Abstract
Recurrent high-grade intracranial malignancies have a grim prognosis and uniform management guidelines are lacking. Re-irradiation is underused due to concerns about irreversible side effects. Pulsed-reduced dose rate radiotherapy (PRDR) aims to reduce toxicity while improving tumor control by exploiting dose-rate effects. We share our initial experience with temporally modulated pulsed proton re-irradiation (TMPPR), focusing on workflow, safety, feasibility, and outcomes for the first patient cohort. TMPPR was administered to patients with recurrent or progressive central nervous system malignancies using intensity modulated proton therapy with three fields. Patient and treatment data were collected, responses categorized using RANO assessment, and toxicities graded using CTCAE v5.0. Five patients received TMPPR between October 2022 and May 2023, with a median age of 54 years (Range: 32–72), and a median time from initial radiotherapy to re-RT of 23 months (Range 14–40). Treatment was completed without delay, with a median dose of 60 GyRBE in 30 fractions. Initial treatment response assessment showed complete (n = 1) or partial (n = 3) responses. Limited toxicity was observed, primarily grade 2 alopecia and one case of radiation necrosis graded at 2. This early experience demonstrates the feasibility of TMPPR delivery, highlighting the importance of prospective evaluations in the re-irradiation setting.
Similar content being viewed by others
Introduction
Intracranial high-grade malignancies, such as glioblastoma—the most common malignant brain tumor in adults—are associated with frequent recurrences and poor prognosis. Salvage strategies such as surgery, re-irradiation (reRT), systemic therapies, and Tumor Treating Fields (TTFields) have not shown significant benefit in overall survival (OS) in the recurrent setting1,2. Out of these options, reRT remains underutilized given the potential for irreversible treatment-related toxicities as tumors typically recur within or adjacent to the radiotherapy field, resulting in frequent direct overlap with a previously treated region of the brain3. Yet, systemic therapies alone in the recurrent setting are associated with poor (< 10%) response rates4. Although no prospective randomized evidence supports one approach over another, a secondary analysis of RTOG 0525 observed a benefit to reRT, although it was limited in scope as few patients underwent a second course of radiotherapy (4% reRT alone and 10% with systemic therapy)5. Therefore, based primarily on retrospective data and limited to selected patients (small recurrence volumes and good performance status), reRT has been delivered as fractionated radiosurgery (FSRS) or hypofractionated schedules to a total dose of 30–35 Gy in 5–15 fractions with variable outcomes and limited randomized data to support its use5,6,7,8. In fact, the best data to date come from the RTOG 1205 trial which compared bevacizumab alone with radiotherapy [35 Gy in 10 fractions] and showed a benefit in 6-month progression-free survival (PFS) from 29 to 52%. However, no benefit in OS was observed9.
Pulsed-reduced dose rate (PRDR) radiotherapy, traditionally delivered with photons, is a reRT technique to reduce toxicity after prior RT in recurrent/progressive central nervous system (CNS) tumors by delivering subfractions at specific time intervals within a single fraction. There are two radiobiological advantages to this technique: first, a low-dose hyper-radiosensitivity of proliferative tumor cells irradiated at doses less than 0.5 Gy; second, non-proliferative normal tissue low-dose rate hypo-radiosensitivity, thereby increasing the therapeutic index as tumor kill is increased and the sub-lethal damage repair occurring in normal cells reduces toxicities10,11,12,13. While Cannon et al. documented a single case of PRDR showing a dramatic response with no associated toxicity14, our primary motivation for exploring proton therapy stems from its potential to further reduce toxicity and better spare normal brain tissue, especially in complex re-irradiation cases. We recently published our experience using photon PRDR in 18 patients and conducted a systematic review and meta-analysis of 188 patients treated with PRDR. With a median PRDR dose of 52 Gy and a median cumulative dose of 110.3 Gy delivered to a median tumor volume of 369.1 cc, 67% of patients experienced grade 2 + toxicities after a median follow-up of 8.7 months15. These results highlight the need for alternative reRT methods, such as proton therapy, which we evaluated to achieve similar benefits with novel radiotherapy technologies.
Proton therapy (PT) has overall dosimetric benefit over photon therapy in CNS malignancies16,17. The latest form of PT, intensity-modulated proton therapy (IMPT) has been dosimetrically demonstrated to result in lower maximum, average, and median doses to critical substructures of the brain compared to passive scatter proton therapy or intensity-modulated radiotherapy (IMRT) delivered using photons18. Our group recently described how IMPT was able to spare the uninvolved brain (V20Gy) and a reduction on D0.03 cc to the brain and optic chiasm when compared to IMRT19. In patients undergoing reRT, these reduced doses to critical substructures can decrease the risks for treatment-related toxicities, allowing the ability to deliver a definitive dose20. Our group has recently reported promising results for our first patient treated with TMPPR21. The objective of this study is to document the institutional workflow, safety, feasibility, acute toxicity, and best tumor response of the first five patients treated with this technique (TMPPR).
Materials and methods
Data acquisition
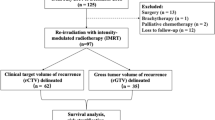
Consecutive patients who underwent a course of TMPPR for recurrent or progressive CNS malignancies in our institution were evaluated. All patients were evaluated in a multidisciplinary CNS tumor conference and selected for this option only when all conventional and clinical trial options were exhausted. Data collected from the electronic medical records included gender, age, tumor histology, Karnofsky Performance Status (KPS) at time of TMPPR, the number and dates of prior interventions, radiotherapy dose and fractionation schedule, and toxicities during treatment and follow-up.
Temporally modulated pulsed proton re-irradiation (TMPPR) workflow
Planning and treatment simulation imaging
At simulation, patients were placed in a supine position and a computerized tomography (CT) scan was performed for treatment planning using 1-mm slice thickness. Patient was immobilized using a thermoplastic mask (Fiberplast®, QFix, Avondale, PA) mask, a custom head rest (MoldCare®, QFix, Avondale, PA), and a knee cushion with hands holding pegs along their side or a ring across their chest for reproducibility and comfort. Diagnostic magnetic resonance (MRI) including a T1 post-Gadolinium (Gd) and a T2-weighted fluid-attenuated inversion recovery (T2/FLAIR) sequences were also performed and co-registered to the treatment planning CT scan for target volume delineation purposes.
Target volumes and organs-at-risk determination and contouring
Planning and simulation images (MR and CT images) were co-registered in the treatment planning system (TPS)—RayStation v.9A (RaySearch Laboratories, Stockholm, Sweden)—for delineation of target volume and organs-at-risk (OARs).
For target volumes, the gross tumor volume (GTV) was defined as the T1 contrast enhanced lesion and resection cavity. For glioma patients, the FLAIR abnormality was also included in the GTV. The clinical target volume (CTV) was defined as a variable GTV expansion, from 0 cm (inclusion of FLAIR only) to 1.5 cm isotropic expansion, limited by anatomical barriers (bone, contralateral brain, brainstem, and optic tract).
For OARs, the uninvolved brain (total brain minus the CTV), brainstem, optic chiasm, ipsilateral and contralateral optic nerves, ipsilateral and contralateral cochlea, and ipsilateral and contralateral hippocampus were contoured.
Treatment planning and delivery
For the TMPPR technique, reRT was planned and delivered using intensity-modulated proton therapy (IMPT) and a modified single field optimization (SFO) approach with 3 fields, where 70% of the total dose was controlled by each beam and the remaining 30% modulated for OAR sparring. Plans were robustly optimized to the CTV and evaluated using 3.5% range uncertainty and 3 mm setup uncertainty on a 2 mm dose grid. Beam angle selection was performed to ensure a large hinge angle among the three fields and at most only one of the three fields end ranging into an OAR. Each field was re-painted once, resulting in six equally weighted sub-fields with each delivering approximately a maximum dose of 0.3 Gy/fraction to the CTV. Beam maximum dose contribution was capped at 0.4 Gy per fraction to limit modulation. Adjusting for the time delay among each field (5 min) yielded an overall time-averaged effective dose rate of < 7 cGy/min.
All CT datasets, dose distributions, and structure sets from previous radiotherapy courses were imported into the RayStation TPS to generate composite dose distributions for dosimetric evaluation. After which, the cumulative equivalent dose in 2-Gy fractions (EQD2) distributions to each OAR were assessed for prior and current treatment plans. The following dose parameters were subsequently extracted: average dose, maximum dose to 0.03 cc, maximum dose to 0.5 cc, and maximum dose to 1 cc in EQD2 values for OARs, including the brain (excluding the CTV), brainstem, optic chiasm, ipsilateral and contralateral optic nerves, ipsilateral and contralateral cochlea, and ipsilateral and contralateral hippocampus, as well as the target volumes.
For daily positioning, a cone-beam CT (CBCT) was performed and aligned to the reference CT for an accurate and precise administration of the dose to the target volumes. Patient position corrections were enabled using a 6 degree of freedom couch. The proton treatment was delivered by Proteus® PLUS Proton Therapy accelerator (Ion Beam Applications S.A.). The delivery time for each subfield and subsequently delay was annotated and documented in the treatment records for each fraction.
Treatment monitoring and follow-up
Weekly monitoring of acute toxicities occurred during treatment. Follow-up evaluations were conducted 4–6 weeks after radiotherapy completion, followed by subsequent assessments every 2–3 months. These evaluations involved clinical examination and the use of contrast-enhanced brain MRI. We employed the Response Assessment in Neuro-Oncology (RANO) radiological response criteria to assess treatment response, including complete response (CR), partial response (CR), stable disease (SD), or progressive disease (PD)22. Toxicity grades were assessed based on the criteria outlined in the National Cancer Institute CTCAE v5.0. Radiation necrosis was defined as the emergence or enlargement of contrast enhancement in the region previously treated with radiotherapy, excluding recurring tumors, and verified by evaluation of multiparametric imaging and advanced options, such as MR perfusion23. These cases were thoroughly discussed in a multidisciplinary tumor conference involving specialists from various fields such as neuroradiology, neurosurgery, neuro-oncology, and radiation oncology. The objective was to reach a consensus among physicians by considering all relevant treatment details and comparing the dose and imaging to differentiate between tumor progression and radiation necrosis.
Pattern of recurrence by localization
We utilized the Dose-Volume Histogram (DVH) to determine the spatial relationship between the recurrent tumor volume and the delivered dose distribution (prior irradiation). The DVH analysis enabled classification of recurrences into different categories based on their location. If more than 95% of the recurrence volume was found within the original high-dose field according to the DVH, it was categorized as a central failure. Recurrences with a distribution of more than 80% to 95%, 20% to 80%, and less than 20% were designated as in-field, marginal, and distant recurrences, respectively24.
Statistical analysis
Descriptive statistics were computed. For continuous variables, the median and range were presented. Sample sizes and percentages were computed for categorical variables.
Ethical statement
The Miami Cancer Institute (Baptist Health South Florida) Institutional Review Board (IRB) approved the protocol study (2083553-1) and waived the need for informed consent from all subjects and/or their legal guardian(s).
Results
Patient’s characteristics
The first five consecutive patients treated with TMPPR for recurrent high-grade intracranial malignancies are included in this analysis (Table 1). The median age was 54 years (range [R]: 32–72 years), and 3 patients (60%) were females.
The first course of radiation was delivered with proton-based RT for three cases and photon-based RT for two cases, with a median dose and dose per fraction of 60 Gy(RBE) (R: 59.4–75 Gy[RBE]) and 2 Gy(RBE) (R: 1.8–2.5 Gy[RBE]), respectively. Four patients received concurrent temozolomide (TMZ). The locations were solely frontal in two cases, two frontotemporal, and one frontoparietal. Four lesions were right-sided, and one left-sided.
The most common histology for prior to reRT was WHO grade 4 glioblastoma (IDH wild-type) in three patients, followed by one with WHO grade 4 astrocytoma (IDH mutated; initially diagnosed as oligodendroglioma/astrocytoma [IDH1 R132H, TP53 and ATRX mutated; negative for 1p/19q]), and one with WHO grade 3 supratentorial ependymoma.
Reirradiation characteristics (TMPPR)
All patients had at least 1 recurrence prior to TMPPR (median 3 recurrences, R: 1–6) for which they underwent one prior course of radiotherapy, at least one surgery (median: 3, R:1–4) and one line of systemic therapy (median: 2, R:1–4). The location of recurrence/progression was marginal to the prior RT field (since the recurrence volume within the high-dose field from previous radiotherapy was between 20 and 80%) in all 5 patients (see Fig. 1). The local treatment was TMPPR alone in 4 cases, and TMPPR following surgical resection in 1 case. The median CTV was 298.9 cc (R: 109.1–442.6 cc), with a median total dose of 54 Gy(RBE) (R: 50.4–59.4 Gy[RBE]) in a median of 30 fractions (range: 28–33). All five patients tolerated TMPPR treatment without any delay or acute complication, demonstrating that the technique is feasible in a clinical setting. No cases were planned but could not proceed due to feasibility, technical, or logistical issues.
Case number 1. Proton isodose distribution in axial, sagital and coronal views for the first (initial) course of proton radiation therapy and re-irradiation with the novel TMPPR technique in a multiprogressive high-grade glioma. As seen, reirradiation volumes completely overlap with prior radiation fields but with limited dose to the surrounding normal brain.
The TMPPR course doses and cumulative doses received by the OARs are described in Table 2. The brainstem received median cumulative doses for D0.03cc, D0.5cc, D1cc, and Dmean of 107.4 Gy(RBE) (range: 0.7–133.1), 100.3 Gy(RBE) (range: 0.6–132.5), 92.6 Gy(RBE) (range: 0.2–131.7), and 28.9 Gy(RBE) (range: 0.1–45.2), respectively. For the uninvolved brain, the median cumulative doses for D0.03cc, D0.5 cc, D1cc, and Dmean were 117.3 Gy(RBE) (range: 107.4–132.4), 116.3 Gy (RBE) (range: 104.5–132.2), 115.8 Gy (RBE) (range: 100.4–131.5), and 30.8 Gy (RBE) (range: 10.6–45.2), respectively. For the optic chiasm, the median cumulative doses for D0.03cc and Dmean were 67.2 Gy (RBE) (range: 0.6–102.9) and 39.5 Gy (RBE) (range: 0.3–98.8), respectively. Finally, for the ipsilateral optic nerve, the median cumulative doses for D0.03 cc and Dmean were 65.9 Gy (RBE) (range: 3.8–102.7) and 44.5 Gy (RBE) (range: 1.1–75.9), respectively.
Daily fractions consisted in the delivery of three fields, each one subdivided in two subfields, with a total of six subfields. The approximated total time for each daily fraction was of 39.5 min, as shown in Fig. 2. Initial positioning and imaging time was 10 min; considering the prolonged time of each fraction, an interim imaging for positioning reassurance was obtained between the third and fourth subfield delivery (overlapping with the 3.5 min of waiting time). Beam-on time for each subfield was approximately 2 min (total beam-on time of 12 min). Finally, the wait time between each beam-on time per subfraction delivered was 3.5 min (total of 17.5 min).
Diagram describing the temporal distribution sequence of each field delivery during a 3-field TMPPR treatment fraction.
Clinical outcomes and treatment-related toxicity
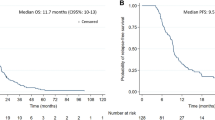
All patients tolerated the TMPPR treatments well. The median follow-up was of 8 months (R: 1–9), and all patients had at least one MR scan after TMPPR completion. The MRI of one of the patients was required 10 days after the end of TMPPR treatment, due to the presence of worsening neurologic symptoms compatible with progression, in central location to the TMPPR. This patient was diagnosed with progressive disease and was referred to a hospice.
Best objective radiological response consisted of one CR, three PR, and one PD. The first patient had the scan while progressing on bevacizumab and ivosidenib and no corticosteroids, revealing a dramatic resolution of the large multi-lobulated enhancing mass, substantial reduction in the infiltrating T2/FLAIR signal, resolution of mass effect on the right lateral ventricle, and absence of elevated relative cerebral blood volume (rCBV) on the perfusion MR (which was previously elevated). These imaging features were consistent with a complete response to treatment.
All the patients (100%) developed grade 2 alopecia (one with associated grade 1 fatigue, and other with headache grade 1). In one case, symptomatic radiation necrosis grade 2 occurred. This patient received bevacizumab with subsequent radiographic improvement on second follow-up MRI (12-week post-TMPPR) compared to the first follow-up MRI (4-week post-TMPPR), as shown in Fig. 3.
Case number 5. Axial slices in three anatomic sequences (T2, T2/FLAIR, and T1-post contrast), and one functional (perfusion, cerebral blood flow [CBV]) demonstrating retraction of the surgical cavity and significant reduction of the heterogenoeus enhacement. Perfusion imaging continued to demonstrate lack of elevated CBV, consistent with radiation necrosis vs. tumor progression.
Discussion
Intracranial reRT presents a challenging situation for which no consensus exists regarding cumulative dose evaluation, or for optimal dose and fractionation25,26. From different reRT techniques, PRDR, classically delivered with photons, has shown promise of delivering reRT by enhancing its lethal effect on tumor cells and allowing normal tissue to recover from sublethal damage, however moderate treatment-related toxicities have been described15. In an effort to reduce these photon PRDR toxicities, we postulated that pulsed-reduced doses delivered with protons (a technique we have designated “temporally modulated proton pulsed re-irradiation” or TMPPR, patent pending 63/484,082) as an alternative for these groups of patients due to the reduced low and intermediate doses afforded by proton therapy to the surrounding (often previously treated) tissues. In this study of our first five patients treated with TMPPR, we have demonstrated the feasibility of TMPPR plan generation, promising outcomes (best overall objective response rate [ORR] of 80%, where one patient achieved a complete response), and limited treatment-related toxicities (grade 2 alopecia for all patients and one case of grade 2 radiation necrosis) as a reRT technique in patients with high grade intracranial disease, validating our previous treatment planning work, and supporting the potential benefit into clinical adoption19.
Currently guidelines consider reRT as a possible approach (low level of evidence), among other options as a local treatment for recurrent glioblastoma. ReRT can be delivered using a variety of techniques, from stereotactic radiosurgery (SRS), fractionated SRS, hypofractionated treatment, conventionally fractionated photon therapy, or even particle therapy6. SRS, supported by multiple retrospective datasets, has been traditionally delivered to recurrent limited target volumes (4–10 cc)27. Furthermore, multiple retrospective and prospective series have investigated fractionated SRS (FSRS) or hypofractionated schedules for recurrent glioblastoma, where the target volumes were fairly modest at 8.5–34 cc (FSRS) and 33–145 cc (hypofractionated reRT)9,27. Conventionally fractionated reRT has also been used (most commonly 36 Gy in 18 fractions) for larger volume recurrences and associated with lower rates of radiation necrosis when compared to more hypofractionated schedules (including SRS/FSRS)26. Taking into account the large treatment volumes involved in our patients (range: 109.1–442.6 cc), too large for hypofractionation, and the close anatomical relation to critical structures, we considered the benefit of combining the radiobiological benefit of delivering TMPPR with the use of protons to deliver meaningful dose to the tumor while sparing the remainder of the uninvolved brain28.
Promising responses were seen in our group of patients (1 CR and 3 PR). One of our patients achieved a CR, as her follow-up showed a clinical improvement associated with imaging findings of a regression of all enhancing and a significant component of the non-enhancing disease (T1 enhancement and associated FLAIR infiltration) as well as resolution of midline shift and mass effect on the adjacent brain parenchyma and the absence of a prior elevated cerebral blood volume (rCBV) on the perfusion MRI. At her current follow-up (now 5 months from TMPPR), she developed leptomeningeal failure in the contralateral untreated brain (previously no disease; Fig. 4). Interestingly, a second patient treated with TMPPR also exhibited a PR to treatment at the area of TMPPR in the right frontal lobe but also has leptomeningeal failure on the contralateral brain on longer follow-up. Whether the enhanced effect of TMPPR can change the pattern of disease progression—from local only to distant failures—deserves further follow-up and study.
Example of a patient with leptomeningeal disease development after prior TMPPR, with development of numerous enhancing nodules in periventricular and parafalcine locations, three of three are highlighted by green arrows.
Overall, three of our patients partially respond to TMPPR and those patients received systemic therapy peri-RT as well. These outcomes resulted in a high overall responses rate not seen in prior studies with other strategies of treatment. Furthermore, the results of clinical trials for systemic therapies in high-grade glioma patients at first recurrence or progression have shown varying levels of success, with ORR ranging from 5 to 41.5%29,30. However, substantial responses or CR to these treatments are uncommon, with none being observed for lomustine alone and only 2.4% for the combination of bevacizumab and irinotecan31,32. When comparing bevacizumab versus nivolumab, Reardon et al. found, for bevacizumab 2.6% CR and 20.5% PR33. Moreover, an ORR of only 35% (CR in one case and 17 cases of PR’s) was seen in another study with bevacizumab alone34. Novel systemic therapies have been studied in clinical trial settings with modest activity in high-grade gliomas as shown in Table 3. Recently, the Checkmate 143 trial, comparing the use of nivolumab vs. bevacizumab at first recurrence did not demonstrate a benefit in OS (9.8 vs. 10 months), moreover, the ORR was higher in bevacizumab arm (7.8 vs. 23.1%), with a CR observed in only 2 of 153 (1.3%) patients receiving nivolumab33. Also, the combination of nivolumab with ipilimumab did not show any CR and almost no difference with nivolumab alone for PR (2 versus 1, respectively)35.
Recently, Ellingson et al. described the following ORRs for different therapies from past clinical trials (68 treatments arms; 4793 patients) for recurrent high-grade gliomas (≤ 3 recurrences, but great majority were first recurrences): chemotherapy 6.1%, biological agents 3.37%, immunotherapies 7.97%, and antiangiogenics 26.8%. The combination of data from chemotherapy, biologics, and immunotherapy suggested a strong correlation between ORR and median OS, with ORR values greater than 25% leading to a median OS of over 15 months4. Given the encouraging response rates in our limited preliminary series of patients and this recently established relationship, a clinical trial for recurrent glioma patients powered for overall survival is currently in development.
There remains a lack of consensus regarding reRT to the brain in terms of tolerance of critical organs and the potential for debilitating or even fatal effects36. Mayer et al. conducted an overview based on clinical data, by comparing several prior studies on the tolerance of the brain to reRT for gliomas. They found an increased risk for radiation necrosis when the equivalent dose in 2-Gy fractions (EQD2) was over 100 Gy. It is important to note that cumulative doses to the normal brain were only reported, not considering the total target volumes involved37. Stiefel et al. conducted a retrospective analysis in 76 patients (including primary tumors and metastasis) with at least two prior courses of radiotherapy. For patients exceeding the D0.1 cc of 100 Gy (EQD2) to the brain, the median D0.1 cc was 114 Gy (range 100–161.5) and resulted in only 2 cases of high-grade (> grade 3) toxicity. Also, they concluded that keeping cumulative doses to the brain up to 120 Gy EQD2, below 100 Gy for brainstem and below 75 Gy EQD2 to chiasm and optic nerves was safe38. Prior studies of photon PRDR have traditionally limited the dose of the brainstem or optic chiasm to 50 Gy in the re-irradiation, regardless the dose received in the prior course of radiotherapy11. In our case, considering the large volumes of reRT (median CTV of 298 cc), and the high median maximal cumulative doses to critical structures (brain [D0.5 cc= 116.3 Gy(RBE), and D1cc = 115.8 Gy(RBE)], brainstem [D0.5 cc = 100.3 Gy(RBE), and D1cc = 92.6 Gy(RBE)], optic chiasm [D0.03 cc = 65.9 Gy(RBE)], and ipsilateral optic nerve [D0.03 cc = 67.2 Gy(RBE)], this novel technique was able to deliver an appropriate definitive dose without any severe treatment-related acute toxicity (alopecia grade 2 and one case of radiation necrosis grade 2), although further follow-up is pending at this time.
Our study has several limitations that need to be considered. Firstly, it is important to note that this retrospective study was conducted in a single institution and involved a limited number of patients. Therefore, due to the heterogeneity of the patients included, the prior treatments received by the patients and treatments after TMPPR were not standardized. This variation in treatment approaches may have influenced the results and should be considered when interpreting the findings. Also, since all patients received systemic therapy peri-TMPPR, the outcomes cannot be ascribed to just a single treatment modality. Lastly, to establish definitive conclusions regarding the long-term safety and efficacy, longer follow-up is necessary and currently ongoing. Additional data collected over an extended period would provide a more comprehensive understanding of the treatment outcomes and potential risks associated with TMPPR.
Conclusion
ReRT with TMPPR for recurrent CNS malignancies workflow and delivery demonstrated to be safe and feasible. Also, it was associated with promising tumor responses and modest treatment-related toxicity, in the first five reported patients treated with this technique in our institution. Further prospective studies are needed to evaluate the outcomes in terms of survival and long-term treatment-related toxicities of patients treated with this technique.
Data availability
The data that support the findings of this study are available from the corresponding author upon reasonable request.
References
Ostrom, Q. T., Price, M., Neff, C., et al. CBTRUS statistical report: Primary brain and other central nervous system tumors diagnosed in the United States in 2015–2019. Neuro-Oncology. 2022; 24(Supplement_5):v1–v95.
Grossman, S. A. et al. Survival of patients with newly diagnosed glioblastoma treated with radiation and temozolomide in research studies in the United States. Clin Cancer Res. 16(8), 2443–2449 (2010).
Chan, J. L. et al. Survival and failure patterns of high-grade gliomas after three-dimensional conformal radiotherapy. J Clin Oncol. 20(6), 1635–1642 (2002).
Ellingson BM, Wen PY, Chang SM, et al. Objective response rate (ORR) targets for recurrent glioblastoma clinical trials based on the historic association between ORR and median overall survival. Neuro Oncol. 2023.
Shi, W. et al. Investigating the effect of reirradiation or systemic therapy in patients with glioblastoma after tumor progression: A secondary analysis of NRG oncology/radiation therapy oncology group trial 0525. Int J Radiat Oncol Biol Phys. 100(1), 38–44 (2018).
National Comprehensive Cancer Network. Central Nervous System Cancers (Version 1.2023). 2023; https://www.nccn.org/professionals/physician_gls/pdf/cns.pdf. Accessed 4 April 2023.
Scoccianti, S. et al. Re-irradiation as salvage treatment in recurrent glioblastoma: A comprehensive literature review to provide practical answers to frequently asked questions. Crit Rev Oncol Hematol. 126, 80–91 (2018).
Straube, C. et al. A second course of radiotherapy in patients with recurrent malignant gliomas: Clinical data on re-irradiation, prognostic factors, and usefulness of digital biomarkers. Curr Treat Options Oncol. 20(9), 71 (2019).
Tsien, C. I., Pugh, S. L., Dicker, A. P., et al. NRG oncology/RTOG1205: A randomized phase II trial of concurrent bevacizumab and reirradiation versus bevacizumab alone as treatment for recurrent glioblastoma. J Clin Oncol. 2022:Jco2200164.
Ma CMC. Pulsed low dose-rate radiotherapy: radiobiology and dosimetry. Phys Med Biol. 2022; 67(3).
Bovi, J., Prah, M., Retzlaff, A., et al. Pulsed reduced dose rate radiotherapy in conjunction with bevacizumab or bevacizumab alone in recurrent high-grade glioma: survival outcomes. Int. J. Radiat. Oncol. Biol. Phys. 2020; 108(4).
Prabhu, A., Lee, M., Galhardo, E., Newkirk, M., Rodriguez, A., Xia, F. Pulsed reduced dose-rate radiotherapy for previously irradiated tumors in the brain and spine. Surg. Neurol. Int. 2021; 12.
Adkison, J. B. et al. Reirradiation of large-volume recurrent glioma with pulsed reduced-dose-rate radiotherapy. Int. J. Radiat. Oncol. 79, 835–841 (2011).
Cannon, G., Tomé, W., Robins, H., Howard, S. Pulsed reduced dose-rate radiotherapy: Case report: A novel re-treatment strategy in the management of recurrent glioblastoma multiforme. J. Neuro-oncology. 2007; 83(3).
Kutuk, T., Tolakanahalli, R., McAllister, N., et al. Pulsed-reduced dose rate (PRDR) radiotherapy for recurrent primary central nervous system malignancies: dosimetric and clinical results. Cancers. 2022; 14(12).
Doyen, J., Falk, A. T., Floquet, V., Hérault, J. & Hannoun-Lévi, J. M. Proton beams in cancer treatments: Clinical outcomes and dosimetric comparisons with photon therapy. Cancer Treat Rev. 43, 104–112 (2016).
Gaito, S. et al. Normal tissue complication probability modelling for toxicity prediction and patient selection in proton beam therapy to the central nervous system: A Literature review. Clin Oncol (R Coll Radiol). 34(6), e225–e237 (2022).
Mohan, R., Das, I. J., Ling, C. C. Empowering intensity modulated proton therapy through physics and technology: An overview. Int. J. Radiat. Oncol.*Biol.*Phys. 2017; 99(2), 304–316.
Wroe, A., Fellows, Z., Kutuk, T., Mehta, M. P., Kotecha, R., Gutierrez, A. Evaluation of pulsed reduced dose rate (PRDR) intensity modulated proton therapy (IMPT) for re-irradiation of CNS malignancies. Int. J. Radiat. Oncol.*Biol.*Phys.. 2022; 114(3, Supplement), e540.
Galle, J. O., McDonald, M. W., Simoneaux, V. & Buchsbaum, J. C. Reirradiation with proton therapy for recurrent gliomas. Int. J. Particle Therapy. 2(1), 11–18 (2015).
La Rosa, A., Gutierrez, A. N., Odia, Y., et al. Temporally modulated pulsed proton re-irradiation (TMPPR) for recurrent high-grade gliomas. Neurooncol Adv. 2023; 5(1):vdad074.
Wen, P. Y. et al. Updated response assessment criteria for high-grade gliomas: Response assessment in neuro-oncology working group. J Clin Oncol. 28(11), 1963–1972 (2010).
Nael, K. et al. Multiparametric MRI for differentiation of radiation necrosis from recurrent tumor in patients with treated glioblastoma. Am. J. Roentgenol. 210(1), 18–23 (2018).
Chan, J. L. et al. Survival and failure patterns of high-grade gliomas after three-dimensional conformal radiotherapy. J. Clin. Oncol. 20(6), 1635–1642 (2002).
Lee, J., Cho, J., Chang, J. H. & Suh, C.-O. Re-irradiation for recurrent gliomas: treatment outcomes and prognostic factors. Yonsei Med J. 57(4), 824–830 (2016).
Howard, S. P., Krauze, A., Chan, M. D., Tsien, C. & Tomé, W. A. The evolving role for re-irradiation in the management of recurrent grade 4 glioma. J. Neuro-Oncol. 134(3), 523–530 (2017).
Minniti, G., Niyazi, M., Alongi, F., Navarria, P., Belka, C. Current status and recent advances in reirradiation of glioblastoma. Radiat. Oncol. (London, England). 2021; 16(1).
La Rosa, A., Gutierrez, A. N., Odia, Y., et al. Temporally-modulated pulsed proton re-irradiation (TMPPR) for recurrent high-grade gliomas. Neuro-Oncol. Adv. 2023.
Taal, W. et al. Single-agent bevacizumab or lomustine versus a combination of bevacizumab plus lomustine in patients with recurrent glioblastoma (BELOB trial): A randomised controlled phase 2 trial. Lancet Oncol. 15(9), 943–953 (2014).
Wick, W. et al. Lomustine and bevacizumab in progressive glioblastoma. N Engl J Med. 377(20), 1954–1963 (2017).
Batchelor, T. T. et al. Phase III randomized trial comparing the efficacy of cediranib as monotherapy, and in combination with lomustine, versus lomustine alone in patients with recurrent glioblastoma. J Clin Oncol. 31(26), 3212–3218 (2013).
Friedman, H. S. et al. Bevacizumab alone and in combination with irinotecan in recurrent glioblastoma. J Clin Oncol. 27(28), 4733–4740 (2009).
Reardon, D. A. et al. Effect of nivolumab vs bevacizumab in patients with recurrent glioblastoma: The CheckMate 143 phase 3 randomized clinical trial. JAMA Oncol. 6(7), 1003–1010 (2020).
Kreisl, T. N. et al. Phase II trial of single-agent bevacizumab followed by bevacizumab plus irinotecan at tumor progression in recurrent glioblastoma. J Clin Oncol. 27(5), 740–745 (2009).
Omuro, A. et al. Nivolumab with or without ipilimumab in patients with recurrent glioblastoma: results from exploratory phase I cohorts of CheckMate 143. Neuro Oncol. 20(5), 674–686 (2018).
Andratschke, N. et al. European Society for Radiotherapy and Oncology and European Organisation for Research and Treatment of Cancer consensus on re-irradiation: Definition, reporting, and clinical decision making. Lancet Oncol. 23(10), e469–e478 (2022).
Mayer, R., Sminia, P. Reirradiation tolerance of the human brain. Int. J. Radiat. Oncol.*Biol.*Phys. 2008; 70(5):1350–1360.
Stiefel, I. et al. High-dose re-irradiation of intracranial lesions: Efficacy and safety including dosimetric analysis based on accumulated EQD2Gy dose EQD calculation. Clin Transl Radiat Oncol. 27, 132–138 (2021).
Perry, J. R. et al. Phase II trial of continuous dose-intense temozolomide in recurrent malignant glioma: RESCUE study. J Clin Oncol. 28(12), 2051–2057 (2010).
Brada, M. et al. Temozolomide versus procarbazine, lomustine, and vincristine in recurrent high-grade glioma. J Clin Oncol. 28(30), 4601–4608 (2010).
Wick, W. et al. Phase III study of enzastaurin compared with lomustine in the treatment of recurrent intracranial glioblastoma. J Clin Oncol. 28(7), 1168–1174 (2010).
Funding
This research received no external funding.
Author information
Authors and Affiliations
Contributions
Conceptualization, A.L.R. and R.K.; methodology, A.L.R., Z.F., N.M., R.T., T.K., A.J.W., L.C., E.P., and R.K.; formal analysis, A.L.R. and R.K..; investigation, A.L.R.; data curation, A.L.R.; writing—original draft preparation, A.L.R. and R.K.; writing—review and editing, A.L.R., Z.F., N.M., R.T., T.K., A.J.W., L.C., E.P., M.D.H., R.H.P., M.W.D., Y.O., M.S.A., M.P.N., A.N.G., and R.K. All authors have read and agreed to the published version of the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Institutional review board statement
The study was conducted in accordance with the Declaration of Helsinki and exempt by the Institutional Review Board of Miami Cancer Institute (protocol code 2083553-1; July 21, 2023).
Patent
Pending 63/484,082.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
La Rosa, A., Fellows, Z., Wroe, A.J. et al. Initial feasibility cohort of temporally modulated pulsed proton re-irradiation (TMPPR) for recurrent high-grade intracranial malignancies. Sci Rep 14, 26685 (2024). https://doi.org/10.1038/s41598-024-78370-x
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-024-78370-x