phenomenon is still worth examining. Generally, these findings suggest that FAERS analyses provide a fair risk assessment of ICI-induced thrombocytopenia.
ICI-induced thrombocytopenia is a diagnosis of exclusion. Chemotherapy-induced thrombocytopenia is often predictable and cyclical, whereas ICI-induced thrombocytopenia has a different natural history with unpredictable onset and no cyclical pattern. Clinicians must identify the onset characteristics and population characteristics of immune thrombocytopenia in a timely manner because ICI-induced thrombocytopenia has the potential to negatively impact clinical outcomes. According to our analysis of the FAERS database, 9.3% of patients died, and 10.8% developed life-threatening conditions. Similarly, Davis et al.25 reported that 11% of patients with immune thrombocytopenia associated with ICI therapy experienced a fatal outcome, and Donald et al.24 reported that death occurred in 19% of patients with immune thrombocytopenia associated with PD-1/PD-L1 inhibitors.
This study has several limitations. Although real-world data samples were used, only partial population data and partial information were collected. Second, the FAERS is a spontaneous reporting system; therefore, reporting and information bias exist. Third, the reports do not confirm causality of the drug-induced event. More studies are needed to identify the risk factors for immune thrombocytopenia associated with ICIs in the future.
Conclusion
In conclusion, there was a significant reporting signal of ICI-induced thrombocytopenia associated with most ICIs, such as pembrolizumab, durvalumab, ipilimumab, nivolumab and atezolizumab, in the FAERS database. Among them, PD-1 inhibitor-induced immune thrombocytopenia is the most common, and CTLA-4 inhibitor-induced immune thrombocytopenia develops earliest. Most ICI-induced thrombocytopenia occurs in the early stages of drug use, such as within the first month. Serial monitoring for immune thrombocytopenia should be considered in patients receiving ICIs, especially for males, elderly individuals and Asian individuals. Additionally, further research is required to gain a better understanding of the frequency and risk factors associated with ICI therapies. An assessment is necessary to determine the most effective approach for managing thrombocytopenia induced by ICIs.
Data availability
The data were obtained from the publicly available FAERS database. The FAERS data were downloaded from https://fis.fda.gov/.
References
Kumar, V. et al. Current diagnosis and management of immune related adverse events (irAEs) induced by immune checkpoint inhibitor therapy. Front. Pharmacol. 8, 49. https://doi.org/10.3389/fphar.2017.00049 (2017).
Wang, D. Y. et al. Fatal toxic effects associated with immune checkpoint inhibitors: a systematic review and meta-analysis. JAMA Oncol. 4(12), 1721–1728. https://doi.org/10.1001/jamaoncol.2018.3923 (2018).
NCCN Clinical Practice. Guidelines in management of immunotherapy-related toxicities (Version 1.2024). nccn.org.
Moore, D. C. et al. Acquired thrombotic thrombocytopenic purpura associated with immune checkpoint inhibitors: a real-world study of the FDA adverse event reporting system. Int Immunopharmacol. 110, 109015. (2022). https://doi.org/10.1016/j.intimp. 109015 (2022).
Haddad, T. C. et al. Immune checkpoint inhibitor-related thrombocytopenia: incidence, risk factors and effect on survival. Cancer Immunol. Immunother. 71(5), 1157–1165. https://doi.org/10.1007/s00262-021-03068-2 (2022).
Song, Y. et al. Fractures due to aromatase inhibitor therapy for breast cancer: a real-world analysis of FAERS Data in the past 15 years. Oncol. Res. Treat. 43(3), 96–102. https://doi.org/10.1159/000505376 (2020).
Kinoshita, S. et al. Time-to-onset analysis of amiodarone-associated thyroid dysfunction. J. Clin. Pharm. Ther. 45(1), 65–71. https://doi.org/10.1111/jcpt.13024 (2020).
Rogers, B. B., Zawislak, C. & Wong, V. Management of hematologic adverse events associated with immune checkpoint inhibitors. J. Adv. Pract. Oncol. 12(4), 392–404. https://doi.org/10.6004/jadpro.2021.12.4.4 (2021).
Kanbour, A. et al. Pancytopenia and limbic encephalopathy complicating immunotherapy for clear cell endometrial cancer with microsatellite instability-high (MSI-H). Onco Targets Ther. 12, 9965–9973. https://doi.org/10.2147/OTT.S223616 (2019).
Amanam, I. et al. Immune thrombocytopenia after immune checkpoint inhibitor therapy. Br. J. Haematol. 193(3), 677–681. https://doi.org/10.1111/bjh.17387 (2021).
Delanoy, N. et al. Haematological immune-related adverse events Induced by anti-pd-1 or anti-Pd-L1 immunotherapy: a descriptive observational study. Lancet Haematol. 6(1), e48–57. https://doi.org/10.1016/S2352-3026(18)30175-3 (2019).
Le Burel, S. et al. Prevalence of immune-related systemic adverse events in patients treated with anti-programmed cell death 1/anti-programmed cell death-ligand 1 agents: a single-centre pharmacovigilance database analysis. Eur. J. Cancer. 82, 34–44. https://doi.org/10.1016/j.ejca.2017.05.032 (2017).
Petrelli, F. et al. Haematological toxicities with immunotherapy in patients with cancer: a systematic review and meta-analysis. Eur. J. Cancer. 103, 7–16. https://doi.org/10.1016/j.ejca.2018.07.129 (2018).
Omar, N. E. et al. Diagnosis and management of hematological adverse events induced by immune checkpoint inhibitors: a systematic review. Front. Immunol. 11, 1354. https://doi.org/10.3389/fimmu.2020.01354 (2020).
Wang, X., NaNa, H. & Lejie, C. Immune thrombocytopenia induced by immune checkpoint inhibitrs in lung cancer: case report and literature review. Front. Immunol. 12, 790051. https://doi.org/10.3389/fimmu.2021.790051 (2021).
Rothman, K. J., Lanes, S. & Sacks, S. T. The reporting odds ratio and its advantages over the proportional reporting ratio. Pharmacoepidemiol Drug Saf. 13(8), 519–523. https://doi.org/10.1002/pds.1001 (2004).
Dumouchel, W. Bayesian data mining in large frequency tables, with an application to the FDA spontaneous reporting system. Am. Stat. 53 (3), 177–190 (1999).
Harada, S. et al. A case of successful treatment with eltrombopag for severe immune-related thrombocytopenia induced by atezolizumab: case report. J. Med. Invest. 70(3.4), 516–520. https://doi.org/10.2152/jmi.70.516 (2023).
Mori, H. et al. Immune thrombocytopenia induced by nivolumab in a patient with non-small cell lung cancer. Respir Med. Case Rep. 28, 100871. https://doi.org/10.1016/j.rmcr.2019.100871 (2019).
Quirk, S. K., Shure, A. K. & Agrawal, D. K. Immune-mediated adverse events of anticytotoxic T lymphocyte-associated antigen 4 antibody therapy in metastatic melanoma. Transl Res. 166(5), 412–424. https://doi.org/10.1016/j.trsl.2015.06.005 (2015).
Nelson, D. et al. Thrombotic thrombocytopenic purpura associated with pembrolizumab. J. Oncol. Pharm. Pract. 28(4), 979–982. https://doi.org/10.1177/1078155221 (2022).
Kanjanapan, Y. et al. Delayed immune-related adverse events in assessment for dose-limiting toxicity in early phase immunotherapy trials. Eur. J. Cancer. 107, 1–7. https://doi.org/10.1016/j.ejca.2018.10.017 (2019).
Jotatsu, T. et al. Immune-mediated thrombocytopenia and hypothyroidism in a lung cancer patient treated with nivolumab. Immunotherapy. 10(2), 85–91. https://doi.org/10.2217/imt-2017-0100 (2018).
Moore, D. C. et al. PD-1/PD-L1 inhibitor-induced immune thrombocytopenia: a pharmacovigilance study and systematic review. Int. Immunopharmacol. 129, 111606. https://doi.org/10.1016/j.intimp.2024.111606 (2024).
Davis, E. J. et al. Hematologic complications of immune checkpoint inhibitors. Oncologist. 24(5), 584–588. https://doi.org/10.1634/theoncologist.2018-0574 (2019).
Funding
This research funded in part by Wu Jieping Medical Foundation (No. 320.6750.2023-6-27).
Author information
Authors and Affiliations
Contributions
D.M. and X.Y. performed data analysis and wrote the main manuscript text. G.X. and X.Z. performed data analysis. K.W. prepared Figs. 1, 2 and 3. L.L. prepared Tables 1, 2 and 3 and X.P. designed the study and revised the manuscript. All authors reviewed and approved the final manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethical approval
Not applicable. This article does not involve animal or human experiments.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Mo, D., Yang, X., Xu, G. et al. Signal detection of immune thrombocytopenia associated with immune checkpoint inhibitors. Sci Rep 14, 23699 (2024). https://doi.org/10.1038/s41598-024-75271-x
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-024-75271-x
Keywords
This article is cited by
-
Safety of immune checkpoint inhibitors: A systematic review of disproportionality analysis studies
European Journal of Clinical Pharmacology (2026)
-
Uncovering prognostic biomarkers through a pharmacovigilance study: the case of RDW
Clinical and Translational Oncology (2025)
-
Cemiplimab
Reactions Weekly (2025)