Abstract
By investigating the correlation between the injection rate and pressure of subretinal tissue plasminogen activator (tPA) and air using a standard Viscous Fluid Control (VFC) system with a 38-gauge cannula, we aimed to establish guidelines for stable injections. We fabricated a retina mimicking model (RMM) with 0.25% agarose solution and an aluminum plate, and substituted submacular hemorrhage (SMH) and tPA with blood-mimicking fluid (BMF) and balanced salt solution (BSS), respectively. The diameter of the pre-bleb mimicking SMH in RMM was 1.30 ± 0.16 cm, increasing to 1.98 ± 0.24 cm and 1.83 ± 0.22 cm after bleb propagation with BSS and air, respectively. BSS injection rates were 2.86 ± 0.04 µl/sec, 6.74 ± 0.48 µl/sec and 8.55 ± 0.16 µl/sec at 8, 12, and 16 psi, respectively. Air injection rates were 37.98 ± 3.11 µl/sec, 79.01 ± 5.13 µl/sec and 156.06 ± 13.72 µl/sec at 2, 3 and 4 psi, respectively. By experimenting with different pressures in the RMM, we found 12 psi to be the minimum for proper BSS injection and 2 psi for air. These findings provide crucial parameters for safer surgery to prevent irreversible damage.
Similar content being viewed by others
Introduction
Extensive submacular hemorrhage (SMH) is a rare but detrimental complication of several retinal diseases, most commonly age-related macular degeneration (AMD), retinal arterial microaneurysm, ocular histoplasmosis, trauma, and high, myopia, which can lead to irreversible retinal changes and vision loss without treatment1,2. Therefore, intervention for SMH is needed to improve its prognosis.
Subretinal injection of tissue plasminogen activator (tPA) in combination with air for the treatment of SMH in AMD is widely used by retinal specialists and has proven to be an effective technique2,3. The surgical procedure consisted of a 25-gauge vitrectomy and subretinal injection of tPA to liquefy coagulated SMH, followed by injection of filtered air to displace the liquefied hemorrhage downward. During the procedure, the surgeon can inject tPA or air by manually pushing the syringe plunger with the assistance of an assistant. In contrast, the surgeon holds the syringe body connected to the vitrectomy cannula to be steady at the subretinal space. However, a manual injection is difficult to precisely control and exclude variables, such as significant shaking by the surgeon and assistant4. Furthermore, there is a possibility of sudden injection during the procedure, which could lead to the risk of causing damage to the retina. Several previous studies in pig or monkey eyes have shown that fast injection rates are associated with significant loss of RPE and photoreceptors in the retina5,6.
To overcome these shortcomings, a method has been developed using a 1 ml syringe connected to a 38-gauge cannula with a standard Viscous Fluid Control (VFC) system using a foot pedal4. A MicroDose Injection Kit7 (MedOne Surgical, Inc., Sarasota, FL) can be used in surgery. However, there is a problem that the product is not available worldwide, especially in South Korea, so a substitute for a MicroDose Injection Kit is needed. Also, as the standard pressure or rate of injection of tPA and air is not fully established, there is still a risk of sudden injection leading to significant damage to the retina. Therefore, it is necessary to investigate the correlation between the injection rate and pressure of subretinal tPA and air injection using a standard VFC system with a 38-gauge cannula. In addition, guidelines for stable injection without a MicroDose Injection Kit should be established. Since it was not feasible to perform experiments in real patients, we performed experiments using simulated models that mimic the actual retinal structure, the retina mimicking model (RMM).
In this study, we aimed to elucidate subretinal injection rate of tPA solution and air at different pressures using a 38-gauge cannula with a controlled injection system in a retina mimicking model.
Materials and methods
Fabrication of the retina mimicking model (RMM)
Based on previous research, agarose has been used in artificial retina studies8, we fabricated a retina mimicking model (RMM) by using agarose (Agarose LE Analytical Grade, Promega). As the artificial retinal layer was composed of 2% agarose gel in the preliminary study8, we first tried to fabricate the RMM with 2% agarose solution. However, it was observed that 2% agarose solution had excessive viscosity, making it challenging to inject the material smoothly without experiencing regurgitation. Moreover, previous studies have indicated that the minimum pressure required for subretinal injection in monkey retinas is 6 psi5, with the mean minimum pressure observed in clinical settings approximating 6 psi (5.8 psi)9. Based on these findings, we tested various RMMs with varying concentrations of agarose solution to identify the appropriate concentration of agarose solution that would facilitate the formation of blebs at a minimum pressure of 6 psi. Consequently, the final concentration of agarose solution for the RMM was established at 0.25%.
The retina mimicking model (RMM) consisted of a 0.25% agarose solution poured along the inner surface of the aluminum plate (LabPlasti, Republic of Korea) (Supplementary Fig. S1a online), which has an inner diameter of 72 mm. Immediately after the agarose solution was poured into the aluminum plates, the plates were floated in a water bath at 25℃ for drying. It was observed that excessive drying time over 30 min makes the agarose brittle. To prevent the loss of a proper viscosity similar to that of the retina, the time from model fabrication to experiments was limited to a maximum of 30 min. In addition, the proper thickness of dried agarose was required, taking into account the depth of needle penetration and the actual thickness of the retina. By experimenting with different amounts of agarose, the volume to be poured along the inner surface of the aluminum plate was determined to be 6 ml (Supplementary Fig. S1b online). To ensure standardization of the fabrication process, all RMMs were prepared by pouring the same amount of agarose solution, prepared simultaneously, onto identical aluminum plates and subjecting them to an identical cooling period.
Preparation of blood-mimicking fluid (BMF)
In order to mimic the conditions of SMH, it was imperative to find appropriate blood substitutes that could faithfully reproduce the environment of SMH. We adopted the concept of “blood-mimicking fluid (BMF)” from previous hemodynamic research. Based on the study, we developed BMF by mixing glycerol with balanced salt solution (BSS; Alcon, Fort Worth, TX ) in a 44:56 ratio10.
Creation of SMH-like structure in the retina mimicking model (RMM)
BMF was subsequently injected into the fabricated retina mimicking model (RMM) (Supplementary Fig. S1b online) to replicate the SMH environment. To create a pre-bleb by SMH, a 1 ml insulin syringe (BD Ultra-Fine II Insulin Syringe, Becton Dickinson Korea) was used to inject 0.1 ml of BMF to create a bleb. In detail, the needle of insulin syringe was bent at an angle of 45 degrees at the midpoint of its total length, with the bevel oriented downward. When inserting the needle into the RMM, the bevel was positioned slightly to the side. The bent portion of the needle was then gently pressed using a cotton swab while the BMF injection was performed (Supplementary Video S1 online). The amount of BMF injected was arbitrarily set as 0.1 ml, because formed bleb in RMM was stable without significant leakage in this volume. This procedure was performed manually by the surgeon (HL), not by the foot pedal-assisted control with a 38-gauge cannula because the needle was easily obstructed due to the viscosity of glycerol.
Subretinal injection with foot pedal system
To quantify the injection rates of tPA and air, we used an Alcon Constellation Viscous Fluid Control (VFC) syringe (Fig. 1a), typically used in surgery, connected to a 38-gauge cannula (inner diameter: 41 gauge) (PolyTip, MedOne Surgical, Sarasota, FL, USA) (Fig. 1b)11. By pressing the foot pedal, we injected fluid and air into the retina mimicking the model with known injection pressure and measure the injection rates.
Pictures of materials for injection. (a) An Alcon Constellation Viscous Fluid Control (VFC) syringe directly connected to a 38-gauge cannula. (b) A 38-gauge cannula (MedOne Surgical, Inc., Sarasota, FL, USA).
With the limited availability of tPA solution, we tested the similarity between tPA solution and BSS. BSS-based tPA solution (12.5 µg/0.1 ml) (Actilyse, Boehringer Ingelheim) and BSS were filled in the VFC syringe, and injected with 16 psi in the air. The injection rates of tPA solution and BSS were 8.12 ± 0.30 µl/sec and 8.50 ± 0.15 µl/sec, respectively, and were not significantly different (N = 3, P = 0.20). Based on this result, we substituted tPA with a balanced salt solution (BSS; Alcon, Fort Worth, TX).
The air injection rate was determined by the time it took to release 1 ml of air filled in the syringe, while the tPA solution injection rate was determined by the time it took to release 0.4 ml of BSS.
Prior to the main experiment, we need to establish the proper range of injection pressure based on the previous study with monkey eyes5. In the study, it was demonstrated that the minimum pressure required to perform subretinal injection was 6 psi, and 20 psi of high injection pressure was shown to cause damage to RPE and photoreceptor5.
In addition, as it has been reported that a continuous flow of fluid is produced in the range of 12–16 psi, we initially tried the subretinal injection of BSS at 8 psi. However, the measured injection rates at 8 psi were too slow, causing the investigator to hold the syringe too long (approximately 140 s to inject 0.4 ml of BSS), which could lead to a significant shaking of the syringe by the investigator and a risk of low stability of the injection. Also, our experiment showed that 12 psi and 16 psi of injection pressures produce a stream of droplets rather than a continuous flow of fluid.
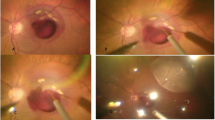
We performed the measurements at pressures of 8, 12 and 16 psi for BSS, and 2, 3 and 4 psi for air. Although the initial injection rate was observed to be at a very slow rate of 8 psi, it was essential to evaluate the injection rate at 8 psi, as the relatively slower rate at this pressure could provide significant insights. Therefore, we included the measurement of the BSS injection rate at 8 psi in our experiments. The injected materials such as BMF and BSS were visualized by the food dye (Eltin food color; Lgreentech, Republic of Korea). A BMF was visualized on the retina mimicking model with a pink food coloring (Fig. 2a). To contrast with the previously injected BMF, a blue food dye was added to the BSS (Fig. 2b).
Pictures of formed bleb. (a) BMF representing SMH with pink food coloring. (b) After the injection of BSS with blue food coloring into the bleb formed by BMF. (c) Injection of air 1 ml after the injection of BMF (0.1 ml) and BSS (0.4 ml).
All injections were performed by the same retinal specialist (HL). Measurement of the target volume was performed by the same investigator (NHK). To ensure the accuracy of the experiments, all experiments were repeated ten times.
A case report of a patient who was diagnosed with SMH and treated by subretinal injection according to this experiment was conducted at Konkuk University Medical Center. The study protocol was approved by the Institutional Review Board (IRB) of Konkuk University Medical Center (IRB number: KUMC 2024-09-020), and was conducted following the tenets of the Declaration of Helsinki. Due to the retrospective nature of the study, Institutional Review Board (IRB) of Konkuk University Medical Center waived the need of obtaining informed consent.
Statistical analyses
Statistical analysis was performed using IBM SPSS Statistics for Windows (version 23.0;IBM-SPSS, Armonk, NY, USA). The sizes of the blebs formed after injection of BMF, BSS, and air, and the injection rates of BSS and air at different pressure were compared using the Kruskal-Wallis test (Post hoc analysis using Mann–Whitney U test, statistically significant P < 0.05/3 = 0.017 by Bonferroni correction).
Results
Formation of submacular hemorrhage (SMH) by blood-mimicking fluid (BMF)
The regularity of the size of the formed bleb determined a successful bleb for artificial SMH. The size of the formed bleb was measured by its ratio to the actual size of the aluminum plate. We photographed the blebs and analyzed the diameter of the bleb using ImageJ program. The diameter of the bleb was calculated based on the measured length of the bleb. The actual sizes of the blebs and plates in the models are shown in Supplementary Table S1 online. The diameter of pre-bleb mimicking SMH formed by 0.1 ml of BMF was 1.30 ± 0.16 cm. Figure 2a shows an example of a bleb for artificial SMH created by BMF. The models with pre-bleb mimicking SMH were selected based on the median value of the diameter of the bleb.
Injection of BSS into the bleb formed by BMF
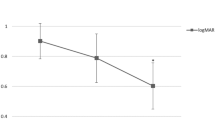
The injection rate of 0.4 ml BSS under different injection pressures is shown in Table 1. The injection rates of BSS into the retina mimicking model, followed by the injection of BMF, were measured to be 2.86 ± 0.04 µl/sec at 8 psi, 6.74 ± 0.48 µl/sec at 12 psi, and 8.55 ± 0.16 µl/sec at 16 psi (Table 1). Increasing the pressure from 8 psi to 12 psi resulted in a 2.36-fold increase in injection rate, and increasing the pressure from 12 psi to 16 psi resulted in a 1.27-fold increase in the injection rate (Fig. 3). After the successful injection of BSS, the color was changed because of a blue food dye mixed into the BSS (Fig. 2b, Supplementary Video S2 online). There was also a significant difference in size after bleb expansion with injection of BSS (P < 0.001; Fig. 4, Supplementary Table S1 online). There was a significant difference in the injection rate of BSS 0.4 ml from the three different pressures (P < 0.001). There was a significant difference in injection rates between the two specific pressure conditions (all P < 0.001, after Bonferroni correction; Table 1).
Change in injection rate of BSS 0.4 ml as a result of variation in injection pressure.
Comparison of size of formed blebs after injection of BMF, BSS, and air. The mean, median, and standard deviation of size of formed blebs are provided in Supplementary Table S1. After subsequent injection of BSS and air into the BMF pre-bleb, the sizes have significantly changed. ***P < 0.05/3 = 0.017.
Injection of air into the mixed solution of BMF and BSS
After injection of both BMF and BSS, the injection rates of air were measured to be 37.98 ± 3.11 µl/sec at 2 psi, 79.01 ± 5.13 µl/sec at 3 psi and 156.06 ± 13.72 µl/sec at 4 psi (Table 1, Supplementary Video S3 online). Increasing the pressure from 2 psi to 3 psi resulted in a 2.08-fold increase in injection rate, and increasing the pressure from 3 psi to 4 psi resulted in a 1.98-fold increase in the injection rate (Figs. 2c and 5). There was no significant difference in size after bleb expansion with injection of air (P = 0.116 after Bonferroni correction; Fig. 4, Supplementary Table S1 online). However, compared with the initial status of bleb form by BMF, there was a significant difference in size after the injection of BSS and air into the BMF compared to be the initial bleb with BMF (P < 0.001; Supplementary Table S1 online, Fig. 4). There was a significant difference in the injection rate of air 1 ml from the three different pressures (P < 0.001). In addition, there was a significant difference in injection rates between the two specific pressure conditions (all P < 0.001, after Bonferroni correction; Table 1).
Change in injection rate of air 1 ml as a result of variation in injection pressure.
In addition, it was observed that when the VFC syringe was filled with more than 8 ml of air, the plunger inside the syringe was pushed back due to the reactive force caused by the filled air.
Discussion
Subretinal macular hemorrhage (SMH), a severe and potentially irreversible complication of age-related macular degeneration (AMD), is typically treated with tissue plasminogen activator (tPA) and air injections during pars plana vitrectomy (PPV) with foot-pedal assisted control1,3,4. However, manual injection using a VFC syringe and a 38-gauge cannula can result in shaking and sudden high-pressure injections, which may potentially damage photoreceptors and the retinal pigment epithelium (RPE). While the MicroDose Injection Kit7 can be used, its limited availability, particularly in South Korea, necessitates the development of a stable procedure using a VFC syringe and a 38-gauge cannula. We performed experiments using a VFC syringe connected to a 38-gauge cannula assisted with foot pedal-assisted control. Furthermore, we devised a retina mimicking model (RMM) by pouring 0.25% agarose solution into the aluminum plate and drying it.
Under the pressure of 12, and 16 psi, BSS, which was selected as a substitute for tPA, was safely injected with the rate of 6.74 ± 0.48, 8.55 ± 0.16 µl/sec, respectively, and under the pressure of 8 psi, the injection rate was too slow to make the surgeon difficult to hold the syringe stably (Table 1). As the pressure sequentially increased from 8 psi to 12 and 16, the injection rate increased by 2.36 times and 1.27 times. Therefore, based on these results, it can be concluded that the minimum pressure required for appropriate BSS injection is 12 psi, with the injection pressure potentially increasing up to a maximum of 16 psi. In addition, the injection rate did not appear to increase proportionally as the pressure increased.
Air was stably injected at a pressure of 2, 3, and 4 psi, with the rate of 37.98 ± 3.11, 79.01 ± 5.13, and 156.06 ± 13.72 µl/sec, respectively (Table 1). As the pressure sequentially increased from 2 psi to 3 and 4, the injection rate increased by 2.08 times and 1.98 times. Based on these results, it can be concluded that the minimum pressure required for appropriate air injection is 2 psi, with the injection pressure potentially increasing up to a maximum of 4 psi. In addition, the injection rate of air did not indicate a non-proportional increase as the pressure increased. As the mean injection rate of air at 4 psi was found to be 156.06 ± 13.72 µl/sec, which indicates that it takes 6.41 s to inject 1 ml of air. These data suggest that the injection of air at pressures exceeding 4 psi is likely to be inadvisable, given the short duration of the injection, which is anticipated to be less than 6.41 s. This may potentially impair the stability of the injection, and the abrupt injection may also result in irreversible damage to retinal structures.
Furthermore, when these methodologies were applied to a real patient with SMH, the injection of tPA and air was successfully performed. During the surgical procedure, it was found that tPA and air were stably injected within the vitreous cavity. The injection of tPA and air into the subretinal space with SMH was also stably performed (Supplementary Video S4 online).
By identifying the injection rate, we can estimate the time needed to inject a given amount of tPA and air into the subretinal space. For example, injecting 0.4 ml of tPA requires 59.33 s at 12 psi, while 16 psi takes 46.78 s. Similarly, injecting 1 ml of air takes 26.33 s at 2 psi, 12.66 s at 3 psi, and 6.41 s at 4 psi. These data, which emphasize time, are significant in various aspects. First, accurately administering minute doses of less than 0.2 ml using only the VFC syringe scale can be challenging since the minimum scale increment is 0.2 ml. In addition, during surgery, neither the surgeon nor the assistant can visually monitor the real-time change in dose, which can lead to loss of stability, and result in irreversible damage to the retina. Quantifying the time required for each pressure level may prevent irreversible damage caused by excessive injection rate or volume. Furthermore, the individual nature of SMH in patients must be considered, as the amount or location of SMH varies between patients, resulting in variable doses of tPA or air required for injection. It is difficult to determine the preoperative amount, and surgeons typically rely on observing changes in subretinal space to determine when to stop the injection. Consequently, they can only consider time as a variable to control. By using the injection rates identified in the experiment and measuring the time during surgery at each pressure level, surgeons can anticipate the amount of tPA or air that will be injected.
However, this study has several limitations. First, it is impossible to determine the exact pressure at the time of formation of the initial bleb as we manually implemented it using an insulin syringe to simulate the environment of SMH, despite previous studies showing that a minimum pressure of 6 psi is required to form a bleb. Second, we did not take into account the change in blood viscosity in the actual SMH environment. In general, changes in viscosity occur over time after a hemorrhage occurs. Therefore, it is clear that the viscosity of actual SMH would vary depending on the time of occurrence. However, our experiment was performed using RMM rather than actual SMH because it is not feasible to perform the experiments on the actual retina of patients with SMH, so we could not analyze the viscosity of all real SMH. Although it will be necessary to analyze the effect of pressure on the injection rate based on different viscosities of SMH, this experiment based on the standard blood viscosity may provide standards when injecting tPA into the subretinal space filled with SMH. Third, we could not exclude the ‘learning effect’ when repeating the experiments. We repeated the experiments ten times in each condition to increase precision, which may lead to improved investigator skill, and then may result in subtle changes in injection rates. Fourth, the experimental subretinal bleb in RMM was set as 0.1 ml. Further study should take account for the variable volume of blebs. Furthermore, the results of the experiment may vary when performed with a more standard tool, such as the MicroDose Injection Kit7. Given that 0.4 ml of BSS is injected using a 10 ml VFC syringe, there is a possibility of a significant loss of tPA, which may have implications for the efficacy of the treatment. Therefore, further studies are required to develop precise guidelines with the use of a standard tool. Despite this limitation, given that at least one vial of tPA is employed for each patient, irrespective of the required dose, with a minimum necessary volume of 0.4 ml, and considering that the limited availability of the MicroDose Injection Kit in certain countries constituted one of the fundamental starting points for our experiments, the 10 ml VFC syringe-based system is deemed to be an efficacious instrumental tool. Finally, the experiment was not performed in animal model or real patients. Given the possibility of a discrepancy between the RMM and the retina of animal or human, the results of this experiment may differ significantly when applied to actual animals or human subjects. Thus, further studies are needed to prove the above results in animal model or patients with SMH.
In conclusion, as subretinal injection is a highly delicate surgical procedure for the treatment of SMH, a precise understanding of its physical properties significantly influences the outcome of the procedure. Therefore, our findings suggest that retinal specialists may utilize these data as a reference. In addition, the finding that the correlation between injection pressure and injection rate may not be linear provides insights into how changes in injection pressure would affect the injection rate. Furthermore, while it is essential to acknowledge the significant discrepancies between the experimental retina mimicking model (RMM) and actual clinical conditions, this study could provide a preliminary basis for subsequent investigations on animal models or human subjects. Additionally, this experiment remains valuable for quantifying surgical variables using time as a parameter, potentially enabling surgeons to more accurately predict and control injection rates or volumes, thereby reducing the risk of irreversible retinal damage due to excessive injection rates or volumes.
Data availability
Further information can be provided by the corresponding author upon request.
References
Scupola, A., Coscas, G., Soubrane, G. & Balestrazzi, E. Natural history of macular subretinal hemorrhage in age-related macular degeneration. Ophthalmologica. 213, 97–102 (1999).
Kadonosono, K. et al. Displacement of submacular hemorrhages in age-related macular degeneration with subretinal tissue plasminogen activator and air. Ophthalmology. 122, 123–128 (2015).
Sharma, S. et al. Pneumatic displacement of submacular hemorrhage with subretinal air and tissue plasminogen activator: initial United States experience. Ophthalmol. Retina. 2, 180–186 (2018).
Novelli, F. J., Preti, R. C., Monteiro, M. L., Nóbrega, M. J. & Takahashi, W. Y. A new method of subretinal injection of tissue plasminogen activator and air in patients with submacular hemorrhage. Retina (Philadelphia Pa). 37, 1607 (2017).
Takahashi, K. et al. The influence of subretinal injection pressure on the microstructure of the monkey retina. PLoS One. 13, e0209996 (2018).
Scruggs, B. A. et al. Optimizing donor cellular dissociation and subretinal injection parameters for stem cell-based treatments. Stem Cells Translational Med.8, 797–809 (2019).
MicroDose Injection Kit, MedOne Surgical, USA, https://medone.com/microdose-injection-kit/.
You, Y. S. et al. An arched micro-injector (ARCMI) for innocuous subretinal injection. PloS One. 9, e104145 (2014).
Scruggs, B. A. et al. Injection pressure levels for creating blebs during subretinal gene therapy. Gene Ther.29, 601–607 (2022).
Yousif, M. Y., Holdsworth, D. W. & Poepping, T. L. A blood-mimicking fluid for particle image velocimetry with silicone vascular models. Exp. Fluids. 50, 769–774 (2011).
Subretinal PolyTip Cannulas, MedOne Surgical, USA, https://medone.com/sub-retinal-cannulas/.
Acknowledgements
This paper was supported by Konkuk University in 2024. The sponsor or funding organization played no role in the design or conduct of this research. None of the authors has any conflicts of interest to disclose.
Author information
Authors and Affiliations
Contributions
H Lee have full access to all the data in the study and take full responsibility for the integrity of the data and the accuracy of the data analyses. Concept and design: NH Kim, H Lee; Data acquisition, analysis, or interpretation: all authors; Drafting of the manuscript: NH Kim, H Lee; Critical revision of the manuscript: all authors; Statistical analysis: NH Kim.The authors have no financial disclosures.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Supplementary Material 3.
Supplementary Material 4.
Supplementary Material 5.
Supplementary Material 6.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Kim, N.H., Lee, M., Chung, H. et al. Correlation between subretinal tissue plasminogen activator and air injection rates with pressure in a retina mimicking model. Sci Rep 14, 26203 (2024). https://doi.org/10.1038/s41598-024-77518-z
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-024-77518-z