Abstract
Resting heart rate (RHR) has prognostic implications in heart failure with reduced ejection fraction, where ≤ 70 bpm is targeted. Whether a RHR > 70 bpm assessed within clinical practice goes along with elevated cardiovascular risk in implantable cardioverter-defibrillator (ICD) / cardiac resynchronization therapy-defibrillator (CRT-D) recipients remains incompletely understood. A total of 1589 patients (ICD n = 1172 / CRT-D n = 417, median age 65 years, 22.6% female) undergoing ICD/CRT-D implantation or revision in the prospective German DEVICE multicenter registry were analyzed. RHR was assessed via a 12-channel electrocardiogram at enrollment. 1-year outcomes (all-cause mortality, major cardio- and cerebrovascular events (MACCE), all-cause hospital admission) were compared between patients with a RHR ≤ 70 bpm and > 70 bpm. 733 patients (46.1%) showed a RHR > 70 bpm. Median RHR was 63 (interquartile range 59; 68) bpm (≤ 70 bpm group) and 80 (75; 89) bpm (> 70 bpm group). Heart failure with reduced ejection fraction was present in 76.3%, a prior myocardial infarction in 32.4% and non-ischemic heart disease in 44.9%. One-year all-cause mortality was similar between RHR groups (≤ 70 bpm 5.4% vs. > 70 bpm 5.4%, p = 0.96), and subgroup analysis regarding patient characteristics and comorbidities revealed only a significantly higher rate of patients with dual chamber ICD in the > 70 bpm group (0.8% vs. 9.2%, p = 0.003). MACCE (5.9% vs. 6.1%, p = 0.87) and defibrillator shock rates (9.9% vs. 9.8%, p = 1.0) were similar. Higher all-cause hospital admission rates were observed in patients with > 70 bpm RHR (23.1% vs. 29.0%, p = 0.027) driven by non-cardiovascular events (6.0% vs. 11.7%, p = 0.001). In conclusion, in ICD and CRT-D recipients a RHR at admission > 70 bpm may indicate patients at increased risk of all-cause hospital admission but not of other adverse cardiovascular events or death at 1-year follow-up.
Similar content being viewed by others
Introduction
Resting heart rate (RHR) reflects a simple non-invasive parameter indicating autonomic nervous tone, which is well known to predict cardiovascular risk in health and cardiovascular diseases including coronary artery disease and heart failure1,2,3,4,5. A therapeutic target has been defined at ≤ 70 bpm for heart failure with reduced ejection fraction (HFrEF) and with a left ventricular ejection fraction (LVEF) of ≤ 35% in current European as well as North American guidelines6,7. Results from the randomized BEAUTIFUL and SHIFT trials contributed to this cut-off value, as a baseline RHR > 70 bpm identified patients with heart failure due to ischemic cardiomyopathy at increased risk for cardiovascular events and death4. Reduction of initially elevated RHR > 70 bpm resulted in fewer events of cardiovascular death or hospital admission due to heart failure8,9.
In patients with an implantable cardioverter-defibrillator (ICD), inconclusive findings have been reported throughout various populations, with RHR cut-off values for prediction of adverse cardiovascular events ranging between 63 and 80 bpm10,11,12. Therefore, we aimed to investigate whether a RHR elevation > 70 bpm measured via a single 12-channel electrocardiogram within clinical practice is associated with an increased risk of adverse cardiovascular events in ICD and cardiac resynchronization therapy-defibrillator (CRT-D) recipients.
Results
Study cohort
A total of 1589 patients were included into the analysis (Fig. 1). Of these, 733 (46.1%) presented with a RHR > 70 bpm. ICD implantation was conducted in a total of 1172 patients (73.8% of all patients, dual-chamber ICD in 18.3% of implanted ICDs) and 417 patients received a CRT-D device. A first-time ICD or CRT-D implantation was conducted in 1495 patients (94.1%), while the remaining patients underwent a revision procedure of an already implanted ICD / CRT-D device or leads during enrollment. The procedure was scheduled electively in 90.7% of patients. HFrEF (LVEF < 40%) was present in 76.3% of patients, with 74.6% of patients displaying a LVEF ≤ 35%. Overall observed cardiac heart disease included prior myocardial infarction (32.4%), dilated cardiomyopathy (33.2%), hypertrophic cardiomyopathy (3.8%), hypertensive cardiomyopathy (7.0%) and valvular heart disease (0.9%). ICD/CRT-D implantation followed a secondary prevention indication in 36.2% of patients. A high MADIT-ICD ventricular tachycardia (VT) / ventricular fibrillation (VF)-score ≥ 7 was present in 50.0% of patients, while the MADIT-ICD non-arrhythmic mortality score was ≥ 3 in 35.5%.
Study flowchart. Main patient selection criteria are displayed. CRT-D cardiac resynchronization therapy-defibrillator, ICD implantable cardioverter-defibrillator.
Patient characteristics
Tables 1, 2 and 3 display baseline characteristics, discharge medication and procedure characteristics comparing patients with a RHR ≤ 70 bpm vs. > 70 bpm. The median RHR was 63 bpm (interquartile range 59; 68 bpm) in the ≤ 70 bpm group and 80 bpm (75; 89 bpm) in the > 70 bpm group. In the ≤ 70 bpm group, there were more male patients (p = 0.049) and patients presented more frequently with a prior myocardial infarction (p = 0.027) or hypertrophic cardiomyopathy (p = 0.042). In the > 70 bpm group, diabetes mellitus (p = 0.007) and dilated cardiomyopathy (p < 0.001) were more frequently present, and the MADIT-ICD VT/VF score and non-arrhythmic mortality score were significantly higher (p < 0.001; p = 0.006). Further baseline characteristics were similar between both groups.
Periprocedural and 1-year outcomes
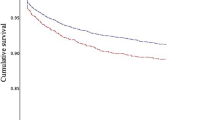
Table 4 shows periprocedural as well as 1-year outcomes in the overall cohort. Periprocedural mortality was low at 0.2% (≤ 70 bpm) vs. 0.1% (> 70 bpm, p = 1.0), with a median hospitalization duration of 3 days in both groups (p = 0.99). The 1-year all-cause mortality did not differ between groups (p = 0.96), and subgroup analysis revealed only a significantly higher rate of 2-chamber ICDs in the > 70 bpm cohort (Table 5). The combined rates of death and defibrillator shocks as well as occurrence of major adverse cardiac and cerebrovascular events (MACCE) were similar between groups (p = 0.87 / p = 0.87). The 1-year all-cause hospital admission rate was higher in the > 70 bpm group (23.1% vs. 29.0%, p = 0.027) with a similar median number of hospitalization days (≤ 70 bpm 7 days (4; 15 days) vs. > 70 bpm 8 days (4; 21 days), p = 0.13). This was driven by hospitalization due to non-cardiovascular causes (≤ 70 bpm 6.0% vs. > 70 bpm 11.7%, p = 0.001) as hospitalization due to cardiovascular causes showed no significant difference between groups. Non-lethal cardiovascular events including survived cardiopulmonary resuscitation, myocardial infarction, stroke, defibrillator shock, electrical storm episodes and necessary ventricular arrhythmia ablation were similarly often observed between groups.
Discussion
In the present study, we analyzed the relationship of the baseline RHR at admission assessed within clinical practice to cardiovascular outcomes in patients undergoing de novo ICD or CRT-D implantation or revision enrolled in the German DEVICE registry. The main findings are that (1) a RHR > 70 bpm was not associated with an increased occurrence of cardiovascular events or death at 1-year follow-up and (2) increased all-cause hospital admission rates were observed in patients with a RHR > 70 bpm in patients with an ICD or CRT-D.
Our results may appear contradicting compared to three large studies previously published, which described an association between RHR elevation and cardiovascular outcomes including mortality and occurrence of ventricular arrhythmias in ICD/CRT-D recipients10,11,12. However, they may be explained with the investigated cohort: Less than 33% of patients presented with a prior myocardial infarction. This is of importance, as event rate and outcomes significantly differ between ischemic and non-ischemic cardiomyopathies. Almost two-third of included patients presented with a primary prevention indication for defibrillator implantation as well as non-ischemic cardiomyopathy, and event rates were lower in this collective as observed in the DANISH trial13. Furthermore, almost 20% of included patients did not present with HFrEF and approximately 25% of patients presented with a LVEF > 35%, which were not included in the SHIFT trial9 and whose percentage appears higher compared to previous investigations of approximately 15%11. Additionally, we only included patients in sinus rhythm and without atrioventricular block, which was not specified in previous studies on RHR in defibrillator recipients11,12. Atrial fibrillation is now well known to represent a possible confounder as affected patients are at increased risk for cardiovascular events14 and a different target RHR of < 110 bpm is recommended for patients with atrial fibrillation where rhythm control is not possible15,16.
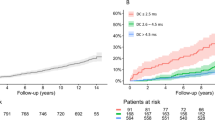
Tailored RHR management has been shown to be of importance also regarding lower heart rate values in ICD and CRT-D recipients depending on the implanted device type and patient comorbidities. Low RHR may indicate chronotropic incompetence, and low heart rate variation in CRT-D patients was described to be an independent predictor of cardiovascular mortality17,18,19. Individualized accelerated pacing was proposed to be beneficial in patients with sick sinus syndrome or heart failure with preserved ejection fraction in previous studies20,21. However, our presented subgroup analysis regarding 1-year mortality shows divergent observations: Patients with a normal systolic left ventricular function did not reveal a significant difference between groups and a negative effect of a RHR > 70 bpm on observed mortality might be more pronounced in the subgroup of patients with a dual chamber ICD. While these observations are hypothesis-generating only and need to be interpreted with caution, there might not be a single “one-size-fits-all” RHR cut-off for ICD as well as CRT-D recipients and individualized RHR management needs to be determined in future investigations.
Limitations
First, as the presented data are derived from a registry, the results are hypothesis-generating only and a selection bias cannot be ruled out. As only patients with sinus rhythm were included, no conclusions can be derived for patients with sustained atrial arrhythmias or atrioventricular block. Baseline RHR may have influenced by the following parameters which were not systematically assessed in the registry: resting time of patients until the baseline 12-channel ECG was recorded, beta blocker dosages and device lower rate settings. However, a previous large analysis showed that the most pronounced heart rate variation during orthostatic changes occurs within the first 30 s in healthy subjects and patients with cardiovascular diseases, which exceeds the time needed for an ECG recording22. Beta blocker dosages were targeted to the highest tolerated dosage in all patients. Lower rate settings may have only minorly have influenced both the ICD cohort as all patients with any atrioventricular block were excluded and the CRT cohort as a baseline RHR of ≤ 70 bpm vs. > 70 bpm was similarly often observed. A follow-up was only available over the course of one year and one might hypothesize that a longer follow-up may have altered observed effects. However, several studies investigating the relationship of heart rate to outcomes in ICD patients report a relevant effect of different HR cut-offs on chosen endpoints within one year which lasted for the remaining follow-up of 2–6 years11,12,17.
Methods
German DEVICE registry
The German DEVICE registry is a national multicenter prospective internet-based database of patients undergoing pacemaker, ICD or CRT device implantation or revision initiated by the ‘Institut für Herzinfarktforschung’ (IHF, Ludwigshafen, Germany) including 50 German centers23,24. A detailed description on patient enrollment and data collection has been published before24,25. In the current analysis, all patients who underwent transvenous ICD or CRT-D de novo implantation or device revision between March 2007 until February 2014 were identified. Device revision included the following procedures: device replacement due to battery depletion or device malfunction, device upgrade to a 2-chamber or CRT-D-system, lead revision due to function defects and device extraction with subsequent implantation of a new device and leads due to pocket or device infection. All patients gave written informed consent for procedures and registry participation. The ethics committee of the Rhineland-Palatinate State Medical Council (Landesärztekammer Rheinland-Pfalz) approved the study, which was conducted in accordance with the provisions of the Declaration of Helsinki, all relevant guidelines and regulations.
Resting heart rate analysis
The baseline rhythm and RHR were determined in supine position via a 12-lead electrocardiogram conducted during the first day of hospitalization of the patient within morning times. Patients presenting with atrial tachyarrhythmia, intermittent / continuous pacemaker stimulation or atrioventricular block of any degree at the time of enrollment were excluded from analysis. Patient characteristics and the impact of baseline RHR on procedural data, periprocedural outcomes and long-term outcomes at 1-year follow-up were investigated. Patients with a RHR ≤ 70 bpm and > 70 bpm were compared. A subgroup analysis was conducted to analyze the influence of comorbidities on 1-year mortality comparing patients with a RHR ≤ 70 bpm vs > 70 bpm.
Follow-up
A detailed description of the follow-up data collection has been provided before24. In short, patients were contacted by the IHF via telephone one year after the index device implantation or device replacement with a dedicated follow-up questionnaire regarding cardiovascular outcomes. Additional device interrogation data were collected for analysis of ICD shocks and ventricular arrhythmia. Data on mortality were gathered from available medical records, outpatient caring physicians or civil registration offices.
Statistical analysis
The patient cohort is characterized by descriptive statistics. Continuous data are provided as medians with 25th and 75th percentiles. For determination of differences of continuous data, the Mann-Whitney-U test was applied. Distributions of categorical variables were compared using Pearson chi-square test. In case of a significant difference, odds ratios with 95%-confidence interval are shown. One-year mortality at 365 days after the index procedure was estimated by the Kaplan-Meier method and compared between groups by log-rank test. Rates of non-lethal follow-up events are reported as percentages among survivors. A p-value < 0.05 is considered statistically significant. Statistical analysis was conducted using the SAS software (release 9.4, SAS Institute, Inc., Cary, NC, USA). All analyses were performed at the Biometrics department of the IHF.
Conclusion
Among patients with an ICD or CRT-D, a RHR > 70 bpm assessed within clinical practice at admission may indicate patients at increased risk of all-cause hospital admission but not of other adverse cardiovascular events or death at 1 year.
Data availability
The datasets analyzed during the current study are available from the corresponding author upon reasonable request.
Abbreviations
- CRT-D:
-
Cardiac resynchronization therapy-defibrillator
- ICD:
-
Implantable cardioverter-defibrillator
- HFrEF:
-
Heart failure with reduced ejection fraction
- IHF:
-
Institut für Herzinfarktforschung
- LVEF:
-
Left ventricular ejection fraction
- RHR:
-
Resting heart rate
- VF:
-
Ventricular fibrillation
- VT:
-
Ventricular tachycardia
References
Kanazawa, H. & Fukuda, K. The plasticity of cardiac sympathetic nerves and its clinical implication in cardiovascular disease. Front. Synaptic Neurosci. 14, 960606. https://doi.org/10.3389/fnsyn.2022.960606 (2022).
Lechat, P. et al. Heart rate and cardiac rhythm relationships with bisoprolol benefit in chronic heart failure in CIBIS II trial. Circulation 103, 1428–1433. https://doi.org/10.1161/01.cir.103.10.1428 (2001).
McAlister, F. A., Wiebe, N., Ezekowitz, J. A., Leung, A. A. & Armstrong, P. W. Meta-analysis: beta-blocker dose, heart rate reduction, and death in patients with heart failure. Ann. Intern. Med. 150, 784–794. https://doi.org/10.7326/0003-4819-150-11-200906020-00006 (2009).
Fox, K. et al. Heart rate as a prognostic risk factor in patients with coronary artery disease and left-ventricular systolic dysfunction (BEAUTIFUL): a subgroup analysis of a randomised controlled trial. Lancet 372, 817–821. https://doi.org/10.1016/s0140-6736(08)61171-x (2008).
Diaz, A., Bourassa, M. G., Guertin, M. C. & Tardif, J. C. Long-term prognostic value of resting heart rate in patients with suspected or proven coronary artery disease. Eur. Heart J. 26, 967–974. https://doi.org/10.1093/eurheartj/ehi190 (2005).
McDonagh, T. A. et al. ESC Guidelines for the diagnosis and treatment of acute and chronic heart failure. Eur. Heart J. 42, 3599–3726. https://doi.org/10.1093/eurheartj/ehab368 (2021).
Heidenreich, P. A. et al. 2022 AHA/ACC/HFSA Guideline for the management of Heart failure: a report of the American College of Cardiology/American Heart Association joint committee on clinical practice guidelines. Circulation https://doi.org/10.1161/cir.0000000000001063 (2022).
Bohm, M. et al. Heart rate as a risk factor in chronic heart failure (SHIFT): the association between heart rate and outcomes in a randomised placebo-controlled trial. Lancet 376, 886–894. https://doi.org/10.1016/S0140-6736(10)61259-7 (2010).
Swedberg, K. et al. Ivabradine and outcomes in chronic heart failure (SHIFT): a randomised placebo-controlled study. Lancet 376, 875–885. https://doi.org/10.1016/s0140-6736(10)61198-1 (2010).
Ahmadi-Kashani, M. et al. Heart rate predicts outcomes in an implantable cardioverter-defibrillator population. Circulation 120, 2040–2045. https://doi.org/10.1161/circulationaha.108.847608 (2009).
Hoogwegt, M. T., Theuns, D. A. M. J., Pedersen, S. S. & Kupper, N. Long-term mortality risk in patients with an implantable cardioverter–defibrillator: influence of heart rate and QRS duration. Int. J. Cardiol. 175, 560–564. https://doi.org/10.1016/j.ijcard.2014.06.030 (2014).
Beinart, R. et al. Relation between resting heart rate and the risk of ventricular tachyarrhythmias in MADIT-RIT. EP Europace 22, 281–287. https://doi.org/10.1093/europace/euz353 (2020).
Køber, L. et al. Defibrillator implantation in patients with nonischemic systolic heart failure. N. Engl. J. Med. 375, 1221–1230. https://doi.org/10.1056/NEJMoa1608029 (2016).
Kirchhof, P. et al. Early rhythm-control therapy in patients with atrial fibrillation. N. Engl. J. Med. 383, 1305–1316. https://doi.org/10.1056/NEJMoa2019422 (2020).
Hindricks, G. et al. ESC Guidelines for the diagnosis and management of atrial fibrillation developed in collaboration with the European association of cardio-thoracic surgery (EACTS). Eur. Heart J. https://doi.org/10.1093/eurheartj/ehaa612 (2020).
Van Gelder, I. C. et al. Lenient versus strict rate control in patients with atrial fibrillation. N Engl. J. Med. 362, 1363–1373. https://doi.org/10.1056/NEJMoa1001337 (2010).
Olshansky, B. et al. Heart rate score predicts mortality independent of shocks in ICD and CRT-D patients. J. Interventional Cardiac. Electrophysiol. 58, 103–111. https://doi.org/10.1007/s10840-019-00688-8 (2020).
Sharma, A. D. et al. Heart rate score, a measure related to chronotropic incompetence in pacemaker patients. Heart Rhythm O2 2, 124–131. https://doi.org/10.1016/j.hroo.2021.02.002 (2021).
Wilkoff, B. L. et al. A device histogram-based simple predictor of Mortality Risk in ICD and CRT-D patients: the heart rate score. Pacing Clin. Electrophysiol. 40, 333–343. https://doi.org/10.1111/pace.13036 (2017).
Infeld, M. et al. Effect of personalized accelerated pacing on quality of life, physical activity, and atrial fibrillation in patients with preclinical and overt heart failure with preserved ejection fraction: the myPACE randomized clinical trial. JAMA Cardiol. https://doi.org/10.1001/jamacardio.2022.5320 (2023).
Kronborg, M. B. et al. Atrial pacing minimization in sinus node dysfunction and risk of incident atrial fibrillation: a randomized trial. Eur. Heart J. https://doi.org/10.1093/eurheartj/ehad564 (2023).
McCrory, C. et al. Speed of heart rate recovery in response to orthostatic challenge. Circul. Res. 119, 666–675. https://doi.org/10.1161/circresaha.116.308577 (2016).
Frommeyer, G. et al. Implantable cardioverter defibrillators in patients with electrical heart disease and hypertrophic cardiomyopathy: data from the German device registry. Clin. Res. Cardiol. 109, 508–512. https://doi.org/10.1007/s00392-019-01532-9 (2020).
Spitzer, S. G. et al. Long-term outcomes after event-free cardioverter defibrillator implantation: comparison between patients discharged within 24 h and routinely hospitalized patients in the German DEVICE registry. Europace 19, 968–975. https://doi.org/10.1093/europace/euw117 (2017).
Bogossian, H. et al. Single chamber implantable cardioverter defibrillator compared to dual chamber implantable cardioverter defibrillator: less is more! data from the German device registry. Clin. Res. Cardiol. 109, 911–917. https://doi.org/10.1007/s00392-019-01584-x (2020).
Funding
The German DEVICE registry is financed by the Institut für Herzinfarktforschung, Ludwigshafen, Germany. The current work received no industry sponsorship.
Author information
Authors and Affiliations
Contributions
Conceptualization: F.A.A., J.S. and C.M.; data acquisition: J.S. and M.H.; data analysis: M.H.; data interpretation: F.A.A., J.S., M.H. and C.M.; funding acquisition: J.S.; project administration: J.S., M.H. and C.M.; supervision: J.S. and C.M.; visualization: F.A.A.; writing—original draft: F.A.A.; writing—review and editing, F.A.A., J.S., S.S., M.H., J.B., T.K., L.E., G.S., M.L., C.S., B.Z., R.B., A.K.K. and C.M. All authors have read and agreed to the published version of the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Alken, FA., Senges, J., Schneider, S. et al. Resting heart rate assessed within clinical practice demonstrates no prognostic relevance for defibrillator recipients in the German DEVICE registry. Sci Rep 14, 29189 (2024). https://doi.org/10.1038/s41598-024-78851-z
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-024-78851-z