Abstract
Background: Neoadjuvant chemoradiotherapy combined with immunotherapy (NICRT) is a new neoadjuvant treatment approach that has raised concerns regarding potential challenges in surgery and postoperative complications. This study aimed to compare the efficacy and safety of neoadjuvant chemoradiotherapy (NCRT) and NICRT for the treatment of resectable locally advanced esophageal squamous cell carcinoma (ESCC). Methods: We retrospectively analyzed 291 patients with locally advanced ESCC who underwent neoadjuvant therapy and esophagectomy at three centers between January 2019 and September 2023 and added the data from PALACE-1 for the analysis, Among these patients, 248 and 61 patients received NCRT and NICRT, respectively. Propensity score matching (PSM) was used to balance the potential bias. Results: After the PSM,the rate of a pathological complete response (pCR) in the NCRT group was not significantly different from that in the NICRT group (46.90% vs 36.36%, P = 0.180). There were no significant differences in the tumor regression grade (TRG) and positive lymph node pCR rates between the two groups (P = 0.233 and P = 0.354, respectively). Treatment-related toxicities and postoperative complications were not significantly different between the NCRT group and the NICRT group (P = 0.199, P = 0.284). Conclusion: Compared with NCRT, NICRT did not lead to the better treatment efficacy. There were no significant differences was observed in the incidence of treatment-related toxicities and postoperative complications.
Similar content being viewed by others
Introduction
Neoadjuvant therapy preceding radical surgery has become the established standard care for patients diagnosed with locally advanced esophageal squamous cell carcinoma (ESCC)1. However, the NEOCRTEC5010 randomized clinical trial indicated that the overall 5-year recurrence rate of ESCC after neoadjuvant chemoradiotherapy (NCRT) treatment exceeds 30%2. Therefore, there is an urgent need to explore novel neoadjuvant treatment models in clinical settings.
The integration of immunotherapy into clinical practice has led to the recommendation of utilizing immune checkpoint inhibitors, such as anti-PD-1/PD-L1, in conjunction with radiotherapy and chemotherapy as the primary therapeutic approach for locally advanced and advanced esophageal cancer3,4,5,6. Recently, several clinical trials have incorporated immunotherapy into perioperative care. Various studies have investigated the efficacy and safety of combining neoadjuvant concurrent chemoradiotherapy with immunotherapy (NICRT) for locally advanced esophageal cancer, demonstrating a potentially enhanced antitumor effect. In the PALACE-1
study7 involving patients with ESCC, pembrolizumab was administered following the CROSS chemoradiotherapy regimen. Of the18 patients who underwent radical surgery, 17 achieved R0 resection, with 10 (55.6%) cases achieving pathological complete response (pCR) in the primary tumor lesions and lymph nodes, and 16 (89%) cases achieving major pathological response (MPR). In the PERFECT study8, 85% of 40 patients completed all cycles of atezolizumab, with a pathological complete response rate of 25% (10/40). NCT028440759demonstrates the effectiveness of pembrolizumab in combination with radiotherapy and chemotherapy for esophageal cancer. The trial included 28 patients, 26 of whom underwent surgery; a pathological complete response rate of approximately 46% was achieved.
NICRT has demonstrated good efficacy and acceptable toxicity as a neoadjuvant treatment. However, there are currently no studies that have directly compared it with traditional NCRT. As a result, the treatment responses,Treatment-related toxicities and postoperative complications associated with NICRT in comparison to traditional NCRT remain unclear and require further investigation.Therefore, we conducted a study involving over 300 patients with locally advanced ESCC who underwent neoadjuvant therapy and radical surgery in three hospitals over the past 5 years to investigate the real-world impact of NCRT and NICRT, and we added the data from PALACE-1 for the analysis,focusing on short-term efficacy and safety.
Methods
Patients
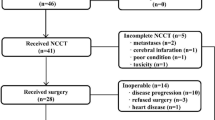
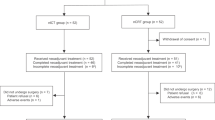
This retrospective study included patients diagnosed with ESCC who underwent neoadjuvant therapy and radical esophagectomy in three hospitals between January 2019 and September 2023. The inclusion criteria were patients aged 18–75 years with pathologically confirmed thoracic ESSC, clinical stage T1N + or T2-4aNany (American Joint Committee on Cancer (AJCC) TNM classification, 8th edition). The exclusion criteria included patients with other tumors, those who did not complete the standard neoadjuvant treatment, and those with incomplete treatment information. To increase the number of patients with NICRT, we included data from PALACE-1 for analysis.A total of 309 patients met the inclusion criteria, including 248 and 61 in the NCRT and NICRT groups, respectively (Fig. 1).
Flow chart of study design. ESCC esophageal squamous cell cancer, NCRT neoadjuvant chemoradiotherapy, NICRT neoadjuvant chemoradiotherapy combined with immunotherapy, PSM propensity score matching.
Neoadjuvant and surgical treatment options
Patients who underwent preoperative radiotherapy received radiation at doses ranging from 30 to 41.4 Gy(mean plus or minus standard deviation).The radiotherapy technique utilized was intensity modulated radiotherapy (IMRT). All patients were positioned under a large-aperture computed tomography (CT) system while in the supine position, with their hands placed on either side of their body. A large mask was then fixed in place, followed by the positioning of laser light and the performance of an enhanced CT scan. Finally, the CT positioning images were transmitted to the planning system. Each patient was positioned by a radiotherapist and a radiotherapy physiotherapist, with the target area primarily determined by the region affected by the lesion.The preoperative chemotherapy regimen consisted of platinum-based drugs in combination with albumin-paclitaxel (TP regimen), with the chemotherapy dose adjusted as needed based on individual patient tolerance. Additionally, the preoperative immunotherapy regimen involved the intravenous administration of various PD-1 (Programmed cell death-1) . All patients included in this study were deemed suitable candidates for radical esophagectomy. Most patients underwent esophagectomy and/or lymph node dissection under general anesthesia, typically 4–8 weeks following completion of the last neoadjuvant treatment.Surgery was performed using either thoracotomy or minimally invasive esophagectomy, which involved two or three incisions, with bifield lymph node dissection established as the standard of care. In cases with suspicious positive lymph nodes, a three-field lymph node dissection was conducted. The esophagus was reconstructed using a gastric tube, followed by either manual or mechanical cervical anastomosis.
Pathological examination
Pathological TNM staging was conducted in accordance with the AJCC/Union of International Cancer Control (UICC) Staging System, version 8. The pathological samples from each patient were assessed by two experienced pathologists. R0 resection signified curative resection with all-negative margins, whereas the pathological complete response (pCR) indicated the absence of viable tumor cells in both the primary tumor area and resected lymph nodes. The tumor regression grade (TRG) was assessed based on the College of American Pathologists (CAP)/AJCC criteria for pathological assessment following neoadjuvant therapy, serving as a measure of the tumor response to preoperative treatment1.
Postoperative complications
Safety and tolerability were assessed through the evaluation of adverse events (AEs), including serious AEs (grade 3 or higher), in accordance with the NCI Common Terminology Criteria for Adverse Events, version 5.0 (NCI-CTCAE v5.0). Additionally, immune-related adverse events (irAEs) were evaluated based on peer-reviewed management guidelines for irAEs10.Documenting all complications and adverse events occur-ring within 30 days of surgery or during the in-hospital stay after esophagectomy.Pulmonary complications include pneumonia treated with antibiotics on the ward,pleural effusion requiring additional drainage procedure,pneumothorax requiring treatment,Anastomotic Leak defined as full thickness GI defect involving esophagus, anastomosis, staple line, or conduit irrespective of presentation or method of identificatio; Cardiac complication include dysrhythmia atrial requiring treatment ,dysrhythmia ventricular requiring treatment congestive heart failure requiring treatment ,pericarditis requiring treatment11,12.
Statistical analysis
Propensity score matching (PSM) was used to create a well-balanced cohort by considering various explanatory factors. The analysis was conducted using R language (version 4.2.3) to perform 1:3 nearest neighbor matching with a caliper of 0.02 between the NICRT and NCRT groups, while adjusting for potential confounding variables.A logistic regression model was used to calculate the propensity score, which included factors such as age, sex, smoking, alcohol consumption, comorbidities, Karnofsky Performance Status (KPS) score, tumor location, and clinical stage. Categorical variables were presented as total numbers and percentages, and group comparisons were conducted using the chi-square test and Fisher’s exact test. Statistical analysis was performed using SPSS for Windows (version 27.0).
Results
Baseline patient characteristics
A total of 309 patients were included in this study, with 248 and 61 patients in the NCRT and NICRT groups, respectively. The baseline characteristics are shown in Table 1. To reduce confounding factors, we conducted 1:3 PSM with a caliper of 0.02. After matching, 145 and 55 patients were assigned to the NCRT and NICRT groups, respectively. Following PSM, there were no significant differences in the characteristics between the two groups (P > 0.05).
Comparison of treatment efficacy and perioperative outcomes between NCRT and NICRT
As shown in Table 2,After the PSM, all patients underwent R0 resection. The pCR was 46.90% in the NCRT group compared to 36.36% in the NICRT group (P = 0.180). Following neoadjuvant therapy for positive lymph nodes, both the NCRT and NICRT groups exhibited no significant difference, with rates of 71.93% and 64.44%, respectively (P = 0.354). Furthermore, there was no difference in postoperative TNM stage between the two groups. In terms of primary tumor withdrawal, over 60% of patients in both groups achieved major pathological response (MPR), and there was no difference in TRG between the two groups (P = 0.233).The perioperative outcomes are presented in Table 3. No significant differences were observed between the two groups regarding Operation time, intraoperative blood loss, postoperative hospitalization duration, ICU readmission rates, readmission rates, and 90-day mortality rates. However, with respect to lymph node yield, the NCRT group exhibited a lower yield compared to the NICRT group (P < 0.001).
Comparison of treatment-related toxicities and postoperative complications between the NCRT and NICRT groups
Following neoadjuvant therapy, the majority of patients will encounter treatment-related toxicity, predominantly in the form of hematological toxicity. After PSM, no significant difference in toxicity was observed between the two treatment methods (P = 0.199, Table 4).In terms of postoperative complications, there was no significant difference between the NICRT and NCRT groups after matching(40.69% vs 49.09% P = 0.284,Table 4) . The most common pulmonary complications included pulmonary infection, pleural effusion, atelectasis, and pulmonary edema. Anastomotic leakage with no significant difference observed between the two groups.(P = 0.151,P = 0.763,Table 4) cardiac-related complications predominantly included arrhythmia and myocardial infarction and showed no differences between the two groups.(P = 0.084,Table 4).
Discussion
In this study, we conduceted the first multicenter retrospective analysis to compare the treatment efficacy, perioperative outcomes,treatment-related toxicities and postoperative complications associated with two neoadjuvant therapies (NCRT and NICRT) following radical surgery for locally advanced ESCC. After the PSM , the pCR rate in the NCRT group was similar to that in the NICRT group, and the pCR rates for positive lymph nodes and TRG were similar between the two groups. In addition to the lymph node yield, perioperative outcomes,treatment-related toxicities and postoperative complications did not differ significantly between the NICRT and NCRT groups.
NCRT is commonly used to treat ESCC. Notable clinical trials in this field include the CROSS13 and NEOCRTEC5010 trial2. Although the CROSS trial demonstrated promising outcomes, it primarily focused on adenocarcinoma, limiting its applicability to locally advanced ESCC. In contrast, findings from the NEOCRTEC5010 trial revealed a pCR rate of 43.2% with neoadjuvant chemoradiotherapy, which is consistent with our trial results. Patients who achieve pCR after surgery tend to have a favorable prognosis and longer disease-free survival (DFS), emphasizing the importance of striving for higher pCR rates with neoadjuvant treatments14.
Immunotherapy is used in ESCC because of the high frequency of mutations15,16; therefore, it is likely to benefit from checkpoint inhibitors targeting programmed cell death protein 1 (PD-1) or PD-1 ligand 1 (PD-L1)17. Additionally, radiotherapy and chemotherapy can trigger immunogenic cell death in tumor cells, stimulate an antitumor immune response, enhance the expression of tumor cell surface antigens, and release tumor-related antigens18,19,20. The combination of these treatments with immunotherapy may lead to more effective antitumor outcomes.
In a phase IB clinical study (PALACE-1)7 involving patients with ESCC, pembrolizumab was administered following the CROSS chemoradiotherapy regimen. Among the 18 patients who underwent surgery, the postoperative complete response rate was 55.6%. Another study (NCT02844075)9 included 28 patients with esophageal cancer (26 who completed surgery), with a pCR rate of approximately 46% (12/26) and a 50% incidence of adverse reactions (14/28). Our trial reported a lower pCR rate than both studies, possibly because of the smaller number of patients enrolled in those trials and the inclusion of patients with stage Ib esophageal cancer in the NCT02844075 trial.
Many may assume that the combination of the three anti-tumor treatments in the NICRT group would lead to more severe postoperative complications. However, our trial analysis indicated no significant difference in the most common chest complications, cardiac complications, and anastomotic leakage between the NCRT group and the control group. The morbidity rates for anastomotic leakage and cardiac complications were found to be within acceptable ranges for both groups. Notably, the incidence of chest complications in our trial was higher than that reported in related studies21.This increase may be attributed to surgical factors, such as anastomotic leakage and recurrent laryngeal nerve injury, as well as to the primary lung disease,as well as the trauma associated with surgery and anesthesia experienced by the patients. Our high complication rate may also be influenced by our small sample size or the fact that most patients included were at locally advanced stages, resulting in greater surgical trauma. Future studies with larger sample sizes will be necessary to further assess these risks.
Although the application of immunotherapy does not demonstrate a superior pathological response compared to neoadjuvant chemoradiotherapy, it does not necessarily correlate with improved OS.Studies22,23 indicate that various neoadjuvant treatments yield differing rates of pCR while demonstrating similar OS outcomes. While the pCR rate among patients receiving neoadjuvant chemotherapy is lower than that of those undergoing neoadjuvant chemoradiotherapy, there is no significant difference in OS between the two treatment modalities, and neoadjuvant chemotherapy remains a recommended treatment option according to current guidelines1. These observations suggest that pCR attained through systemic therapy may more effectively translate into improved OS for patients compared to pCR achieved via local therapies, such as radiotherapy.The incorporation of immunotherapy into neoadjuvant treatment may enhance systemic anti-tumor efficacy and theoretically lead to improved OS for patients. However, this potential benefit requires validation through follow-up studies and large-scale, multi-center clinical trials.
However, it is important to note that this study has several limitations. Specifically, As a retrospective study, this research may be subject to potential data collection bias. Consequently,PSM was employed to mitigate any effects of such bias. Furthermore, there was insufficient clinical diagnosis of positive lymph nodes, and histological confirmation of each suspicious lymph node was not conducted via endoscopic ultrasound fine needle aspiration (EUS-FNA). the NICRT group had fewer patients and a shorter follow-up period, which prevented us from obtaining long-term results to compare the efficacy of the two treatments. Ma et al.24 examined the effectiveness and safety of anti-PD-1 antibodies in combination with chemoradiotherapy in locally advanced ESCC; the median progression-free survival in the anti-PD-1+CRT group was nearly double that of the CRT group, and the median overall survival was significantly higher in the anti-PD-1+CRT group. However, it is important to acknowledge that the patients in this study did not undergo surgical intervention and the sample size was small. Additionally, variations in chemotherapy and immunotherapy regimens between the groups may have influenced the neoadjuvant efficacy, although this is an inherent challenge.
Conclusion
Patients with ESCC who underwent NICRT did not show higher rates of a pCR, positive lymph node response, or improved TRG of the primary tumor than those who received NCRT. Furthermore, aside from the lymph node yield, no statistically significant differences observed in treatment-related toxicities, perioperative outcomes, or postoperative complication rates between the two groups. These findings suggest that the short-term efficacy and safety profile of NICRT is promising. However, a substantial number of subsequent phase III clinical trials are required to validate these results and to assess long-term survival outcomes.
Data availability
The raw data supporting the conclusions of this article will be made available by the authors, without undue reservation.
References
Ajani, J.A. et al. Esophageal and esophagogastric junction Cancers, version 2.2019, NCCN clinical practice guidelines in oncology. J. Natl. Comprehens. Cancer Netw. 17(7), 855–883 (2019).
Yang, H. et al. Neoadjuvant chemoradiotherapy followed by surgery versus surgery alone for locally advanced squamous cell carcinoma of the esophagus (NEOCRTEC5010): A phase III multicenter, randomized, open-label clinical trial. J. Clin. Oncol. 36(27), 2796–2803 (2018).
Zhang, B. et al. Phase II clinical trial using camrelizumab combined with apatinib and chemotherapy as the first-line treatment of advanced esophageal squamous cell carcinoma. Cancer Commun. 40(12), 711–720 (2020).
Shah, M. A. et al. KEYNOTE-975 study design: A phase III study of definitive chemoradiotherapy plus pembrolizumab in patients with esophageal carcinoma. Future Oncol. 17(10), 1143–1153 (2021).
Zhang, W. et al. Safety and feasibility of radiotherapy plus camrelizumab for locally advanced esophageal squamous cell carcinoma. Oncologist 26(7), e1110–e1124 (2021).
Doki, Y. et al. Nivolumab combination therapy in advanced esophageal squamous-cell carcinoma. N. Engl. J. Med. 386(5), 449–462 (2022).
Li, C. et al. Preoperative pembrolizumab combined with chemoradiotherapy for oesophageal squamous cell carcinoma (PALACE-1). Eur. J. Cancer 144, 232–241 (2021).
van den Ende, T. et al. Neoadjuvant chemoradiotherapy combined with atezolizumab for resectable esophageal adenocarcinoma: A single-arm phase II feasibility trial (PERFECT). Clin. Cancer Res. 27(12), 3351–3359 (2021).
Lee, S. et al. A phase II trial of preoperative chemoradiotherapy and pembrolizumab for locally advanced esophageal squamous cell carcinoma (ESCC). Ann. Oncol. 30 (2019).
Martins, F. et al. Adverse effects of immune-checkpoint inhibitors: Epidemiology, management and surveillance. Nat. Rev. Clin. Oncol. 16(9), 563–580 (2019).
Low, D. E. et al. International consensus on standardization of data collection for complications associated with esophagectomy. Ann. Surg. 262(2), 286–294 (2015).
Dindo, D., Demartines, N. & Clavien, P.-A. Classification of surgical complications. Ann. Surg. 240(2), 205–213 (2004).
Shapiro, J. et al. Neoadjuvant chemoradiotherapy plus surgery versus surgery alone for oesophageal or junctional cancer (CROSS): Long-term results of a randomised controlled trial. Lancet Oncol. 16(9), 1090–1098 (2015).
Wan, T. et al. The prognostic value of a pathologic complete response after neoadjuvant therapy for digestive cancer: Systematic review and meta-analysis of 21 studies. Ann. Surg. Oncol. 26(5), 1412–1420 (2019).
Integrated genomic characterization of oesophageal carcinoma. Nature 541(7636), 169–175 (2017).
Zhang, L. et al. Genomic analyses reveal mutational signatures and frequently altered genes in esophageal squamous cell carcinoma. Am. J. Hum. Genet. 96(4), 597–611 (2015).
Yarchoan, M. et al. Targeting neoantigens to augment antitumour immunity. Nat. Rev. Cancer 17(4), 209–222 (2017).
Brandmaier, A. & Formenti, S. C. The impact of radiation therapy on innate and adaptive tumor immunity. Semin. Radiat. Oncol. 30(2), 139–144 (2020).
Reynders, K. et al. The abscopal effect of local radiotherapy: Using immunotherapy to make a rare event clinically relevant. Cancer Treat. Rev. 41(6), 503–510 (2015).
Krysko, D. V. et al. Immunogenic cell death and DAMPs in cancer therapy. Nat. Rev. Cancer 12(12), 860–875 (2012).
Low, D. E. et al. Benchmarking complications associated with esophagectomy. Ann. Surg. 269(2), 291–298 (2019).
Cools-Lartigue, J. et al. An international cohort study of prognosis associated with pathologically complete response following neoadjuvant chemotherapy versus chemoradiotherapy of surgical treated esophageal adenocarcinoma. Ann. Surg. 276(5), 799–805 (2022).
von Döbeln, G.A. et al. Neoadjuvant chemotherapy versus neoadjuvant chemoradiotherapy for cancer of the esophagus or gastroesophageal junction: long-term results of a randomized clinical trial. Dis. Esophagus 32(2) (2018).
Ma, J. et al. Efficacy and safety of anti-PD-1 antibody plus chemoradiotherapy in locally advanced esophageal squamous cancer. Front. Oncol. 13 (2023).
Author information
Authors and Affiliations
Contributions
All authors made a significant contribution to the work reported, whether that is in the conception, study design,execution, acquisition of data, analysis, and interpretation, or in all these areas; took part in drafting, revising, or critically reviewing the article; gave final approval of the version to be published; have agreed on the journal to which the article has been submitted; and agree to be accountable for all aspects of the work.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethical approval and consent to participate
This study has been approved by the Ethics Committee of Cancer Hospital Affiliated to Shandong First Medical University. Given its retrospective nature, the committee has waived the informed consent requirement for this study.We declare that patients information will be kept confidential and that we adhere to the principles of the Declaration of Helsinki.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Shi, S., Zhou, H., Li, L. et al. Comparison of neoadjuvant chemoradiotherapy versus chemoradiotherapy plus immunotherapy for esophageal squamous cell carcinoma in a real-world multicenter cohort: a propensity score matching study. Sci Rep 14, 24738 (2024). https://doi.org/10.1038/s41598-024-76097-3
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-024-76097-3
Keywords
This article is cited by
-
A nomogram for postoperative pulmonary infections in esophageal cancer patients: a two-center retrospective clinical study
BMC Surgery (2025)
-
Propensity score matching analysis of the association between physical activity and multimorbidity in middle-aged and elderly Chinese
Scientific Reports (2024)