Abstract
Obesity is associated with dysfunctional electrocardiographic and cardiac autonomic parameters, which may lead to increased cardiovascular morbidity. Novel electrocardiographic repolarization markers such as Tpeak-Tend (Tpe) interval have not yet been deeply studied in obese patients. We aimed to investigate the association between ventricular repolarization parameters and heart rate variability (HRV) and how they are affected by changes occurring in the cardiac autonomic nervous system. Ninety subjects categorized by Southeast Asian BMI (kg/m2) standards - normal (18-22.9), overweight (23-24.9), and obese (> 24.9), underwent assessment of ventricular repolarization parameters and HRV. Linear correlation between different parameters was also conducted. Obese subjects exhibited longer QTc and Tpe intervals compared to normal-weight subjects (p-value < 0.001, 0.026 respectively). The QTc interval showed a significant correlation (p < 0.05) with all HRV parameters by linear correlation, while the Tpe interval did not. Anthropometric parameters (BMI, WC, and WHR) were also correlated to both ventricular repolarization variables and HRV. While changes in the QTc interval may be due to obesity and/or autonomic changes occurring in the obese state, the Tpe interval does not show a relation with autonomic parameters. Thus, implicating that a change in the Tpe interval is primarily due to the direct effect of either cardiac or visceral obesity. The observed associations between ECG parameters, obesity indices, and HRV parameters suggest a role for ECG in screening for cardiovascular morbidity.
Similar content being viewed by others
Introduction
Obesity is a well-established major risk factor for cardiovascular morbidity and is often linked to various cardiovascular events1. In Southeast Asian regions, obesity is emerging as a direct risk factor for cardiovascular and other systemic morbidities1. Obesity causes alterations in the structure of cardiac tissue, such as ventricular hypertrophy, left atrial enlargement, and cardiac remodeling at the molecular/cellular and interstitial level2,3. These changes affect the electrical events of depolarization and repolarization during the cardiac cycle, which are then reflected as changes in surface ECG, including prolonged duration of various segments (PR interval and QT interval) and decreased amplitude of the QRS complex. Recently, newer parameters like the Tpeak-to-Tend (Tpe) interval and JT interval are being recognized as better markers for ventricular repolarization4,5. This is because the Tpe interval, the time between the peak and the end of the T wave, has been established as a measure of transmural dispersion of ventricular repolarization6. The prolongation of the Tpe interval may be attributed to the structural effects of obesity on the heart, which profoundly impacts M cells in the myocardium (the mid-myocardial layer of the heart). A prolonged Tpe interval also implies that the myocardium spends more time in repolarization, making it more susceptible to arrhythmogenic stimuli since it is during this phase that the heart is in a state of relative refractory period (Fig. 1).
Section of myocardium showing different layers of the cardiac myocardium (A), ECG characteristics measured in our study, QT interval and Tpe interval (B), Effect of different layers of myocardium on Tpe interval (B).
Obesity is linked to widespread enlargement of fat depots, systemic inflammation, and uncontrolled release of fatty acids (FAs) into the bloodstream7. These characteristics contribute to the development of cardiac adiposity, characterized by an increase in intramyocardial triglyceride content and the expansion of fat volume surrounding the heart and major blood vessels. Moreover, the metabolically active adipose tissue secretes pro-inflammatory and arrhythmogenic adipocytokines, which modulate ionic channel activity and prolong the action potential duration (APD) of the ventricles8.
Abnormalities in ventricular repolarization have been correlated with susceptibility to sudden cardiac death in patients who have recovered from myocardial infarction, patients with QT interval prolongation due to specific drugs, users of liquid protein-modified diets, individuals with idiopathic mitral valve prolapse, and patients afflicted with congenital long-QT syndrome9. These factors also pose risks for various arrhythmogenic states, such as long QT syndrome and Brugada syndrome9. Researchers have also demonstrated that the prolongation of ventricular repolarization is a concerning factor in the origin of life-threatening ventricular arrhythmias in both acquired and congenital conditions. An increased risk of arrhythmia leading to ventricular dysfunction is characterized by a prolonged QT interval, abnormal T-wave configuration, and increased dispersion of the QT interval, generally termed “long QT syndromes” (LQTS)10. It has also been shown that analyzing different components of the QT interval is more useful for recognizing the risk of sudden cardiac death than measuring the QT interval alone in electrocardiographic parameters11.
The autonomic nervous system (ANS) plays a critical role in maintaining cardiovascular homeostasis12. Autonomic nerve discharges (as in physiological conditions) lead to the local release of neurotransmitters that shorten the refractory period, thereby increasing repolarization13. Autonomic changes have a direct effect on the ventricular myocardium and therefore, impact the duration of cardiac repolarization14. The Framingham Offspring cohort revealed that individuals with abnormal heart rate variability (HRV), an indicator of sympathetic and parasympathetic activity in the heart, have a greater risk of developing clinical features of metabolic syndrome12. These individuals also faced a heightened incidence of diabetes, cardiovascular events, and mortality risk over a 20-year period. Obesity increases sympathetic activity while reducing parasympathetic (vagal) tone, indicating poor autonomic control of cardiac rhythm in obese individuals. The waist-to-hip ratio (WHR) serves as an indicator of visceral adiposity and is strongly correlated with increased sympathetic activity and reduced parasympathetic activity15. Pathology affecting the sympathetic and parasympathetic nerve fibers supplying the blood vessels and heart can lead to autonomic dysfunction16. As neuropathy tends to occur earlier in the longest nerve fibers, the first manifestations of autonomic neuropathy in metabolic disorders stem from parasympathetic nervous system (PNS) denervation and alterations in heart rate variability (HRV)17. These abnormalities in heart rate control and vascular tone typically manifest as resting tachycardia in the advanced stages of autonomic dysfunction.
With further advancement of this autonomic dysfunction, the signs of abnormal heart rate control and vascular tone become more prominent, manifesting as resting tachycardia. This progression eventually disrupts normal blood pressure regulation, resulting in presyncope symptoms, exercise intolerance, and palpitations. These symptoms not only significantly impair quality of life but also reflect cardiovascular instability in these groups of people18.
In recent times the development of cardiovascular symptoms and sudden cardiac events have been increasing in the younger population at an alarming rate19. This is a cause of concern and necessitates further research for a better understanding to tackle the emerging epidemic of cardiovascular morbidity in this age group. Thus, this study proposes to investigate the interplay between altered autonomic functions and cardiac ventricular repolarization parameters in populations of normal, overweight, and obese individuals. This could also provide insight into how obesity impacts cardiovascular parameters early in an obese person’s life.
Methods
Our study was a cross-sectional study and sample size was calculated by a previous study done by Hussain et al.20.
Formula used –
T = number of pairwise comparison, if we have k groups then, k(k-1)/2 possible comparison: µA = 374.83 µB = 390.17 pooled σ = 18.4 T = 3, n = 31 so, per group = 16.
We took n = 30 in each group (BMI (kg/m2)- 18-22.9, 23-24.9 and > 24.9) to increase the power of our study further to 90%. This was in accordance with Mishra et al.21 who determined obesity cut-offs in the Southeast Asia region and suggested that BMI levels above 22.9 indicate a higher cardiovascular risk, different from Western populations where obesity is defined as a BMI of > 25.
The inclusion criteria for the study group were individuals within the range of 19–35 years of age. Only people with morning shifts of not more than 8 h, were considered for participation22. We excluded individuals who were on medications affecting ECG parameters, such as antimalarials (chloroquine, mefloquine), antipsychotics, ketoconazole, and antiarrhythmics. Subjects with chronic kidney disease, liver disease, endocrine abnormalities, cardiac abnormalities, severe anemia, substance abuse, hypertension, diabetes mellitus were also excluded from the study.
Study protocol
Prior informed consent was obtained after explaining the study procedure to the subjects. Participants were advised to avoid caffeine or tea on the investigation day and to have a light breakfast at least three hours before testing. Subjects were referred to the Exercise Physiology Lab, Department of Physiology, Vardhman Mahavir Medical College. Investigations were conducted between 9 a.m. and 11:30 a.m. to account for variations in autonomic tone throughout the day23. The lab temperature was maintained between 23 °C and 25 °C. Participants were allowed to acclimatize to lab conditions for 20 min before testing began. Tests were performed uniformly across all participants to minimize bias.
Parameters measured
Anthropometric measurements included height (using a stadiometer, in cm), weight (using an automatic weighing scale (Crown)™ in kg), and body mass index (BMI) calculated as weight (in kg) / height (in meters) ². Additional measurements included waist circumference (WC) at the umbilical level, hip circumference (HC) at the pubic symphysis and the largest outward part of the hip, and waist-hip ratio (WHR) calculated as WC/HC. ECG intervals namely Tpe, QT and JT intervals were recorded automatically using the ML 870B80 POWERLAB (8/30) (A.D. Instruments) in lead V524. Tpe/QT, Tpe/QTc and Tpec/QTc ratios were also calculated manually, in lead v5 which is the lead preferred for measuring the Tpe interval as recommended by Emori et al.25. The correction of QT interval for heart rate was done automatically by the machine and the formula used was Bazette’s formula that is QTc = QT /√RR.
Heart rate variability (HRV) parameters were measured and analyzed using the built-in HRV module of ML 870B80 (A.D. Instruments). A 5-minute ECG recording was used to calculate time-domain parameters such as mean heart rate, SDRR (square root of the variance of the RR interval), SDSD (standard deviation of square root deviations of RR intervals), RMSSD (root mean square of standard deviation), MedRR (median of all RR intervals), and pRR50 (percentage of successive beats with a difference of more than 50 ms). Frequency domain parameters like total power (TP), very low frequency (VLF), low frequency (LF), high frequency (HF), and the LF/HF ratio were expressed as power (in ms²), normalized units (nu), and percentages (%).
Statistical analysis
Data was analyzed using GraphPad Prism version 9.3.0 (GraphPad Software, San Diego, California, USA). The Kolmogorov-Smirnov (K-S) test was used to assess the normality of data distribution. Continuous variables were presented as Mean ± Standard deviation (SD). ANOVA was performed to assess differences among the normal weight, overweight, and obese groups. Post-hoc Tukey’s test was applied to determine statistically significant differences between groups. Pearson’s linear correlation was used to analyze relationships among anthropometric, ventricular repolarization, and HRV parameters. Significance was set at p < 0.05 with a 95% confidence interval and β = 0.2, providing a power of 80%.
Ethical policy and institutional ethics committee
The study protocol was approved by the Institutional Ethics Committee (IEC/VMMC/SJH/Thesis/2019-10/CC-256) on August 17, 2021. The procedures adhered to the guidelines outlined in the Declaration of Helsinki 2013. Informed consent was obtained from all participants before enrollment.
Results
Ninety subjects aged 19–35 years, were assessed according to the inclusion and exclusion criteria.
Anthropometric parameters
Measured anthropometric parameters included weight, height, BMI, WC, HC, and WHR. Most of the anthropometric parameters (except Height) were statistically different between the three groups with higher values in the obese group when compared with the other two groups (Table 1).
Electrocardiographic and heart rate variability parameters
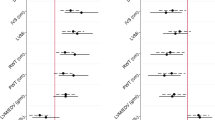
Electrocardiographic measurements showed higher heart rates in the obese and overweight groups when compared to the normal weight group (Table 2). The Tpe interval was also significantly different in the three groups with higher values in the obese group. The QTc interval also varied notably across the three study groups (Table 2).
Statistically significant differences were also observed in time-domain and frequency-domain parameters, including the average RR interval, median RR interval, LF power%, and VLF power% in the three groups (Table 2).
Pearson linear correlation of anthropometric and ventricular repolarization parameters with HRV parameters
Anthropometric parameters like BMI, WC, and WHR showed significant negative correlations with HF power in ms², nu, and %. Additionally, WHR and WC were correlated with time-domain HRV parameters (Table 3). While the QTc interval showed significant correlations with total power, HF power (ms²), HF power%, LF power (nu), HF power (nu), LF/HF ratio, average RR, MedRR, SDRR, SDSD, RMSSD, and pRR50, no correlation was observed between the Tpeak-Tend interval and any HRV parameters in this study (Table 3).
Discussion
Relationship between ventricular repolarization parameters and obesity
Prolonged QT and Tpe intervals in obese individuals suggest altered cardiac electrophysiology, as indicated by increased durations of both ventricular depolarization and repolarization. However, since the Tpe/QT ratio is also higher in obese people, this may imply that the repolarization duration of the M cells of the myocardium contributes more to the total duration of the QT interval. A recent study done by Dykeirt et al.26 has shown that an increase in Tpe interval and its dispersion was longer in the obese group. Yamaguchi et al.5 documented that longer Tpe interval values are directly related to increased chances of cardiac morbidity (arrhythmia). Additionally, a study by Panikkath et al.27 stated that the risk of cardiac mortality (sudden cardiac death) increases with longer Tpe interval durations. Our study also corroborates this finding of longer Tpe values in obese individuals. There is not a clear consensus about normal Tpe values in different populations, however a relative increase in this interval has been associated with many cardiovascular comorbidities such as coronary heart disease, cerebrovascular disease, rheumatic heart disease28. The QTc interval was significantly longer in the obese on the electrocardiogram. Other ventricular repolarization parameters, such as the QT interval, Tpe/QTc ratio, Tpe/QT ratio, and JT interval, had longer durations in the obese but were not statistically significant between the three groups. A significant positive correlation was found between obesity indices (such as BMI, WC, and WHR) and ventricular repolarization parameters (such as the TPE interval and QTc interval) when all 90 subjects were analyzed together using Pearson linear correlation as shown in Table 3.
Relationship between heart rate variability and obesity
In our study, the average RR interval [normal weight group (902.26 ± 133.78 ms), overweight group (837.94 ± 100.11 ms), and obese group (817.15 ± 89.92 ms)], was shorter in obese subjects compared to normal-weight subjects and overweight subjects. We found similar results with MedRR. The simple linear correlation of the average RR and MedRR intervals was also statistically significantly negatively correlated with BMI, WC, and WHR, further reinforcing that heart rate increases with body weight.
In our study, SDSD was significantly negatively correlated with both WC (r = -0.432, p-value = 0.017) and WHR (r = -0.437, p-value = 0.016). This has also been documented by Poliakova et al.29, who studied 97 subjects with no endocrinological or cardiovascular abnormalities and related obesity indices to heart rate variability. We also found a negative correlation between SDRR and WHR (r = -0.397, p-value = 0.033). SDSD and SDRR represent both SNS and PNS activity in humans and are highly correlated with VLF band power, LF band power, and total power in the measurement of short-term heart rate variability30. Thus, a negative correlation of these parameters with obesity indices indicates decreased activity of the autonomic nervous system. A previous study by Kleiger et al.31 indicates that higher SDRR values reduce the risk of mortality in acute myocardial infarction patients (5.3 times) compared to those with lower SDRR. We also documented a negative correlation between WHR and RMSSD (r = -0.437, p-value = 0.016), which has been previously documented by Kim et al.32, who found a strong negative correlation between WHR and RMSSD. Additionally, in our study, RMSSD was negatively associated with WC (r = -0.864, p-value = 0.017), consistent with findings from previous studies by Windham et al.33. Since RMSSD is an index for parasympathetic function or vagal influence on the heart, lower RMSSD values indicate decreased PNS activity in the obese. The pRR50 is an index for measuring high-frequency parameters in the heart and is closely correlated with PNS activity. This is considered a more reliable index than short-term SDNN measurements for short samples of 5-minute duration34. We also found that WHR and WC were significantly negatively correlated with pRR50 (r = -0.438, p-value = 0.022; r = -0.415, p-value = 0.031, respectively). Similar observations have also been documented in a study by Farah et al.35.
The observations of reduced indices of parasympathetic activity, such as RMSSD and pRR50 in the time domain and HF in the frequency domain, imply that obesity significantly reduces parasympathetic activity compared to normal-weight and overweight individuals. A higher LF/HF ratio despite significantly lower LF and LF power% in obese people also suggests that there was a derangement of both sympathetic and parasympathetic limbs of the autonomic nervous system in obesity, with parasympathetic derangement being more pronounced than sympathetic derangement.
A previous study by Rajalakshmi et al.36 showed that HF power (ms²) was negatively correlated with BMI (r = -0.40), WC (r = -0.37), and WHR (r = 0.37), as well as the negative correlation of HF (nu) with BMI (r = 0.32) and WC (r = 0.28), like what we found in our study (Table 3). The HF band, also known as the respiratory band, correlates with the respiratory cycle and reflects parasympathetic activity. It has been observed that total vagal blockade effectively eliminates HF oscillations37. The modulation of vagal tone thus plays a vital role in maintaining dynamic autonomic regulation crucial for cardiovascular health. Subsequently, deficient vagal inhibition has been causally linked to increased cardiovascular morbidity38. Therefore, the findings observed in our study show that as obesity increases, parameters measured in the HF band, such as HF power, HF power%, and HF nu, decrease. This suggests that obese subjects have reduced parasympathetic activity and increased sympathetic activity compared to normal-weight and overweight subjects as seen in our study.
Relationship between heart rate variability and ventricular repolarization parameters
We also documented a statistically significant linear correlation between the QTc interval and various HRV parameters. A statistically significant negative correlation was found between the QTc interval and average RR, median RR, SDRR, SDSD, RMSSD, and pRR50 in time domain parameters. Additionally, a statistically significant negative correlation was found between the QTc interval and total power, HF power (ms²), HF power%, and HF nu in frequency domain parameters. A statistically significant positive correlation of the QTc interval was found with VLF power%, LF nu, and the LF/HF ratio. These findings suggest that the QTc interval is affected by autonomic activity in the heart, as shown by Maule et al.39 and Seon40. While no significant correlation between these HRV parameters and the TPE interval was observed, the potential mechanism explaining the association between a prolonged QTc interval and decreased HRV has not been fully established. Proposed mechanisms for QTc interval prolongation include gene mutations affecting myocardial ion channels involved in cardiac repolarization, an imbalance of the cardiovascular autonomic nervous system, hyperglycemia or hypoglycemia, hyperinsulinemia, obesity, post-myocardial infarction scar tissue, and ventricular hypertrophy41,42,43,44,45,46,47. A proposed mechanism of the effect of obesity on various parameters measured in our study is depicted in Fig. 2 which may be useful tool for further studies on the subject and can be validated in larger populations.
The effect of Obesity on Cardiac autonomic function (A), the effect of obesity on ventricular repolarisation parameters both QTc interval and Tpe interval (B), the effect of autonomic dysfunction on QT interval alone (C).
In developing countries screening the patients with ECG is an accessible and affordable tool for the masses. As we have seen that the Southeast Asian population is at greater risk for cardiovascular morbidities even at lower BMI levels. Therefore, using ventricular repolarization parameters is a cost-effective approach, especially in low- to middle-income countries, to assess whether a person might be at risk for cardiovascular disease, even in the absence of any genetic or predisposing conditions that could inherently cause an increase in ventricular repolarization parameters.
Limitations
Future studies with larger cohorts may provide more insights into the causal relationship between QTc prolongation and HRV. This study did not report the prevalence of death or future cardiovascular disease associated with the studied variables. This cross-sectional study cannot determine cause and effect and could be influenced by other unmeasured factors. Echocardiography of the study population could provide more information on the structural changes in the heart and help better understand the relationship between the autonomic and structural effects of obesity. We chose to focus on Ventricular repolarization parameters as they are more accessible and affordable with decent reliability. Further studies using these specific tests could shed more light on these findings.
Conclusion
The present study demonstrates a significant impact of obesity on the electrocardiographic and autonomic changes within the cardiovascular system. Such changes may result from hemodynamic, structural, and other alterations associated with obesity. The observed effects of obesity on the QT and QTc intervals appear to be linked to these changes, as evidenced by correlations with both anthropometric and heart rate variability (HRV) parameters. This suggests that a reduction in parasympathetic activity, coupled with relatively increased sympathetic activity, may contribute to QTc interval prolongation. Although the Tpe interval is less influenced by autonomic modulation, the data indicates that it serves as a crucial marker of altered electrophysiological function in M cells of the myocardium, directly impacted by obesity. These findings underscore the need for further studies to explore potential clinical applications.
Data availability
The datasets used and/or analysed during the current study available from the corresponding author on reasonable request.
References
Poirier, P. & Eckel, R. H. Obesity and cardiovascular disease. Curr. Atheroscler Rep. 4(6), 448–453 (2002).
Alpert, M. A., Omran, J. & Bostick, B. P. Effects of obesity on cardiovascular hemodynamics, cardiac morphology, and ventricular function. Curr. Obes. Rep. 5(4), 424–434 (2016).
Lazzarin, T. et al. Role of nutrients and foods in attenuation of cardiac remodeling through oxidative stress pathways. Antioxid. (Basel). 11(10), 2064 (2022).
Shimizu, M. et al. T-peak to T-end interval may be a better predictor of high-risk patients with hypertrophic cardiomyopathy associated with a cardiac troponin I mutation than QT dispersion. Clin. Cardiol. 25(7), 335–339 (2002).
Yamaguchi, M. et al. T wave peak-to-end interval and QT dispersion in acquired long QT syndrome: A new index for arrhythmogenicity. Clin. Sci. Lond. Engl. 1979. 105(6), 671–676 (2003).
Locati, E. T., Bagliani, G. & Padeletti, L. Normal ventricular repolarization and QT interval. Card Electrophysiol. Clin. 9(3), 487–513 (2017).
Guzzardi, M. A., Iozzo, P. Fatty & heart cardiac damage, and inflammation. Rev. Diabet. Stud. RDS. 8(3), 403–417 (2011).
Patel, K. H. K. et al. Increasing adiposity is associated with QTc interval prolongation and increased ventricular arrhythmic risk in the context of metabolic dysfunction: results from the UK Biobank. Front. Cardiovasc. Med. 9, 939156 (2022).
Omran, J., Bostick, B. P., Chan, A. K. & Alpert, M. A. Obesity and ventricular repolarization: A comprehensive review. Prog Cardiovasc. Dis. 61(2), 124–135 (2018).
Jackman, W. M. et al. The long QT syndromes: a critical review, new clinical observations and a unifying hypothesis. Prog Cardiovasc. Dis. 31(2), 115–172 (1988).
O’Neal, W. T. et al. Association between QT-Interval components and sudden Cardiac death: the ARIC Study (Atherosclerosis Risk in communities). Circ. Arrhythm. Electrophysiol. 10(10), (2017).
Wulsin, L. R., Horn, P. S., Perry, J. L., Massaro, J. M. & D’Agostino, R. B. Autonomic imbalance as a predictor of metabolic risks, cardiovascular disease, diabetes, and mortality. J. Clin. Endocrinol. Metab. 100(6), 2443–2448 (2015).
Shen, M. J. & Zipes, D. P. Role of the autonomic nervous system in modulating Cardiac Arrhythmias. Circ. Res. 114(6), 1004–1021 (2014).
Magnano, A. R., Holleran, S., Ramakrishnan, R., Reiffel, J. A. & Bloomfield, D. M. Autonomic nervous system influences on qt interval in normal subjects. J. Am. Coll. Cardiol. 39(11), 1820–1826 (2002).
Yadav, R. L. et al. Association between obesity and heart rate variability indices: an intuition toward cardiac autonomic alteration – a risk of CVD. Diabetes Metab Syndr Obes Targets Ther. 10, 57–64 (2017).
Vinik, A. I., Maser, R. E., Mitchell, B. D. & Freeman, R. Diabetic autonomic neuropathy. Diabetes Care. 26(5), 1553–1579 (2003).
Benichou, T. et al. Heart rate variability in type 2 diabetes mellitus: A systematic review and meta–analysis. PLOS ONE. 13(4), e0195166 (2018).
Pop-Busui, R. et al. Diabetic neuropathy: A position statement by the American Diabetes Association. Diabetes Care. 40(1), 136–154 (2017).
Abbas, R. et al. Sudden cardiac death in young individuals: A current review of evaluation, screening and prevention. J. Clin. Med. Res. 15(1), 1–9 (2023).
Hussain, G. & Farooque, I. Effect of obesity on electrocardiographic parameters of ventricular repolarization in healthy adults. J. Evid. Based Med. Healthc. 4(95), 5915–5920 (2017).
Misra, A. et al. Consensus statement for diagnosis of obesity, abdominal obesity and the metabolic syndrome for Asian indians and recommendations for physical activity, medical and surgical management. J. Assoc. Physicians India. 57, 163–170 (2009).
Mozos, I. & Filimon, L. QT and tpeak-tend intervals in shift workers. J. Electrocardiol. 46(1), 60–65 (2013).
Baschieri, F. & Cortelli, P. Circadian rhythms of cardiovascular autonomic function: physiology and clinical implications in neurodegenerative diseases. Auton. Neurosci. Basic. Clin. 217, 91–101 (2019).
Li, Z. D. et al. Association between ventricular repolarization variables and cardiac diastolic function: A cross-sectional study of a healthy Chinese population. World J. Clin. Cases. 7(8), 940–950 (2019).
Emori, T. & Antzelevitch, C. Cellular basis for complex T waves and arrhythmic activity following combined I(kr) and I(Ks) block. J. Cardiovasc. Electrophysiol. 12(12), 1369–1378 (2001).
Dykiert, I. A. et al. The effect of obesity on repolarization and other ECG parameters. J. Clin. Med. 13(12), 3587 (2024).
Panikkath, R. et al. Prolonged tpeak-to-tend interval on the resting ECG is associated with increased risk of sudden cardiac death. Circ. Arrhythm. Electrophysiol. 4(4), 441–447 (2011).
Cardiovascular diseases (CVDs). [Internet]. [cited 2024 Aug 30]. https://www.who.int/news-room/fact-sheets/detail/cardiovascular-diseases-(cvds).
Poliakova, N. et al. Influence of obesity indices, metabolic parameters and age on cardiac autonomic function in abdominally obese men. Metabolism. 61(9), 1270–1279 (2012).
Shaffer, F. & Ginsberg, J. P. An overview of heart rate variability metrics and norms. Front. Public. Health. 5, 258 (2017).
Kleiger, R. E., Miller, J. P., Bigger, J. T. & Moss, A. J. Decreased heart rate variability and its association with increased mortality after acute myocardial infarction. Am J Cardiol. 59(4), 256–62 (1987).
Kim, J. A. et al. Heart rate variability and obesity indices: Emphasis on the response to noise and standing. J. Am. Board. Fam Pract. 18(2), 97–103 (2005).
Windham, B. G. et al. The relationship between heart rate variability and adiposity differs for central and overall adiposity. J. Obes. 2012, e149516 (2012).
Malik, M. et al. Heart rate variability: Standards of measurement, physiological interpretation, and clinical use. Eur. Heart J. 17(3), 354–381 (1996).
Farah, B. Q., Prado, W. L., do, Tenório, T. R. & dos Ritti-Dias, S. Heart rate variability and its relationship with central and general obesity in obese normotensive adolescents. Einstein Sao Paulo Braz. 11(3), 285–290 (2013).
SM, R. R. Y. V., N. & CG, M. S. Heart rate variability in Indian obese young adults. PJP. 8(1), 39–34 (2018).
Shaffer, F., McCraty, R. & Zerr, C. L. A healthy heart is not a metronome: An integrative review of the heart’s anatomy and heart rate variability. Front. Psychol. 5, 1040 (2014).
Thayer, J. F., Yamamoto, S. S. & Brosschot, J. F. The relationship of autonomic imbalance, heart rate variability and cardiovascular disease risk factors. Int. J. Cardiol. 141(2), 122–131 (2010).
Maule, S. et al. Prolonged QT interval and reduced heart rate variability in patients with uncomplicated essential hypertension. Hypertens. Res. Off J. Jpn Soc. Hypertens. 31(11), 2003–2010 (2008).
Cha, S. A. Heart rate-corrected QT interval prolongation is associated with decreased heart rate variability in patients with type 2 diabetes. Med. (Baltim). 101(45), e31511 (2022).
Roden, D. M. & Spooner, P. M. Inherited long QT syndromes: A paradigm for understanding arrhythmogenesis. J. Cardiovasc. Electrophysiol. 10(12), 1664–1683 (1999).
Peters, R. W., Byington, R. P., Barker, A. & Yusuf, S. Prognostic value of prolonged ventricular repolarization following myocardial infarction: The BHAT experience. The BHAT Study Group. J. Clin. Epidemiol. 43(2), 167–172 (1990).
Marfella, R. et al. The effect of acute hyperglycaemia on QTc duration in healthy man. Diabetologia. 43(5), 571–575 (2000).
Dekker, J. M. et al. QTc duration is associated with levels of insulin and glucose intolerance. The Zutphen Elderly Study. Diabetes. 45(3), 376–380 (1996).
Rautaharju, P. M., Park, L. P., Chaitman, B. R., Rautaharju, F. & Zhang, Z. M. The Novacode criteria for classification of ECG abnormalities and their clinically significant progression and regression. J. Electrocardiol. 31(3), 157–187 (1998).
Gastaldelli, A., Emdin, M., Conforti, F., Camastra, S. & Ferrannini, E. Insulin prolongs the QTc interval in humans. Am. J. Physiol. Regul. Integr. Comp. Physiol. 279(6), R2022–2025 (2000).
Carella, M. J. et al. Obesity, adiposity, and lengthening of the QT interval: Improvement after weight loss. Int. J. Obes. Relat. Metab. Disord J. Int. Assoc. Study Obes. 20(10), 938–942 (1996).
Funding
Open access funding provided by Manipal Academy of Higher Education, Manipal
This research did not receive any specific grant from funding agencies in the public, commercial, or non-profit sectors. Manipal Academy of Higher Education will cover the costs for APC (Article Processing Charges) and/or Open Access publication fees.
Author information
Authors and Affiliations
Contributions
HA, SG and HS gave the concept and design, AT, IY and PK wrote the main manuscript text and did statistical analysis, AT prepared Figs. 1 and 2. All authors reviewed the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Tomar, A., Ahluwalia, H., Isser, H.S. et al. Analysis of ventricular repolarization parameters and heart rate variability in obesity: a comparative study. Sci Rep 14, 25855 (2024). https://doi.org/10.1038/s41598-024-76580-x
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-024-76580-x